Article Search
Volume 27.1
January–April 2024
Full table of contents
ISSN: 1094-8074, web version;
1935-3952, print version
Recent Research Articles
See all articles in 27.1 January-April 2024
See all articles in 26.3 September-December 2023
See all articles in 26.2 May-August 2023
See all articles in 26.1 January-April 2023
 Myriam Boivin. Laboratoire de Paléontologie, Institut des Sciences de l’Évolution de Montpellier (ISE-M, UMR 5554, CNRS/UM/IRD/EPHE), c.c. 064, Université de Montpellier, place Eugène Bataillon, F-34095 Montpellier Cedex 05, France. myriam.boivin@umontpellier.fr
Myriam Boivin. Laboratoire de Paléontologie, Institut des Sciences de l’Évolution de Montpellier (ISE-M, UMR 5554, CNRS/UM/IRD/EPHE), c.c. 064, Université de Montpellier, place Eugène Bataillon, F-34095 Montpellier Cedex 05, France. myriam.boivin@umontpellier.fr
Myriam Boivin is a PhD student and teaching assistant at the University of Montpellier, France, supervised by Pierre-Olivier Antoine and Laurent Marivaux. Her works focus on morphology, phylogeny, and paleobiogeography of Paleogene caviomorph rodents from Peruvian Amazonia.

 Laurent Marivaux. Laboratoire de Paléontologie, Institut des Sciences de l’Évolution de Montpellier (ISE-M, UMR 5554, CNRS/UM/IRD/EPHE), c.c. 064, Université de Montpellier, place Eugène Bataillon, F-34095 Montpellier Cedex 05, France. laurent.marivaux@umontpellier.fr
Laurent Marivaux. Laboratoire de Paléontologie, Institut des Sciences de l’Évolution de Montpellier (ISE-M, UMR 5554, CNRS/UM/IRD/EPHE), c.c. 064, Université de Montpellier, place Eugène Bataillon, F-34095 Montpellier Cedex 05, France. laurent.marivaux@umontpellier.fr
Laurent Marivaux is a CNRS researcher, paleontologist and head of the Paleontology Department at the "Institut des Sciences de l'Evolution de Montpellier". He has a PhD in Vertebrate Paleontology from the University of Montpellier. His research focuses on macroevolutionary, phylogenetic, historical biogeography and paleobiological aspects of the early Tertiary adaptive radiations of some mammal groups (especially Rodentia, Primates, Dermoptera and Chiroptera, and others in collaboration), with a special emphasis on the role of the geodynamic and paleoenvironment changes on their emergence, dispersals and evolution. His is primarily a field paleontologist working in Paleo- and Neotropics.

 Maëva J. Orliac. Laboratoire de Paléontologie, Institut des Sciences de l’Évolution de Montpellier (ISE-M, UMR 5554, CNRS/UM/IRD/EPHE), c.c. 064, Université de Montpellier, place Eugène Bataillon, F-34095 Montpellier Cedex 05, France. maeva.orliac@umontpellier.fr
Maëva J. Orliac. Laboratoire de Paléontologie, Institut des Sciences de l’Évolution de Montpellier (ISE-M, UMR 5554, CNRS/UM/IRD/EPHE), c.c. 064, Université de Montpellier, place Eugène Bataillon, F-34095 Montpellier Cedex 05, France. maeva.orliac@umontpellier.fr
Maëva J. Orliac is a CNRS researcher at the University of Montpellier, France. She is a vertebrate paleontologist devoted to Cenozoic mammal evolution and phylogeny. She completed her undergraduate training and her PhD at the University Pierre et Marie Curie, in Paris. Her work primarily focused on the morphology and systematics of suoids (pigs and peccaries) and hippos, and their relationships within Artiodactyla. She is currently working on endocranial features of extinct and extant ungulates and primates, such as the middle and inner ear, and brain morphology. Recent fieldwork has taken her to Cenozoic deposits of Turkey and Peruvian Amazonia.

 Francois Pujos. Instituto Argentino de Nivología, Glaciología y Ciencias Ambientales (IANIGLA), CCT-CONICET-Mendoza, Av. Ruiz Leal s/n, Parque Gral. San Martín, 5500 Mendoza, Argentina. fpujos@mendoza-conicet.gob.ar
Francois Pujos. Instituto Argentino de Nivología, Glaciología y Ciencias Ambientales (IANIGLA), CCT-CONICET-Mendoza, Av. Ruiz Leal s/n, Parque Gral. San Martín, 5500 Mendoza, Argentina. fpujos@mendoza-conicet.gob.ar
François Pujos is a former Researcher of the French Institute of Andean Studies (IFEA) and currently Senior Researcher of the Argentinean Scientific and Technical National Research Council (CONICET). As a geologist and paleomammalogist, he focuses on systematics, evolution, and paleobiogeography of South American Cenozoic mammals (e.g., Xenarthra). He has organized 25 field missions in several countries of South America (i.e., Argentina, Bolivia, Brazil, and Peru). As a collaborator on a multidisciplinary team, he has participated in the discovery of several new vertebrate communities in the Bolivian Altiplano and Peruvian Amazon and described new taxa, including several ground sloths.

 Rodolfo Salas-Gismondi. Departamento de Paleontología de Vertebrados, Museo de Historia Natural - Universidad Nacional Mayor San Marcos (MUSM), Av. Arenales 1256, Lima 11, Peru. rsalasgismondi@gmail.com
Rodolfo Salas-Gismondi. Departamento de Paleontología de Vertebrados, Museo de Historia Natural - Universidad Nacional Mayor San Marcos (MUSM), Av. Arenales 1256, Lima 11, Peru. rsalasgismondi@gmail.com
Rodolfo Salas-Gismondi is founder and head of the Vertebrate Paleontology Department of the San Marcos University Natural History Museum in Lima, Peru. He has a PhD from the Institut des Sciences de l'Evolution, University of Montpellier, France. He is currently at the Smithsonian Tropical Research Institute, Panama. His research was first devoted to the evolution of South American vertebrates, mainly terrestrial and aquatic mammals (Pacific Coast, Andean highlands, and Amazonia). His current research is focused in the systematics, ecology, and evolution of the crocodylians from the Tertiary of Amazonia.

 Julia V. Tejada-Lara. Departamento de Paleontología de Vertebrados, Museo de Historia Natural - Universidad Nacional Mayor San Marcos (MUSM), Av. Arenales 1256, Lima 11, Peru, and Department of Earth and Environmental Sciences, Columbia University in the City of New York and Division of Paleontology, American Museum of Natural History, Central Park West at 79th Street, New York, NY 10024, USA. julia.tejada@columbia.edu
Julia V. Tejada-Lara. Departamento de Paleontología de Vertebrados, Museo de Historia Natural - Universidad Nacional Mayor San Marcos (MUSM), Av. Arenales 1256, Lima 11, Peru, and Department of Earth and Environmental Sciences, Columbia University in the City of New York and Division of Paleontology, American Museum of Natural History, Central Park West at 79th Street, New York, NY 10024, USA. julia.tejada@columbia.edu
Julia Victoria Tejada-Lara is a PhD student at the Department of Earth and Environmental Sciences at Columbia University, New York. She holds a Master's degree from the Department of Zoology (University of Florida) and a BSc from the National University of San Marcos, Peru. She is Associate Researcher at the French Institute of Andean Studies and at the Natural History Museum in Lima, Peru. Her research has focused on the evolution of Cenozoic mammal communities and ecosystems from Proto-Amazonia and the Andean Plateau. Current research combines systematics, paleogeography, stable isotope geochemistry, and paleoecology of extinct and modern mammals from South America.

 Pierre-Olivier Antoine. Laboratoire de Paléontologie, Institut des Sciences de l’Évolution de Montpellier (ISE-M, UMR 5554, CNRS/UM/IRD/EPHE), c.c. 064, Université de Montpellier, place Eugène Bataillon, F-34095 Montpellier Cedex 05, France. pierre-olivier.antoine@umontpellier.fr
Pierre-Olivier Antoine. Laboratoire de Paléontologie, Institut des Sciences de l’Évolution de Montpellier (ISE-M, UMR 5554, CNRS/UM/IRD/EPHE), c.c. 064, Université de Montpellier, place Eugène Bataillon, F-34095 Montpellier Cedex 05, France. pierre-olivier.antoine@umontpellier.fr
Pierre-Olivier Antoine is a Professor of Paleontology at the University of Montpellier, France. He has a PhD from the Muséum National d'Histoire Naturelle, Paris and he was formerly Associate Professor in the University of Toulouse, France. His research was primarily devoted to Cenozoic mammal evolution, combining phylogenetic analyses and fieldwork in remote areas from Eurasia and South America. Major focuses of his current research are balanced between Cenozoic stratigraphy, biochronology, and paleogeography in Eurasia, and Proto-Amazonian biodiversity and ecosystem dynamics in South America. He has participated in more than 40 field expeditions in Pakistan, Turkey, Peru, and Bolivia.
APPENDIX 1.
Evolution of some Eocene caviomorphs-bearing localities between 2011 and 2016 in the Contamana area. 1, CTA-51; 2, CTA-29; 3, CTA-27.

APPENDIX 2.
