Article Search
Volume 27.1
January–April 2024
Full table of contents
ISSN: 1094-8074, web version;
1935-3952, print version
Recent Research Articles
See all articles in 27.1 January-April 2024
See all articles in 26.3 September-December 2023
See all articles in 26.2 May-August 2023
See all articles in 26.1 January-April 2023
SUPPLEMENTARY ANIMATION 1. Animation (.mov format; 24 sec) compiled using Avizo software showing the rotation of an isosurface rendering of a Burejospermum seymourense cocoon-wall fragment (NRMS089729) generated from Synchrotron-radiation-based X-Ray tomographic microscopy (SRXTM) at the TOMCAT beamline of the Swiss Light Source at the Paul Scherrer Institute, Switzerland, using a beam energy of 10 kV and a 20× objective. Note the relatively smooth (inner) alytine and filamentous (outer) hapsine with embedded mineral matter. Click on image to see animation.
SUPPLEMENTARY ANIMATION 2. Animation (.mov format; 24 sec) compiled using Avizo software showing the rotation of a volume rendering of a Burejospermum seymourense cocoon wall fragment (NRMS089729) generated from Synchrotron-radiation-based X-ray tomographic microscopy (SRXTM) at the TOMCAT beamline of the Swiss Light Source at the Paul Scherrer Institute, Switzerland, using a beam energy of 10 kV and a 20× objective. Note the irregular distribution of entombed mineral matter (white) and the hollow tube in the lower portion of the wall fragment that may represent the original position of a withdrawn seta. Click on image to see animation.
SUPPLEMENTARY ANIMATION 3. Composite orthoslice animation (.mov format; 24 sec) of 1461 steps through a Burejospermum seymourense cocoon wall fragment (NRMS089729) using Avizo software. Orthoslice data obtained via synchrotron-radiation-based X-ray tomographic microscopy at the TOMCAT beamline of the Swiss Light Source at the Paul Scherrer Institute, Switzerland, using a beam energy of 10 kV and a 20× objective. Note the similar composition of the dense (inner) alytine and filamentous (outer) hapsine; the hollow pseudo-segmented tube in the lower left portion parallel to the cocoon surface may represent the original position of a withdrawn seta; some smaller cavities in the alytine represent moulds of encased spermatozoa and micro-organisms. Click on image to see animation.
SUPPLEMENTARY ANIMATION 4. Animation of a three-dimensional volume rendering (.mov format; 24 sec) of a Burejospermum seymourense cocoon wall fragment (NRMS089729) compiled from segmented computed tomographic data to illustrate an enclosed tubular structure (possible setal anchoring trace) highlighted in yellow that is flared at one end and tapers distally. Click on image to see animation.
FIGURE 1. Diagrammatic sections through the walls of fossil clitellate annelid cocoons showing the general structure and representation of layers. 1, Burejospermum. 2, Pilothylakos. 3, Dictyothylakos. 4, Pegmatothylakos.
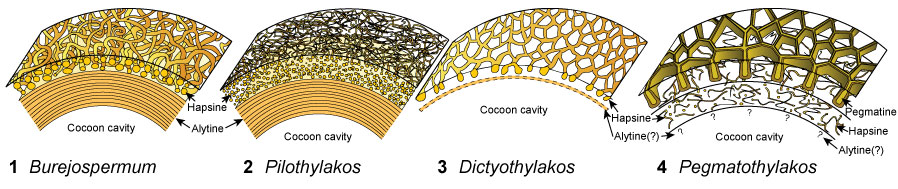
FIGURE 2. Locality maps and stratigraphic column of the studied succession. 1, Map of Antarctica showing the location of the study area. 2, Map of the northern Antarctic Peninsula showing the location of Seymour Island. 3, Geological sketch map of Seymour Island, Antarctic Peninsula, showing the positions of fossil localities IAA 1/90 and IAA 2/95. 4, Stratigraphic column of the La Meseta Formation on Seymour Island (from Reguero et al., 2013). Strontium date values from Dingle and Lavelle (1998), Dutton et al. (2002), Reguero et al. (2002), and Ivany et al. (2008).
