SYSTEMATIC PALEONTOLOGY
(continued)
Caenophidia Hoffstetter, 1939
Acrochordus
Hornstedt, 1787
Acrochordus dehmi
Hoffstetter, 1964
Figure 4,
Figure 5
Referred specimens. 1289 vertebrae and
ribs representing all regions of the axial skeleton.
Localities and ages. Lower and middle
Siwalik Group of the Potwar Plateau, Pakistan, as well as middle Siwalik Group
of Nepal and middle-upper Siwalik Group of Jammu, India (West et al. 1991;
Rage
et al. 2001).
Revised diagnosis. Large snake assigned
to the genus Acrochordus based on the following characters (Hoffstetter, 1964;
Hoffstetter and Gayrard 1965;
McDowell 1979): presence of parazygosphenal
foramina, accessory processes consisting of vertically oriented blades with
convex lateral margins, absence of tuberae costae on ribs, presence of small
pterapophyses, synapophyses low-slung and ventrally elongate. Differs from all
other members of the genus in larger size, possession of lymphapophyseal
foramen, and tall neural spines with straight dorsal margins.
Description. Acrochordus dehmi is
represented by vertebrae from all regions of the column and by fragmentary ribs.
No identifiable cranial elements have been recovered from the Pakistan Siwalik
Group, but a distal quadrate was described from Nepal (West et al. 1991). Precloacal vertebrae from all post-embryonic ontogenetic stages were recovered
from Pakistan. This description augments that of
Hoffstetter (1964) and
describes cloacal, postcloacal, and costal morphology, and subadult ontogenetic
stages.
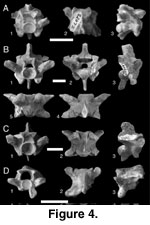 Precloacal vertebrae (Figure 4). In
anterior view, the cotyle is approximately circular in outline. Two to three
paracotylar foramina are present on either side of the cotyle. Small, paired
ventrolateral cotylar processes are present below the ventral margin of the
cotyle, forming a thin paralymphatic channel in most specimens. In posterior
precloacal vertebrae, the ventrolateral processes are greatly reduced, and the
paralymphatic channels extend along the medial surface of the synapophysis and
extend onto the ventrolateral margins of the cotyle (Figure 4C.1, 2). The prezygapophyses are robust and diverge from the body of the element at a high
angle. The prezygapophyseal accessory processes are strongly rounded in outline
and slightly discontinuous with the ventral angle of the prezygapophyses. The
synapophyses are low slung, with anteroposteriorly wide and anteriorly angled
parapophyses. The neural canal is relatively smaller than in more derived
snakes, with smoothly curved lateral and dorsal margins. The zygosphene is
robust and dorsoventrally tall. Parazygosphenal foramina are present at the
bases of the zygosphenal articular facets. Small pterapophyses are present on
the dorsal margin of the postzygapophyses (Figure
4A, B).
Precloacal vertebrae (Figure 4). In
anterior view, the cotyle is approximately circular in outline. Two to three
paracotylar foramina are present on either side of the cotyle. Small, paired
ventrolateral cotylar processes are present below the ventral margin of the
cotyle, forming a thin paralymphatic channel in most specimens. In posterior
precloacal vertebrae, the ventrolateral processes are greatly reduced, and the
paralymphatic channels extend along the medial surface of the synapophysis and
extend onto the ventrolateral margins of the cotyle (Figure 4C.1, 2). The prezygapophyses are robust and diverge from the body of the element at a high
angle. The prezygapophyseal accessory processes are strongly rounded in outline
and slightly discontinuous with the ventral angle of the prezygapophyses. The
synapophyses are low slung, with anteroposteriorly wide and anteriorly angled
parapophyses. The neural canal is relatively smaller than in more derived
snakes, with smoothly curved lateral and dorsal margins. The zygosphene is
robust and dorsoventrally tall. Parazygosphenal foramina are present at the
bases of the zygosphenal articular facets. Small pterapophyses are present on
the dorsal margin of the postzygapophyses (Figure
4A, B).
