|
|
|
SYSTEMATIC PALEONTOLOGY
Order DECAPODA Latreille, 1802 Type Species. Clytiopsis argentoratensis Bill, 1914 Included Species. Clytiopsis argentoratensis Bill, 1914; C. thuringica Förster, 1967 Discussion. Bill (1914) erected a genus and two species: Clytiopsis argentoratensis and Clytiopsis elegans. Differences between the two were minimal which led Gall and Fisher (1965) to conclude that C. elegans is synonymous with C. argentoratensis. Förster (1966, 1967) and Gall (1971) supported that view. Clytiopsis argentoratensis and Clytiopsis thuringica are the only two species described within Clytiopsis so far. Several authors have reported Clytiopsis sp. (e.g., Bill 1914; Förster 1966, 1967; Diedrich and Schulz 2003).
Clytiopsis argentoratensis Bill, 1914
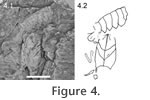
1914 Clytiopsis elegans Bill, p. 300, pl. 10, fig. 3, pl. 11, fig. 2, pl. 12, figs. 2, 3. 1928 Clytiopsis argentoratensis Bill; Schmidt, p. 323, fig. 883. 1928 Clytiopsis elegans Bill; Schmidt, p. 324, fig. 884. 1965 Clytiopsis argentoratense Bill; Gall and Fischer, p. 44, fig. 1, pl. 1, 2. 1966 Clytiopsis argentoratensis Bill; Förster, p. 83, fig. 10, pl. 13, figs. 1, 2, 3. 1967 Clytiopsis argentoratensis Bill; Förster, p. 146, fig. 4, pl. 9, fig. 3. 1971 Clytiopsis argentoratensis Bill; Gall, p. 52, figs. 12, 13, pl. 12, fig. 4, pl. 13, figs. 1, 2. 1999 Clytiopsis argentoratensis Bill; Hauschke and Wilde, fig. 10. Diagnosis. Cephalothorax cylindrical; rostrum triangular; faint median groove; postorbital ridge and antennal ridge with spines; strong cervical groove; faint postcervical groove partly parallels branchiocardiac region; strong branchiocardiac groove and cervical groove connected by two-lobed hepatic groove; ventral groove connects to branchiocardial groove; antennal groove curves forward from cervical groove. Mostly pits on branchial region, sometimes granules; granules on cardiac region, gastric and antennal region. Abdomen longer than cephalothorax; first somite small; epimeres rounded with pointed tips. Telson spade-shaped; exopods with diaeresis. First three pereiopods with opposing dactylus and propodus. Description. Cephalothorax cylindrical; wider posteriorly. Front narrowing anteriorly. Rostrum not preserved. Median line is faint furrow. No intercalated plate observed. Boundary of gastric and antennal region is postorbital ridge composed of small spines. Cervical groove marks end of both regions. Posteriorly directed postorbital ridge makes a 40–50° angle to median line. Anteriorly directed cervical groove arises at median line with an angle of about 30°, curving to about 40°; stronger than median line. Gastroorbital groove very faint to invisible. Postcervical groove does not reach median line; in between cervical and branchiocardiac groove; weaker than cervical and branchiocardiac grooves; extends parallel to branchiocardiac groove in middle part but curves to cervical groove ventrally; approaches branchiocardiac groove near median line. Branchiocardiac and cervical grooves define a trapezoid region consisting of medial cardiac and ventral hepatic regions. Posteriorly directed branchiocardiac groove originates at about median line; starts at a very low angle to median line, curves to an angle of 30–40° to median line, then curves slightly forward at about midlength in dorsal view; stronger than postcervical groove; slightly weaker than cervical groove. Two-lobed hepatic furrow connects cervical and branchiocardiac groove. Branchial region ended by groove along rim of cephalothorax. Groove about as strong as branchiocardiac groove; starts about perpendicular to median line; more ventrally it curves posteriorly. Mostly pits on branchial region, sometimes granules; granules on cardiac region, gastric and antennal region. Abdomen only partly preserved in one specimen (Figure 2.1); same for a part of an appendage; one part of uropod preserved; no telson preserved. Few granules or pits visible on abdominal segments. State of preservation of epimeres does not allow further description. For measurements see Table 1. Material examined. Four specimens (MAB k2855–2858) deposited at Oertijdmuseum De Groene Poort, Boxtel, The Netherlands. The exact stratigraphic level at which the specimens were collected is unknown. Discussion. The cephalothorax of four specimens can be assigned to Clytiopsis argentoratensis. Although not all details as described by Förster (1966) are visible, all the characters that are visible are the same. The type species description of Bill (1914) is not useful because it does not describe the cephalothorax in detail. The antennal ridge, the ventral groove, and the bifurcation at the most dorsal position of the postcervical groove were invisible due to the preservation state. Differences with Clytiopsis thuringica from the Ladinian of Straußfurt (Germany) are numerous. The postcervical groove does not approach the cervical groove as it does in C. thuringica. The postorbital ridge is smaller than that of C. thuringica. The ornamentation of the specimens from Winterswijk does not show pits followed by larger granules posteriorly, as is the case for C. thuringica. Of special note is specimen MAB k2856 (Figure 2.3). The trifurcated ridge in the gastric was probably caused by diagenetic pressure differences. Ridges are rare on lobsters from the Triassic and, if present, are usually formed by aligned bumps, which is not the case here. The median groove does not follow the anteriormost branch of the trifurcation closely. Lastly, the two posteriormost ridges are unequal in size and length. Therefore, the trifurcated ridge is considered as having been formed after burial.
