Article Search
Volume 27.1
January–April 2024
Full table of contents
ISSN: 1094-8074, web version;
1935-3952, print version
Recent Research Articles
See all articles in 27.1 January-April 2024
See all articles in 26.3 September-December 2023
See all articles in 26.2 May-August 2023
See all articles in 26.1 January-April 2023
For all supplemental material see 492_supplemental.zip.
SUPPLEMENTARY MOVIE
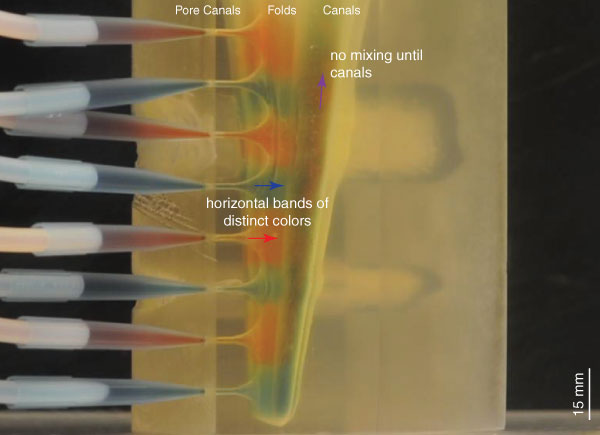
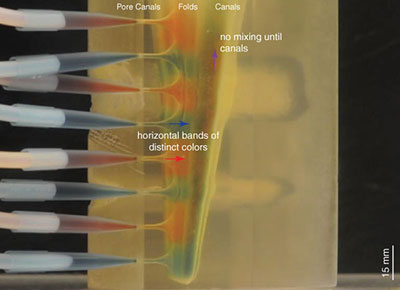
Visualization of the flow within the 3D printed model (Re = 0.376, see Table 1). Flow in the folds consists of horizontal bands of distinct red and blue color, indicating no adoral component to flow and no mixing within the folds, consistent with Hypothesis 2 (see text, Figure 2.2). The animation is sped up 16x. Figure 4 shows a single frame from the flow pattern observed, showing the steady-state flow pattern after nine minutes of flow. Click on image to activate animation.
SUPPLEMENTARY TABLE S1.
Full calculations for in vivo flow conditions.
| What is the Re for blastoid Pentremites rusticus in vivo? | ||
| Assumed values shown in blue along with source | ||
| Environmental data | ||
| nu, kinematic viscosity, m^2/s | 1.00E-006 | Use 1e-6 for 25C, 1.4 for 12C, 1.8 for 0C |
| Case 1: Cilia in pore canals | ||
| At the pore | ||
| Pore diameter, m | 2.54E-005 | Measured from Schmidtling and Marshall 2010 |
| Pore area, m^2 | 0.0000000005 | |
| Clia velocity, m/s | 6.00E-004 | Paul 1978 |
| Volumetric flow rate m^3/s | 0.0 | |
| Re,D pore | 0.01524 | Implies laminar flow |
| In the folds | ||
| Spacing between folds, m | 4.26E-005 | |
| Spacing between pores, m | 2.65E-004 | Schmidtling and Marshall 2010 |
| Number of folds per pore | 3 | From Schmidtling and Marshall, figure 6 |
| Fold area, m^2 | 0.0000000339 | 66.809793371 |
| Volumetric flow rate m^3/s | 0.0 | |
| Velocity in fold, m/s | 0.0000089807 | m/s |
| Dh fold, 4*FA/WP | 0.0000733744 | for finite channels |
| Dh fold, 2*spacing | 0.0000851648 | for infinite array of closely spaced plates |
| Re,Dh fold | 0.0006589551 | |
| Re,Dh2 fold | 0.0007648415 | Implies laminar flow |
| Case 2: Cilia in folds | ||
| At the pore | ||
| Pore diameter, m | 2.54E-005 | Measured from Schmidtling and Marshall 2010 |
| Pore area, m^2 | 0.0000000005 | |
| Volumetric flow rate m^3/s | 0.0 | |
| Pore velocity, m/s | 0.040085876 | |
| Re,D pore | 1.018181251 | Implies laminar flow |
| In the folds | ||
| Spacing between folds, m | 4.26E-005 | NOT measured in Schmidtling and Marshall; Tony measured in x-ray image |
| Spacing between pores, m | 2.65E-004 | 7x pore diameter |
| Number of folds per pore | 3 | From Schmidtling and Marshall, figure 6 |
| Fold area, m^2 | 0.0000000339 | |
| Cilia velocity, m/s | 6.00E-004 | Paul 1978 |
| Volumetric flow rate m^3/s | 0.0 | |
| Dh fold, 4*FA/WP | 0.0000733744 | For finite channels |
| Dh fold, 2*spacing | 0.0000851648 | For infinite array of closely spaced plates |
| Re,Dh fold | 0.0440246517 | |
| Re,Dh2 fold | 0.0510989011 | Implies laminar flow |
| Case 2 is the controlling case (higher Re, highest mixing expected there). | ||
SUPPLEMENTARY TABLE S2.