Palaeogene caviomorph taxa used for comparisons in this study.
| Genus | Species | Locality and/or Formation (Fm.) | Age | Observed material (original or cast) | Primary references |
| Llitun | L. notuca | Cabeza Blanca (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | UM (cast) | Vucetich et al., 2015 |
| Leucokephalos | L. zeffiae | Cabeza Blanca (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | UM (cast) | Vucetich et al., 2015 |
| Octodontoidea | |||||
| Eosallamys | E. paulacoutoi | Santa Rosa (Yahuarango Fm.), Peru | ?Late Eocene / Early Oligocene | INGEMMET (holotype) | Frailey and Campbell, 2004 |
| E. simpsoni | Santa Rosa (Yahuarango Fm.), Peru | ?Late Eocene / Early Oligocene | INGEMMET (holotype) | Frailey and Campbell, 2004 | |
| Eoespina | E. woodi | Santa Rosa (Yahuarango Fm.), Peru | ?Late Eocene / Early Oligocene | INGEMMET (holotype) | Frailey and Campbell, 2004 |
| Eosachacui | E. lavocati | Santa Rosa (Yahuarango Fm.), Peru | ?Late Eocene / Early Oligocene | INGEMMET (holotype) | Frailey and Campbell, 2004 |
| Draconomys | D.verai | La Cantera (Sarmiento Fm.), Argentina | late Early Oligocene | UM (cast) | Vucetich et al., 2010 |
| Vallehermosomys | V. mazzonii | La Cantera (Sarmiento Fm.), Argentina | late Early Oligocene | UM (cast) | Vucetich et al., 2010 |
| ?Vallehermosomys | ?V. merlinae | La Cantera (Sarmiento Fm.), Argentina | late Early Oligocene | UM (cast) | Vucetich et al., 2010 |
| Migraveramus | M. beatus | Salla (Salla Beds), Bolivia | late Early Oligocene - Late Oligocene | Patterson and Wood 1982 | |
| Sallamys | S. pascuali | Salla (Salla Beds), Bolivia | late Early Oligocene - Late Oligocene | MNHN and UM (original) | Hoffstetter and Lavocat, 1970; Lavocat, 1976; Hartenberger, 1975; Patterson and Wood, 1982 |
| S. cf. pascuali | Salla (Salla Beds), Bolivia | late Early Oligocene - Late Oligocene | Shockey et al., 2009 | ||
| S. quispea | Cerro Pan de Azúcar (Moquega Fm.), Peru | late Early Oligocene - Late Oligocene | MUSM (original) | Shockey et al., 2009 | |
| Paulacoutomys | P. paulista | Fazenda Santa Fé (Tremembé Fm.), Brazil | late Early Oligocene - Late Oligocene | MLP (cast) | Vucetich et al., 1993 |
| Platypittamys | P. brachyodon | Scaritt Pocket (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | UM (cast) | Wood, 1949; Wood and Patterson, 1959 |
| Deseadomys | D. arambourgi | Cabeza Blanca (Sarmiento Fm.) and La Flecha, Argentina | late Early Oligocene - Late Oligocene | MHNH (original) | Wood and Patterson, 1959 |
| Galileomys | G. baios | Cabeza Blanca (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | UM (cast) | Vucetich et al., 2015 |
| Acarechimys | A. leucotheae | Cabeza Blanca (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | UM (cast) | Vucetich et al., 2015 |
| Chinchilloidea | |||||
| Branisamys | B. luribayensis | Salla (Salla Beds), Bolivia | late Early Oligocene - Late Oligocene | MNHN and UM (original) | Hoffstetter and Lavocat, 1970; Lavocat, 1976; Patterson and Wood, 1982 |
| Incamys | I. bolivianus | Salla (Salla Beds), Bolivia | late Early Oligocene - Late Oligocene | MNHN and UM (original) | Hoffstetter and Lavocat, 1970; Lavocat, 1976; Patterson and Wood, 1982 |
| I. menniorum | Cabeza Blanca (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | UM (cast) | Vucetich et al., 2015 | |
| Loncolicu | L. tretos | Cabeza Blanca (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | UM (cast) | Vucetich et al., 2015 |
| Scotamys | S. antiquus | Cabeza Blanca and La Flecha (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | Loomis, 1914; Wood and Patterson, 1959 | |
| Cephalomys | C. arcidens | Cabeza Blanca and La Flecha (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | MACN (original) | Ameghino, 1897; Wood and Patterson, 1959 |
| C. plexus | Cabeza Blanca and La Flecha (Sarmiento Fm.), Argentina | late Early Oligocene - Late Oligocene | MACN (original) | Ameghino, 1897; Wood and Patterson, 1959 | |
| C. bolivianus | Salla (Salla Beds), Bolivia | late Early Oligocene - Late Oligocene | MNHN and UM (original) | Lavocat, 1976; Patterson and Wood, 1982 | |
| Cavioidea | |||||
| Eobranisamys | E. romeropittmanae | Santa Rosa (Yahuarango Fm.), Peru | ?Late Eocene / Early Oligocene | INGEMMET (holotype) | Frailey and Campbell, 2004 |
| E. riverai | Santa Rosa (Yahuarango Fm.), Peru | ?Late Eocene / Early Oligocene | INGEMMET (holotype) | Frailey and Campbell, 2004 | |
| Eoincamys | E. ameghinoi | Santa Rosa (Yahuarango Fm.), Peru | ?Late Eocene / Early Oligocene | INGEMMET (holotype) | Frailey and Campbell, 2004 |
| E. pascuali | Santa Rosa (Yahuarango Fm.), Peru | ?Late Eocene / Early Oligocene | INGEMMET (holotype) | Frailey and Campbell, 2004 | |
| Andemys | A. termasi | Tinguiririca Termas del Flaco (Abanico Fm.), Chili | late Early Oligocene - Late Oligocene | Bertrand et al., 2012 | |
Fm., Formation; INGEMMET, Instituto Geológico Minero y Metalúrgico, Lima; MACN, Museo Argentino de Ciencias Naturales, Buenos Aires; MLP, Museo de Ciencias Naturales de La Plata; MUSM, Museo de Historia Natural de la Universidad Nacional Mayor San Marcos, Lima; MNHN, Musée National d'Histoire Naturelle, Paris; UM, Université de Montpellier. |
|||||
APPENDIX 3.
Dental measurements (in millimetres) of rodent material from CTA-47, CTA- 51, CTA-73, CTA-27, CTA-66, and CTA-29, Middle Eocene - late Middle Eocene, Peruvian Amazonia.
| Collection number | Locality | Taxon | Locus | ML | MW | Hg | Hb | HIg | HIb |
| MUSM 2648 | CTA 47 | ?Cachiyacuy kummeli | l. dP4 | 1.48 | 0.50 | 0.34 | |||
| MUSM 2645 | CTA 47 | ?Canaanimys sp. | r. dp4 | ||||||
| MUSM 2646 | CTA 47 | ?Canaanimys sp. | l. lower molar | ||||||
| MUSM 2647 | CTA 47 | Caviomorpha indet. 1 | r. lower molar | ||||||
| MUSM 2651 | CTA 51 | Cachiyacuy cf. contamanensis 1 | r. dp4 | ||||||
| MUSM 2652 | CTA 51 | Cachiyacuy cf. contamanensis 1 | l. m1 | 1.86 | 1.75 | ||||
| MUSM 2653 | CTA 51 | Cachiyacuy cf. contamanensis 1 | l. m3 | 2.19 | 0.76 | 0.96 | 0.35 | 0.44 | |