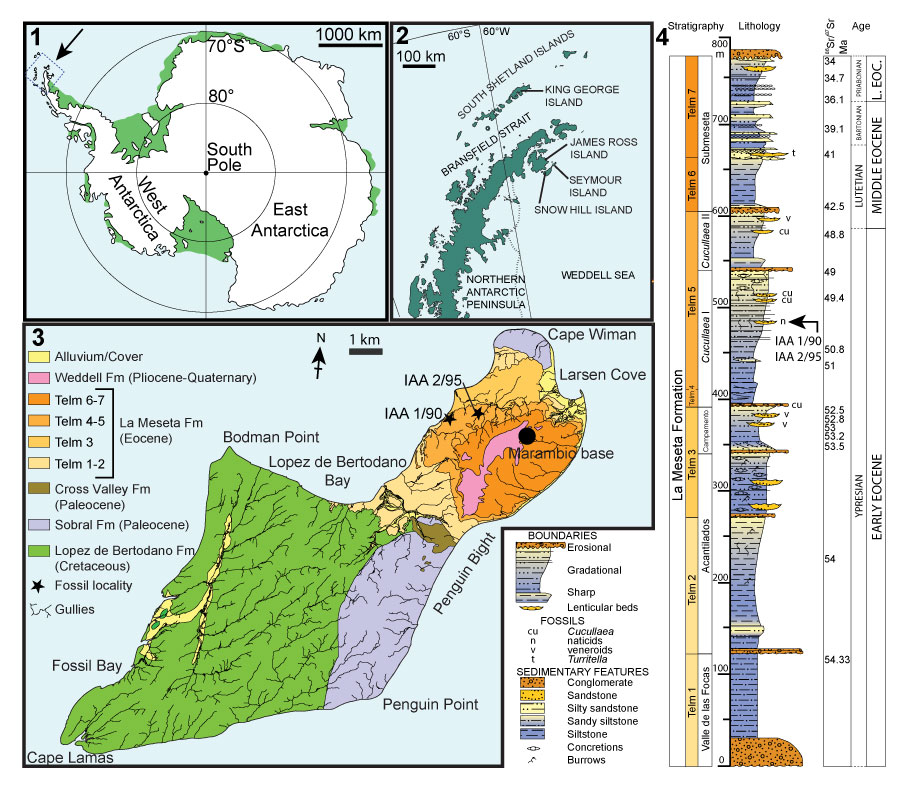
FIGURE 3. Photographs of sampling localities. 1, Aerial view of ‘Ungulate site’ (IAA 1/90, 64°14'04.67"S, 56°39'56.38"W) with ‘ Natica horizon’ marked by arrow. 2, Detail of section between Cucullaea 1 bed (below quad bike) and ‘ Natica horizon’ at IAA 1/90 (arrowed) showing dominance of poorly consolidated sandstones, mudstones and shell-rich conglomerates. 3, Panoramic view of ‘Marsupial site’ (IAA 2/95: 64°13'58"S, 56°39'06"W) with ‘ Natica horizon’ exposed along foreground ridge, and Cockburn Island in background. 4, ‘ Natica horizon’ near site IAA 2/95 showing incised base and lenticular character of the bed. Photographs by T. Mörs (1, 2), F. Degrange (3), and J. Hagström (4).

FIGURE 4. Light micrographs of early Eocene clitellate annelid cocoons from Seymour Island. 1-3, Burejospermum seymourense sp. nov. (NRMS089727). 4-6, Burejospermum punctatum sp. nov. (NRMS089728). 7-9, Pegmatothylakos manumii sp. nov. (NRMS089730). 1, 4, 7, Lateral views of cocoons (posterior to left); g = equatorial girdle. 2, 5, 8, Views of posterior end of cocoons showing opening left by detachment of operculum. 3, 6, 9, Enlargements of cocoon wall exterior showing pale felt-like hapsine covering in B. seymourense (3), relatively smooth surface in B. punctatum ( 6) and reticulate pegmatine of P. manumii (9). Scale bars represent 1 mm in 1, 2, 4, 5, 7, 8; 100 µm in 3, 6, 9.

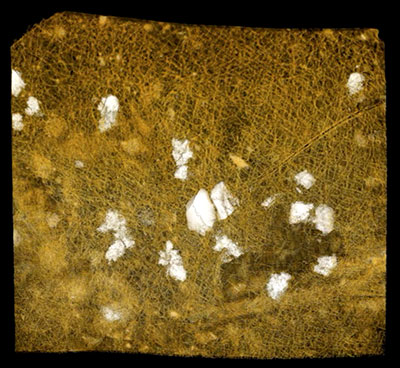
FIGURE 5. Scanning electron micrographs of Burejospermum seymourense sp. nov. (NRMS089727). 1, Cocoon in lateral view. 2, Details of typical hapsine threads. 3, Detail of dense hapsine threads over girdle region. 4, Elliptical secreted plate-like structures on exterior of hapsine. 5, Broken cocoon wall in cross-section showing solid alytine (lower) and thread-like hapsine (upper). 6, Detail of plate-like secretion on exterior of hapsine. Scale bars represent 1 mm in 1; 100 µm in 2; 50 µm in 3, 4; 25 µm in 5, 6.
