Article Search
Volume 27.1
January–April 2024
Full table of contents
ISSN: 1094-8074, web version;
1935-3952, print version
Recent Research Articles
See all articles in 27.1 January-April 2024
See all articles in 26.3 September-December 2023
See all articles in 26.2 May-August 2023
See all articles in 26.1 January-April 2023
Daniel I. Hembree Department of Geological Sciences
Department of Geological Sciences
Ohio University
316 Clippinger Laboratories
Athens, Ohio, 45701
USA
Daniel Hembree is an Assistant Professor in the Department of Geological Sciences at Ohio University. His research interests primarily lie with animal-substrate interactions in ancient and modern continental environments. These interactions are preserved in the fossil record as trace fossils. Trace fossils provide an in situ record of ancient biodiversity, ecology, and environment. The study of trace fossils, therefore, provides vital information for accurate paleoenvironmental reconstructions. This involves not only the study of paleosols and continental trace fossils throughout geologic time, but also the experimental study of burrowing behaviors of extant terrestrial annelids, arthropods, amphibians, and reptiles. Current research projects involve the study of the influence of climate changes on ancient soils and soil ecosystems including those of the Pennsylvanian and Permian of southeast Ohio, Permian of eastern Kansas, and the Eocene to Miocene of Colorado and Wyoming.
Lee M. Johnson
Department of Geological Sciences
Ohio University
316 Clippinger Laboratories
Athens, Ohio, 45701
USA
Lee Johnson completed his Bachelors of Science degree in the Department of Geological Sciences at Ohio University in 2011. He worked in Dr. Hembree's Continental Ichnology Research Laboratory for two years and assisted in a number of experimental projects with burrowing animals.
Robert W. Tenwalde Department of Geological Sciences
Department of Geological Sciences
Ohio University
316 Clippinger Laboratories
Athens, Ohio, 45701
USA
Robert Tenwalde completed his Bachelors of Science degree in the Department of Geological Sciences at Ohio University in 2011. He worked in Dr. Hembree's Continental Ichnology Research Laboratory for two years and assisted in a number of experimental projects with burrowing animals.
FIGURE 1. 1) General scorpion morphology. ct: cephalothorax; ms: mesosoma; mt: metasoma; pd: pedipalp; ch: chelicerae; te: telson; w1-4: walking legs. 2) Specimen of Hadrurus arizonensis used in this study. 3) Known H. arizonensis distribution in the United States.

FIGURE 2. Quantitative burrow description models. 1) Burrows were described by their maximum depth (D), tunnel and shaft width (w), height (h), and circumference (c), total length (L), and branching angle (BA). Burrows were divided into segments (s), surface openings (e), and chambers (h). 2) Complexity is a measure of the number of segments, chambers, and surface openings (e) within a single burrow system. 3) Tortuosity is a measure of the average sinuosity of all of the segments of a burrow system. The tortuosity of a single segment is found by dividing the total length (u) by the straight line distance (v).
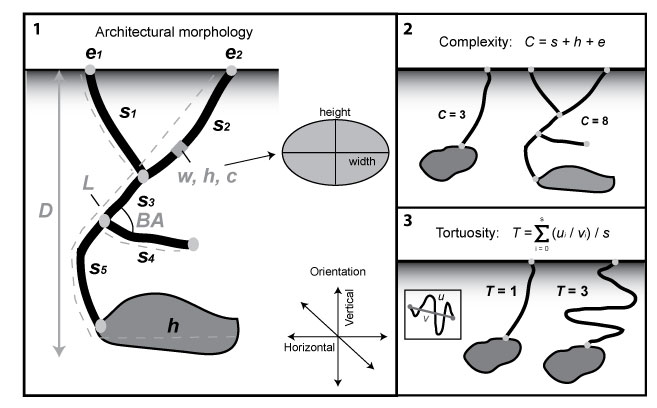
FIGURE 3. Burrowing methods used by H. arizonensis. 1) Excavation by kicking sediment behind the body using the first two walking legs. The pedipalps are held up and are not involved in burrowing. 2) Excavation by kicking sediment using the first three walking legs. 3) Excavation by gathering sediment with the first three walking legs and dragging it away from the site of active excavation.

FIGURE 4. Burrowing methods used by H. arizonensis. 1) Excavation by kicking with the first two walking legs, oblique view. 2) Excavation by kicking with the first three walking legs, overhead view. 3) Excavation by dragging with the walking legs, side view.
FIGURE 5. Surface morphology produced by H. arizonensis burrowing. 1) Experimental tank containing 75% sand and 25% clay with a flat, even surface. 2) Experimental tank containing 25% sand and 75% clay with a flat, even surface. 3) Ripples and depressions produced in low density sediment after seven days. 4) Ripples and depressions produced in low density sediment after 14 days; surface relief is approximately 15 cm. 5) Overhead views of a burrow opening beneath a rock (A) and within an excavated pit (B). 6) Oblique view of a burrow opening within an excavated pit.

FIGURE 6. Subvertical ramps. 1) Left oblique view of a typical subvertical ramp (DHS2). 2) Right oblique view of a typical subvertical ramp (DHS17). 3) Overhead view of a subvertical ramp with a sharp curve (DHS10).

FIGURE 7. Subvertical ramps. 1) Left oblique view of an elongate, curved subvertical ramp (DHS17). 2) Side view of a subvertical ramp with a short branch (DHS16). 3) Overhead view of a subvertical ramp with a short branch and a chamber near the burrow entrance (DHS16).
