Article Search
Volume 27.1
January–April 2024
Full table of contents
ISSN: 1094-8074, web version;
1935-3952, print version
Recent Research Articles
See all articles in 27.1 January-April 2024
See all articles in 26.3 September-December 2023
See all articles in 26.2 May-August 2023
See all articles in 26.1 January-April 2023
APPENDIX 1.
Character matrix of wing and tegmina characters. Taken from Rehn (1951). We read through the original document and extracted character data from the taxonomic keys and descriptions of taxa. When conflicting information was encountered, we recorded both accounts and noted the conflict with an "or" statement or with a "?". In some cases, character information was inferred and marked with a "?", but most often missing data was left blank. (Supplied as XLS file.)
APPENDIX 2.
Comment on use of Rehn, JWH. (1951). “Classification of the Blattaria as indicated by their wings (Orthoptera)”. Memoirs of the American Entomological Society. 14. 1-134
As his doctoral dissertation, John W.H. Rehn (not to be confused with J.A.G. Rehn, his father) did an extensive review of the wing and tegminal morphology of cockroaches. Given that fossil cockroaches are largely classified based on their wings, this publication is a major point of reference. However, cockroach wing morphology is currently known to give erroneous phylogenetic signal (Princis, 1960; Klass, 2001) and thus fossil classifications may suffer from this error. That is not to say that Rehn (1951) is not an important work. It is a major resource for wing morphology and it set many of the conventions used today. However, as we discuss below, the many errors in this paper make improper use of the work potentially misleading.
In historical context Rehn’s paper was important, although perhaps not as influential as McKittrick (1964) or Princis (1963). At first it was received positively. Gurney (1951) reviewed Rehn (1951) and said, “Fundamentally, the classification which he offers appears to be very sound, and modifications which specialists will undoubtedly suggest will in no way detract from the basic value of the work or the regularity with which it will be consulted for many decades”. However, soon the work’s classifications were largely rejected (Princis, 1960; McKittrick, 1964) with some exception (for examples see [Roth and Willis 1957; Roth, 1977, 1996]). In the modern era, phylogenetic studies generally do not reference “Classification of the Blattaria as indicated by their wings” (Grandcolas, 1996; Maekawa et al., 1999; Deitz et al., 2003; Maekawa et al., 2003; Ware et al., 2008; Djernæs et al., 2012; Djernæs et al., 2015) with the exception of a single morphological character cited by Thorne and Carpenter (1992) or references to its illustrations (Klass and Meier, 2006).
The paper consists mainly of morphological descriptions of wings and tegmina at various taxonomic levels (superfamilies, families, subfamilies and tribes), and a series of wing and tegminal illustrations. Although the paper represents an exhaustive synthesis of data, much of the written work is unreliable. The language of the work is highly subjective, often vague and sometimes inconsistent. Illustrating all three of these points, the dissertation frequently refers to the length of the subcosta as simply “long” or “short”. This is sometimes expanded upon by giving the subcostal length relative to wing length. However, references to a subcostal length equivalent to half the wing length are described as both “long” or “short” at different times. There are many other instances where the assertions about comparative morphology are often incorrect when simply cross referenced with other parts of this work (in particular see the examples at the end of this section). Finally, Klass and Meier (2006) point out that Rehn (1951) is unable to thoroughly define vein homologies because he omits detailed analysis of the wing base.
The characters described in the paper are infrequently uniformly distributed within the clades defined, and they are almost always plesiomorphic or homoplastic. Maybe most unfortunate of all, the paper fails to describe the state of many of characters in a large proportion of the taxa discussed (57% empty cells in Appendix 1).
Rehn did complete an extremely ambitious undertaking in his 1951 paper, and much of this contribution is still valuable. Many authors still use the terminology in Rehn (1951) for the wing and tegminal veins (e.g., Anisyutkin, 2008, 2014; Wang et al., 2014; Evangelista et al., 2016). The illustrations are a common point of reference and are reproduced in multiple works (e.g., Klass, 2001). However, relying solely on the written descriptions of wing venation can be misleading.