| MUSM 2654 | CTA 51 | Cachiyacuy cf. kummeli | l. M1 | ||||||
| MUSM 2655 | CTA 51 | Eoespina sp. | r. M2 | 1.38 | 1.41 | 0.80 | 0.44 | 0.57 | 0.31 |
| MUSM 2656 | CTA 51 | Caviomorpha indet. 2 | r. lower molar | ||||||
| MUSM 2657 | CTA 51 | Caviomorpha indet. 2 | l. dP4 | 1.50 | 1.36 | 0.68 | 0.42 | 0.45 | 0.31 |
| MUSM 2658 | CTA 51 | Caviomorpha indet. 2 | l. upper tooth | 1.35 | 1.23 | ||||
| MUSM 2659 | CTA 73 | Caviomorpha indet. 3 | r. lower molar | ||||||
| MUSM 2660 | CTA 73 | Caviomorpha indet. 4 | l. upper molar | 1.49 | 0.68 | 0.46 | |||
| MUSM 2661 | CTA 27 | Cachiyacuy contamanensis | l. dp4 | 2.61 | 1.78 | 1.06 | 0.87 | 0.41 | 0.33 |
| MUSM 2662 | CTA 27 | Cachiyacuy contamanensis | l. dp4 | 2.16 | 1.51 | 0.68 | 0.58 | 0.32 | 0.27 |
| MUSM 2663 | CTA 27 | Cachiyacuy contamanensis | l. dp4 | 1.46 | 0.72 | 0.80 | |||
| MUSM 2664 | CTA 27 | Cachiyacuy contamanensis | l. dp4 | ||||||
| MUSM 2665 | CTA 27 | Cachiyacuy contamanensis | l. dp4 | 2.22 | 1.48 | 0.86 | 0.70 | 0.39 | 0.32 |
| MUSM 2666 | CTA 27 | Cachiyacuy contamanensis | l. dp4 | ||||||
| MUSM 2667 | CTA 27 | Cachiyacuy contamanensis | l. dp4 | ||||||
| MUSM 1880 | CTA 27 | Cachiyacuy contamanensis | r. dp4 | 2.31 | 1.53 | ||||
| MUSM 2668 | CTA 27 | Cachiyacuy contamanensis | r. dp4 | 1.34 | |||||
| MUSM 2669 | CTA 27 | Cachiyacuy contamanensis | r. dp4 | 1.66 | |||||
| MUSM 2670 | CTA 27 | Cachiyacuy contamanensis | r. dp4 | 1.53 | |||||
| MUSM 2671 | CTA 27 | Cachiyacuy contamanensis | r. dp4 | 1.60 | |||||
| MUSM 2672 | CTA 27 | Cachiyacuy contamanensis | r. dp4 | 1.48 | |||||
| MUSM 2673 | CTA 27 | Cachiyacuy contamanensis | r. dp4 | ||||||
| MUSM 2674 | CTA 27 | Cachiyacuy contamanensis | l. p4 | 1.94 | 1.90 | 1.30 | 0.67 | ||
| MUSM 2675 | CTA 27 | Cachiyacuy contamanensis | l. p4 | ||||||
| MUSM 1879 | CTA 27 | Cachiyacuy contamanensis | r. p4 | 1.99 | 2.00 | ||||
| MUSM 2676 | CTA 27 | Cachiyacuy contamanensis | r. p4 | 2.04 | 2.03 | 1.09 | 1.29 | 0.53 | 0.63 |
| MUSM 2677 | CTA 27 | Cachiyacuy contamanensis | r. p4 | 2.14 | 1.99 | 0.90 | 0.96 | 0.42 | 0.45 |
| MUSM 2678 | CTA 27 | Cachiyacuy contamanensis | r. p4 | 2.00 | |||||
| MUSM 2679 | CTA 27 | Cachiyacuy contamanensis | l. m1 | ||||||
| MUSM 2680 | CTA 27 | Cachiyacuy contamanensis | l. m1 | 2.13 | 2.07 | 0.81 | 1.17 | 0.38 | 0.55 |
| MUSM 2681 | CTA 27 | Cachiyacuy contamanensis | l. m1 | 2.07 | 0.67 | 0.32 | |||
| MUSM 2682 | CTA 27 | Cachiyacuy contamanensis | l. m1 | 1.94 | 1.87 | 0.64 | 0.33 | ||
| MUSM 1878 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.15 | 1.99 | ||||
| MUSM 2683 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 1.66 | |||||
| MUSM 2684 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.11 | 2.02 | 0.78 | 0.37 | ||
| MUSM 2685 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.09 | 2.07 | 1.11 | 0.53 | ||
| MUSM 2686 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.03 | 1.96 | 0.91 | 0.89 | 0.45 | 0.44 |
| MUSM 2687 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.20 | 2.10 | 0.52 | 0.55 | 0.24 | 0.25 |
| MUSM 2688 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.15 | 1.98 | 1.00 | 0.83 | 0.47 | 0.39 |
| MUSM 2689 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.18 | 1.99 | 1.20 | 1.30 | 0.55 | 0.60 |
| MUSM 2690 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.17 | 2.11 | 0.68 | 0.31 | ||
| MUSM 2691 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.18 | 2.04 | 0.54 | 0.62 | 0.25 | 0.28 |
| MUSM 2692 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.13 | 2.02 | 0.59 | 1.00 | 0.28 | 0.47 |
| MUSM 2693 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.02 | 1.94 | ||||
| MUSM 2694 | CTA 27 | Cachiyacuy contamanensis | r. m1 | 2.08 | 1.91 | 0.77 | 0.94 | 0.37 | 0.45 |
| MUSM 2695 | CTA 27 | Cachiyacuy contamanensis | l. m2 | 2.08 | |||||
| MUSM 2696 | CTA 27 | Cachiyacuy contamanensis | l. m2 | ||||||
| MUSM 2697 | CTA 27 | Cachiyacuy contamanensis | l. m2 | ||||||
| MUSM 2698 | CTA 27 | Cachiyacuy contamanensis | l. m2 | ||||||
| MUSM 2699 | CTA 27 | Cachiyacuy contamanensis | l. m2 | 2.29 | 2.24 | ||||
| MUSM 2700 | CTA 27 | Cachiyacuy contamanensis | l. m2 | 2.27 | 2.28 | ||||
| MUSM 2701 | CTA 27 | Cachiyacuy contamanensis | l. m2 | 2.36 | 2.41 | 1.07 | 1.46 | 0.45 | 0.62 |
| MUSM 2702 | CTA 27 | Cachiyacuy contamanensis | l. m2 | 2.21 | 2.39 | ||||
| MUSM 2703 | CTA 27 | Cachiyacuy contamanensis | l. m2 | 2.18 | 2.35 | ||||
| MUSM 1877 | CTA 27 | Cachiyacuy contamanensis | r. m2 | 2.44 | 2.36 | ||||
| MUSM 1914 | CTA 27 | Cachiyacuy contamanensis | r. m2 | 2.45 | 2.33 | ||||
| MUSM 1915 | CTA 27 | Cachiyacuy contamanensis | r. m2 | 2.37 | 2.18 | ||||
| MUSM 2704 | CTA 27 | Cachiyacuy contamanensis | r. m2 | 2.21 | 2.17 | 0.88 | 1.02 | 0.40 | 0.46 |
| MUSM 2705 | CTA 27 | Cachiyacuy contamanensis | r. m2 | 2.21 | 2.09 | 0.86 | 0.39 | ||
| MUSM 2706 | CTA 27 | Cachiyacuy contamanensis | r. m2 | ||||||
| MUSM 2707 | CTA 27 | Cachiyacuy contamanensis | r. m2 | ||||||
| MUSM 2708 | CTA 27 | Cachiyacuy contamanensis | r. m2 | 2.49 | 2.52 | ||||
| MUSM 2709 | CTA 27 | Cachiyacuy contamanensis | r. m2 | 2.27 | 2.24 | 1.05 | 0.83 | 0.46 | 0.37 |
| MUSM 2710 | CTA 27 | Cachiyacuy contamanensis | r. m2 | 2.29 | 2.29 | 0.95 | 0.41 | ||
| MUSM 2711 | CTA 27 | Cachiyacuy contamanensis | l. m3 | 0.91 | |||||
| MUSM 1876 | CTA 27 | Cachiyacuy contamanensis | r. m3 | 2.46 | 2.24 | ||||
| MUSM 2712 | CTA 27 | Cachiyacuy contamanensis | r. m3 | 2.46 | 2.07 | 0.79 | 0.32 | ||
| MUSM 2713 | CTA 27 | Cachiyacuy contamanensis | r. m3 | 2.39 | 1.93 | ||||
| MUSM 2714 | CTA 27 | Cachiyacuy contamanensis | r. m3 | 2.35 | 2.14 | 1.13 | 0.80 | 0.48 | 0.34 |
| MUSM 1875 | CTA 27 | Cachiyacuy contamanensis | P3 or DP3 | 0.77 | 0.82 | ||||
| MUSM 1874 | CTA 27 | Cachiyacuy contamanensis | l.dP4 | 2.03 | 1.98 | ||||
| MUSM 2715 | CTA 27 | Cachiyacuy contamanensis | l. dP4 | 1.83 | 1.84 | 0.71 | 0.49 | 0.39 | 0.27 |