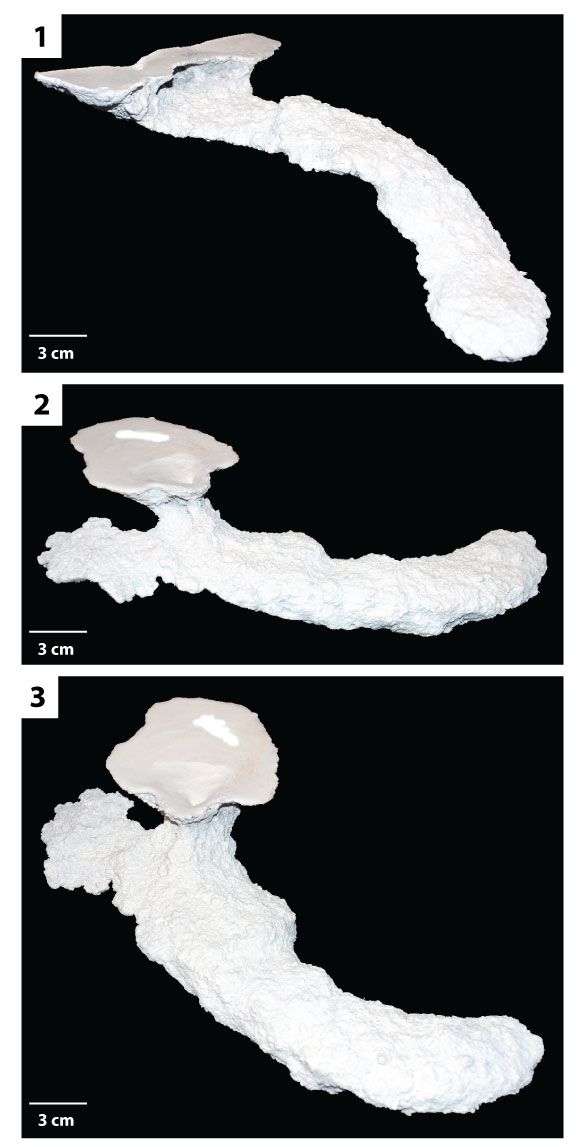
FIGURE 8. U-shaped burrows. 1) Side view of a typical U-shaped burrow (DHS4). 2) Side view of a U-shaped burrow (DHS9) showing the large difference in angles between each shaft (40° and 90°) typical of H. arizonensis burrows of this type.
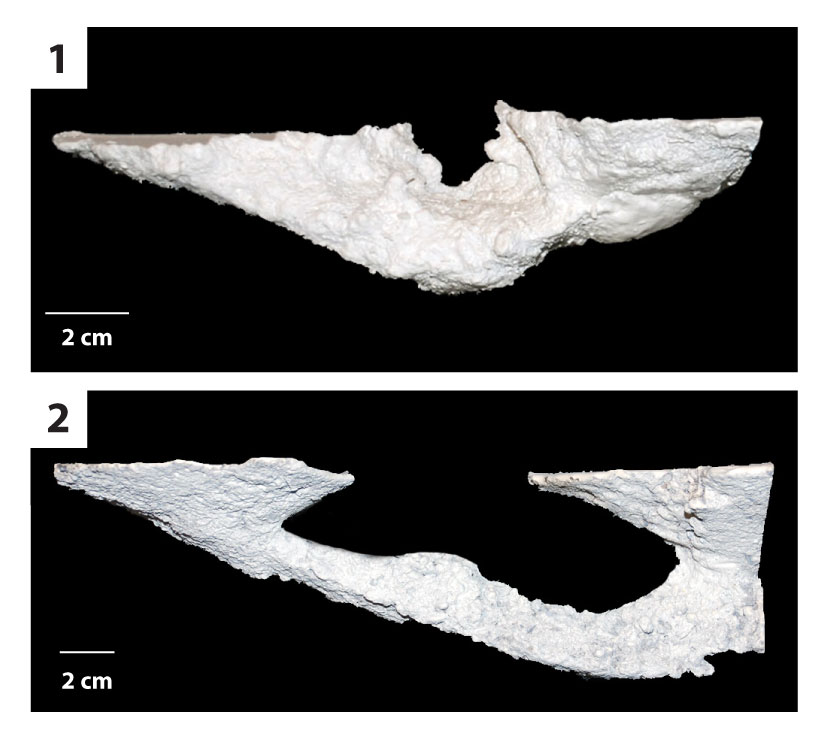
FIGURE 9. Helical burrow casts. 1) Oblique view of a short, highly tortuous helical burrow (DHS3). The terminal chamber is directly below the burrow opening. 2) Side view of an elongate (76 cm) helical burrow with a low tortuosity (DHS15). 3) Side view of a highly tortuous helical burrow (DHS3). 4) Oblique view of an elongate helical burrow with a low tortuosity (DHS15).
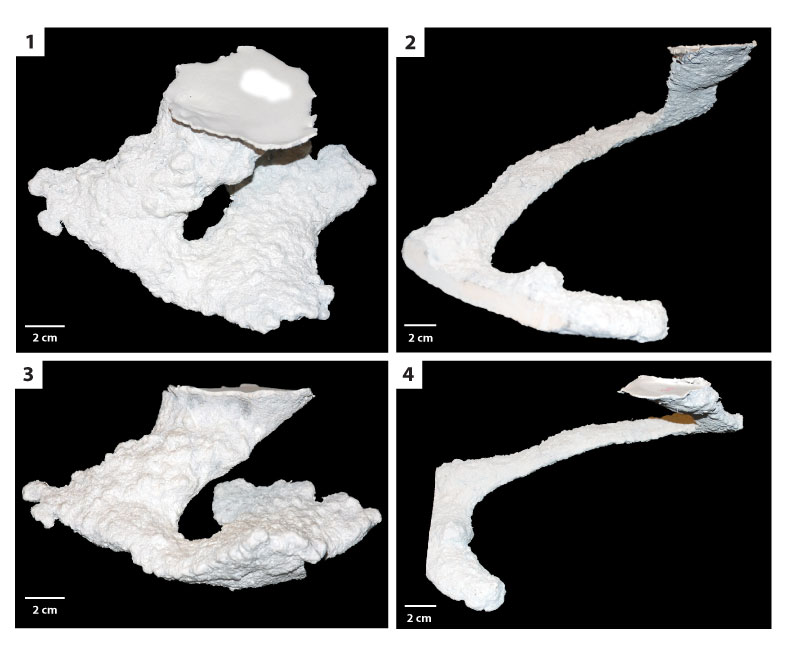
FIGURE 10. Mazeworks. 1) Overhead view of a mazework consisting of a U-shaped burrow and two subvertical ramps (DHS8). 2, 3) Side views of mazework consisting of a U-shaped burrow and two subvertical ramps (DHS8). 4) Underside of a mazework (DHS4); a short tunnel (A) connects a subvertical ramp (B) and a U-shaped burrow (C).

FIGURE 11. Mazeworks. 1) Side view of a mazework consisting of three U-shaped burrows (DHS5). 2) Side view of a mazework consisting of two U-shaped burrows and a subvertical ramp (DHS4).