Gall and Fischer (1965) studied the 50 specimens of Clytiopsis from the Anisian of the Vosges Mountains in France on which
Bill (1914) based his descriptions. They rectified the groove pattern of C. argentoratensis (see their figure 1). The main difference between the reconstruction of
Förster (1966,
1967), who did not address
Gall and Fischer's (1965) reconstruction, and theirs is the postcervical groove. Their figure 1 shows that the dorsal bifurcation is far more extended than in
Förster (1966,
1967).
Occurrence and age. The studied specimens are all from the Middle Triassic (Anisian) strata from the Winterswijk quarry complex, the eastern Netherlands. Specimens MAB k28562858 were found in Quarry III; MAB k2855 was found in Quarry II. Other specimens have been found in Anisian strata in the Vosges Mountains of France (Figure 3).
Subfamily ERYMINAE
van Straelen, 1924 Diagnosis. Cephalothorax cylindrical; triangular rostrum; median line present; intercalated plate on gastric part; strong cervical groove; small, faint postcervical groove parallels stronger branchiocardiac groove; cephalothorax partly smooth or pitted. Terga and epimeres pitted; epimeres with pointed, slightly posteriorly directed tip. Spade-shaped telson with longitudinal groove but without bristle structure; exopod with diaeresis. Description. As for type species. Etymology. The name is derived from a specialist on the geology and paleontology of the Winterswijk quarry complex, and collector of the specimens described herein: Henk Oosterink.
Oosterinkia neerlandica n. sp.
1979 Clytiopsis sp., Oosterink, fig. 1. 1986 Clytiopsis sp., Oosterink, p. 57, fig. 17. Diagnosis. As for genus. Description. Cephalothorax cylindrical; widest part at half to two thirds of length. Front narrowing. Rostrum triangular. Median line is faint groove from posterior to slightly anterior of cervical groove; on anterior half of gastric region median line becomes a 0.3 mm wide ridge, which is an intercalated plate; becoming groove again on rostrum. Postorbital ridge covered with a row of four more or less equal-sized, forwardly oriented spines. Left antennal ridge extends more posteriorly than postorbital ridge; approaches cervical groove; extends parallel to postorbital ridge; exhibits strong spines. Strong cervical groove arises at median line at an angle of about 30° and curves ventrally to about 40°; becomes stronger ventrally. Small, very faint postcervical groove parallels branchiocardiac groove; neither reaches median line nor curves to cervical groove. Sinuous branchiocardiac groove arises at median line at a very low angle, increases to about 40°, then increases to about 50°; about as pronounced as cervical groove; more pronounced ventrally. Branchial region ends in groove along rim of cephalothorax. Groove about as strong as branchiocardiac groove; starts near and about perpendicular to median line; ventrally curving more posteriorly and more pronounced ventrally. Cephalothorax party smooth and pitted except few isolated, aligned nodes just posterior of cervical groove. Pits on branchial, posterodorsal part of branchiocardiac region, and anterior part of antennal and gastric region. First of six somites reduced; epimere not visible. Sixth somite longest. Epimeres convex in anterior part and initially convex and becoming downward more concave at posterior part; sides merge to form an apex directed slightly posteriorly. Epimere of sixth somite smallest. Epimeres and most ventral part of terga with pits. Spade-shaped telson with small row of nodes in middle part surrounded by smaller ones; some pits on the sides; longitudinal groove in middle; no bristles visible at distal rim. Right uropod only partly preserved. Endopod with longitudinal lines at distalmost part and small, faint, transverse ridges in the middle. Exopod with transversal, curved diaeresis. Part of exopod or antennae close to posteriormost left part of branchial region. At least two parts of thoracic appendage to left of cephalothorax. For measurements see Table 1. Etymology. The name is derived from the country in which the specimen was found. Type. The holotype (MAB k2854) and sole specimen is stored at Oertijdmuseum De Groene Poort, Boxtel, The Netherlands. The exact stratigraphic level at which the specimen was collected is unknown. Discussion. Oosterinkia neerlandica exhibits a deep groove anterior to the cervical groove which