Below are only some of the errors and inconsistencies in “Classification of the Blattaria as indicated by their wings”. “Conflicts” or “contradictions” refer to instances where the author made irreconcilable statements about the state of a morphological character in a taxon.
Holocompsinae - Contradiction between the key and the description. Key says they don’t have psuedostigma on the wings but the description says they do.
Corydiinae - Numerous conflicting statements about the number of branches in the tegmina’s media, regularity of the radius, and minimum number of anal veins in the wings.
Latindiinae - The description of the subfamily conflicts with the descriptions of the tribes it contains. The number of branches on R1 in the wings is unclear.
Latindiini - There is a conflict about the number of anal veins in the wings.
Compsodiini - Conflict in the number of anal veins in tegmina.
Panesthiidae - Contradiction with earlier sections about the length of the anal area in tegmina and some other characters. The description contradicts itself about the maximum number of branches in the radial sector.
Nyctiborinae - Contradiction about the regularity of the radial veins.
Epilamprinae - Contradiction about the branching of the media vein, and the distance between p3 and the branched axillary.
Perisphaerini - Two contradictions (one with the previous description of Epilamprinae and one within the description) about the shape of the plical furrow of the tegmina. There are also contradictions about the shape of the anterior margin.
Nauphoetini - There is a conflict about the minimum number of branches on the branched axillary vein of the wings. It is also unclear if there is an apical posterior branch on the radius or not.
Panchlorini - There is a contradiction about the regularity of branches in the subcosta of tegmina.
Thoracini - There is a conflict in the number of branches in the branched axillary veins in the wings.
Parcoblattini - Many of the characters he discusses in other sections are completely missing from this description.
Eustegastini - Conflicting statements about the number of branches of the media of the wings.
Euandroblattini - Conflicting statements about the weight of the plical furrow in the tegmina.
Ischnopterini - Conflicting statements about the presence of apical radial rami of tegmina. There is a contradiction of terminology about the apical or anterior subcostal rami. Finally, a contradiction about the number of cubital veins going to the plical furrow tegmina.
Plectopterini - contradiction in the direction of the branches coming off of the fused media and cubitus of tegmina.
APPENDIX 3.
Reflectance Transformation Image (RTI) of “Gyna” obesa (Piton, 1940). High resolution photographs with hemispherical lighting with a focus on the pronotum and thorax. Various light angles emphasize different ridges in the fossil and make it difficult to discern which path the posterior edge of the pronotum takes. The more posterior candidate for the edge of the pronotum could be sculpturing of the middle thoracic segment. The file can be viewed with RTI viewer 1.1.0 (http://culturalheritageimaging.org/What_We_Offer/Downloads/View/) and as zipped RTI file.

APPENDIX 4.
Fossil Blattodea s.s. from Fossil Works database. A list of fossil cockroaches based on the fossil works database (Alroy et al., 2016). The ages and phylogenetic determinations in the list are presented without our scrutiny. We discuss many of these fossils in the paper and consider many of them to be unreliable. (Available as xls file.)
 Dominic A. Evangelista, Muséum National d'Histoire Naturelle, 45 Rue Buffon CP50, Paris, France 75005. DominicEv@gmail.com
Dominic A. Evangelista, Muséum National d'Histoire Naturelle, 45 Rue Buffon CP50, Paris, France 75005. DominicEv@gmail.com
Dominic Anthony Evangelista received his PhD in 2016 in Ecology and Evolutionary Biology from Rutgers, The State University of New Jersey. He has published on cockroach systematics and ecology. He is currently an NSF postdoctoral research fellow at le Muséum national d'Histoire naturelle in Paris, France. His scientific interests include: phylogenomics, the origins of biodiversity, and Blaberoidea.