| MUSM 2716 | CTA 27 | Cachiyacuy contamanensis | l.dP4 | 1.85 | 1.71 | 0.83 | 0.58 | 0.45 | 0.31 |
| MUSM 2717 | CTA 27 | Cachiyacuy contamanensis | l. dP4 | 1.90 | 1.89 | 0.91 | 0.65 | 0.48 | 0.34 |
| MUSM 2718 | CTA 27 | Cachiyacuy contamanensis | r. dP4 | ||||||
| MUSM 2719 | CTA 27 | Cachiyacuy contamanensis | r. dP4 | 2.06 | 1.88 | 1.26 | 0.77 | 0.61 | 0.38 |
| MUSM 2720 | CTA 27 | Cachiyacuy contamanensis | r. dP4 | 1.93 | 1.82 | 0.94 | 0.74 | 0.49 | 0.38 |
| MUSM 2721 | CTA 27 | Cachiyacuy contamanensis | r. dP4 | 1.90 | 1.66 | 0.78 | 0.61 | 0.41 | 0.32 |
| MUSM 2722 | CTA 27 | Cachiyacuy contamanensis | r. dP4 | 1.96 | 1.84 | 1.07 | 0.63 | 0.55 | 0.32 |
| MUSM 2723 | CTA 27 | Cachiyacuy contamanensis | r. dP4 | ||||||
| MUSM 2724 | CTA 27 | Cachiyacuy contamanensis | l. P4 | 2.24 | |||||
| MUSM 1873 | CTA 27 | Cachiyacuy contamanensis | r. P4 | 1.82 | 2.38 | ||||
| MUSM 2725 | CTA 27 | Cachiyacuy contamanensis | r. P4 | 1.64 | 2.18 | 1.32 | 0.99 | 0.80 | 0.60 |
| MUSM 2726 | CTA 27 | Cachiyacuy contamanensis | r. P4 | 1.72 | 2.23 | 1.26 | 1.08 | 0.74 | 0.63 |
| MUSM 2727 | CTA 27 | Cachiyacuy contamanensis | r. P4 | ||||||
| MUSM 2728 | CTA 27 | Cachiyacuy contamanensis | r. P4 | ||||||
| MUSM 2729 | CTA 27 | Cachiyacuy contamanensis | r. P4 | 1.68 | 1.94 | 1.24 | 0.74 | ||
| MUSM 1872 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 2.07 | 2.22 | ||||
| MUSM 2730 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 1.94 | 1.92 | 1.04 | 0.39 | 0.53 | 0.20 |
| MUSM 2731 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 1.89 | 2.00 | 1.28 | 0.68 | ||
| MUSM 2732 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 2.12 | 2.22 | ||||
| MUSM 2733 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 2.10 | 2.35 | 1.50 | 0.71 | ||
| MUSM 2734 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 1.96 | 2.20 | 1.44 | 0.73 | ||
| MUSM 2735 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 1.88 | 2.12 | ||||
| MUSM 2736 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 1.89 | 2.11 | 1.06 | 0.67 | 0.56 | 0.35 |
| MUSM 2737 | CTA 27 | Cachiyacuy contamanensis | l. M1 | 1.90 | |||||
| MUSM 2738 | CTA 27 | Cachiyacuy contamanensis | l. M1 | ||||||
| MUSM 2739 | CTA 27 | Cachiyacuy contamanensis | r. M1 | 1.82 | 2.12 | 1.51 | 0.83 | ||
| MUSM 2740 | CTA 27 | Cachiyacuy contamanensis | r. M1 | 1.95 | |||||
| MUSM 2741 | CTA 27 | Cachiyacuy contamanensis | r. M1 | 1.95 | |||||
| MUSM 2742 | CTA 27 | Cachiyacuy contamanensis | r. M1 | 1.93 | 2.31 | ||||
| MUSM 2743 | CTA 27 | Cachiyacuy contamanensis | r. M1 | 1.85 | 2.01 | 1.04 | 0.82 | 0.56 | 0.44 |
| MUSM 2744 | CTA 27 | Cachiyacuy contamanensis | r. M1 | 2.01 | 1.79 | ||||
| MUSM 2745 | CTA 27 | Cachiyacuy contamanensis | r. M1 | ||||||
| MUSM 2746 | CTA 27 | Cachiyacuy contamanensis | r. M1 | ||||||
| MUSM 2747 | CTA 27 | Cachiyacuy contamanensis | l. M2 | 2.29 | 2.44 | ||||
| MUSM 2748 | CTA 27 | Cachiyacuy contamanensis | l. M2 | 2.15 | 2.53 | 1.05 | 0.71 | 0.49 | 0.33 |
| MUSM 2749 | CTA 27 | Cachiyacuy contamanensis | l. M2 | 2.05 | 2.43 | 0.93 | 0.69 | 0.45 | 0.33 |
| MUSM 2750 | CTA 27 | Cachiyacuy contamanensis | l. M2 | 2.18 | 2.39 | 1.45 | 0.67 | ||
| MUSM 1871 | CTA 27 | Cachiyacuy contamanensis | r. M2 | 2.22 | 2.69 | ||||
| MUSM 2751 | CTA 27 | Cachiyacuy contamanensis | r. M2 | 2.17 | 2.70 | ||||
| MUSM 2752 | CTA 27 | Cachiyacuy contamanensis | r. M2 | 2.18 | 2.61 | 1.31 | 0.86 | 0.60 | 0.40 |
| MUSM 2753 | CTA 27 | Cachiyacuy contamanensis | r. M2 | 2.21 | 2.62 | 0.89 | 0.40 | ||
| MUSM 2754 | CTA 27 | Cachiyacuy contamanensis | r. M2 | 2.21 | 1.29 | 0.58 | |||
| MUSM 2755 | CTA 27 | Cachiyacuy contamanensis | r. M2 | 2.14 | |||||
| MUSM 2756 | CTA 27 | Cachiyacuy contamanensis | r. M2 | 2.06 | |||||
| MUSM 2757 | CTA 27 | Cachiyacuy contamanensis | r. M2 | 1.91 | 0.48 | 0.25 | |||
| MUSM 2758 | CTA 27 | Cachiyacuy contamanensis | l. M3 | 2.10 | 2.38 | 1.41 | 1.00 | 0.67 | 0.48 |
| MUSM 2759 | CTA 27 | Cachiyacuy contamanensis | l. M3 | 2.09 | 2.32 | ||||
| MUSM 2760 | CTA 27 | Cachiyacuy contamanensis | l. M3 | 2.04 | 2.32 | 1.21 | 0.59 | ||
| MUSM 2761 | CTA 27 | Cachiyacuy contamanensis | l. M3 | 2.08 | 2.35 | 1.19 | 0.63 | 0.57 | 0.30 |
| MUSM 1870 | CTA 27 | Cachiyacuy contamanensis | r. M3 | 2.34 | 2.44 | ||||
| MUSM 1888 | CTA 27 | Cachiyacuy kummeli | l. dp4 | 1.53 | 1.20 | ||||
| MUSM 2762 | CTA 27 | Cachiyacuy kummeli | l. dp4 | 1.72 | 1.17 | ||||
| MUSM 2763 | CTA 27 | Cachiyacuy kummeli | l. dp4 | 1.59 | 1.15 | 0.44 | 0.67 | 0.28 | 0.42 |
| MUSM 2764 | CTA 27 | Cachiyacuy kummeli | r. dp4 | 1.47 | 1.15 | 0.50 | 0.55 | 0.34 | 0.37 |
| MUSM 2765 | CTA 27 | Cachiyacuy kummeli | r. dp4 | 1.45 | 1.05 | 0.60 | 0.59 | 0.41 | 0.40 |
| MUSM 2766 | CTA 27 | Cachiyacuy kummeli | l. p4 | 1.24 | 1.22 | 0.70 | 0.57 | ||
| MUSM 2767 | CTA 27 | Cachiyacuy kummeli | l. m1 | 1.51 | 1.51 | 0.59 | 0.39 | ||
| MUSM 2768 | CTA 27 | Cachiyacuy kummeli | l. m1 | 1.50 | 1.51 | 0.81 | 0.54 | ||
| MUSM 1887 | CTA 27 | Cachiyacuy kummeli | r. m1 | 1.59 | 1.54 | ||||
| MUSM 2769 | CTA 27 | Cachiyacuy kummeli | r. m1 | 1.48 | 1.51 | 0.76 | 0.51 | ||
| MUSM 2770 | CTA 27 | Cachiyacuy kummeli | r. m1 | 1.55 | 1.55 | 0.90 | 0.58 | ||
| MUSM 2771 | CTA 27 | Cachiyacuy kummeli | r. m1 | 1.45 | 1.41 | 0.61 | 0.42 | ||
| MUSM 2772 | CTA 27 | Cachiyacuy kummeli | l. m2 | 1.54 | 0.76 | 0.49 | |||
| MUSM 1886 | CTA 27 | Cachiyacuy kummeli | r. m2 | 1.83 | 1.67 | ||||
| MUSM 2773 | CTA 27 | Cachiyacuy kummeli | r. m2 | 1.60 | 1.56 | 0.64 | 0.62 | 0.40 | 0.39 |
| MUSM 2774 | CTA 27 | Cachiyacuy kummeli | r. m2 | 1.68 | 1.70 | 0.70 | 0.61 | 0.42 | 0.36 |
| MUSM 2775 | CTA 27 | Cachiyacuy kummeli | r. m2 | ||||||
| MUSM 2776 | CTA 27 | Cachiyacuy kummeli | r. m2? | ||||||
| MUSM 2777 | CTA 27 | Cachiyacuy kummeli | l. m3 | 1.59 | 1.32 | ||||
| MUSM 2778 | CTA 27 | Cachiyacuy kummeli | l. m3 | 1.77 | 1.58 | ||||
| MUSM 2779 | CTA 27 | Cachiyacuy kummeli | l. m3 | 1.29 | 1.25 | 0.56 | 0.56 | 0.44 | 0.44 |