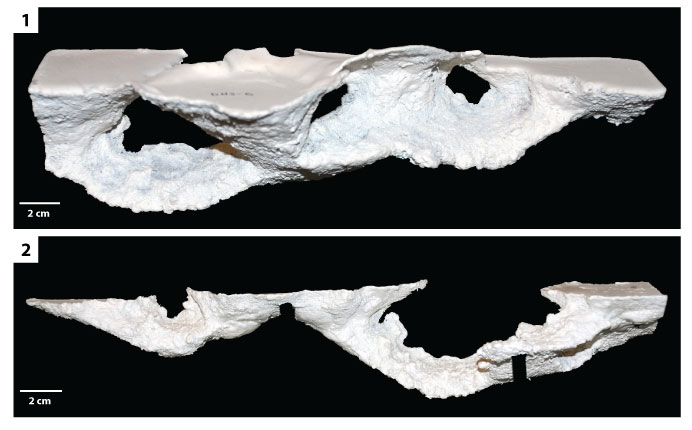
FIGURE 12. Biogenic cross lamination. 1) Production of laminae (at arrow) by a specimen of H.arizonensis during excavation by the kick method. 2) Ripple forms produced by H. arizonensis (at arrows) with cross lamination visible in cross section. 3) Two stacked cross-laminated ripple forms. 4, 5) Stacked ripples show in (3) after two days; the upper ripple has grown and migrated to the left. 6) Steeply dipping set of biogenic cross laminae.

FIGURE 13. Burrows produce by other arachnids. 1) Oblique view of a large helical burrow produced by Pandinus imperator. 2) Oblique view of a mazework produced by Mastigoproctus giganteus.
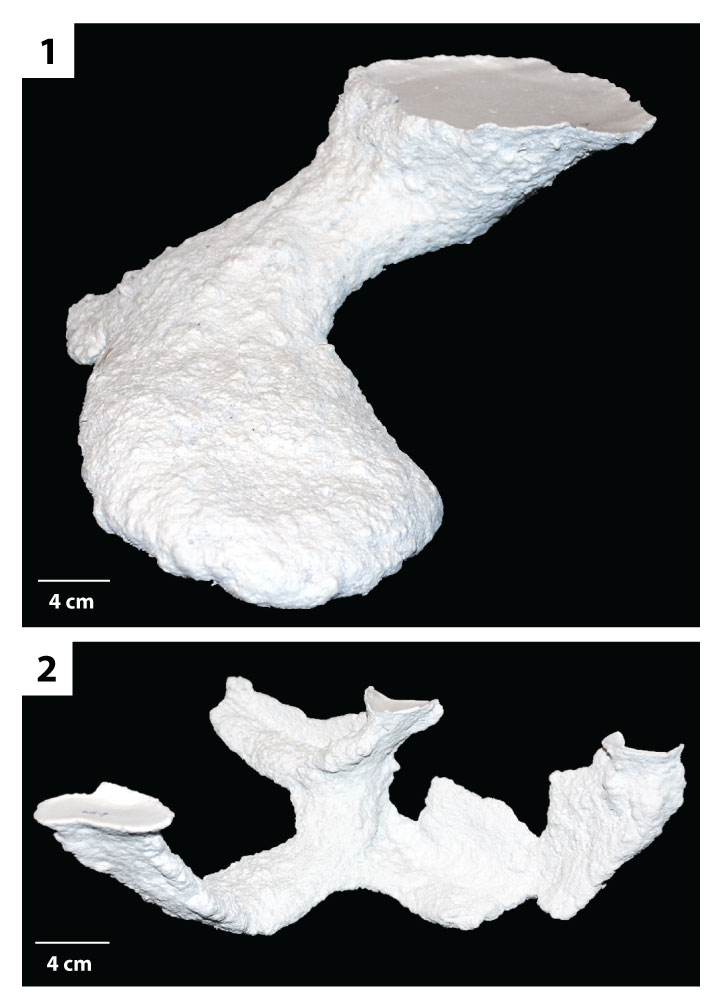
TABLE 1. Experimental parameters and resulting burrow morphologies. SR: subvertical ramp, UB: U-shaped burrow, HB: helical burrow, MW: mazework, XL: cross lamination. Sediment density values are in kgf/cm2. Sediment depths are in cm. Sediment moisture values are in percent total volume.
|
Tank Size |
Sediment Composition |
Sediment Depth |
Sediment Density |
Sediment Moisture |
Burrow Architecture |
|
10 |
75-25% clay-sand |
20 |
2.1-3.2 |
0 |
None |
|
10 |
50-50% clay-sand |
20 |
1.1-1.4 |
0 |
None |
|
10 |
25-75% clay-sand |
20 |
0.4-0.7 |
0 |
XL |
|
|
|
|
|
|
|
|
10 |
75-25% clay-sand |
20 |
2.1-3.2 |
20 |
SR, UB, HB, MW |
|
10 |
50-50% clay-sand |
20 |
1.1-1.4 |
20 |
SR, UB |
|
10 |
25-75% clay-sand |
20 |
0.4-0.7 |
20 |
SR |
|
|
|
|
|
|
|
|
10 |
75-25% clay-sand |
20 |
2.1-3.2 |
50 |
SR, UB, HB, MW |
|
10 |
50-50% clay-sand |
20 |
1.1-1.4 |
50 |
SR, UB |
|
10 |
25-75% clay-sand |
20 |
0.4-0.7 |
50 |
SR |
|
|
|
|
|
|
|
|
20 |
75-25% clay-sand |
35 |
2.1-3.2 |
0 |
None |
|
20 |
50-50% clay-sand |
35 |
1.1-1.4 |
0 |
None |
|
20 |
25-75% clay-sand |
35 |
0.4-0.7 |
0 |
XL |
|
|
|
|
|
|
|
|
20 |
75-25% clay-sand |
35 |
2.1-3.2 |
20 |
SR, UB, HB, MW |
|
20 |
50-50% clay-sand |
35 |
1.1-1.4 |
20 |
SR, UB |
|
20 |
25-75% clay-sand |
35 |
0.4-0.7 |
20 |
SR |
|
|
|
|
|
|
|
|
20 |
75-25% clay-sand |
35 |
2.1-3.2 |
50 |
SR, UB, HB, MW |
|
20 |
50-50% clay-sand |
35 |
1.1-1.4 |
50 |
SR, UB |
|
20 |
25-75% clay-sand |
35 |
0.4-0.7 |
50 |
SR |
|
|
|
|
|
|
|
|
56 |
75-25% clay-sand |
55 |
2.1-3.2 |
0 |
None |
|
56 |
50-50% clay-sand |
55 |
1.1-1.4 |
0 |
None |
|
56 |
25-75% clay-sand |
55 |
0.4-0.7 |
0 |
XL |
|
|
|
|
|
|
|
|
56 |
75-25% clay-sand |
55 |
2.1-3.2 |
20 |
SR, UB, HB, MW |
|
56 |
50-50% clay-sand |
55 |
1.1-1.4 |
20 |
SR, UB |
|
56 |
25-75% clay-sand |
55 |
0.4-0.7 |
20 |
SR |
|
|
|
|
|
|
|
|
56 |
75-25% clay-sand |
55 |
2.1-3.2 |
50 |
SR, UB, HB, MW |
|
56 |
50-50% clay-sand |
55 |
1.1-1.4 |
50 |
SR, UB |
|
56 |
25-75% clay-sand |
55 |
0.4-0.7 |
50 |
SR |
TABLE 2. Measurements of representative three dimensional casts of burrows produced by Hadrurus arizonensis (DHS), Pandinus imperator (ES), and Mastigoproctus giganteus(WS). All measurements are in cm.