is present on both sides of the median line. The groove approaches very close to the cervical groove on the left side of the cephalothorax and bifurcates on the right side. This groove is not symmetrical and, therefore, must be considered to be a taphonomic feature. There are several genera within the family Erymidae: Clytiella Glaessner 1931; Clytiopsis Bill 1914; Enoploclytia McCoy 1849; Eryma von Meyer 1840; Galicia Garassino and Krobicki 2002; Lissocardia von Meyer 1851; Palaeastacus Bell 1850; Paraclytiopsis Oravecz 1962; Protoclytiopsis Birshtein 1958; Pustulina Quenstedt 1857. At first sight Oosterinkia neerlandica resembles Clytiopsis spp. However, differences from Clytiopsis thuringica may be observed. First, the postcervical groove is longer and more pronounced in C. thuringica. The ornamentation differs: C. thuringica has nodes posterior to pits, while O. neerlandica has only pits in the gastric region. The postorbital ridge of C. thuringica has spines that increase in height posteriorly, while O. neerlandica shows no spines. The median furrow is continuous in C. thuringica but is not continuous in O. neerlandica. The furrow becomes an intercalated plate in the posterior part of the gastric region. Differences in the postcephalothracic parts cannot be given because the description of the sole specimen of C. thuringica is based on a cephalothorax only. Oosterinkia neerlandica differs from Clytiopsis argentoratensis in several aspects. The median line is only present as a faint groove posterior from about the cervical groove and is a ridge on the anteriormost part of the gastric. The median groove does not turn into an intercalated plate in specimens of C. argentoratensis; it remains a groove. The postcervical groove of O. neerlandica is smaller and fainter than in C. argentoratensis. The cervical and branchiocardiac groove are very clear in O. neerlandica, even more pronounced than in other specimens. Therefore, a clearer postcervical groove would be expected if it were to be C. argentoratensis. Furthermore, the cephalothorax is pitted in the gastric and antennal region, but exhibits granules in C. argentoratensis. The telson shows no bristle structure that is present in C. argentoratensis according to Gall (1971). Gall and Fisher (1965), however, mentioned that it might not have been present in some specimens of C. argentoratensis due to the nature of fossilization. The telson shows a small groove, a feature not reported on C. argentoratensis. The new genus differs from Paraclytiopsis by the presence of a median line and the intercalated plate, which are present in Oosterinkia neerlandica. Clytiella also does not exhibit an intercalated plate, and, moreover, has a row of spines on the median keel. This is not observed in O. neerlandica. It differs from Protoclytiopsis by its stronger branchiocardiac groove compared to the postcervical groove, which is the opposite in Protoclytiopsis. Enoploclytia and Palaeastacus have a more pronounced ornamentation with nodes on the gastic and cardiac regions. Oosterinkia neerlandica is smooth to pitted on the whole cephalothorax. The new genus differs from Eryma by the branchiocardiac groove that is about as strong as the cervical groove, the pitted abdomen, and the smooth to pitted cephalothorax. Eryma has a weaker branchiocardiac groove compared to the cervical groove, a smooth abdomen, and a granulated cephalothorax. Lissocardia differs from Oosterinkia by having longitudinal ridges in the gastric region instead of an oblique ridge and a weaker branchiocardiac groove. Galicia is granulated on the cephalothorax and has a stronger postcervical groove that joins the branchiocardiac groove, while the new genus has a smooth to pitted cephalothorax and a very weak postcervical groove that does not join the branchiocardiac groove. Pustulina bears a strong gastroorbital groove, has a granulated cephalothorax, a strong postcervical groove, and a small branchiocardaic groove. On the other hand, O. neerlandica exhibits no gastroorbital groove, has a smooth to pitted cephalothorax, and bears a strong branchiocardiac, but a faint postcervical groove. The specimen, thus, shows numerous differences compared to the known genera and species. We, therefore, erect Oosterinkia neerlandica n. gen., n. sp. Occurrence. One specimen from Anisian sediments from Quarry I of the Winterswijk quarry complex, eastern Netherlands.