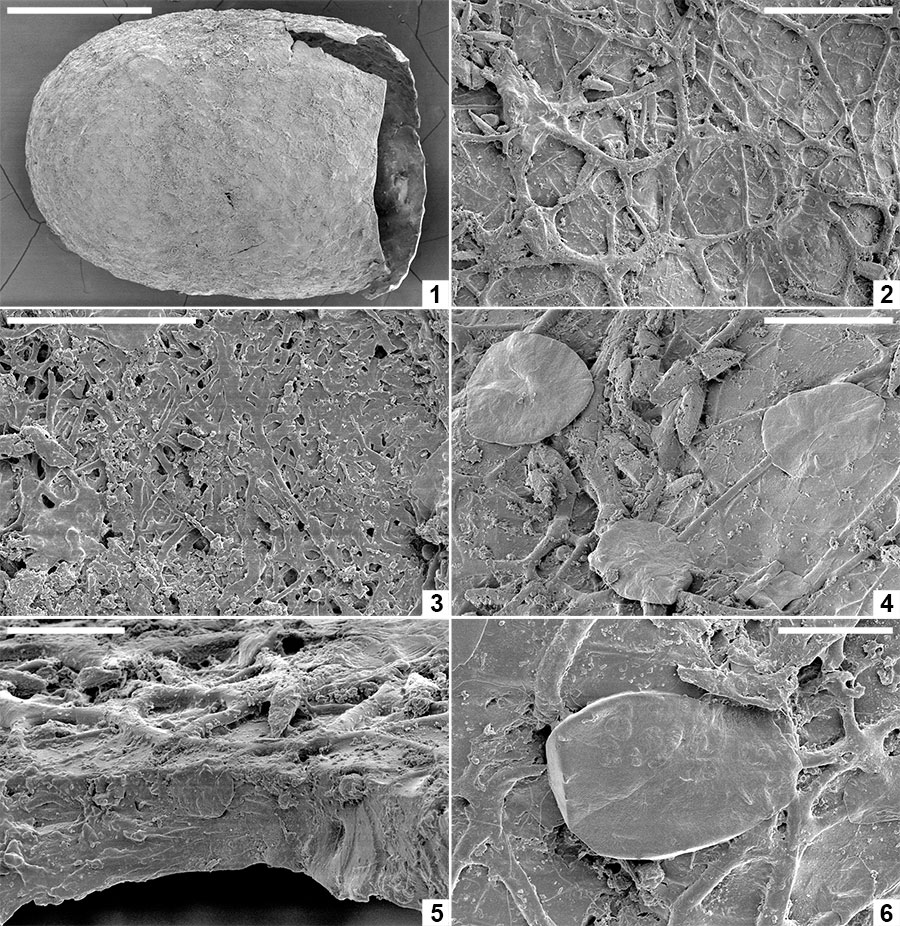
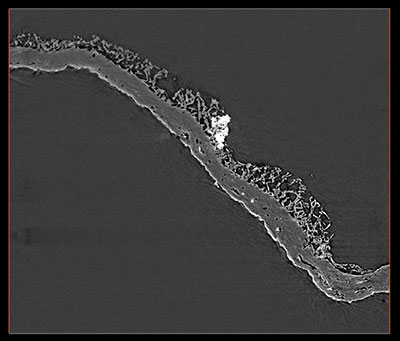
FIGURE 6. Computed tomographic images of a Burejospermum seymourense cocoon wall (NRMS089729) generated from Synchrotron-radiation-based X-Ray tomographic microscopy (SRXTM) at the TOMCAT beamline of the Swiss Light Source at the Paul Scherrer Institute, Switzerland. 1, Isosurface rendering of cocoon’s external surface showing mineral grains embedded in hapsine. 2, Isosurface rendering of cocoon’s internal surface showing creasing caused by alytine threads. Arrow indicates the position of a hollow tube within the alytine. 3, 4, Longitudinal orthoslice sections, at different focal planes, of a tube within the alytine showing pseudosegmentation. 5-8, Translucent volume rendering of a tube within the alytine imaged at different focal planes and magnifications to highlight the transverse to chevron-shaped thread arrangement lining the tube wall. 9, Isosurface rendering of hapsine outer surface showing details of the complex arrangement of hirudoin threads. 10, Transverse orthoslice section of the cocoon wall showing the near-solid alytine layer (bottom) and mesh-like hapsine layer (top). 11, Isosurface rendering of interior alytine wall showing an embedded spermatozoal nuclear region. 12, Colour-inverted rendering of Figure 6.11. 13, Volume rendering of the cocoon wall showing numerous mineral (white-yellow) inclusions. 14, Oblique translucent volume rendering of the cocoon wall showing the distribution of mineral grains mainly in the outer hapsine layer. Scale bars represent 100 µm in 1, 2, 10, 13, 14; 50 µm in 9; 25 µm in 3-7; 10 µm in 8; 5 µm in 11, 12.