|
|
DHS1 |
DHS2 |
DHS3 |
DHS4 |
DHS5 |
DHS6 |
DHS7 |
DHS8 |
DHS9 |
|
Architecture |
SR |
SR |
HB |
MW |
MW |
SR |
HB |
MW |
UB |
|
Surface openings |
1 |
1 |
1 |
5 |
4 |
1 |
1 |
3 |
2 |
|
Maximum depth |
3.9 |
2.8 |
7.2 |
7.4 |
6.7 |
4.3 |
4.6 |
8.4 |
8.0 |
|
Total length |
11 |
9 |
27 |
57 |
46 |
7 |
13 |
31 |
30 |
|
Maximum width |
4.3 |
6.0 |
7.7 |
4.6 |
6.4 |
5.9 |
3.3 |
7.2 |
4.2 |
|
Minimum width |
3.2 |
3.0 |
2.8 |
1.9 |
2.6 |
2.9 |
2.7 |
2.9 |
3.0 |
|
Average width |
3.9 |
4.8 |
4.5 |
3.6 |
4.2 |
4.2 |
3.1 |
4.7 |
3.6 |
|
Maximum height |
2.8 |
1.9 |
3.2 |
2.3 |
3.1 |
1.6 |
1.4 |
2.6 |
2.5 |
|
Minimum height |
1.0 |
1.1 |
0.8 |
0.8 |
1.4 |
1.0 |
0.6 |
1.4 |
1.5 |
|
Average height |
1.9 |
1.6 |
2.0 |
1.8 |
1.9 |
1.4 |
1.0 |
1.9 |
2.1 |
|
Average W/H ratio |
2.1 |
3.0 |
2.3 |
2.0 |
2.2 |
3.0 |
3.1 |
2.5 |
1.7 |
|
Maximum circumference |
12.1 |
14.0 |
18.0 |
14.5 |
17.0 |
11.5 |
10.5 |
16.0 |
11.5 |
|
Minimum circumference |
10.0 |
11.2 |
6.5 |
9.5 |
10.5 |
6.0 |
8.5 |
9.5 |
10.5 |
|
Average circumference |
11.2 |
12.7 |
14.5 |
13.1 |
12.2 |
9.4 |
9.0 |
12.3 |
11.3 |
|
Maximum slope |
40° |
10° |
30° |
50° |
90° |
30° |
40° |
50° |
90° |
|
Minimum slope |
0° |
0° |
0° |
0° |
0° |
0° |
0° |
0° |
0° |
|
Average slope |
20° |
5° |
15° |
27° |
37° |
15° |
20° |
30° |
45° |
|
Branching angles |
NA |
NA |
NA |
75 |
75 |
NA |
NA |
70 |
NA |
|
Complexity |
3 |
3 |
3 |
9 |
9 |
3 |
3 |
7 |
4 |
|
Tortuosity |
1.1 |
1.1 |
9.0 |
1.4 |
1.8 |
1.1 |
3.3 |
1.1 |
1.3 |
|
Sediment Density |
L |
L |
H |
H |
H |
L |
H |
H |
M |
TABLE 2 (continued).
|
|
DHS10 |
DHS11 |
DHS12 |
DHS13 |
DHS14 |
DHS15 |
DHS16 |
DHS17 |
ES1 |
WS1 |
|
Architecture |
SR |
SR |
SR |
SR |
SR |
HB |
SR |
SR |
HB |
MW |
|
Surface openings |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
4 |
|
Maximum depth |
3.0 |
4.2 |
3.4 |
4.0 |
4.2 |
16.0 |
5.3 |
6.8 |
12.5 |
12.4 |
|
Total length |
16 |
11 |
19 |
19 |
23 |
76 |
37 |
26 |
36 |
66 |
|
Maximum width |
5.2 |
4.4 |
6.5 |
4.9 |
6.7 |
5.3 |
6.2 |
5.4 |
12.0 |
6.2 |
|
Minimum width |
3.8 |
4.1 |
4.2 |
3.1 |
4.1 |
3.7 |
3.1 |
4.0 |
7.9 |
3.7 |
|
Average width |
4.4 |
4.3 |
5.2 |
4.1 |
4.9 |
4.5 |
4.8 |
4.9 |
9.7 |
4.6 |
|
Maximum height |
2.1 |
2.0 |
3.3 |
2.6 |
2.3 |
3.3 |
2.8 |
2.2 |
5.1 |
2.0 |
|
Minimum height |
1.3 |
0.9 |
1.9 |
1.8 |
1.4 |
1.6 |
1.7 |
1.5 |
2.8 |
1.7 |
|
Average height |
1.8 |
1.6 |
2.2 |
2.2 |
1.7 |
2.3 |
2.4 |
1.8 |
3.9 |
1.9 |
|
Average W/H ratio |
2.4 |
2.7 |
2.4 |
1.9 |
2.9 |
2.0 |
2.0 |
2.7 |
2.5 |
2.4 |
|
Maximum circumference |
13.6 |
11.2 |
16.5 |
13.0 |
15.5 |
14.0 |
15.0 |
13.0 |
27.0 |
14.5 |
|
Minimum circumference |
9.7 |
6.8 |
11.5 |
9.9 |
10.5 |
10.0 |
11.0 |
11.5 |
20.5 |
9.0 |
|
Average circumference |
11.9 |
10.0 |
13.9 |
11.5 |
12.5 |
17.3 |
12.9 |
12.6 |
23.8 |
11.3 |
|
Maximum slope |
10° |
40° |
10° |
20° |
20° |
30° |
20° |
20° |
40° |
80° |
|
Minimum slope |
0° |
0° |
0° |
0° |
0° |
0° |
0° |
0° |
0° |
0° |
|
Average slope |
5° |
20° |
5° |
10° |
10° |
13° |
10° |
8° |
20° |
45° |
|
Branching angles |
NA |
NA |
NA |
NA |
NA |
NA |
65 |
NA |
NA |
80 |
|
Complexity |
3 |
3 |
3 |
3 |
3 |
3 |
4 |
3 |
3 |
10 |
|
Tortuosity |
1.3 |
1.5 |
1.1 |
1.2 |
1.4 |
3.3 |
1.4 |
1.2 |
1.7 |
1.4 |
|
Sediment Density |
M |
L |
M |
M |
M |
H |
H |
M |
NA |
NA |
TABLE 3. Bray Curtis Similarity measure results. 1. Comparison matrix of all burrow casts. 2. Comparison matrix of subvertical ramps. 3. Comparison matrix of mazeworks. 4. Comparison matrix of helical burrows. DHS: Hadrurus arizonensis, ES: Pandinus imperator, WS: Mastigoproctus giganteus, SR: subvertical ramp, UB: U-shaped burrow, HB: helical burrow, MW: mazework (PDF format).