Full calculations for model flow conditions as tested.
| What is the Re for the model as tested? | |||
| Assumed values shown in blue along with source | |||
| Environmental data | |||
| nu, kinematic viscosity, m^2/s | 1.00E-006 | Use 1e-6 for freshwater at 25 C | |
| Use 27e-6 for mineral oil | |||
| At the faucet | |||
| Time, s | 4034 | Tony Huynh measured | |
| Level change, m | 0.0025 | Tony Huynh measured | |
| Area, m^2 | 0.1114302763 | Tony Huynh measured, for McMurdo portable tank with black screens | |
| Volumetric flow rate m^3/s | 0.0000000691 | ||
| At the pore | |||
| pPore diameter, m | 1.85E-003 | TH design, CM, DE measured | |
| Pore area, m^2 | 2.69E-006 | ||
| Number of pores connected | 8 | ||
| Volumetric flow rate m^3/s | 0.0000000086 | 1/8 of flow from tap | |
| Velocity at pore, m/s | 0.0032113231 | ||
| Re,D pore | 5.9409476461 | transition, less than 10 | |
| In the folds | |||
| Spacing between folds, m | 3.10E-003 | TH design, CM DE measured | |
| Spacing between pores, m | 0.0153 | TH designed, DE meas STL | |
| Number of folds per pore | 3 | Tony Huynh designed into model, Fig 3 | |
| Fold area, m^2 | 1.42E-004 | 52.9347713135 | |
| Volumetric flow rate m^3/s | 0.0000000086 | ||
| Velocity in fold, m/s | 0.0000606657 | m/s | |
| Dh fold, 4*FA/WP | 0.0051554348 | for finite channels | |
| Dh fold, 2*spacing | 0.0062 | for infinite array of closely spaced plates | |
| Re,Dh fold | 0.3127578744 | ||
| Re,Dh2 fold | 0.376127117 | laminar | |
| 7.4 | x in vivo Re | ||
SUPPLEMENTARY TABLE S3.
Full calculations for model operated to match lowest Re in vivo case.
| What speed would we have to run at to match in vivo | ||
| Assumed values shown in blue along with source | ||
| Environmental data | ||
| nu, kinematic viscosity, m^2/s | 1.00E-006 | Use 1e-6 for freshwater at 25 C |
| Use 30e-6 for mineral oil | ||
| At the pore | ||
| Pore diameter, m | 1.85E-003 | Tony Huynh designed |
| Pore area, m^2 | 0.000002688 | |
| Volumetric flow rate m^3/s | 0.0000000016 | |
| Velocity at pore, m/s | 5.85E-004 | |
| Re,D pore | 1.081961112 | Transition, less than 10 |
| In the folds | ||
| Spacing between folds, m | 3.10E-003 | Tony Huynh designed |
| Spacing between pores, m | 0.0153 | Tony Huynh designed |
| Number of folds per pore | 3 | Tony Huynh designed into model, Fig 3 |
| Fold area, m^2 | 0.00014229 | |
| Volumetric flow rate m^3/s | 0.0000000016 | |
| Velocity in fold, m/s | 0.0000110484 | m/s |
| Dh fold, 4*FA/WP | 0.0051554348 | For finite channels |
| Dh fold, 2*spacing | 0.0062 | For infinite array of closely spaced plates |
| Re,Dh fold | 0.0569592391 | |
| Re,Dh2 fold | 0.0685 | Implies laminar flow |
| Re in vivo, case 1 | 0.0007648415 | |
| Re in vivo, case 2 | 0.0510989011 | |
SUPPLEMENTARY FILE.
Stereolithography (STL) file of model of the distal end of the hydrospire in Pentremites rusticus (see palaeo-electronica.org/content/2015/1073-blastoid-hydrospire-fluid-flow) (see 492_supplemental.zip).