In
posterior view, the zygantrum is wide with thick margins and divided by the
posterior median notch to the base of the neural spine. Within the zygosphene,
large, paired endozygantral foramina are located at the ventromedial surfaces of
the articular facets. The anterior margins of spinal accessory nerve fenestrae
are present at the posterior margin of the neural canal.
In
lateral view, the neural spine originates just behind the zygosphene and is
posteriorly angled. In more posterior vertebrae (Figure
4C.3), the dorsal
margins of the neural spine are rounded and the anterior margin is depressed, as
in Acrochordus javanicus (Hoffstetter and Gayrard 1965). In vertebrae
from the middle portion of the trunk, however, the spine is robust and tall with
squared dorsal edges (Figure 4B.3). A shallow fossa is present on the lateral
surface of the neural arch in the same position as the lateral foramen of other
taxa. Minute lateral foramina are present at the posteroventral margin of the
fossa. The synapophyseal articular surface consists of distinct diapophyseal and
parapophyseal facets that are strongly angled relative to each other. Ventrally,
the hypapophysis is thin and elongate. It is ventrally deflected, but does not
extend posteriorly beyond the condyle.
In
ventral view (Figure 4B.5, C.2), the centrum is triangular in outline. The
hypapophysis is ovoid and elongate in cross section. The relatively small,
osseous paralymphatic channels are present at the medial margins of the
synapophyses. In dorsal view (Figure 4A.2, B.4), the interzygapophyseal ridge is
concave with a smoothly curving margin. The pre-and postzygapophyses are
elongate and diverge from the main body of the element at high angles.
Prezygapophyseal articular facets are ovoid and anterolaterally angled, becoming
more laterally than anteriorly angled in vertebrae from the middle to posterior
regions of the column. The accessory processes are anteroposteriorly compressed
and are laterally angled with respect to the articular facets. The lateral
margins of the zygosphene are curved, but the anterior margin is straight. The
spinous process is transversely wide in cross section with a rounded posterior
pillar tapering anteriorly to a thin lamina. The neural spine is relatively
short anteroposteriorly in vertebrae from the anterior region of the column
(Figure 4A.1, A.3).
Several incomplete specimens reveal internal vertebral morphology, including the
patterns of communication between vertebral foramina. The internal structure of
vertebrae in A. dehmi consists of a series of interconnected marrow
cavities throughout the neural arch and processes, and bisecting the vertebral
centrum. The cavities are paired and symmetrical around the sagittal plane of
the element, with a main chamber within the neural arch, dorsal to the centrum
body. Extensions radiate out from the main chamber to the base of the
prezygapophyses and the synapophyses anteriorly and through the central body
posteriorly, where they are connected. This connection possesses the same
general shape as the primary lacuna described for Python by
Hoffstetter
and Gasc (1969), but is slightly more posterior. Both the paracotylar and
parazygosphenal foramina communicate with the main chamber anteriorly.
Posterodorsally, the endozygantral foramina (zygantral foramina,
Rage 2001)
communicate with the main chamber, and a smaller, medial channel branches off
from the base of the foramina to form a dorsal communication between the two
sides of the element. This channel has a small anteriorly directed vacuity at
the base of the neural spine. The pattern of placement and communication between
marrow cavities in Acrochordus dehmi is generally similar to that
described for Pterosphenus (Hutchison 1985), but is more extensive than
that described for other taxa (Sood 1948).
Subadults (Figure 4D). Precloacal
vertebrae of subadult Acrochordus dehmi were recovered from 14 screen-washed localities throughout the Siwalik sequence. Subadult stages possess
characters that typify juvenile growth stages in snakes (e.g.,
LaDuke 1991).