Superfamily GLYPHEOIDEA Winckler, 1882 Emended diagnosis. Subcylindrical carapace; long rostrum; cephalic region with longitudinal ridges reduced or absent; well marked cervical groove; postcervical and branchiocardiac grooves closely spaced and parallel, approaching or extending to median line; well-marked hepatic groove extends in smooth arc defining posterior, ventral, and anterior margins of "adductor testis" muscle attachment; inferior groove absent or weakly marked; strong and subchelate pereiopod I; subchelate pereiopods IIIII; abdominal terga pitted, smooth, or with granules; uropodal exopod with or without diaeresis (modified from Feldmann et al. 2002 and Garassino and Rigo 2008). Type Species. Glyphea grandis von Meyer, 1837, by original designation Included Species. Pseudoglyphea grandis (von Meyer 1837); P. numismalis (Oppel 1853); P. eximia Oppel 1861; P. amalthea Oppel 1861; P. terquemi Oppel 1861; P. etalloni Oppel 1862; P. ancylochelis (Woodward 1863); P. jourdani (Dumortier 1867); P. paronae (Colosi 1921); P. spinosa (Assmann 1927); P. mulleri (van Straelen 1936); P. straeleni (Théobald 1953); P. alpina (Förster 1971); P. gigantea Garassino and Teruzzi 1993; P. foersteri Feldmann, Crisp, and Pirrie 2002; P. friulana Garassino and Rigo 2008.
Pseudoglyphea cf. P. spinosa
1928 Pseudopemphix spinosus Assmann; Schmidt, p. 319, fig. 878. 1932 Pseudoglyphea spinosa (Assmann); Glaessner, p. 113-114, fig. 2C. 1960 Pseudoglyphea spinosa (Assmann); Glaessner, p. 40, fig. 19.1. 1966 Pseudoglyphea spinosa (Assmann); Förster, fig. 36. 1967 Pseudoglyphea ? spinosa (Assmann); Förster, p. 163, fig. 10a, pl. 11, fig. 1. 1979 Pseudoglyphea cf. spinosa (Assmann); Oosterink, p. 115. 1986 Pseudoglyphea cf. spinosa (Assmann); Oosterink, p. 62. Description. Left part of cephalothorax best preserved; only cardiac region preserved on right part. Possible part from gastric and/or rostrum with small granules, possibly accompanied by bases of antennules. Longitudinal axis is faint ridge on branchial region; on cardiac region it is a furrow formed by position of large nodes on both sides. Strong cervical groove (only visible on right side) forms about a 40° angle with longitudinal axis. Strong postcervical groove parallels weaker branchiocardiac groove dorsally, then curves slightly toward it and parallels it again; ventralmost part as strong as cervical groove; dorsalmost part less pronounced; does not connect to longitudinal axis. Branchiocardiac groove arises near axis and gradually curves forward from 30° to about 50°. Faint groove posterior and parallel to branchiocardiac groove distal from longitudinal axis. Marginal groove marks end of branchial region; arises perpendicular to longitudinal axis, curves forward followed by sharp curve backwards. Strong nodes on cardiac region and in region between postcervical and branchiocardiac groove. Smaller granules on branchial region. Five granular terga visible of which first is mostly hidden beneath the cephalothorax. Second and third visible terga separated. Last tergum longest. One part of epimere preserved on right side of last or next to last somite; at least partly granular. Telson broken into many pieces. Left uropod granular; endopod overlapping exopod; furrow in middle does not reach diaeresis; diaeresis with small spines directed posteriorly; distalmost part of uropod not preserved. Exopod with thickened lateral margin exhibiting pits. Granular endopod only preserved on right uropod. Part of possible thoracic appendage preserved right of abdomen. For measurements see Table 1. Material examined. This specimen (MAB k2859) is stored at Oertijdmuseum De Groene Poort, Boxtel, The Netherlands. The exact level at which the specimen was collected is unknown. Discussion. The described specimen resembles Pseudopemphix albertii (von Meyer 1840) due to the fact that only a part of this specimen is preserved. Förster (1967) pointed out that Pseudopemphix and Pseudoglyphea have similarities, and Assmann (1927) assigned P. spinosa to Pseudopemphix when he erected the species. However, there are some major differences between P. spinosa and Pseudopemphix albertii, type species of the genus. Schulz (2002) mentioned that the abdomen of Pseudopemphix albertii has pits, which is not the case in this specimen from the Netherlands. Unfortunately, the type specimen of Pseudoglyphea spinosa is known only from the cephalothorax. The distribution and number of