TABLE 1. Calculation of the Re in the most distal (aboral) portion of the hydrospire, corresponding to the first eight hydrospire pores, in living Pentremites rusticus and in the 72x scale model.
FIGURE 1. Anatomy of the hydrospires of the blastoid Pentremites rusticus. 1.1, Location of one of the five radially distributed hydrospires within the calyx, showing incurrent hydrospire pores, and excurrent spiracle (inferred direction of water flow indicated by the arrows). 1.2, Oblique view of a section of a hydrospire and associated structures. Modified from Schmidtling and Marshall (2010).
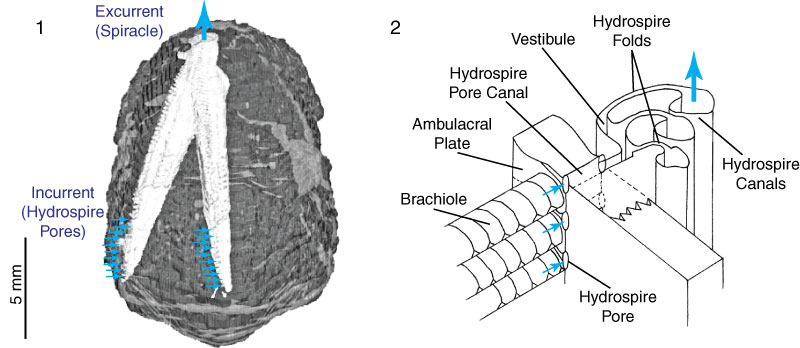
FIGURE 2. Schematic showing hypothesized flow patterns within the hydrospire folds. 2.1, In Hypothesis 1, the flow has an adoral component representing respiratory leakage. 2.2, In Hypothesis 2, the flow is entirely radial, without leakage. See text for further discussion.
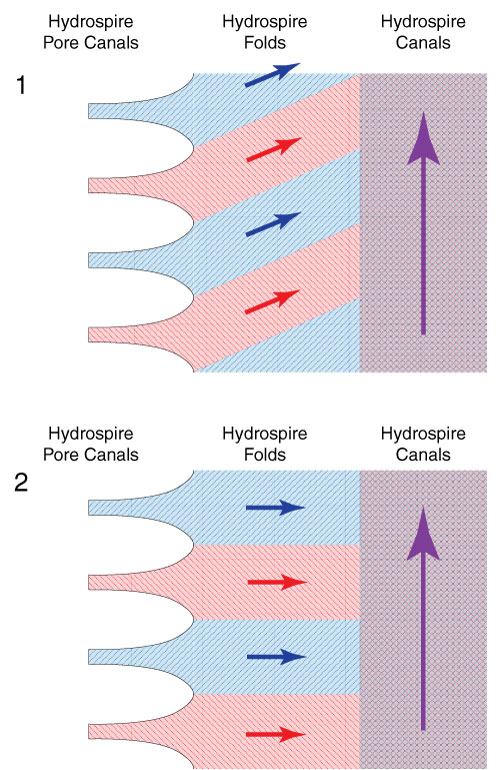
FIGURE 3. Digital and physical models use to visualize fluid flow. 3.1, Digital solid model of approximately the lower quarter of a hydrospire of Pentremites rusticus, using Blender (see text). 3.2, 3D-printed rendering of the digital model, shown with inlet headers connected.
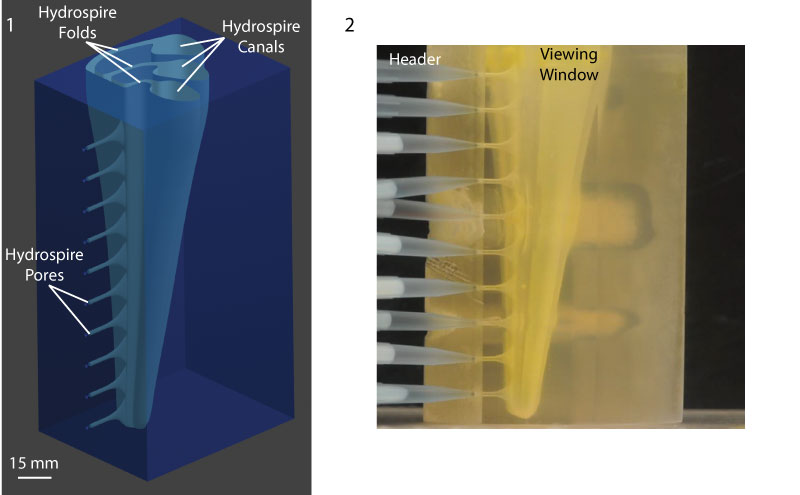
FIGURE 4. Visualization of the flow within the 3D printed model (Re = 0.376, see Table 1). Flow in the folds consists of horizontal bands of distinct red and blue color, indicating no adoral component to flow and no mixing within the folds, consistent with Hypothesis 2 (see text, Figure 2.2). The still used in the print version of this paper is a single frame from the flow pattern observed, showing the steady-state flow pattern after nine minutes of flow. The animation is sped up 16x (for video see supplementary material).