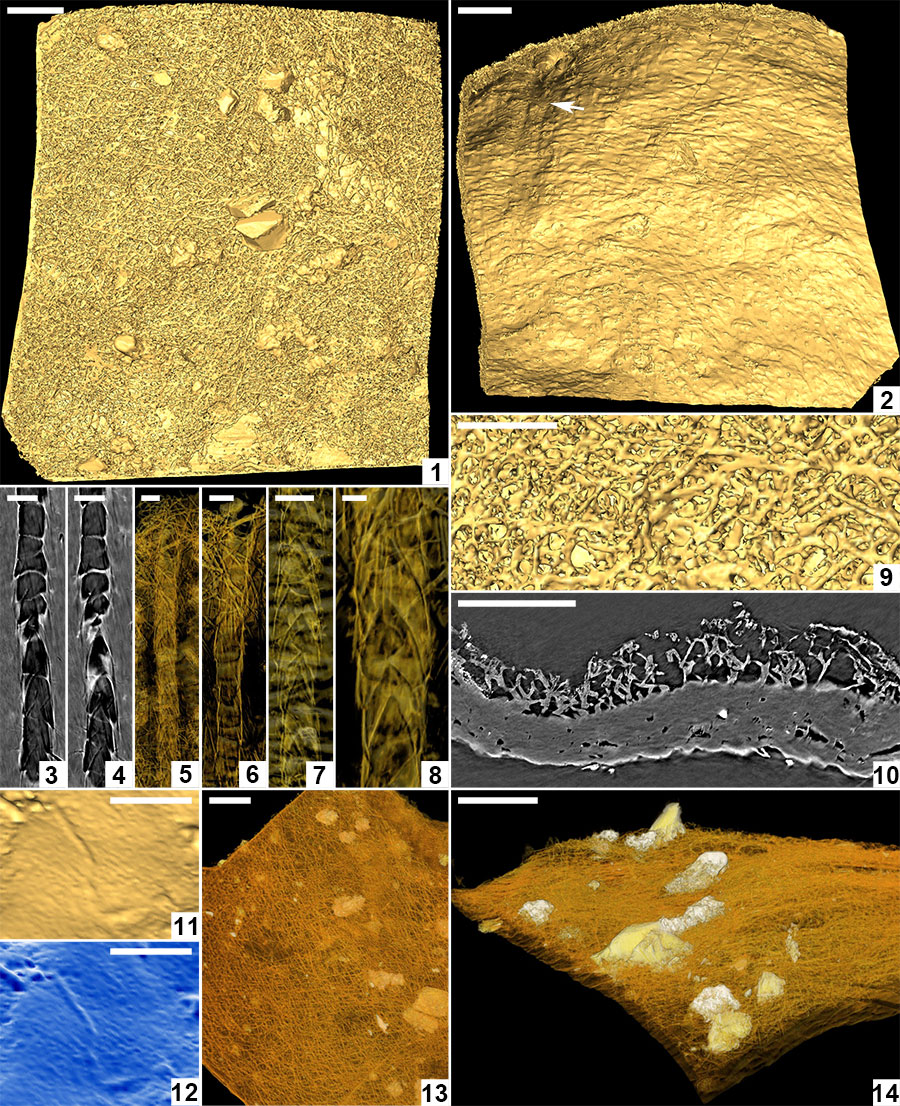
FIGURE 7. Scanning electron micrographs of Burejospermum punctatum sp. nov. (NRMS089728). 1, Cocoon in lateral view. 2, Details of sparse hapsine threads embedded in alytine surface. 3, Detail of typical cocoon surface showing pitting. 4, Detail of girdle region showing granular microtexture and a single surface pit. 5, Detail of outer alytine surface showing cleft and fold structures (arrowed). 6, Cross-section of cocoon wall showing laminated architecture. Scale bars represent 1 mm in 1; 100 µm in 3, 5; 10 µm in 2, 4, 6.
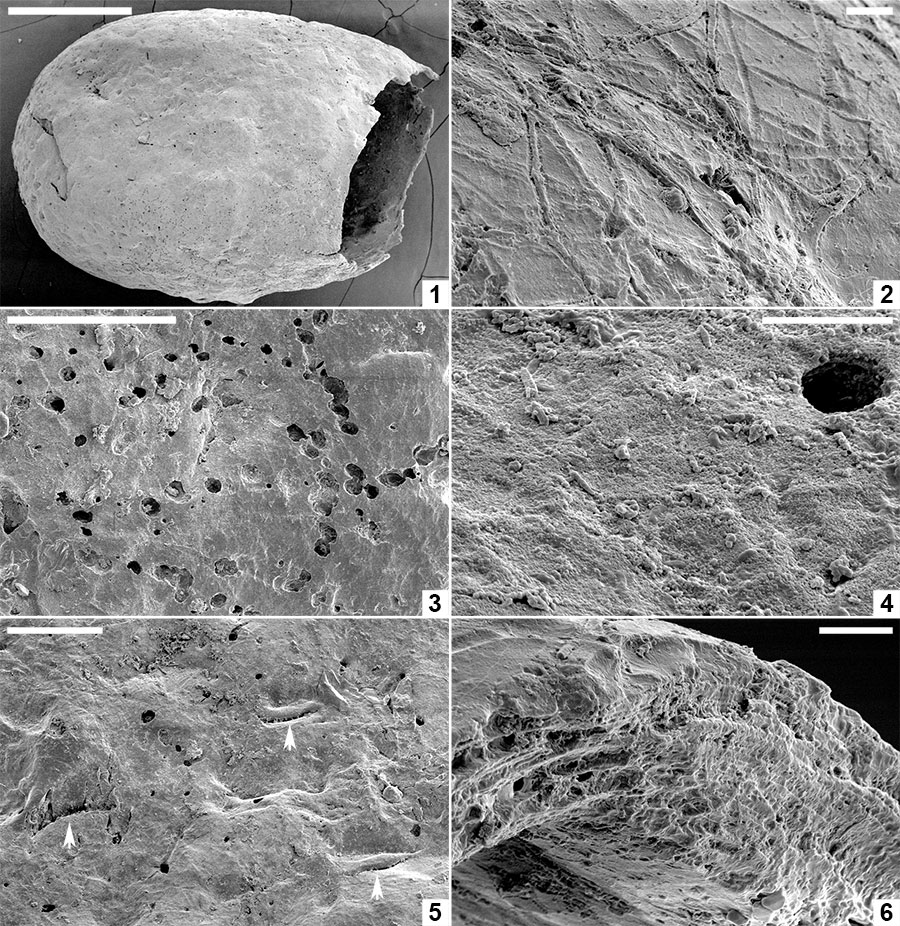
FIGURE 8. Scanning electron micrographs of Pegmatothylakos manumii sp. nov. (NRMS089730). 1, Cocoon in lateral view. 2, Enlargement of robust reticulate pegmatine covering thread-like hapsine. 3, Enlargement of areole in pegmatine reticulum showing underlying mesh-like hapsine. 4, Enlargement of girders forming pegmatine showing hackly inner zone and smoothly welded threads lining girder margins. 5, Detail of hackly and smooth threads in pegmatine girders. 6, Broken pegmatine girder showing irregular internal structure. 7, Enlargement of interior surface of pegmatine girder showing smooth threads with irregular branching and orientation. 8, Enlargement of hapsine threads. Scale bars represent 1 mm in 1; 250 μm in 2; 100 μm in 3, 4; 25 μm in 5-8.