TABLE 4. Spearman's rank order analysis results comparing eight quantitative burrow properties (X) with sediment density (Y). Sediment density is ranked from 1 (0.4–0.7 kgf/cm2), 2 (1.1–1.4 kgf/cm2), and 3 (2.1–3.2 kgf/cm2).
|
1. Complexity vs. Sediment Density |
|
2. Tortuosity vs. Sediment Density |
||||||||
|
Raw Score |
|
|
Rank |
|
|
Raw Score |
|
|
Rank |
|
|
X |
Y |
|
X |
Y |
|
X |
Y |
|
X |
Y |
|
3 |
3 |
|
6.5 |
14 |
|
9 |
3 |
|
17 |
14 |
|
9 |
3 |
|
16.5 |
14 |
|
1.4 |
3 |
|
11 |
14 |
|
9 |
3 |
|
16.5 |
14 |
|
1.8 |
3 |
|
13.5 |
14 |
|
3 |
3 |
|
6.5 |
14 |
|
3.3 |
3 |
|
15.5 |
14 |
|
7 |
3 |
|
15 |
14 |
|
1.1 |
3 |
|
3 |
14 |
|
3 |
3 |
|
6.5 |
14 |
|
3.3 |
3 |
|
15.5 |
14 |
|
4 |
3 |
|
13.5 |
14 |
|
1.4 |
3 |
|
11 |
14 |
|
4 |
2 |
|
13.5 |
7.5 |
|
1.3 |
2 |
|
8.5 |
7.5 |
|
3 |
2 |
|
6.5 |
7.5 |
|
1.3 |
2 |
|
8.5 |
7.5 |
|
3 |
2 |
|
6.5 |
7.5 |
|
1.1 |
2 |
|
3 |
7.5 |
|
3 |
2 |
|
6.5 |
7.5 |
|
1.2 |
2 |
|
6.5 |
7.5 |
|
3 |
2 |
|
6.5 |
7.5 |
|
1.4 |
2 |
|
11 |
7.5 |
|
3 |
2 |
|
6.5 |
7.5 |
|
1.2 |
2 |
|
6.5 |
7.5 |
|
3 |
1 |
|
6.5 |
2.5 |
|
1.1 |
1 |
|
3 |
2.5 |
|
3 |
1 |
|
6.5 |
2.5 |
|
1.1 |
1 |
|
3 |
2.5 |
|
3 |
1 |
|
6.5 |
2.5 |
|
1.1 |
1 |
|
3 |
2.5 |
|
3 |
1 |
|
6.5 |
2.5 |
|
1.5 |
1 |
|
13.5 |
2.5 |
|
|
Rs=0.54 |
|
|
|
|
|
Rs=0.58 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3. Circumference vs. Sediment Density |
|
4. Total Length vs. Sediment Density |
||||||||
|
Raw Score |
|
|
Rank |
|
|
Raw Score |
|
|
Rank |
|
|
X |
Y |
|
X |
Y |
|
X |
Y |
|
X |
Y |
|
15 |
3 |
|
16 |
14 |
|
27 |
3 |
|
11 |
14 |
|
13.1 |
3 |
|
14 |
14 |
|
57 |
3 |
|
16 |
14 |
|
12.2 |
3 |
|
8 |
14 |
|
46 |
3 |
|
15 |
14 |
|
9 |
3 |
|
1 |
14 |
|
13 |
3 |
|
5 |
14 |
|
12.3 |
3 |
|
9 |
14 |
|
31 |
3 |
|
13 |
14 |
|
17.3 |
3 |
|
17 |
14 |
|
76 |
3 |
|
17 |
14 |
|
12.9 |
3 |
|
13 |
14 |
|
37 |
3 |
|
14 |
14 |
|
11 |
2 |
|
4 |
7.5 |
|
30 |
2 |
|
12 |
7.5 |
|
11.9 |
2 |
|
7 |
7.5 |
|
16 |
2 |
|
6 |
7.5 |
|
13.9 |
2 |
|
15 |
7.5 |
|
19 |
2 |
|
7.5 |
7.5 |
|
11.5 |
2 |
|
6 |
7.5 |
|
19 |
2 |
|
7.5 |
7.5 |
|
12.5 |
2 |
|
10 |
7.5 |
|
23 |
2 |
|
9 |
7.5 |
|
12.6 |
2 |
|
11 |
7.5 |
|
26 |
2 |
|
10 |
7.5 |
|
11.2 |
1 |
|
5 |
2.5 |
|
11 |
1 |
|
3.5 |
2.5 |
|
12.7 |
1 |
|
12 |
2.5 |
|
9 |
1 |
|
2 |
2.5 |
|
9.4 |
1 |
|
2 |
2.5 |
|
7 |
1 |
|
1 |
2.5 |
|
10 |
1 |
|
3 |
2.5 |
|
11 |
1 |
|
3.5 |
2.5 |
|
|
Rs=0.44 |
|
|
|
|
|
Rs=0.82 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
5. Maximum Width vs. Sediment Density |
|
6. Maximum Height vs. Sediment Density |
||||||||
|
Raw Score |
|
|
Rank |
|
|
Raw Score |
|
|
Rank |
|
|
X |
Y |
|
X |
Y |
|
X |
Y |
|
X |
Y |
|
7.7 |
3 |
|
17 |
14 |
|
3.2 |
3 |
|
15 |
14 |
|
4.6 |
3 |
|
5 |
14 |
|
2.3 |
3 |
|
7.5 |
14 |
|
6.4 |
3 |
|
13 |
14 |
|
3.1 |
3 |
|
14 |
14 |
|
3.3 |
3 |
|
1 |
14 |
|
1.4 |
3 |
|
1 |
14 |
|
7.2 |
3 |
|
16 |
14 |
|
2.6 |
3 |
|
10.5 |
14 |
|
5.3 |
3 |
|
8 |
14 |
|
3.3 |
3 |
|
16.5 |
14 |
|
6.2 |
3 |
|
12 |
14 |
|
2.8 |
3 |
|
12.5 |
14 |
|
4.2 |
2 |
|
2 |
7.5 |
|
2.5 |
2 |
|
9 |
7.5 |
|
5.2 |
2 |
|
7 |
7.5 |
|
2.1 |
2 |
|
5 |
7.5 |
|
6.5 |
2 |
|
14 |
7.5 |
|
3.3 |
2 |
|
16.5 |
7.5 |
|
4.9 |
2 |
|
6 |
7.5 |
|
2.6 |
2 |
|
10.5 |
7.5 |
|
6.7 |
2 |
|
15 |
7.5 |
|
2.3 |
2 |
|
7.5 |
7.5 |
|
5.4 |
2 |
|
9 |
7.5 |
|
2.2 |
2 |
|
6 |
7.5 |
|
4.3 |
1 |
|
3 |
2.5 |
|
2.8 |
1 |
|
12.5 |
2.5 |
|
6 |
1 |
|
11 |
2.5 |
|
1.9 |
1 |
|
3 |
2.5 |
|
5.9 |
1 |
|
10 |
2.5 |
|
1.6 |
1 |
|
2 |
2.5 |
|
4.4 |
1 |
|
4 |
2.5 |
|
2 |
1 |
|
4 |
2.5 |
|
|