These include an enlarged neural canal (canal diameter exceeds cotylar
diameter), relatively small prezygapophyses, synapophyses with poorly
differentiated para- and diapophyses that are relatively large and more
ventrally deflected than in adult specimens, and a neural spine that consists
only of a small posterodorsally angled process extending from the posterior
margin of the neural canal. Despite these differences from adult specimens,
generic assignment can be based on the possession of vertical, blade-like
accessory processes and multiple pairs of paracotylar foramina. Additionally,
the ventral deflection of the synapophyses characteristic of Acrochordus
is greater than seen in adults.
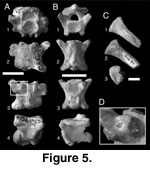 Cloacal vertebrae (Figure 5A, D). A
single element referable to the cloacal region of the vertebral column was
recovered. It is comparatively shorter and wider than precloacal and caudal
vertebrae, as in extant snakes (Hoffstetter and Gayrard 1965;
Thireau 1967), and
possesses the diagnostic prezygapophyseal accessory processes of Acrochordus.
In anterior view (Figure 5A.1), bases of the dorsal and ventral lymphapophyseal
processes are preserved and are separated by a wide, concave arc. Well-developed
osseous paralymphatic channels are present medial to the ventral lymphapophyseal
bases. In dorsal view (Figure 5A.2), the prezygapophysis is proportionally
smaller relative to the neural arch than in precloacal elements. The neural
spine is restricted to the posterior margin of the neural arch and is triangular
in cross section. In ventral view (Figure 5A.3), the hypapophysis extends nearly
the length of the centrum. The lymphapophyseal bases are transversely wide, and
a large foramen is present on the anteroventral face of the lymphapophysis
between the two bases (Figure 5A.3, D). The foramen is well developed, rounded,
and symmetrically present on both lymphapophyses (Figure 5D). In lateral view
(Figure 5A.4), the neural spine is reduced, with a gradually sloping anterodorsal margin. The spine does not extend beyond the posterior margin of
the neural arch.
Cloacal vertebrae (Figure 5A, D). A
single element referable to the cloacal region of the vertebral column was
recovered. It is comparatively shorter and wider than precloacal and caudal
vertebrae, as in extant snakes (Hoffstetter and Gayrard 1965;
Thireau 1967), and
possesses the diagnostic prezygapophyseal accessory processes of Acrochordus.
In anterior view (Figure 5A.1), bases of the dorsal and ventral lymphapophyseal
processes are preserved and are separated by a wide, concave arc. Well-developed
osseous paralymphatic channels are present medial to the ventral lymphapophyseal
bases. In dorsal view (Figure 5A.2), the prezygapophysis is proportionally
smaller relative to the neural arch than in precloacal elements. The neural
spine is restricted to the posterior margin of the neural arch and is triangular
in cross section. In ventral view (Figure 5A.3), the hypapophysis extends nearly
the length of the centrum. The lymphapophyseal bases are transversely wide, and
a large foramen is present on the anteroventral face of the lymphapophysis
between the two bases (Figure 5A.3, D). The foramen is well developed, rounded,
and symmetrically present on both lymphapophyses (Figure 5D). In lateral view
(Figure 5A.4), the neural spine is reduced, with a gradually sloping anterodorsal margin. The spine does not extend beyond the posterior margin of
the neural arch.
Caudal vertebrae (Figure 5B). Seventeen
caudal vertebrae of Acrochordus dehmi were recovered. In anterior view
(Figure 5B.1), the cotyle is relatively wider than in precloacal and cloacal
elements. The prezygapophyses diverge at a steep angle and extend to the dorsal
margin of the zygosphene. Prezygapophyseal accessory processes are smaller than
in precloacal elements. Ventrally angled, pillar-like pleurapophyseal bases
diverge from the centrum on either side of the cotyle. The zygosphene is small
with weakly developed articular facets. In dorsal view (Figure 5B.2), caudal
vertebrae are elongate, with a sharply convex zygosphenal ridge. The neural
spine is ovoid in cross section and restricted to the posterior margin of the
neural arch. In ventral view (Figure 5B.3), preserved portions of the haemapophyses indicate that they were restricted to just anterior of the
condyle. The haemapophyses are divided at their contact with the central body,
as in A. javanicus but unlike A. granulatus, in
which the haemapophyses are only distally forked (Hoffstetter and Gayrard 1965).