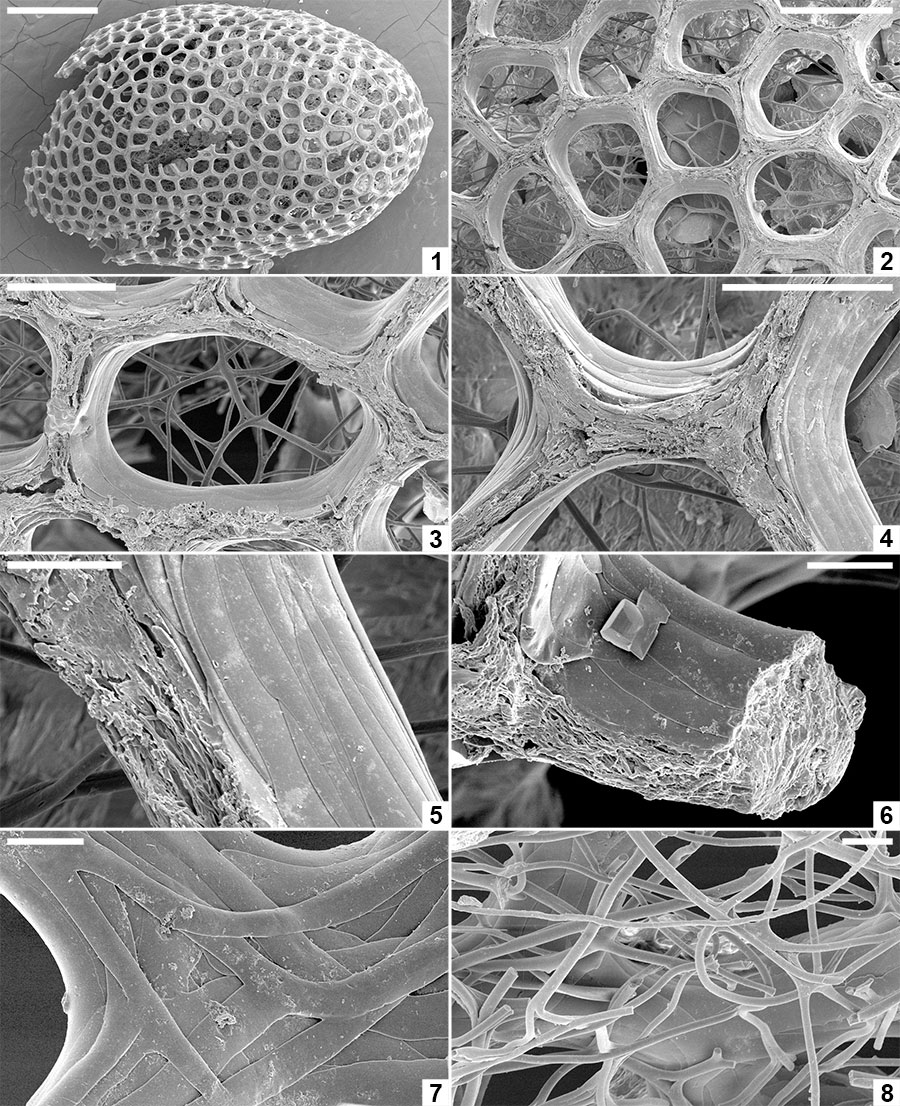
FIGURE 9. Spermatozoa and bacteria preserved embedded in the innermost layer of the alytine wall of Burejospermum seymourense sp. nov., NRMS089729 ( 1-12); Burejospermum punctatum sp. nov., NRMS089728 ( 13); and Pegmatothylakos manumii sp. nov., NRMS089730 (14). 1, Overview of cocoon inner surface showing a range of elongate spermatozoa and elliptical-reniform bacteria. 2, Spermatozoon acrosome (white arrow) with swollen tip (probably containing the acrosome vesicle) and apical button (black arrow). 3, Enlargement of a spirally organized detached acrosome. 4, Thin sheet-like innermost layer of alytine draped over mineral inclusion (top) and entombing numerous elliptical bacteria. 5, Several spermatozoal nuclear regions, one with an attached whip-like flagellum (white arrow). A pair of dimpled elliptical bacteria are also present in the upper left (black arrow). 6, Enlargement of a portion of a spermatozoon flagellum showing slightly beaded structure. 7, Enlargement of the nuclear and mitochondrial region of a spermatozoon showing granular ornamentation and swellings in the anterior portion of the attached flagellum. Reniform bacterium also present in upper right (white arrow). 8, Several spermatozoal flagella showing indistinct spiral architecture. 9, Nuclear (white arrow), mitochondrial (black arrow) and anterior flagellar (hollow arrow) regions of a spermatozoon. 10, Enlargement of a flagellum showing beaded structure that probably reflects helical architecture. 11, Enlargement of Figure 9.9 showing details of nuclear (n), mitochondrial (m) and anterior flagellar (f) regions. 12, Overlapping elliptical bacterium (b) and flagellate spermatozoon. 13, Elongate coiled acrosome (a) and slender flagellum (f) of separate spermatozoa. 14, Nuclear (n) and flagellar (f) regions of a spermatozoon. Scale bars represent 10 µm in 1, 4, 5, 13, 14; 2 µm in 2, 3, 7-9; 1 µm in 6, 10-12.