Rs=0.26 |
|
|
|
|
|
Rs=0.43 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7. Maximum Depth vs. Sediment Density |
|
8. Average Slope vs. Sediment Density |
||||||||
|
Raw Score |
|
|
Rank |
|
|
Raw Score |
|
|
Rank |
|
|
X |
Y |
|
X |
Y |
|
X |
Y |
|
X |
Y |
|
7.2 |
3 |
|
13 |
14 |
|
15 |
3 |
|
9.5 |
14 |
|
7.4 |
3 |
|
14 |
14 |
|
27 |
3 |
|
14 |
14 |
|
6.7 |
3 |
|
11 |
14 |
|
37 |
3 |
|
16 |
14 |
|
4.6 |
3 |
|
9 |
14 |
|
20 |
3 |
|
12 |
14 |
|
8.4 |
3 |
|
16 |
14 |
|
30 |
3 |
|
15 |
14 |
|
16 |
3 |
|
17 |
14 |
|
13 |
3 |
|
8 |
14 |
|
5.3 |
3 |
|
10 |
14 |
|
10 |
3 |
|
6 |
14 |
|
8 |
2 |
|
15 |
7.5 |
|
45 |
2 |
|
17 |
7.5 |
|
3 |
2 |
|
2 |
7.5 |
|
5 |
2 |
|
2 |
7.5 |
|
3.4 |
2 |
|
3 |
7.5 |
|
5 |
2 |
|
2 |
7.5 |
|
4 |
2 |
|
5 |
7.5 |
|
10 |
2 |
|
6 |
7.5 |
|
4.2 |
2 |
|
6.5 |
7.5 |
|
10 |
2 |
|
6 |
7.5 |
|
6.8 |
2 |
|
12 |
7.5 |
|
8 |
2 |
|
4 |
7.5 |
|
3.9 |
1 |
|
4 |
2.5 |
|
20 |
1 |
|
12 |
2.5 |
|
2.8 |
1 |
|
1 |
2.5 |
|
5 |
1 |
|
2 |
2.5 |
|
4.3 |
1 |
|
8 |
2.5 |
|
15 |
1 |
|
9.5 |
2.5 |
|
4.2 |
1 |
|
6.5 |
2.5 |
|
20 |
1 |
|
12 |
2.5 |
|
|
Rs=0.68 |
|
|
|
|
|
Rs=0.31 |
|
|
|
Neoichnology of the desert scorpion Hadrurus arizonensis: burrows to biogenic cross lamination
Plain Language Abstract
Burrowing by terrestrial animals is common in arid and semi-arid environments. Scorpions have comprised a significant portion of the diversity of predatory arthropods in these environments for the last 320 million years. Most modern scorpions are active burrowers and therefore it is likely that ancient scorpions have left a substantial, if rarely recognized, record of fossil burrows. A major problem with recognizing fossil scorpion burrows is the limited understanding of burrows produced by modern scorpions. This project involved the study of the burrowing behaviors and burrow morphologies of the North American desert scorpion Hadrurus arizonensis. Individual animals were placed into sediment-filled terrariums for two- to three-week periods after which burrows were cast, excavated, and described. Descriptions of the burrows included their overall form (straight, sinuous, helical, or branching), dimensions (depth, length, width, height, and circumference), cross-sectional shape, orientation in the sediment, and the type and amount of branching. Experiments were run with variations in sediment composition, density, and moisture to evaluate the animal’s behavioral response to different environmental conditions. Specimens of H. arizonensis were found to burrow by scratching and kicking loose sediment from the subsurface with the first two to three pairs of walking legs. The burrows produced consisted of subvertical ramps, U-shaped burrows, helical burrows, and interconnected branching networks. In the process of excavating the burrows, the desert scorpions also produced an irregular surface topography as well as structures in dry, sandy sediment that resembled crossbedding. Increasing clay content and sediment density increased the complexity of burrow architectures produced. Reducing these variables limited the complexity of the burrows, reduced their likelihood of preservation, and increased the abundance of biogenic cross-lamination. The results of these and similar experimental studies of burrowing animals can be applied to the interpretation of fossil burrow assemblages in order to better interpret the types of organisms present within ancient soil ecosystems.