| MUSM 1885 | CTA 27 | Cachiyacuy kummeli | r. m3 | 1.60 | 1.47 | ||||
| MUSM 2780 | CTA 27 | Cachiyacuy kummeli | r. m3 | 1.81 | 1.80 | ||||
| MUSM 2781 | CTA 27 | Cachiyacuy kummeli | r. m3 | 1.70 | 1.51 | ||||
| MUSM 2783 | CTA 27 | Cachiyacuy kummeli | l. P4 | ||||||
| MUSM 1881 | CTA 27 | Cachiyacuy kummeli | r. P4 | 1.16 | |||||
| MUSM 2784 | CTA 27 | Cachiyacuy kummeli | r. M1 or M2 | 1.47 | |||||
| MUSM 1882 | CTA 27 | Cachiyacuy kummeli | l. M1 | 1.45 | 1.63 | ||||
| MUSM 2785 | CTA 27 | Cachiyacuy kummeli | l. M1 | 1.55 | 1.61 | ||||
| MUSM 1883 | CTA 27 | Cachiyacuy kummeli | l. M2 | 1.69 | 1.87 | ||||
| MUSM 2786 | CTA 27 | Cachiyacuy kummeli | l. M2 | 1.52 | 1.80 | 0.88 | 0.58 | ||
| MUSM 1884 | CTA 27 | Cachiyacuy kummeli | l. M3 | 1.67 | 1.71 | ||||
| MUSM 1895 | CTA 27 | Canaanimys maquiensis | r. dp4 | 1.07 | |||||
| MUSM 1894 | CTA 27 | Canaanimys maquiensis | l. m1 | 1.54 | 1.57 | ||||
| MUSM 2787 | CTA 27 | Canaanimys maquiensis | l. m1 | 1.42 | 1.47 | ||||
| MUSM 2788 | CTA 27 | Canaanimys maquiensis | l. m1 | ||||||
| MUSM 2789 | CTA 27 | Canaanimys maquiensis | r. m1 | 1.52 | 1.53 | 0.83 | 0.55 | ||
| MUSM 2790 | CTA 27 | Canaanimys maquiensis | r. m1 | 1.70 | 1.45 | 0.83 | 0.49 | ||
| MUSM 1893 | CTA 27 | Canaanimys maquiensis | l. m2 | 1.80 | 1.79 | ||||
| MUSM 1892 | CTA 27 | Canaanimys maquiensis | r. m3 | 1.71 | 1.36 | ||||
| MUSM 2791 | CTA 27 | Canaanimys maquiensis | r. m3 | 1.73 | 1.44 | 0.80 | 0.74 | 0.46 | 0.43 |
| MUSM 1891 | CTA 27 | Canaanimys maquiensis | l. M1 | 1.46 | 1.82 | ||||
| MUSM 1890 | CTA 27 | Canaanimys maquiensis | r. M2 | 1.63 | 1.95 | ||||
| MUSM 2792 | CTA 27 | Canaanimys maquiensis | l. M3 | 1.49 | 1.78 | ||||
| MUSM 1889 | CTA 27 | Canaanimys maquiensis | r. M3 | 1.50 | 1.87 | ||||
| MUSM 2793 | CTA 27 | Canaanimys maquiensis | r. M3 | 1.47 | 1.72 | ||||
| MUSM 2794 | CTA 27 | Canaanimys maquiensis | r. M3 | 1.40 | 1.84 | ||||
| MUSM 2795 | CTA 27 | Eobranisamys javierpradoi sp. nov. | l. m1 or m2 | ||||||
| MUSM 1898 | CTA 27 | Eobranisamys javierpradoi sp. nov. | r. m2 | 2.23 | |||||
| MUSM 1899 | CTA 27 | Eobranisamys javierpradoi sp. nov. | l. m3 | 2.47 | 2.06 | ||||
| MUSM 2796 | CTA 27 | Eobranisamys javierpradoi sp. nov. | r. m3 | ||||||
| MUSM 2797 | CTA 27 | Eobranisamys javierpradoi sp. nov. | r. dP4 | 2.11 | 1.91 | ||||
| MUSM 1896 | CTA 27 | Eobranisamys javierpradoi sp. nov. | l. P4 | 1.86 | 2.33 | ||||
| MUSM 2798 | CTA 27 | Eobranisamys javierpradoi sp. nov. | l. P4 | 1.65 | 2.15 | 0.75 | 0.45 | ||
| MUSM 2799 | CTA 27 | Eobranisamys javierpradoi sp. nov. | r. M1 or M2 | 2.68 | 1.03 | ||||
| MUSM 1897 | CTA 27 | Eobranisamys javierpradoi sp. nov. | l. M1 | 2.39 | 2.49 | ||||
| MUSM 2800 | CTA 27 | Eobranisamys javierpradoi sp. nov. | l. M1 | 2.23 | 2.38 | ||||
| MUSM 2801 | CTA 27 | Eobranisamys javierpradoi sp. nov. | l. M3 | 2.27 | |||||
| MUSM 1912 | CTA 27 | cf. Eoespina sp. | l. M2 | 1.51 | 1.77 | ||||
| MUSM 2802 | CTA 27 | cf. Eoespina sp. | l. M2 | ||||||
| MUSM 1913 | CTA 27 | cf. Eoespina sp. | r. M2 | 1.48 | 1.85 | ||||
| MUSM 2841 | CTA 66 | Eobranisamys sp. | l. upper molar? | ||||||
| MUSM 2842 | CTA 66 | Eobranisamys sp. | l. upper molar | ||||||
| MUSM 2819 | CTA 29 | Pozomys ucayaliensis gen. et sp. nov. | r. M3 | ||||||
| MUSM 2820 | CTA 29 | Pozomys ucayaliensis gen. et sp. nov. | r. p4 | ||||||
| MUSM 2821 | CTA 29 | Pozomys ucayaliensis gen. et sp. nov. | l. p4 | 1.34 | 1.33 | 0.56 | 0.73 | 0.42 | 0.55 |
| MUSM 2822 | CTA 29 | Pozomys ucayaliensis gen. et sp. nov. | r. m1 | 1.58 | 1.56 | 0.64 | 0.75 | 0.40 | 0.48 |
| MUSM 2822 | CTA 29 | Pozomys ucayaliensis gen. et sp. nov. | r. m2 | 1.93 | 1.63 | 0.59 | 0.76 | 0.31 | 0.39 |
| MUSM 2833 | CTA 29 | Pozomys ucayaliensis gen. et sp. nov. | r. M2 | 1.45 | 1.81 | 0.89 | 0.42 | 0.62 | 0.29 |
| MUSM 2823 | CTA 29 | Cachiyacuy cf. contamanensis 2 | r. dp4 or p4 | 1.43 | 0.66 | 0.62 | |||
| MUSM 2824 | CTA 29 | Cachiyacuy cf. contamanensis 2 | r. dp4 | ||||||
| MUSM 2825 | CTA 29 | Cachiyacuy cf. contamanensis 2 | l. dp4 | 2.22 | 1.49 | 0.73 | 0.64 | 0.33 | 0.29 |
| MUSM 2826 | CTA 29 | Cachiyacuy cf. contamanensis 2 | r. m1 | 2.19 | 1.99 | 1.03 | 0.47 | ||
| MUSM 2827 | CTA 29 | Cachiyacuy cf. contamanensis 2 | r. m1 | 1.03 | 2.03 | 0.54 | 0.52 | ||
| MUSM 2828 | CTA 29 | Cachiyacuy cf. contamanensis 2 | r. dP4 | 1.63 | 1.69 | ||||
| MUSM 2829 | CTA 29 | Cachiyacuy cf. contamanensis 2 | r. upper molar | 1.90 | 1.03 | ||||
| MUSM 2830 | CTA 29 | Cachiyacuy cf. contamanensis 2 | r. upper molar | 1.22 | 1.65 | 0.65 | 0.53 | ||
| MUSM 2831 | CTA 29 | Cachiyacuy cf. contamanensis 2 | l. M1 | 2.02 | 2.20 | ||||
| MUSM 2832 | CTA 29 | Cachiyacuy cf. contamanensis 2 | l. M2 | 2.33 | 2.77 | ||||
| MUSM 2563 | CTA 29 | Cachiyacuy cf. contamanensis 2 | r. M2 | 2.00 | 2.52 | 1.19 | 0.78 | 0.59 | 0.39 |
| MUSM 2834 | CTA 29 | Cavioidea or Chinchilloidea indet. | r. dp4 | 2.66 | 1.85 | ||||
| MUSM 2835 | CTA 29 | Cavioidea or Chinchilloidea indet. | r. m2 | 2.68 | 2.81 | 1.09 | 0.69 | 0.41 | 0.26 |
| MUSM 2836 | CTA 29 | Cavioidea or Chinchilloidea indet. | l. upper molar | 2.55 | 1.20 | 0.47 | |||
| MUSM 2837 | CTA 29 | Cavioidea or Chinchilloidea indet. | l. upper molar | ||||||
| MUSM 2838 | CTA 29 | Caviomorpha indet. 5 | r. dp4 | 1.87 | 1.14 | 0.44 | 0.23 | ||
| MUSM 2839 | CTA 29 | Caviomorpha indet. 6 | r. upper molar | 1.30 | 1.41 | 0.45 | 0.30 | 0.35 | 0.23 |
Hb, maximum labial crown height; Hg, maximum lingual crown; HIb, hypsodont index (calculated from Hb); HIg, hypsodont index (calculated from Hg); l., left; ML, maximum anteroposterior length; MW, maximum linguolabial width; r., right. |
|||||||||
TABLE 1. List of described taxa (in bold) depending the studied Eocene localities of Contamana (classed from the oldest to the most recent) and the comparison with Santa Rosa (Peru; Frailey and Campbell, 2004). The color ranges correspond to the stratigraphic distribution of a taxon or a set of taxa closely related (at a generic or specific level).