 Marie Djernæs. Natural History Museum, Cromwell Road, London SW7 5BD, United Kingdom. marie_djernaes@hotmail.com
Marie Djernæs. Natural History Museum, Cromwell Road, London SW7 5BD, United Kingdom. marie_djernaes@hotmail.com
Marie Djernæs received her Ph.D. at University of Alberta and has worked both on Dictyoptera phylogeny (molecular and morphological) and on comparative morphological studies of Trichoptera and Lepidoptera. She is author of the chapter on Blattodea in the forthcoming Insect Diversity II: Science and Society. Her scientific interests include: Dictyoptera phylogeny, detailed studies of morphological structures in insects, evolution of insects and their morphology.

 Manpreet Kaur Kohli. Rutgers University, 195 University Ave, Newark, NJ, USA 07102. mkk24@njit.edu
Manpreet Kaur Kohli. Rutgers University, 195 University Ave, Newark, NJ, USA 07102. mkk24@njit.edu
Manpreet Kohli is pursuing her PhD in Ecology and Evolutionary Biology at Rutgers, The State University of New Jersey. She is interested in studying different aspects of insect evolution. In here current research she is exploring biogeographic history of dragonflies. She also works on how fossil choice, fossil placement and missing data affects the results of divergence time analysis.
FIGURE 1. Cladogram of Blattodea phylogeny synthesized from eight previous studies (figure 2 in Maekawa et al., 2003; figure 4 in Klass and Meier, 2006; figure 1 in Inward et al., 2007; figure 3 in Pellens et al., 2007; figure 2 in Murienne, 2009; figure 2 in Djernæs et al., 2012; figure 3 in Djernæs et al., 2015; figures 4, 5, and 6 in Legendre et al., 2015). Support for a clade is represented by circles, each colored to indicate the studies in which that clade was recovered. Inward et al. (2007) and Pellens et al. (2007) do not report all support values for nodes on their trees so support from the topology of these studies may be over-represented here. The numbering next to Anaplectidae represents different possible positions of this group, or the potential that this group is polyphyletic. Legendre et al. (2015) supports Blattoidea and Blattoidea + Corydioidea but without Anaplectidae - 1, yet will still note their support for these clades with this one exception. Single terminals are shown for other taxa even though some may prove paraphyletic or polyphyletic with further study (e.g., Pseudophyllodromiinae and Epilamprinae). We did not find any reliably supported relationships within most of the Blaberidae, and thus its phylogeny is largely a polytomy. In some cases, a clade was recovered but the study had insufficient taxon sampling to support that clade (e.g., Maekawa et al., 2003 recovered Blattodea as monophyletic by necessity because it did not include Mantodea or other outgroup taxa).
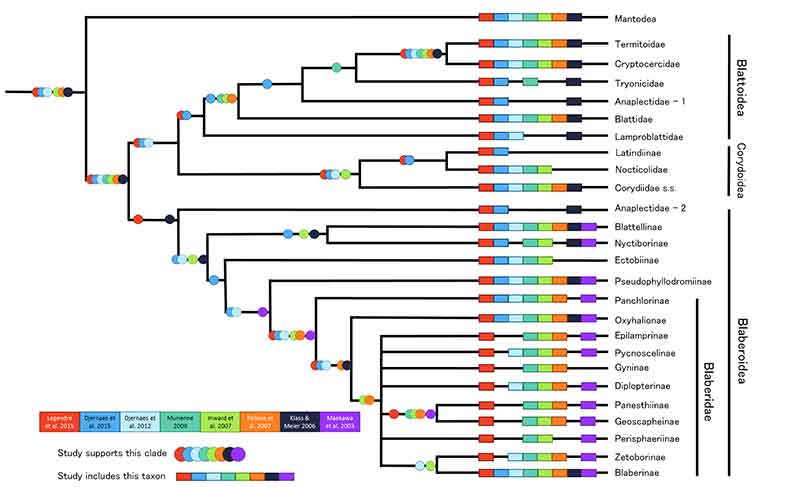
FIGURE 2. Simplified topology of Blattodea with fossil calibrations. Tree topology is simplified from Figure 1. Fossils are shown in grey with dashed lines to represent uncertain position within containing clade. Red symbol indicates the nodes each fossil calibrates with roman numerals indicating the fossil that corresponds to each node calibration. The minimum dates for each of the calibration points are: i - 125.45 Ma; ii - 52 Ma; iii - 48.14 Ma; iv - 48.14 Ma. Corydiidae s.s. consists of Holocompsinae, Tiviinae, and Euthyrrhaphinae. Corydiidae s.l. consists of Corydiidae s.s., Latindiinae and Nocticolinae. Note that no Ectobiinae other than Ectobius have been used in molecular phylogenetic studies to date.