Resumen en Español
Neoicnología del escorpión del desierto Hadrurus arizonensis: madrigueras y laminación cruzada biogénica
En los ambientes continentales áridos y semiáridos es frecuente la bioturbación debida a animales terrestres. Los escorpiones han constituido una parte significativa de la diversidad de los artrópodos depredadores en esos ambientes desde el Paleozoico superior hasta la actualidad. Muchos escorpiones son excavadores activos y es probable que su registro icnofósil, aunque raramente puesto de manifiesto, sea considerable. En el presente trabajo se estudian los comportamientos de excavación y morfología de las trazas del escorpión Hadrurus arizonensis (Scorpiones: Caraboctonidae). Se colocaron varios individuos de la especie en sendos terrarios llenos de sedimento durante períodos de dos a tres semanas, tras los cuales se sacaron moldes de las madrigueras excavadas, se aislaron y se describieron. Las descripciones de las estructuras subterráneas incluyen aspectos como arquitectura, dimensiones, bioglifos, complejidad y tortuosidad. También se llevaron a cabo pruebas adicionales cambiando la composición del sedimento, su densidad y su humedad para evaluar la respuesta del animal a diferentes condiciones ambientales. Los ejemplares de H. arizonensis excavan el sedimento con los dos o tres primeros pares de patas marchadoras. Las estructuras biogénicas subterráneas incluyen galerías inclinadas, excavaciones en forma de U y helicoidales y estructuras laberínticas. En el proceso de excavación de las madrigueras, los escorpiones del desierto producen una topografía de superficie de tipo hummocky, así como estructuras en sedimentos secos y arenosos que semejan laminaciones y laminaciones cruzadas de tipo ripple. Un incremento en el contenido en arcilla y en la densidad del sedimento da lugar a una complejidad mayor en la arquitectura de las excavaciones. Por el contrario, una disminución en esas variables disminuye la complejidad de las madrigueras y su probabilidad de preservación, al tiempo que aumenta la abundancia de la laminación cruzada biogénica. Los datos obtenidos en este estudio y en otros similares pueden ser aplicados al análisis de las asociaciones de icnofósiles terrestres con objeto de mejorar las interpretaciones paleoecológicas de los antiguos ecosistemas del suelo.
Palabras clave: icnofósiles; trazas fósiles; bioturbación; continental; artrópodo; paleoecología
Traducción: Miguel Company
Résumé en Français
Néoichnologie du scorpion du désert Hadrurus arizonensis: des terriers à une stratification biogénique croisée
La bioturbation par des animaux terrestres est commune dans les environnements continentaux aride et semi-aride. Les scorpions constituent un part significative de la diversité des prédateurs arthropodes dans ces environnements depuis Paléozoïque supérieur jusqu'à aujourd’hui. De nombreux scorpions sont des fouisseurs actifs et ont probablement un registre ichnofossile important, bien que rarement reconnu. Ce projet consiste en une étude des comportements de fouissage et de morphologie des traces du scorpion Hadrurus arizonensis (Scorpiones: Caraboctonidae). Des individus on été placés dans des terrariums remplis de sédiment pour des périodes allant de 2 à 3 semaines, après quoi les terriers ont été moulés, extraits et décrits. La description de sub-surface des structures comprend l’architecture, la taille, les bioglyphes, la complexité et la tortuosité. De plus les expérimentations ont été réalisées avec des sédiments de composition, densité et humidité différentes, dans le but d’évaluer la réponse du comportement de ces animaux aux changements des conditions environnementales. Les spécimens de H. arizonensis ont creusé leurs terriers en grattant et évacuant le sédiment libre de la sub-surface avec leurs deux ou trois premières paires de pattes. Les structures biogéniques de sub-surface produites correspondent à des rampes sub-verticales, des terriers en forme de U, des terriers hélicoïdaux, et des labyrinthes. Associé au creusement des terriers, les scorpions du désert ont produit des monticules de surface, ainsi que des structures ressemblant à des stratifications et des rides à stratification croisée dans les sédiments sec et sableux. L’architecture des terriers formés s’est complexifiée avec augmentation de la densité et de la portion d’argile dans les sédiments. La réduction de ces variables a entraîné une limitation de la complexité des terriers, une diminution des chances de préservation des terriers, et une augmentation de l’abondances des stratifications croisées biogéniques. Les données collectées à partir de ces expérimentations et à partir d’études similaires peuvent être appliquées aux assemblages d’ichnofossiles terrestres dans le but de mieux interpréter la paléoécologie des anciens écosystèmes du sol.
Mots clés : ichnofossiles ; traces fossiles ; bioturbations ; continental ; arthropode ; paléoécologie
Translator: Olivier Maridet
Deutsche Zusammenfassung
Neoichnologie des Wüstenskorpions Hadrurus arizonensis: Bauten bis zu biogener Kreuzschichtung
Bioturbation terrestrischer Tiere kommt häufig in ariden und semi-ariden kontinentalen Milieus vor. Skorpione machten in einer solchen Umgebung einen erheblichen Teil der Diversität unter den räuberischen Arthropoden vom späten Paläozoikum bis heute aus. Viele Skorpione graben aktiv und haben wahrscheinlich einen maßgeblichen, wenn auch selten bemerkten Ichnofossilbeleg. In dieser Studie geht es um das Grabverhalten und die Spurenmorphologie des Skorpions Hadrurus arizonensis (Scorpiones: Caraboctonidae). Es wurden einzelne Tiere für zwei bis drei Wochen in sedimentbefüllte Terrarien gesetzt. Danach wurden die Bauten abgegossen, ausgegraben und beschrieben. Die Beschreibung der Untergrundstrukturen beinhaltet Architektur, Dimensionen, Bioglyphen, Komplexität und Tortuosität. Es wurden zusätzliche Experimente mit unterschiedlichem Sediment, Zusammensetzung, Dichte und Feuchtigkeit durchgeführt um die Reaktion der Tiere auf unterschiedliche Umweltbedingungen zu testen. Tiere der Art H. arizonensis gruben indem sie mit ihren ersten beiden bis ersten drei Beinpaaren loses Sediment vom Untergrund wegkratzen oder kickten. Die auf diese Weise entstandenen Bauten setzten sich aus subvertikalen Rampen, U-förmigen, spiralförmigen und labyrinth-artigen Bauten zusammen. Beim Anfertigen der Bauten produzierten die Wüstenskorpione außerdem eine hügelige Topographie zusammen mit Strukturen, die im trockenen, sandigen Sediment Lamination oder Rippelkreuzschichtung ähnlich sehen. Eine Zunahme der Tonkomponente und Sedimentdichte erhöhten die Komplexität der Bautenarchitektur. Eine Reduzierung dieser Variablen beschränkte die Komplexität der Bauten, verringerte die Erhaltungswahrscheinlichkeit und erhöhte die Häufigkeit der biogenen Kreuzschichtung. Daten aus dieser und ähnlichen experimentellen Untersuchungen können auf terrestrische Ichnofossil-Ansammlungen angewendet werden um die Paläoökologie ehemaliger Boden-Ökosysteme besser deuten zu können.