| CTA 47 | CTA 51 | CTA 73 | CTA 27 | CTA 66 | CTA 29 | Snta Rosa | |
| Cachiyacuy contamanensis | X | ||||||
| Cachiyacuy cf. contamanensis 1 | X | ||||||
| Cachiyacuy cf. contamanensis 2 | X | ||||||
| Cachiyacuy kummeli | X | ||||||
| Cachiyacuy cf. kummeli | X | ||||||
| ?Cachiyacuy kummeli | X | ||||||
| Canaanimys maquiensis | X | ||||||
| ?Canaanimys sp. | X | ||||||
| Eobranisamys javierpradoi sp. nov. | X | ||||||
| Eobranisamys sp. | X | ||||||
| Eobranisamys romeropittmanae | X | ||||||
| Eobranisamys riverai | X | ||||||
| Eoespina sp. | X | ||||||
| cf. Eoespina sp. | X | ||||||
| Eoespina woodi | X | ||||||
| Pozomys ucayaliensis gen. et sp. nov. | X | ||||||
| Cavioidea or Chinchilloidea indet. | X | ||||||
| Caviomorpha indet. 1 | X | ||||||
| Caviomorpha indet. 2 | X | ||||||
| Caviomorpha indet. 3 | X | ||||||
| Caviomorpha indet. 4 | X | ||||||
| Caviomorpha indet. 5 | X | ||||||
| Caviomorpha indet. 6 | X |
FIGURE 1. Geographic location and Cenozoic stratigraphy of the Contamana area, Peru. 1, location map of the Contamana area in Peruvian Amazonia (Loreto Department). 2-3, synthetic stratigraphic units of the complete Contamana Cenozoic sequence along the Cachiyacu stream (modified after Antoine et al., 2016, figure 3), including fossil-bearing levels, among which Eocene rodent-yielding localities CTA-47, CTA-51, CTA-73, CTA-27, CTA-66, and CTA-29. Note also in the same section, the location of the other rodent-bearing localities, designated by an asterisk, in the Pozo, Chambira and Pebas Fm.; 2, NE Flank of the Maquía Anticline; 3, SW Flank of the Maquía Anticline. Modified from Antoine et al. (2012, 2016). Fm., Formation.
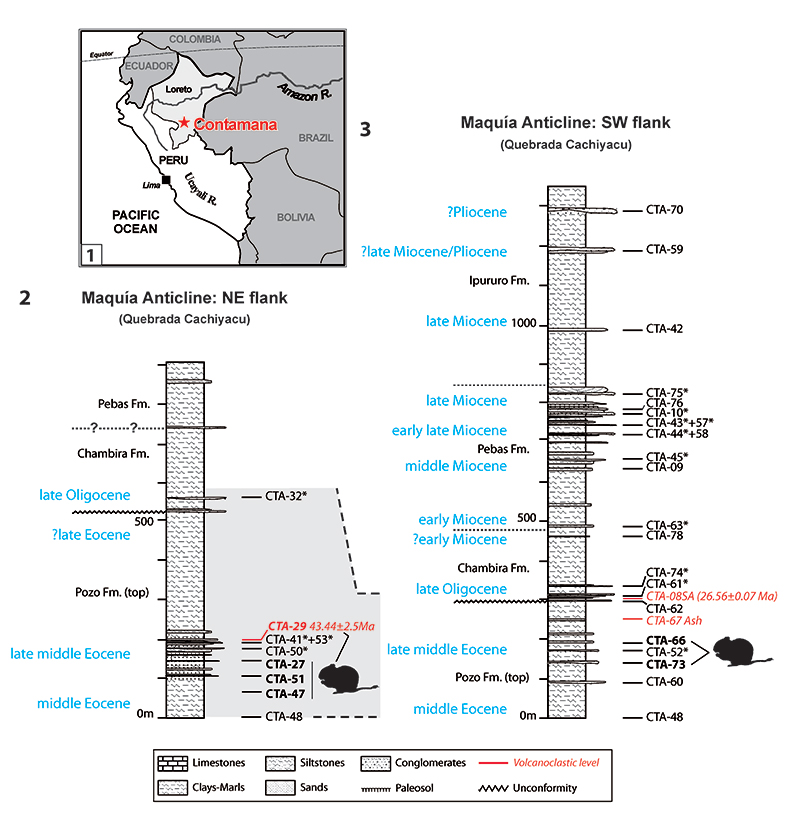
FIGURE 2. Dental nomenclature for upper teeth in occlusal view. 1, upper molar; 2, P4; 3, dP4. 1, paracone; 2, protocone; 3, metacone; 4, hypocone; 5, parastyle; 6, mesostyle; 7, anteroloph; 8, anterior arm of the protocone; 9, posterior arm of the protocone (= lingual protoloph); 10, posterior outgrowth of the protocone; 11, protoloph (= labial protoloph); 12, mure; 13, third transverse crest (= central transverse crest); 14, mesolophule; 15, mesoloph; 16, anterior arm of the hypocone; 17, metaloph; 18, posteroloph; 19, paraflexus; 20, hypoflexus/hypofossette; 21, confluence of the paraflexus with the hypoflexus; 22, mesoflexus/mesofossette; 23, metaflexus; 24, confluence of the metaflexus with the posteroflexus; 25, posteroflexus. Based on observations made on the new material, the dental terminology is modified after Wood and Wilson (1936), Fields (1957), Marivaux et al. (2004, 2017) and Antoine et al. (2012).
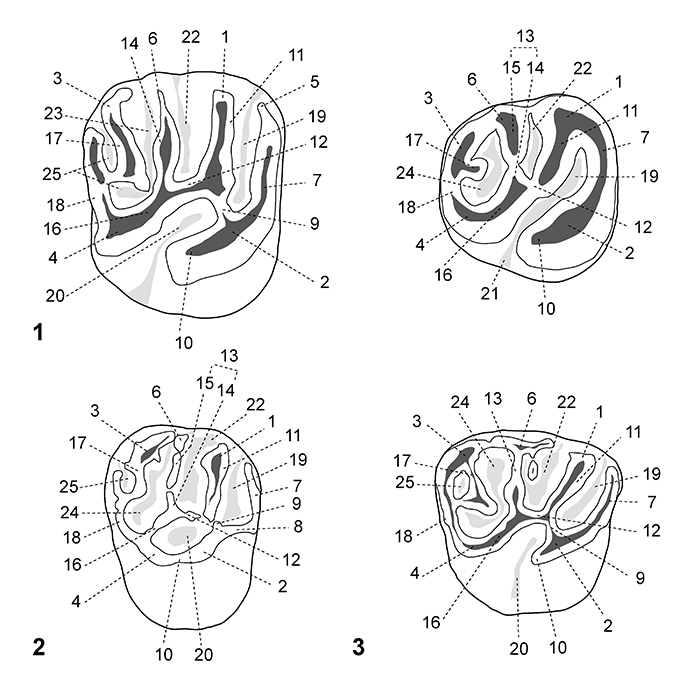
FIGURE 3. Dental nomenclature for lower teeth in occlusal view. 1, lower molar; 2, p4; 3, dp4. 1, protoconid; 2, metaconid; 3, mesoconid; 4, entoconid; 5, hypoconid; 6, mesostylid; 7, metalophulid I; 8, posterior arm of the metaconid; 9, posterior arm of the protoconid; 10, neomesolophid; 11, second transverse cristid; 12, mesolophid; 13, rest of the mesolophid?; 14, ectolophid; 15, mesial ectolophid; 16, distal ectolophid; 17, hypolophid; 18, anterior arm of the entoconid; 19, posterior arm of the entoconid; 20, anterior arm of the hypoconid; 21, posterior arm of the hypoconid; 22, anterior outgrowth of the hypoconid; 23, posterolophid; 24, anteroflexid/anterofossettid; 25, mesoflexid; 26, mesial mesoflexid; 27, distal mesoflexid; 28, confluence of the anteroflexid with the mesoflexid; 29, hypoflexid; 30, metaflexid; 31, confluence of the hypoflexid with the metaflexid. Based on observations made on the new material, the dental terminology is modified after Wood and Wilson (1936), Fields (1957), Marivaux et al. (2004, 2017) and Antoine et al. (2012).
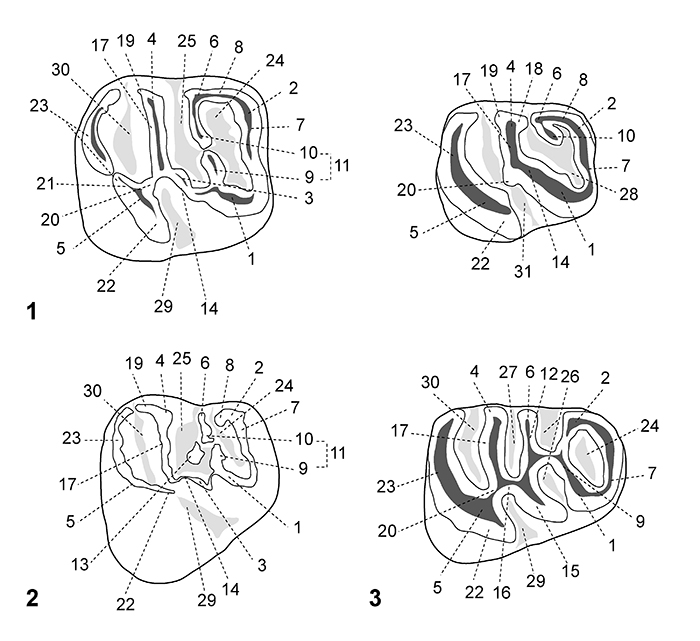
FIGURE 4. Scanning electron microscope images (in occlusal view) of fossil caviomorph teeth from CTA-27. Cachiyacuy contamanensis (1-6), Eobranisamys javierpradoi sp. nov. (7-8), Cachiyacuy kummeli (9-13), Canaanimys maquiensis (13-16) and cf. Eoespina sp. (17). 1, right m3 (MUSM 2713); 2, right m2 (MUSM 1914); 3, right m2 (MUSM 1915); 4, right p4 (MUSM 2678); 5, fragmentary right dp4 (MUSM 2670); 6, left M3 (MUSM 2758); 7, right dP4 (MUSM 2797); 8, fragmentary left M3 (MUSM 2801); 9, left dp4 (MUSM 2762); 10, left p4 (MUSM 2766); 11, right m3 (MUSM 2780, reversed); 12, left M1 (MUSM 2785); 13, left M2 (MUSM 2786); 14, fragmentary right M3 (MUSM 2794); 15, right M3 (MUSM 2793); 16, left M3 (MUSM 2792, reversed); 17, fragmentary left M2 (MUSM 2802). Top scale for 1-13, bottom scale for 14-17.