In lateral view (Figure 5B.4), caudal vertebrae are tall and broad with a deeper
body and shorter neural arch than in precloacal vertebrae. The neural spine is
low with a sloping anterior margin.
Ribs (Figure 5C). Articulated and
associated ribs were recovered from a single locality (Y-935). Ribs of
Acrochordus dehmi are robust and relatively thicker than in A.
javanicus or A. granulatus (Hoffstetter and Gasc 1969). In posterodorsal and anteroventral views, the proximal end is unexpanded relative
to other snakes, and there is no evidence of a pseudotuberculum process. In
articular view, the capitular and tubercular articular surfaces are continuous
and differentiated only by a pronounced recess for the attachment of costal
ligaments on the anteroventral margin of the element. This gives the element a
pronounced “C” shape in articular view (Figure 5C.3), corresponding to the recurved synapophyseal articular facets of precloacal vertebrae.
Discussion. Hoffstetter’s (1964) specific
diagnosis of A. dehmi was based on the following characters in
comparison with A. javanicus: large size; relatively taller, less
inclined, and rounder cotyle; dorsoventrally elongate synapophyses with robust
parapophyseal facets, synapophyses closely appressed in anterior view; and
thickened margins of zygosphene and zygantrum. The use of body size as a
diagnostic character for taxa possessing indeterminate growth is tenuous, and
previous attempts using vertebral size to differentiate fossil snake species
have been falsified (Christman 1975;
Prange and Christman 1976). Nevertheless,
the vast majority of the A. dehmi hypodigm consists of specimens
that are considerably larger than reported for living Acrochordus
(Hoffstetter 1964;
Hoffstetter and Gayrard 1965;
Shine 1986a,
1986b). The more
rounded cotyle and smaller parapophyseal processes of A. dehmi are
highly variable in both the Siwalik collection and in A. javanicus.
They are not considered diagnostic in this study. The increased thickness of
both the zygosphene and zygantral margins in A. dehmi appear
consistent in the Siwalik sample; however, these characters were determined to
be ontogenetically variable in other taxa (Auffenberg 1963). In comparison with
A. javanicus, the majority of characters used previously to
diagnose A. dehmi are either highly variable or are size
dependent. The sample of this study allows for a survey of all regions in the
vertebral column, and two characters here are used to diagnose Acrochordus
dehmi, the presence of a lymphatic foramen on cloacal vertebrae and neural
spines of nonposterior precloacal vertebrae with straight anterodorsal margins.
Acrochordus dehmi is one of the few snake
taxa whose species level interrelationships can be determined by vertebral
characters. Similarities between A. dehmi and A.
javanicus were noted (Hoffstetter 1964;
Rage 1987), and the presence of parazygosphenal foramina unambiguously unites the two as sister taxa to the
exclusion of A. granulatus and potentially A. arafurae
(McDowell 1979). Greatly reduced lateral foramina in A. dehmi
are also similar to the condition in A. javanicus, where
well-developed foramina are absent (Hoffstetter and Gayrard 1965). The complete
bifurcation of caudal haemapophyses in A. dehmi and A.
javanicus relative to the limited bifurcation in A. granulatus
supports this hypothesis, but the polarity of this character has not been
determined.
Acrochordus dehmi is the most abundant
Siwalik reptile taxon, and is represented by 1,289 specimens from 113 localities
throughout the entire temporal interval represented by the lower and middle
Siwalik Group. Hoffstetter (1964) suggested that that Acrochordus in the
Siwalik record may represent anagenetic evolution, with a second, younger
species replacing A. dehmi in the Dhok Pathan Formation, but was
unable to test this hypothesis due to sample size limitations.