FIGURE 10. Organic inclusions embedded in the innermost layer of the alytine wall of Burejospermum seymourense sp. nov. (NRMS089729) b = bacteria in a chain, n = detached spermatozoal nuclear regions, a = detached spermatozoal coiled acrosome regions, u = unidentified vermiform bodies. Scale bar represents 5 µm.
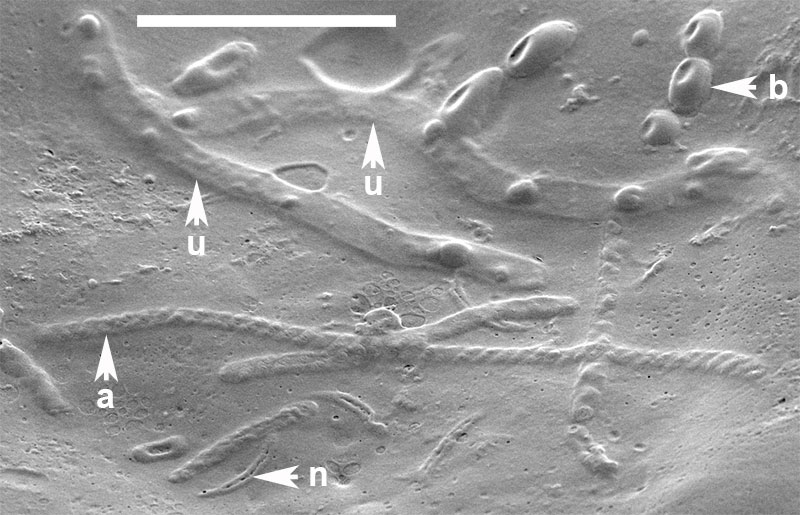
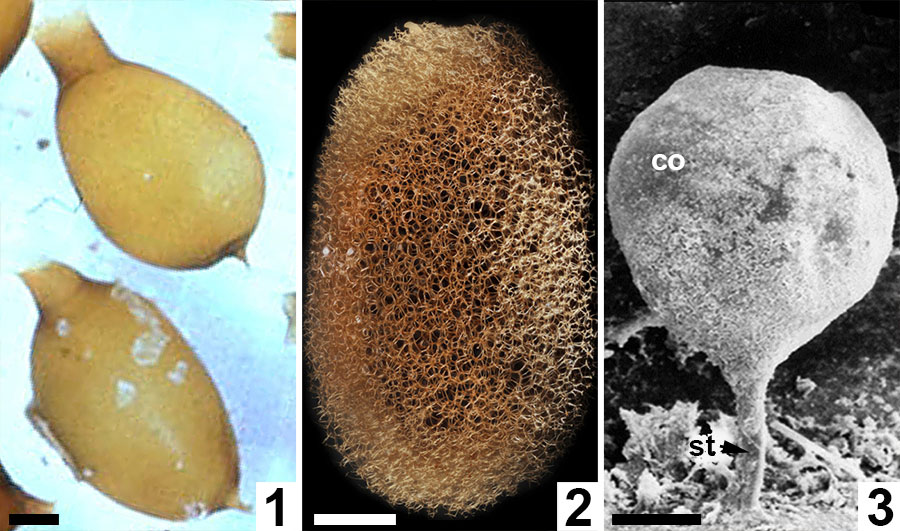
FIGURE 11. Comparative morphologies of extant clitellate annelid cocoons. 1, Light micrograph of two earthworm (Oligochaeta: Lumbricus terrestris) cocoons with prominent polar extensions and walls constructed almost entirely of the solid alytine layer (from Clive A. Edwards, The Ohio State University, Columbus, USA, Public domain via Wikimedia Commons). 2, Light micrograph of a true leech (Hirudinea: ? Macrobdella decora) cocoon with a wall dominated by a Dictyothylakos -like meshed hapsine layer (courtesy of Macroscopic Solutions, LLC. www.macroscopicsolutions.com). 3, Scanning electron micrograph of a crayfish worm (Branchiobdellida: Cambarincola macrocephelus or C. fallax) cocoon with a thin scabrate hapsine layer overlying solid alytine (courtesy of Dr Naglaa M SH Geasa, Tanta University, Egypt; after Geasa, 2014, figure 6G); co = cocoon body lacking an operculum; st = attachment stalk. Scale bars represent 1 mm in 1, 2; 50 µm in 3.