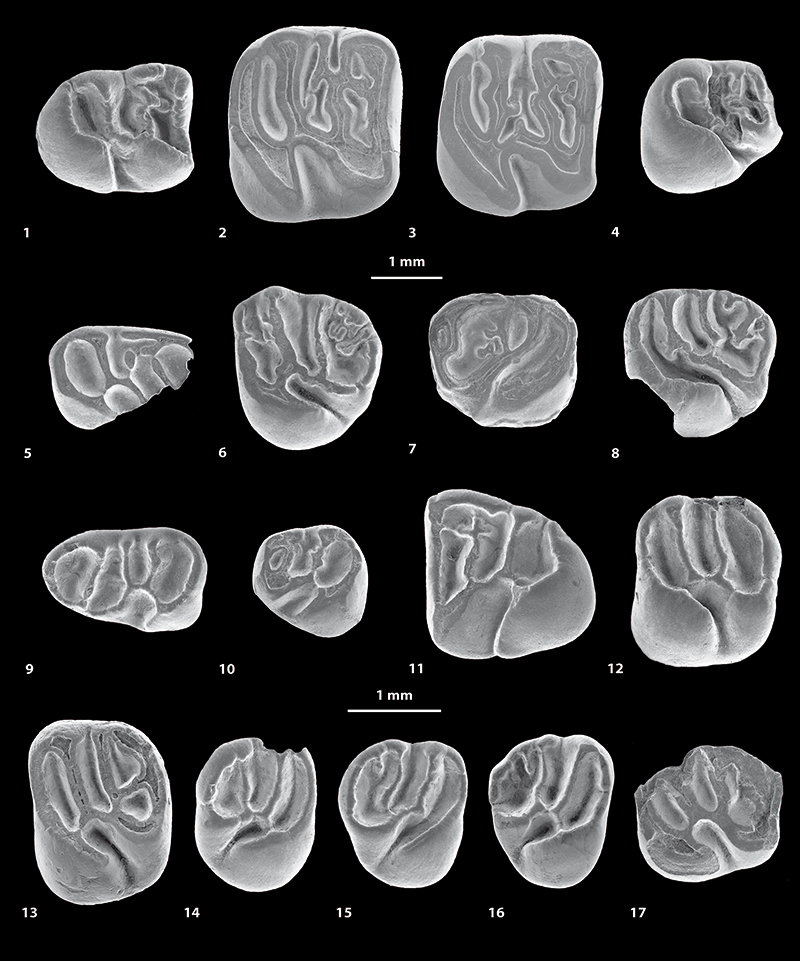
FIGURE 5. Scanning electron microscope images (in occlusal view) of fossil caviomorph teeth from CTA-47 (1-4), CTA-51 (5-12), CTA-73 (13-14) and CTA-66 (15-16). ?Canaanimys sp. (1-2), ?Cachiyacuy kummeli (3), Caviomorpha indet. 1 (4), Cachiyacuy cf. contamanensis 1 (5-7), Caviomorpha indet 2 (8-10), Cachiyacuy cf. kummeli (11), Eoespina sp. (12), Caviomorpha indet 3 (13), Caviomorpha indet 4 (14) and Eobranisamys sp. (15-16). 1, fragmentary right dp4 (MUSM 2645); 2, fragmentary left lower molar (MUSM 2646); 3, fragmentary left dP4 (MUSM 2648); 4, fragmentary right lower molar (MUSM 2647); 5, fragmentary right dp4 (MUSM 2651, reversed); 6, left m1 (MUSM 2652); 7, left m3 (MUSM 2653); 8, fragmentary right lower molar (MUSM 2656); 9, fragmentary left dP4 (MUSM 2657); 10, fragmentary left upper molar (MUSM 2658); 11, fragmentary left M1 (MUSM 2654); 12, right M2 (MUSM 2655); 13, fragmentary right lower molar (MUSM 2659); 14, fragmentary left upper molar (MUSM 2660); 15, fragmentary left upper molar? (MUSM 2841); 16, fragmentary left upper molar (MUSM 2842). Top scale for 1-12, bottom scale for 13-16.
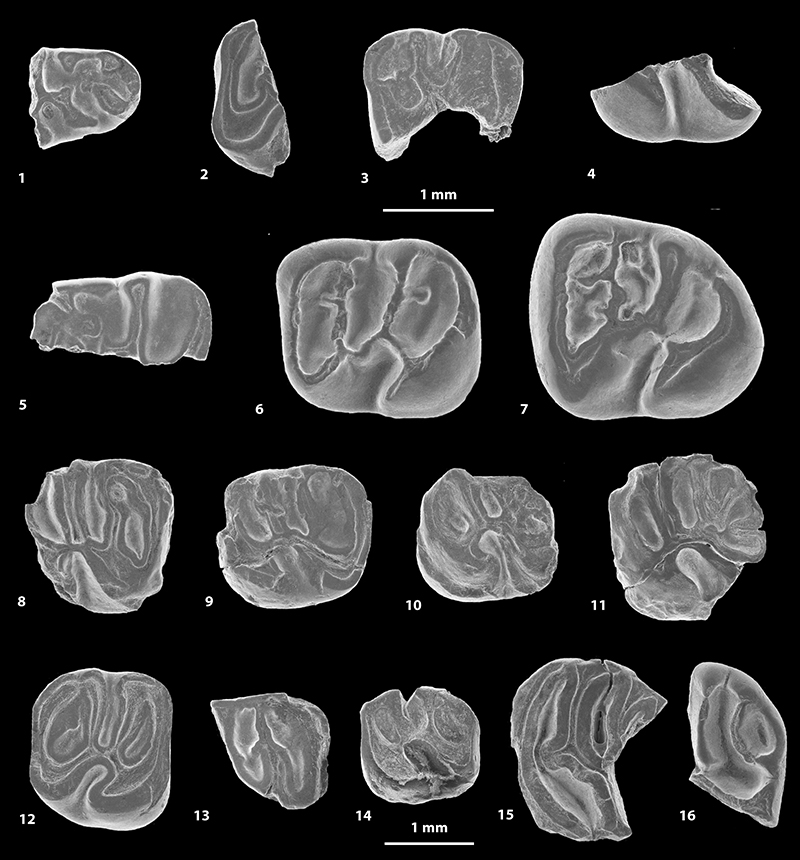
FIGURE 6. Scanning electron microscope images of fossil caviomorph teeth from CTA-29. Pozomys ucayaliensis gen. et sp. nov. (1-6), Cachiyacuy cf. contamanensis 2 (7-12), Caviomorpha indet. 5 (13), Caviomorpha indet. 6 (14) and Cavioidea or Chinchilloidea indet. (15-17). 1, right m1-2, occlusal view (MUSM 2822); 2, left p4, occlusal view (MUSM 2821, reversed); 3, fragmentary right M3, occlusal view (MUSM 2819); 4, right M2, occlusal view (MUSM 2833); 5, right m1-2, labial view (MUSM 2822); 6, right m1-2, lingual view (MUSM 2822); 7, left dp4, occlusal view (MUSM 2825); 8, right m1, occlusal view (MUSM 2827, reversed); 9, right dP4, occlusal view (MUSM 2828, reversed); 10, left M1, occlusal view (MUSM 2831); 11, right M2, occlusal view (MUSM 2563, reversed); 12, fragmentary left M2, occlusal view (MUSM 2832); 13, right dp4, occlusal view (MUSM 2838); 14, fragmentary right upper molar, occlusal view (MUSM 2839); 15, right m2, occlusal view (MUSM 2835); 16, right dp4, occlusal view (MUSM 2834); 17, fragmentary right upper molar, occlusal view (MUSM 2836, reversed).

FIGURE 7. Morphological variation of the p4 (in occlusal view) of Cachiyacuy contamanensis from CTA-27. 1, MUSM 2674; 2, MUSM 2676; 3, MUSM 2677; 4, MUSM 1879; 5, MUSM 2678. Original computerized schemas (1-5) by Myriam Boivin.