Rage (1987)
echoed Hoffstetter (1964) in stating that two species of Acrochordus are
present- A. dehmi in the lower and middle Siwalik Group, and a
second taxon closely related to A. javanicus in the middle and
upper Siwalik Group.
Examination of the Acrochordus record from the Potwar Plateau does not
reveal differences between younger and older samples that can be separated from
intracolumnar variability. Thus, all specimens are referred to Acrochordus
dehmi in this study.
Despite a wide geographic distribution of extant species throughout southern
Asia to Australia (e.g.,
McDowell 1979), there is only a single definitive
fossil record of Acrochordus within its current geographic range, from
the early Miocene of Thailand (Rage and Ginsburg 1997). Reports of
Acrochordus from the Pliocene of northern Australia (Smith and Plane 1985)
now represent an elapid (Scanlon
et al. 2003). As a result, fossil
distributions suggest an Asian origin of the genus with subsequent dispersal
through Indonesia to northern Australia. The absence of Acrochordus from
any Australian Miocene fossil localities, despite intense study of the snake
record, suggests that immigration did not occur prior to the Pliocene. The
minimum divergence timing for Acrochordus can be constrained as no
younger than the first occurrence of its sister taxon Colubroidea in the early
Eocene (e.g., Rage et al. 2003;
Head et al. 2005). The fossil record of
Acrochordus dehmi extends throughout the lower and middle Siwalik Group, and
the timing of extinction for the taxon can only be limited to younger than 6.35
Ma.

 Precloacal vertebrae (Figure 4). In
anterior view, the cotyle is approximately circular in outline. Two to three
paracotylar foramina are present on either side of the cotyle. Small, paired
ventrolateral cotylar processes are present below the ventral margin of the
cotyle, forming a thin paralymphatic channel in most specimens. In posterior
precloacal vertebrae, the ventrolateral processes are greatly reduced, and the
paralymphatic channels extend along the medial surface of the synapophysis and
extend onto the ventrolateral margins of the cotyle (Figure 4C.1, 2). The prezygapophyses are robust and diverge from the body of the element at a high
angle. The prezygapophyseal accessory processes are strongly rounded in outline
and slightly discontinuous with the ventral angle of the prezygapophyses. The
synapophyses are low slung, with anteroposteriorly wide and anteriorly angled
parapophyses. The neural canal is relatively smaller than in more derived
snakes, with smoothly curved lateral and dorsal margins. The zygosphene is
robust and dorsoventrally tall. Parazygosphenal foramina are present at the
bases of the zygosphenal articular facets. Small pterapophyses are present on
the dorsal margin of the postzygapophyses (Figure
4A, B).
Precloacal vertebrae (Figure 4). In
anterior view, the cotyle is approximately circular in outline. Two to three
paracotylar foramina are present on either side of the cotyle. Small, paired
ventrolateral cotylar processes are present below the ventral margin of the
cotyle, forming a thin paralymphatic channel in most specimens. In posterior
precloacal vertebrae, the ventrolateral processes are greatly reduced, and the
paralymphatic channels extend along the medial surface of the synapophysis and
extend onto the ventrolateral margins of the cotyle (Figure 4C.1, 2). The prezygapophyses are robust and diverge from the body of the element at a high
angle. The prezygapophyseal accessory processes are strongly rounded in outline
and slightly discontinuous with the ventral angle of the prezygapophyses. The
synapophyses are low slung, with anteroposteriorly wide and anteriorly angled
parapophyses. The neural canal is relatively smaller than in more derived
snakes, with smoothly curved lateral and dorsal margins. The zygosphene is
robust and dorsoventrally tall. Parazygosphenal foramina are present at the
bases of the zygosphenal articular facets. Small pterapophyses are present on
the dorsal margin of the postzygapophyses (Figure
4A, B).