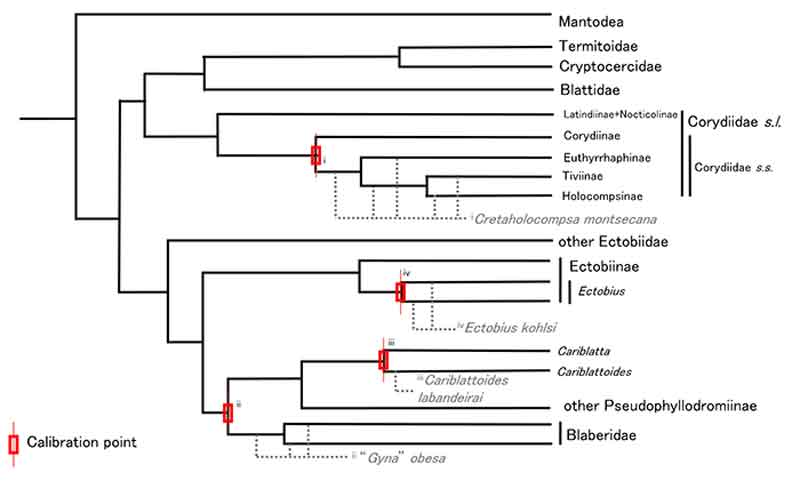
FIGURE 3. Illustrations of calibrating fossils. All illustrations reproduced with permission. Scale bars equal 1 mm. 1, Cretaholocompsa montsecana Martinez-Delclos, 1993 (in Martinez-Delclos, 1993, figure 8). 2, Cariblattoides labandeirai Vršanský, Vidlička, Čiampor, Jr. and Marsh 2012 (in Vršanský et al., 2012, figure 12). 3, Ectobius kohlsi Vršanský, Vidlička, and Labandeira 2014 (in Vršanský et al., 2014, figure 3).
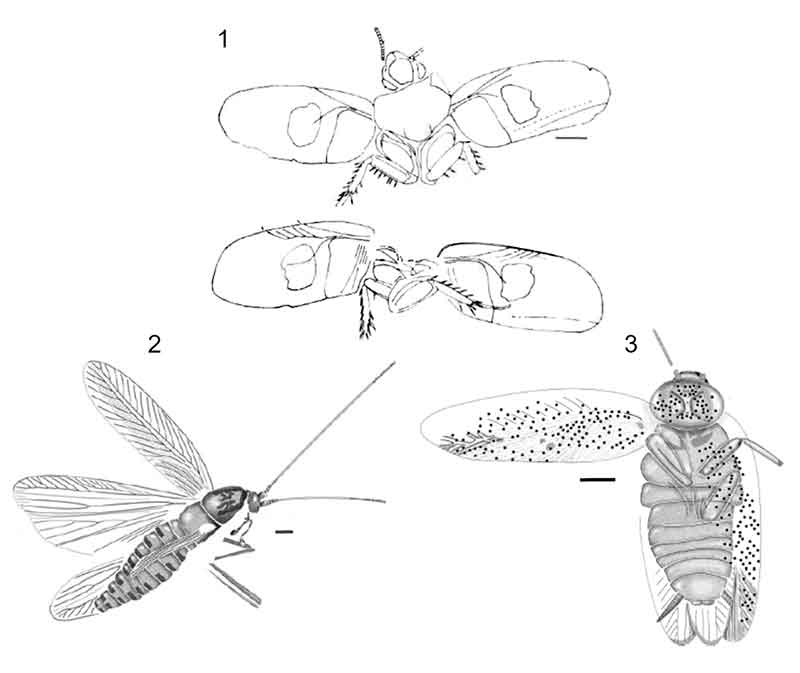
FIGURE 4. “Gyna” obesa (Piton, 1940) with ventral perspective. 1, Illustration of holotype. Hashed line on pronotum denotes two possible paths delimiting the posterior margin of the pronotum. Broken lines elsewhere denote damage or incomplete preservation. Scale bar equals 10 mm. 2, Photo of holotype MNHN.F.R06689. Photo is a composite of two images taken with polarized lighting at different angles. Scale 10 mm total. 3, Higher magnification of posterior end showing stout, segmented cerci and details of abdominal termination. Scale 3 mm total.