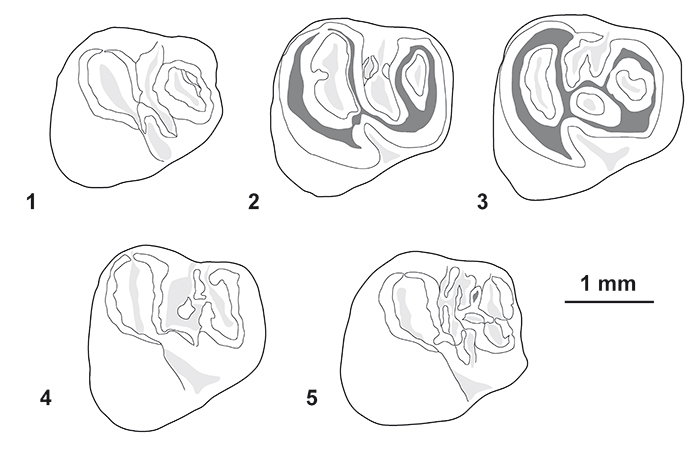
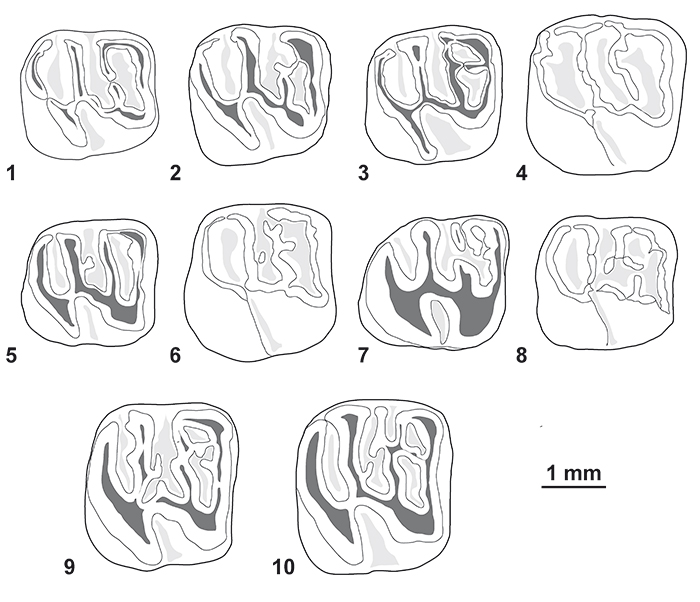
FIGURE 8. Morphological variation of the lower molars (in occlusal view) of Cachiyacuy contamanensis from CTA-27. 1, MUSM 1878; 2, MUSM 2704; 3, MUSM 2684; 4, MUSM 2708; 5, MUSM 2692; 6, MUSM 2701; 7, MUSM 2714; 8, MUSM 2689; 9, MUSM 1915; 10, MUSM 1914. Original computerized schemas (1-10) by Myriam Boivin.
Late middle Eocene caviomorph rodents from Contamana, Peruvian Amazonia
Plain Language Abstract
A recently described caviomorph rodent assemblage from Eocene deposits of Peruvian Amazonia (Contamana, CTA-27) is revised along with the description of five new other assemblages from the same area and stratigraphic interval (CTA-47, CTA-51, CTA-73, CTA-66, and CTA-29). These fossils shed new light on the paleodiversity and early evolutionary history of a pivotal mammalian group during a time window poorly documented paleontologically in tropical-equatorial South America.
Resumen en Español
Roedores caviomorfos del Eoceno medio tardío de Contamana, Amazonía peruana
Los roedores caviomorfos representan uno de los grupos más exitosos de mamíferos placentarios de América del Sur. A pesar de su alta diversidad actual, y también durante el Neógeno y Paleógeno tardío, su historia evolutiva temprana ha permanecido poco conocida durante mucho tiempo. Las recientes expediciones de campo en la Amazonía peruana han proporcionado ejemplares que se cuentan entre los primeros representantes del grupo para depósitos datados del Eoceno medio tardío (Contamana, yacimiento CTA-27, ~ 41 millones de años). Aquí se presenta un análisis exhaustivo del material de roedores de CTA-27 y de nuevas localidades subcoetáneas descubiertas en la misma área y formación geológica (Formación Pozo): CTA-47, CTA-51, CTA-73, CTA-66 y CTA-29. Para estas localidades se ha identificado un total de 20 taxones de roedores, entre los cuales uno procedente de CTA-29 (Pozomys ucayaliensis gen. et sp. nov.) permanece con afinidades suprafamiliares inciertas. Adicionalmente, el material de CTA-27 previamente atribuido a Eobranisamys sp. se asigna aquí a la nueva especie Eobranisamys javierpradoi. En términos de composición taxonómica, las localidades del Eoceno del área de Contamana presentan muchos taxones en común (Cachiyacuy, Canaanimys, Eobranisamys, y Eoespina, o parientes muy cercanos). Estas asociaciones del Eoceno son claramente distintas de las del Oligoceno, la mayoría documentadas en latitudes medias y altas. Por el contrario, comparten algunas afinidades con la localidad de Santa Rosa del Eoceno tardío-Oligoceno más temprano (Amazonía peruana), a partir de cuyos fósiles originalmente se describieron los dos géneros Eobranisamys y Eoespina. Esta similitud faunística podría estar reflejando más bien afinidades biogeográficas que contemporaneidad. Además, el patrón oclusal de algunos molares superiores de Eosallamys de Santa Rosa recuerda los patrones de Cachiyacuy y Canaanimys. Estas asociaciones de caviomorfos de baja latitud proporcionan nuevas perspectivas sobre la historia evolutiva temprana, biogeografía y paleodiversidad del grupo.
Palabras clave: Barrancan; Sudamérica; Sistemática; Bioestratigrafía; nuevo género; nueva especie
Traducción: Enrique Peñalver (Sociedad Española de Paleontología)
Résumé en Français
Rongeurs caviomorphes de l'Eocène moyen terminal de Contamana, Amazonie péruvienne
Une faune de rongeurs caviomorphes provenant de dépôts éocènes d'Amazonie péruvienne et récemment décrite (Contamana, CTA-27) est ici révisée. Cinq nouvelles faunes provenant de la même zone géographique et issues du même intervalle stratigraphique (CTA-47, CTA-51, CTA-73, CTA-66, and CTA-29) sont également décrites. Ces fossiles apportent un nouvel éclairage sur la paléodiversité et l'histoire évolutive précoce d'un groupe de mammifères majeur durant une fenêtre de temps peu documentée paléontologiquement en Amérique de Sud tropicale-équatoriale.
Mots-clés: Barrancien, Amérique du Sud, Systématique, Biostratigraphie, nouveau genre, nouvelle espèce.
Translator: Author
Deutsche Zusammenfassung
Spätmitteleozäne caviomorphe Nagetiere aus Contamana, peruanisches Amzonien
Caviomorphe Nagetiere sind eine der erfolgreichsten Gruppen von Plazentatieren aus Südamerika. Ungeachtet ihrer großen Vielfalt heute, im Neogen und im Paläogen, war ihre frühe Evolutionsgeschichte lange unklar. Auf jüngsten Forschungsreisen im peruanischen Amazonien wurden die frühesten Mitglieder dieser Gruppe in Gesteinen aus dem mittleren Eozän (Contamana, CTA-27 Lokalität, ~41 Ma) gefunden. Hier stellen wir eine umfassende Analyse des Nagetier-Materials aus CTA-27 und neuer zeitgleicher Zwischenschichten aus dem gleichen Gebiet und der gleichen geologischen Formation (Pozo Formation) zur Verfügung: CTA-47, CTA-51, CTA-73, CTA-66 und CTA-29. Insgesamt wurden in diesen Fundstellen 20 Nagetier-Taxa identifiziert, von denen eines aus CTA-29 (Pozomys ucayaliensis gen. et sp. nov.) unklare suprafamiliäre Verwandtschaftsbeziehungen hat. Zusätzlich wird Material aus CTA-27, das zuvor Eobranisamys sp. zugeschrieben wurde, der neuen Art Eobranisamys javierpradoi zugeteilt. Was die Zusammensetzung der Taxa angeht, haben eozäne Fundstellen aus dem Contamara-Gebiet viele Taxa gemeinsam (Cachiyacuy, Canaanimys, Eobranisamys und Eoespina, oder sehr nahe Verwandte). Diese eozänen Fundstellen unterscheiden sich deutlich von denen aus dem Oligozän, hauptsächlich dokumentiert in mittleren und hohen Breitengraden. Demgegenüber teilen sie einige Ähnlichkeiten mit der späteozänen bis früholigozänen Santa Rosa Fundstelle (peruanisches Amazonien) von wo die beiden Gattungen Eobranisamys and Eoespina im Original beschrieben wurden. Diese Faunennähe spiegelt wahrscheinlich eher biogeographische Ähnlichkeiten als Gleichzeitigkeit. Zusätzlich bildet das Okklusionsmuster einiger oberen Molaren von Eosallamys aus Santa Rosa das von Cachiyacuy und Canaanimys ab. Diese caviomorphen Assemblagen aus niedrigen Breiten liefern neue Erkenntnisse über die frühe Evolutionsgeschichte, Biogeographie und Paläodiversität dieser Gruppe.
Schlüsselwörter: Barrancan; Südamerika; Systematik; Biostratigraphie; neue Gattung; neue Art
Translator: Eva Gebauer
Arabic

Translator: Ashraf M.T. Elewa
-
-
-
Review: The Princeton Field Guide to Mesozoic Sea Reptiles
 The Princeton Field Guide to Mesozoic Sea Reptiles
The Princeton Field Guide to Mesozoic Sea ReptilesArticle number: 26.1.1R
April 2023


 Poster Winners 2024
Poster Winners 2024