 Stephen McLoughlin, Department of Palaeobiology, Swedish Museum of Natural History, P.O. Box 50007, S-104 05 Stockholm, Sweden. steve.mcloughlin@nrm.se
Stephen McLoughlin, Department of Palaeobiology, Swedish Museum of Natural History, P.O. Box 50007, S-104 05 Stockholm, Sweden. steve.mcloughlin@nrm.se
Stephen McLoughlin is a Professor of Palaeobotany in the Department of Palaeobiology at the Swedish Museum of Natural History. He has previously held research and teaching positions in palaeobotany and sedimentology at various institutions in Australia. His main research interests are the late Palaeozoic and Mesozoic floras of the Southern Hemisphere, particularly the systematics, evolution and palaeobiology of glossopterids and seed-ferns, and plant-animal interactions.

 Benjamin Bomfleur, Department of Palaeobiology, Swedish Museum of Natural History, P.O. Box 50007, S-104 05 Stockholm, Sweden. Benjamin.Bomfleur@nrm.se
Benjamin Bomfleur, Department of Palaeobiology, Swedish Museum of Natural History, P.O. Box 50007, S-104 05 Stockholm, Sweden. Benjamin.Bomfleur@nrm.se
Benjamin Bomfleur is a researcher in Palaeobiology at the Swedish Museum of Natural History in Stockholm, Sweden. His research focus lies in studying the biology, ecology, and evolution of Mesozoic plants and vegetation, with special emphasis on Antarctica and past Polar ecosystems. However, his general interest in anything unusual has led him to work on a wide variety of subjects and fossil curiosities, including Jurassic fern chromosomes, a fossilized "bell-animal", and 50-million-year-old leech sperm.

 Thomas Mörs. Department of Palaeobiology, Swedish Museum of Natural History, P.O. Box 50007, S-104 05 Stockholm, Sweden. Thomas.Mors@nrm.se
Thomas Mörs. Department of Palaeobiology, Swedish Museum of Natural History, P.O. Box 50007, S-104 05 Stockholm, Sweden. Thomas.Mors@nrm.se
Thomas Mörs is a Researcher and Senior curator at the Swedish Museum of Natural History in Stockholm. At the Department of Palaeobiology he is responsible for the curation of fossil vertebrates. Additionally, he is Adjunct professor for Historical Geology and Palaeontology at the University of Uppsala. His main research interests are the evolution, systematics, biogeography and palaeoecology of Cenozoic rodents of the Northern Hemisphere. His interest in Polar Palaeontology has resulted in six expeditions to Svalbard and Antarctica.

 Marcelo Reguero. División Paleontología de Vertebrados, Museo de La Plata, Paseo del Bosque s/n, B1900FWA La Plata, Argentina. mreguero@dna.gov.ar
Marcelo Reguero. División Paleontología de Vertebrados, Museo de La Plata, Paseo del Bosque s/n, B1900FWA La Plata, Argentina. mreguero@dna.gov.ar
Marcelo A. Reguero is a Curator in the Division of Vertebrate Palaeontology at the Natural History Museum in La Plata. He is a researcher at the Argentine National Council of Science and Technology (CONICET). His main research interests are the evolution, systematics, palaeoecology and biogeography of Cenozoic mammals of South America and Antarctica. As a scientific coordinator at the Argentine Antarctic Institute (IAA) he has, for many years, organized expeditions to the Antarctic Peninsula with a focus on vertebrate palaeontology, and has been involved in more than 35 campaigns in Antarctica.

Fossil clitellate annelid cocoons and their microbiological inclusions from the Eocene of Seymour Island, Antarctic
Plain Language Abstract
Three new types of fossil cocoons belonging to clitellate annelids (the group including crayfish worms, leeches and earthworms) are described from 50-million-year-old sedimentary rocks on Seymour Island, Antarctica. The cocoons were probably produced by freshwater animals but were washed into, and buried within, nearshore marine sediments. These cocoons, composed of robust protein threads arranged in various architectures, provide an opportunity to investigate the evolutionary history of clitellate annelids—a group that otherwise has a meagre body-fossil record. Moreover, a range of fossil micro-organisms and the oldest preserved examples of animal sperm cells are fossilized as moulds within the hardened walls of the cocoons in a manner similar to entombment in amber. Thus, fossil cocoons potentially provide a window into the evolutionary history of associated soil and freshwater micro-organisms. They also may enhance palaeoenvironmental interpretations of sediments, advance correlations between continental and shallow marine strata, and improve understanding of the evolution of clitellate annelids.