 Tony L. Huynh
Tony L. Huynh
Department of Integrative Biology
University of California, Berkeley
Berkeley, California 94720-3140 USA
huynhtony@berkeley.edu
Tony L. Huynh received an undergraduate degree from the department of Integrative Biology at UC Berkeley in 2012. He likes using tools and approaches from mechanical engineering, computer-aided 3D design, and computed tomography (CT imaging) to answer evolutionary and ecological questions related to functional morphology. He is always on the hunt for another specimen to put into a CT scanner and has worked with vertebrates, invertebrates, and plants, along the entire range of geologic time. He is currently the lab manager for a metabolic physiology lab specializing in invertebrates at UC Berkeley.

 Dennis Evangelista
Dennis Evangelista
Department of Biology
University of North Carolina at Chapel Hill
Chapel Hill, North Carolina 27599-328
USA
devangel@live.unc.edu
Dennis Evangelista is a postdoctoral researcher in comparative biomechanics. He studied mechanical and electrical engineering at MIT and Naval Postgraduate School before earning a PhD in Integrative Biology at UC Berkeley. He enjoys working with undergraduates, flight, swimming, maneuvering and control, behavior of aggregations, evolution, and biomechanics in extreme environments, and has worked on vertebrates, invertebrates, and plants, both extinct and extant. He is a good cook and is raising a guide dog puppy.

 Charles R. Marshall
Charles R. Marshall
University of California Museum of Paleontology and Department of Integrative Biology
University of California, Berkeley
Berkeley, California, 94720-478
USA
crmarshall@berkeley.edu
Charles Marshall is currently the Director of the University of California Museum of Paleontology and a Professor in the Department of Integrative Biology at the University of California, Berkeley. His undergraduate degree from the Australian National University was in geology (especially in palaeontology), zoology, applied mathematics, physical chemistry, and history, and included the equivalent of a Masters thesis on lungfish. His Ph.D. (on sand dollars) was from the Committee on Evolutionary Biology at the University of Chicago, followed by an NIH postdoc in evo-devo with Rudy Raff at Indiana University (where he shifted to regular echinoids). His research interests have been described as being pathologically broad, which does not seem to be an unfair description.
Visualizing the fluid flow through the complex skeletonized respiratory structures of a blastoid echinoderm
Plain Language Abstract
Our goal is to understand the functional effectiveness of the remarkably complex skeletonized respiratory structures in blastoids, an extinct group of bottom-dwelling stalked echinoderm. The complexity of the structures required a modeling approach, and we used a scaled-up 3D printed model of part of one of these structures to directly observe the pattern of water flow within it. Our results show that the path of water flow within the structures is consistent with a respiratory system able to function with high effectiveness. While it is notoriously difficult to determine the exact reasons for the evolutionary success of one group over other groups, it is possible that the hydrospires also helped the blastoids with some other function beyond respiration, for example, feeding.
Resumen en Español
Visualizando el flujo de fluido a través de las complejas estructuras esqueléticas respiratorias de un equinodermo blastoideo
Los blastoideos del grupo Spiraculata tienen extraordinarias estructuras respiratorias esqueléticas internas, las hidrospiras ("hydrospires"). Sin embargo, el patrón detallado del flujo de agua de mar dentro de las hidrospiras es desconocido, por lo que es difícil evaluar su eficacia respiratoria. Con el uso de un modelo físico impreso 3D de mayor escala (72x), para visualizar el flujo de agua a través de la parte más distal (aboral) de la hidrospira de Pentremites rusticus, mostramos que el flujo era coherente con el intercambio respiratorio eficaz en los pliegues de la hidrospira- el flujo continuaba horizontalmente dentro de los pliegues de la hidrospira pasando después a través de los canales de poros de la hidrospira y solo desarrollaba un componente adoral a su velocidad una vez que había entrado en los canales de la hidrospira. El flujo laminar ordenado observado es compatible con los valores de Reynolds que estimamos para un blastoideo vivo (Re = 0,0008-0,05). Aunque la mayoría de los análisis funcionales de hidrospiras espiraculadas se han centrado en su función respiratoria, también es posible que estas jugasen un papel importante en la alimentación, ayudando a sacar agua más allá de las braquiolas, lo cual constituye una hipótesis que puede ser objeto de futuras comprobaciones.