SCHLÜSSELWÖRTER: Ichnofossilien; Spurenfossilien; Bioturbation; kontinental; Arthropode; Paläoökologie
Translators: Eva Gebauer and Anke Konietzka
Arabic

Translator: Ashraf M.T. Elewa
Polski Abstrakt
Neoichnologia skorpiona pustynnego Hadrurus arizonensis: nory biogenicznym powodem laminacji skośnej
Bioturbacja przez zwierzęta lądowe jest częsta w suchych i półsuchych środowiskach kontynentalnych. Skorpiony stanowią spory wkład w różnorodność drapieżnych stawonogów w tych środowiskach już od późnego paleozoiku. Wiele skorpionów to aktywni kopacze nor i zapewne mają duży, gdy z rzadka rozpoznawany, udział w zapisie ichnologicznym. W naszym badaniu zajęliśmy się etologią kopania nor i morfologią śladów skorpiona Hadrurus arizonensis (Scorpiones: Caraboctonidae). Pojedyncze zwierzęta umieszczaliśmy w wyłożonych osadem terrariach na okres dwóch do trzech tygodni, po który nory były odlewany, a odlewy wykopywane i analizowane. Na opisy podziemnych struktur składały się: architektura, wymiary, bioglify, złożoność i krętość. Dodatkowe eksperymenty przebiegały przy odmiennym składzie podłoża, jego gęstości i wilgotności, by przetestować odpowiedź zwierzęcia na zmianę warunków środowiskowych. Okazy H. arizonensis kopały przez drapanie i wyrzucanie luźnego osadu z głębi przy użyciu pierwszych dwóch do trzech par odnóży krocznych. Wyprodukowane podziemne struktury biogeniczne składały się z subwertykalnych ramp, nor U-kształtnych i heliakalnych, oraz złożonych sieci. Podczas wykopywania nor, skorpiony pustynne produkowały też zaburzoną topografię powierzchni, jak i struktury w suchym piaszczystym osadzie, które przypominały laminację i przekątną laminację zmarszczek. Zwiększona zawartość iłu i gęstość osadu zwiększały skomplikowanie architektoniczne produkowanych nor. Obniżanie tych zmiennych ograniczało złożoność nor, zmniejszało prawdopodobieństwo zachowania i zwiększało ilość biogenicznej laminacji przekątnej. Dane zebrane z poniższego i podobnych badań eksperymentalnych mogą znaleźć zastosowanie kopalnych ichnozespołów lądowych i pomóc lepiej zinterpretować paleoekologię dawnych ekosystemów glebowych.
Słowa kluczowe: ichnoskamieniałości; skamieniałości śladowe; bioturbacja; kontynentalne; stawonóg; paleoekologia
Translators: Dawid Mazurek and Robert Bronowicz
Riassunto in Italiano
Neoicnologia dello scorpione del deserto Hadrurus arizonensis
Fenomeni di bioturbazione legata all’azione di animali terrestri sono comuni in ambienti continentali aridi e semiaridi. Sin dal tardo Paleozoico fino ad epoche attuali gli scorpioni costituiscono una parte importante della diversità degli artropodi predatori. Molti scorpioni sono scavatori attivi e con tutta probabilità hanno un record a livello paleoichnologico significativo, seppur esso è raramente riconosciuto come tale. Il presente progetto ha interessato lo studio delle abitudini “scavatorie” e della morfologia delle tracce corrispondenti di Hadrurus arizonensis (Scorpiones: Caraboctonidae). I singoli esemplari sono stati posti in terrari riempiti di sedimento per un periodo di due-tre settimane nelle quali sono stati osservati e descritti i fori da essi scavati. La descrizione delle strutture sotterranee ha compreso i seguenti parametri: architettura, dimensioni, bioglifi, complessità e tortuosità. Ulteriori prove sono state condotte utilizzando sedimenti di differente composizione, densità e umidità al fine di stimare la risposta comportamentale degli esemplari a differenti condizioni ambientali. H. arizonensis scava “grattando” e “calciando via” sedimenti sciolti dal sottosuolo con le prime due paia delle appendici locomotorie. Le strutture biogeniche sotterranee prodotte consistevano in rampe subverticali, fori ad U, elicoidali e labirintiformi. E’ da mettere in evidenza che durante il processo di scavo in sedimenti sabbiosi ed asciutti gli scorpioni del deserto hanno prodotto una superficie esterna ondulata e strutture con aspetto di tipo laminato e a laminazione incrociata. All’aumentare della componente argillosa e della densità del sedimento è aumentata anche la complessità dell’architettura del foro. All’estremo contrario abbiamo notato una ridotta complessità dei fori, una minore probabilità di conservazione e un’aumentata abbondanza di stratificazione incrociata biogenica. I dati ottenuti con la presente ricerca e quelli ottenuti da ricerche sperimentali simili possono essere applicati allo studio degli ichnofossili terrestri al fine di ottenere una migliore interpretazione paleoecologica degli ecosistemi legati ai suoli.
Parole chiave: ichnofossili; tracce fossili; bioturbazione; continentale; artropodi; paleoecologia
Translator: Chiara Angelone
-
-
-
Review: The Princeton Field Guide to Mesozoic Sea Reptiles
 The Princeton Field Guide to Mesozoic Sea Reptiles
The Princeton Field Guide to Mesozoic Sea ReptilesArticle number: 26.1.1R
April 2023





 Poster Winners 2024
Poster Winners 2024