Fossil calibrations for the cockroach phylogeny (Insecta, Dictyoptera, Blattodea), comments on the use of wings for their identification, and a redescription of the oldest Blaberidae
Plain Language Abstract
Determining the dates of major evolutionary events requires the use of fossils. However, many fossils are described using insufficient data or are poorly curated. We review the literature of fossil descriptions in order to determine the oldest cockroach fossils that have strong data supporting their identifications. We give the names of four fossils that can be reliably used for determining the ages of major events in cockroach evolutionary history. The oldest non-termite fossil is about 125.5 million years old. We also discuss the use of wing morphology in cockroach taxonomy and emphasize the unreliability of this character system.
Resumen en Español
Calibraciones fósiles para la filogenia de las cucarachas (Insecta, Dictyoptera, Blattodea), comentarios sobre el uso de las alas para su identificación, y una redescripción de los Blaberidae más antiguos
Se proporciona la primera evaluación exhaustiva de las calibraciones fósiles para la estimación del tiempo de divergencia de las cucarachas. A través de una revisión de descripciones publicadas de fósiles, evaluamos los fósiles más antiguos para varios nodos en el grupo corona Blattodea de acuerdo con los mejores métodos propuestos recientemente. Dado que la mayoría de las descripciones de las cucarachas fósiles dependen en gran medida de las venaciones del ala y de la tegmina, también proporcionamos una evaluación crítica de lo indicado en Rehn (1951), que es el trabajo más extenso sobre estos caracteres. Consideramos que Rehn (1951) estableció incorrectamente el estado de numerosos caracteres. Esto, combinado con el bajo número de caracteres informativos en las alas de las cucarachas, afecta negativamente a las justificaciones filogenéticas para algunas de las supuestas cucarachas fósiles más antiguas. Concluimos que actualmente los mejores fósiles para usar en la calibración del árbol de las cucarachas son: Cretaholocompsa montsecana, “Gyna” obesa, Cariblattoides labandeirai, y Ectobius kohlsi. Uno de estos, “Gyna” obesa, fue estudiado de manera insuficiente en su descripción original, por lo que brindamos una redescripción facilitada por imágenes de alta resolución y por un conocimiento moderno en sistemática. Comentamos la dificultad de utilizar las llamadas cucarachas fósiles (fossil roachoids) porque su posición en la base de Dictyoptera está bajo disputa y no se puede verificar de manera fiable. No incluimos calibraciones para linajes de termitas.