Palabras clave: Blastoidea; Echinodermata; Morfología funcional; Flujo de fluido; Hidrospira; Modelo impreso tridimensional
Traducción: Enrique Peñalver
Résumé en Français
Visualisation de l'écoulement du fluide à travers les structures respiratoires squelettisées complexes d'un échinoderme blastoïde
Les blastoïdes spiraculés ont des structures respiratoires squelettisées internes extraordinaires, les hydrospires. Cependant, le modèle détaillé de l'écoulement de l'eau de mer en leur sein n'est pas connu, ce qui rend difficile l'évaluation de l'efficacité respiratoire. En utilisant un modèle physique imprimé en 3D agrandis (72x) afin de visualiser le flux d'eau à travers la partie la plus distale (aborale) de l'hydrospire de Pentremites rusticus, nous montrons que le flux était consistent avec un échange respiratoire efficace dans les plis d'hydrospire - le flux a continué horizontalement dans les plis d'hydrospire après le passage à travers les canaux de pores d'hydrospire et a seulement développé un composant adorale à sa vitesse une fois qu'il était entré dans les canaux d'hydrospire. Le flux laminaire et ordonné observé est consistent avec les nombres de Reynolds que nous estimons pour un blastoïde vivant (Re = 0,0008 à 0,05). Alors que la plupart des analyses fonctionnelles des hydrospires spiraculés sont concentrées sur leur fonction respiratoire, il est également possible qu'ils ont joué un rôle dans l'alimentation, en aidant à puiser de l'eau au-delà des brachioles, ce qui est une hypothèse qui est prêtée à une analyse future.
Mots-clés: Blastoidea; Echinodermata; Morphologie fonctionnelle; Écoulement de fluide; Hydrospire; modèle imprimé en trois dimensions
Translator: Kenny J. Travouillon
Deutsche Zusammenfassung
Sichtbarmachung des Flüssigkeitsstroms durch die komplexen skelettierten Atemwegsstrukturen eines blastoiden Echinodermen
Spirakulate Blastoide haben außergewöhnliche interne skelettierte Atemwegsstrukturen, die Hydrospire. Jedoch ist nicht bekannt wie genau das Meerwasser hindurchströmt, was die Bestimmung der respiratorischen Wirksamkeit der Blastoide erschwert. Mit einem vergrößerten (72x) 3D-Druckmodell visualisierten wir den Wasserfluss durch den am weitesten distal (aboral) gelegenen Teil der Hydrospire von Pentremites rusticus. Wir zeigten, dass der Durchfluss bei effektivem respiratorischen Austausch in den Falten der Hydrospire gleich bleibend war – der Durchfluss setzte sich horizontal fort innerhalb der Falten der Hydrospire nachdem die Porenkanäle passiert waren und nahm erst eine adorale Geschwindigkeitskomponente an als die Hydrospiren-Kanäle passiert waren. Die beobachtete geordnete laminare Strömung stimmt mit der Reynoldszahl die bei einem lebenden Blastoiden (Re = 0.0008–0.05) zu erwarten ist überein. Während die meisten Funktionsanalysen über spirakulate Hydrospire auf die Atemwegsfunktion fokussiert sind, ist es ebenso möglich, dass diese eine Rolle bei der Nahrungsaufnahme spielten indem sie Wasser hinter die Brachiolen brachten, was eine Hypothese für weitere Untersuchungen ist.
Schlüsselwörter: Blastoidea; Echinodermata; Funktionsanalyse; Flüssigkeitsstrom; Hydrospire; dreidimensionales Druckmodell
Translator: Eva Gebauer
Arabic
in progress
Translator: Ashraf M.T. Elewa
-
-
-
Review: The Princeton Field Guide to Mesozoic Sea Reptiles
 The Princeton Field Guide to Mesozoic Sea Reptiles
The Princeton Field Guide to Mesozoic Sea ReptilesArticle number: 26.1.1R
April 2023


 Poster Winners 2024
Poster Winners 2024