Resumen en Español
Capullos fósiles de anélidos clitelados y sus inclusiones microbiológicas del Eoceno de la Isla Seymour, la Antártida
Los anélidos clitelados tienen un registro escaso de fósiles corporales, pero secretan capullos proteínicos para la protección de los huevos, los cuales, después del endurecimiento, tienen un cierto potencial de fosilización y pueden constituir una fuente no explorada para la evaluación de la historia evolutiva de este grupo. Se describen tres especies de capullos de clitelados (a saber, Burejospermum seymourense sp. nov., B. punctatum sp. nov. y Pegmatothylakos manumii gen. et sp. nov.) del Eoceno inferior de la Formación La Meseta, Isla Seymour, la Antártida. Los capullos probablemente provienen de medios continentales y fueron transportados a ambientes marinos costeros o estuarinos. Los capullos proporcionan la primera evidencia de relaciones de comensalismo o parasitismo en los ecosistemas continentales del Eoceno de la Antártida. Por otra parte, dentro de las paredes consolidadas de los capullos se encuentran en forma de moldes numerosos microorganismos y los ejemplos fosilizados más antiguos de espermatozoides animales. Los capullos fósiles de anélidos potencialmente ofrecen una mejor interpretación paleoambiental de los sedimentos, la correlación entre los estratos continentales y los marinos de ambiente somero, y una mejor comprensión del desarrollo de los rasgos reproductivos de los anélidos clitelados y la historia evolutiva de microorganismos de cuerpo blando en general.
Palabras clave: Branchiobdellida, nuevo género, nuevas especies, espermatozoides, Formación La Meseta, Península Antártica
Traducción: Enrique Peñalver (Sociedad Española de Paleontología)
Résumé en Français
Des cocons d'annélides clitellates fossiles de l'Éocène de l'île Seymour, Antarctique, et leurs inclusions microbiologiques
Le registre fossile du corps des annélides clitellates est pauvre mais les cocons protéiques qu'ils secrètent pour protéger leurs œufs sont facilement fossilisés après durcissement et offrent une ressource largement inexploitée pour évaluer l'histoire évolutive de ce groupe. Nous décrivons ici trois espèces de cocons de clitellates (Burejospermum seymourense sp. nov., B. punctatum sp. nov., et Pegmatothylakos manumii gen. et sp. nov.) provenant de l'Éocène inférieur de la formation de La Meseta, île Seymour, Antarctique. Les cocons dérivent probablement de milieux continentaux et ont été transportés vers des environnements marins côtiers à estuariens au sein desquels ils ont été préservés. Les cocons constituent les premières preuves de relations commensales ou parasitaires dans les écosystèmes continentaux d'Antarctique à l'Éocène. De plus, de nombreux micro-organismes et les plus anciens exemples de spermatozoïdes animaux fossilisés sont préservés sous forme de moules au sein des murs consolidés des cocons. Les cocons d'annélides ont le potentiel pour améliorer les interprétations paléoenvironnementales des sédiments et les corrélations entre les strates continentales et marines de faible profondeur, et pour mieux comprendre le développement des traits reproductifs des annélides clitellates et, de manière plus générale, l'histoire évolutive des micro-organismes à corps mou.
Mots-clés : branchiobdellidés ; nouveau genre ; nouvelle espèce : spermatozoïdes ; formation de La Meseta ; péninsule Antarctique
Translator: Antoine Souron
Deutsche Zusammenfassung
Fossile clitellate Annelidenkokons und deren mikrobiologische Einschlüsse aus dem Eozän von Seymour Island, Antarktika
Clitellate Anneliden haben einen spärlichen Nachweis an Körperfossilien. Sie scheiden jedoch eiweißhaltige Kokons zum Schutz ihrer Eier ab, die nach der Aushärtung gut fossilisieren und eine noch weitgehend ungenutzte Quelle zur Beurteilung der Evolutionsgeschichte dieser Gruppe sind. Wir beschreiben drei Arten clitellater Kokons (Burejospermum seymourense sp. nov., B. punctatum sp. nov und Pegmatothylakos manumii gen. et sp. nov.) aus dem unteren Eozän der La Meseta Formation, Seymour Island, Antarktika. Die Kokons entstanden vermutlich auf dem Kontinent und wurden zu küstennahen Meeres- oder Mündungsgebieten transportiert, wo sie auch konserviert wurden. Die Kokons sind der erste Nachweis einer kommensalen oder parasitischen Beziehung in einem eozänen kontinentalen Ökosystem von Antarktika. Darüber hinaus sind zahleiche Mikroorganismen und die ältesten fossilen Beispiele von tierischen Spermatozoa innerhalb der verfestigten Kokonwände erhalten. Fossile Annelidenkokons bieten die Möglichkeit einer erweiterten Interpretation der Paläoumwelt durch Sedimente, der Korrelation zwischen kontinentalen und flachmarinen Schichten und eine bessere Kenntnis über die Entwicklung der Reproduktionsmerkmale von clitellaten Anneliden und der Evolutionsgeschichte von Weichkörper-Mikroorganismen im Allgemeinen.
Schlüsselwörter: Branchiobdelliden, neue Gattung, neue Art, Spermatozoa, La Meseta Formation, antarktische Halbinsel
Translator: Eva Gebauer
Arabic

Translator: Ashraf M.T. Elewa
-
-
-
Review: The Princeton Field Guide to Mesozoic Sea Reptiles
 The Princeton Field Guide to Mesozoic Sea Reptiles
The Princeton Field Guide to Mesozoic Sea ReptilesArticle number: 26.1.1R
April 2023




 Poster Winners 2024
Poster Winners 2024