Palabras clave: Filogenia; morfología; datación; divergencia; sistemática; paleoentomología
Traducción: Enrique Peñalver (Sociedad Española de Paleontología)
Résumé en Français
Des calibrations fossiles pour la phylogénie des blattes (Insecta, Dictyoptera, Blattodea), avec des commentaires sur l'utilisation des ailes pour leur identification, et une redescription des plus anciens Blaberidae
Dans cet article, nous fournissons la première évaluation détaillée des points de calibration fossile pour estimer les temps de divergence des blattes. En utilisant une synthèse des descriptions de fossiles publiées et en suivant les pratiques les mieux adaptées récemment proposées, nous évaluons les plus anciens fossiles pour plusieurs nœuds du groupe-couronne des Blattodea. Comme la plupart des descriptions de cafards fossiles s'appuient fortement sur la vénation des ailes et du tegmen, nous fournissons aussi une réévaluation critique de Rehn (1951), le travail le plus exhaustif sur ces caractères. Nous concluons que Rehn (1951) a décrit de manière incorrecte l'état de nombreux caractères. Combiné avec le faible nombre de caractères informatifs sur les ailes des blattes, cela affecte négativement les justifications phylogénétiques de certains des plus anciennes blattes présumées. Nous concluons que les meilleurs fossiles connus actuellement pour calibrer l'arbre des blattes sont : Cretaholocompsa montsecana, "Gyna" obesa, Cariblattoides labandeirai, et Ectobius kohlsi. Un de ces taxons, "Gyna" obesa, n'avait pas été traité avec suffisamment de détails dans sa description originelle. Nous fournissons donc une redescription facilitée par l'imagerie à haute résolution et la connaissance de la systématique moderne. Nous commentons sur la difficulté d'utiliser les prétendus fossiles de roachoïdes car leur position à la base de Dictyoptera est débattue et ne peut pas être vérifiée de manière fiable. Nous n'incluons pas de calibrations pour les clades de termites.
Mots-clés : phylogénétique ; morphologie ; datation ; divergence ; systématique ; paléoentomologie
Translator: Antoine Souron
Deutsche Zusammenfassung
Fossil- Kalibrierung für die Schaben-Phylogenie (Insecta, Dictyoptera, Blattodea), Anmerkungen zur Identifikation über Flügelmerkmale und eine Neu-Beschreibung der ältesten Blaberidae
Wir stellen hier die erste Beurteilung von Fossil-Kalibrierung zur Kalkulation des Divergenz-Zeitpunktes der Schaben bereit. Durch eine Überprüfung publizierter Fossilbeschreibungen evaluieren wir älteste Fossilien an verschiedenen Knotenpunkten innerhalb der Kronen-Gruppe Blattodea in Übereinstimmung mit kürzlich vorgeschlagenen Best Practises. Da sich die meisten Beschreibungen fossiler Schaben stark auf die Flügel-und tegminale Nervatur stützen, legen wir auch eine kritische Überprüfung von Rehn (1951) vor, dem umfangreichsten Werk über diese Merkmale. Wir stellen fest, dass Rehn (1951) fehlerhaft über den Zustand einiger Merkmale berichtet hat. Zusammen mit der geringen Anzahl an Merkmalen im Schaben-Flügel beeinflusst dies die phylogenetischen Begründungen einiger der ältesten mutmaßlichen fossilen Schaben. Wir folgern, dass momentan folgende Fossilien am besten geeignet für eine Kalibrierung des Schaben-Stambaumes sind: Cretaholocompsa montsecana, "Gyna" obesa, Cariblattoides labandeirai und Ectobius kohlsi. "Gyna" obesa wurde in der Originalbeschreibung nur unzureichend behandelt, weshalb wir eine Neubeschreibung vorgenommen haben, welche durch hochauflösende Bildgebung und die Kenntnis der modernen Systematik erleichtert wurde. Wir kommentieren die Problematik mit den sogenannten fossilen Roachoiden, da deren Position an der Basis der Dictyoptera umstritten ist und nicht zuverlässig nachgewiesen werden kann. Wir beziehen keine Kalibrierungen für Terminen-Linien mit ein.
Schlüsselwörter: Phylogenetik; Morphologie; Datierung; Divergenz; Systematik; Paläoentomologie
Translator: Eva Gebauer
Arabic

Translator: Ashraf M.T. Elewa
-
-
-
Review: The Princeton Field Guide to Mesozoic Sea Reptiles
 The Princeton Field Guide to Mesozoic Sea Reptiles
The Princeton Field Guide to Mesozoic Sea ReptilesArticle number: 26.1.1R
April 2023


 Poster Winners 2024
Poster Winners 2024