
| Case study 3 - Morphometrics of Ordovician trilobites |
Data files: glaber.dat,
linnars1.dat og
linnars2.dat
 |
Norwegian illaenid trilobite (Paleontological Museum, Oslo). Length approx. 6 cm. |
Although distinctive and often common in Ordovician faunas, illaenid trilobites have relatively featureless, smooth exoskeletons; taxonomic discrimination within the group is therefore difficult. While a number of authors have attempted a qualitative approach to illaenid systematics, Bruton & Owen (1988) described the Norwegian Upper Ordovician members of the family in terms of measurements and statistics. This study seeks to discriminate between at least two different species of the illaenid trilobite Stenopareia.

Four measurements made on the cranidia of the Norwegian illaenid trilobites are defined on the figure. L2, W1, W2 and W3 have been measured on Stenopareia glaber from the Ashgill of Norway (glaber.dat) together with S. linnarssoni from both Norway (linnars1.dat) and Sweden (linnars2.dat). A previous study of S. glaber recognized long and short forms. The first specimen recorded in glaber.dat is a typical long form, the second a typical short form and the third, the lectotype of S. glaber. The remaining measurements relate to further specimens of S. glaber.
Open glaber.dat, and 'commatize' if necessary. Using 'Row color/symbol' in the Edit menu, assign the symbol '+' to row 1 (typical long form), square to row 2 (typical short) and 'x' to row 3 (lectotype). Select the L2 and W1 columns, and choose 'Linear' in the Model menu:
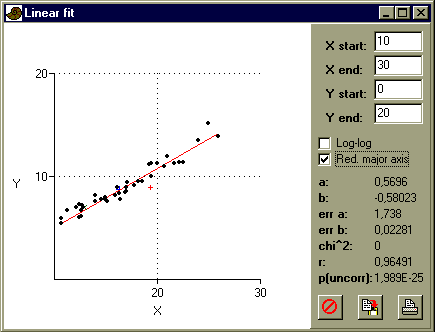
Repeat the exercise with different pairs of variates (L2/W1/W2/W3) plotted against each other. You may have to move columns by dragging the column labels. How do the typical long, short and lectotypic individuals relate to the sample as a whole? Is there any justification for splitting the sample into long and short forms?
Repeat the plotting procedure above (linear fit) for the concatenated data set. For the variety of measurements available, is there a partition between samples on any of the possible XY plots?

All four variables have relatively similar, positive loadings on the first component. Can you interpret this first component in morphological terms? Also look at the loadings for component 2 and 3, and interpret these.
Now choose 'View scatter' in the Principal Components window to see the specimen scores on the two first components:
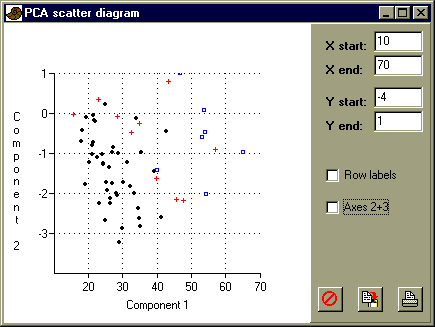
Are the groups well separated? Tick the 'Row labels' box to identify specimen 1 (long form) and 2 (short form) in the S. glaber group. Does their placement on the diagram correspond with your interpretation of component 2?
More information about Principal Components analysis can be found in the manual.
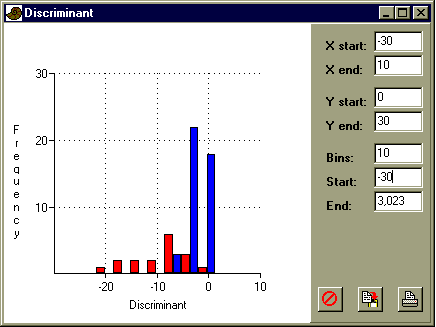
More information about discriminant analysis can be found in the manual.