large nodes is critical. There are fewer large nodes on Pseudopemphix albertii (see Schmidt 1928, figure 876; Förster 1967, figure 12; Schulz 2002) than on the specimen at hand. Moreover, the large nodes are also present between the postcervical and the branchiocardiac groove in this specimen. This applies to P. spinosa, but not to the type of Pseudopemphix (see Schmidt 1928, figure 878; Förster 1967, figure 10, 12; Schulz 2002). In addition, the cardiac region of Pseudopemphix albertii bears both large and small nodes, which is not the case in this specimen. It bears large nodes only. Furthermore, this specimen does bear a thickening just before the end of the branchial region which is not observed in Pseudopemphix. In conclusion, this specimen is better referred to Pseudoglyphea than to Pseudopemphix. We favor assignment to Pseudoglyphea over other genera within the Mecochiridae and over Pseudopemphix. The specimen bears all the characters described by Förster (1967), Feldmann et al. (2002), and Garassino and Rigo (2008) as far as they are visible in this specimen. The only exceptions are the terga, which would be pitted or smooth in most species of Pseudoglyphea, but have nodes in this specimen. It clearly differs from Mecochirus Germar 1827, Meyeria McCoy 1849, Huhatanka Feldmann and West 1978, and Jabaloya Garassino et al. 2009 by its less oblique grooves posteriorly from the cervical grooves. It differs from Pseudopemphix by exhibiting spines on the cardiac region of Pseudoglyphea that do not appear to form rows and the absence of pits on the cephalothorax. This specimen can, thus, best be assigned to Pseudoglyphea. It differs from Pseudoglyphea foersteri by its large spines on the cardiac region, which are absent in P. foersteri. Pseudoglyphea friulana has weaker postcervical and branchiocardiac grooves. The postcervical groove is more pronounced than in specimens of P. grandis and P. alpina. The cardiac region has stronger spines than on specimens of P. mulleri, P. amalthea, P. terquemi, and P. gigantea. The postcervical and branchiocardiac groove of P. staeleni, P. numismalis, and P. jourdani do not approach the median line, which can be observed from this specimen. The preservation of the cephalothorax of P. paronae allowed identification of a strong cervical groove, the hepatic groove, and a part of the postcervical groove only (van Straelen 1924). The assignment of that species to Pseudoglyphea, thus, is doubtful. The postcervical appears to be fairly weak and, as a result, differs from the described specimen. Pseudoglyphea etalloni differs from the specimen by possessing a distinct bifurcation of the postcervical groove dorsally and by the presence of larger nodes on the dorsal part of the branchiocardiac region. The described specimen does not show a distinct bifurcation in the postcervical groove and has a weak granulation on the branchiocardiac region. Pseudoglyphea eximia has a smaller branchiocardiac region and has weaker nodes on the cardiac region. The specimen has more large nodes in the cardiac region than specimens of P. ancylochelis. It is very similar to P. spinosa based on the cardiac region being covered by strong nodes, the presence of strong nodes between the postcervical and branchiocardiac groove, the relative strength of the groove, and the relatively large, granulated branchiocardiac region. In addition, Assmann (1927) and Schmidt (1928) mentioned that the holotype of Pseudoglyphea spinosa has a thickening just before the end of the branchial region. The thickening is observed in the specimen from Winterswijk. There are, however, some minor differences. Förster (1967) mentioned that the longitudinal axis of Pseudoglyphea (?) spinosa has large spines and Assmann (1927) stated that they appeared on the anterior part of the cardiac region. This is not observed in this specimen. It has a ridge in the branchial region and a groove on the cardiac region. Furthermore, Assmann (1927) mentioned that the nodes at the anterior of the cardiac region are larger in P. spinosa, which is not clear from this specimen. The differences are, however, too few to warrant erecting a new species. Therefore, we refer to it as Pseudoglyphea cf. P. spinosa. Occurrence and age. One specimen found in Anisian sediments from Quarry III of the Winterswijk quarry complex, the eastern Netherlands. The holotype was found in Anisian sediments of Dinkelberg, Germany (Figure 3). |
|