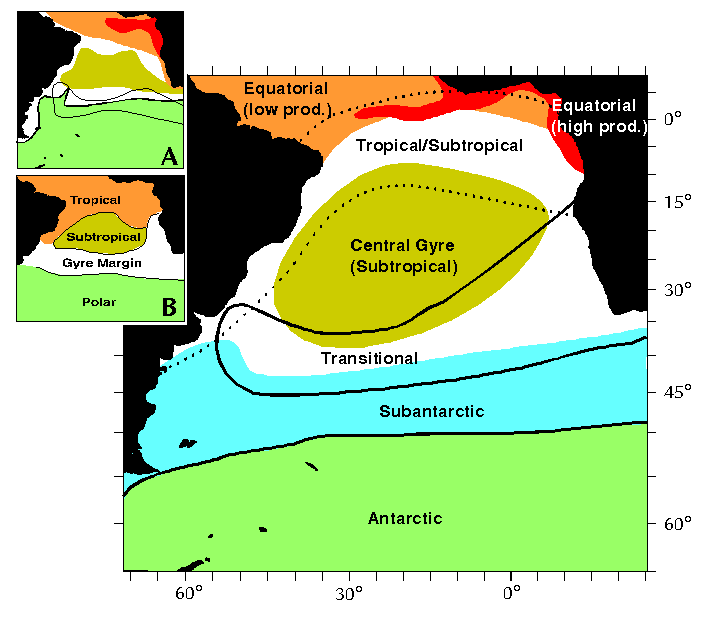
| Figure 1A. Main features of a polycystine spumellarian cell. Slightly modified from Kling (1978) and Boltovskoy (1981e). |
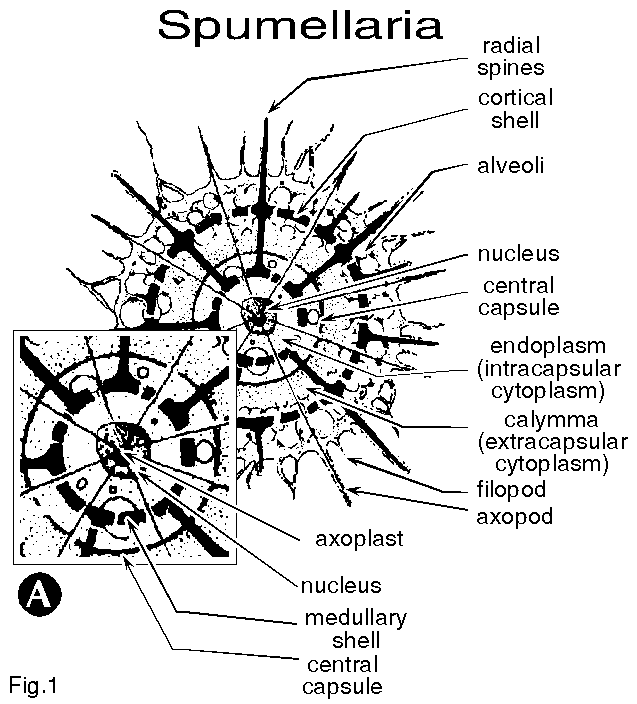 |
| Figure 1B. Main features of a polycystine nassellarian cell. From Hollande and Enjumet (1960). |
 |
| Figure 1C-E. General view of entire radiolarian colonies. C, D: by courtesy of Neil Swanberg (from Swanberg 1979); E: from Strelkov and Reshetnjak (1971). |
 |
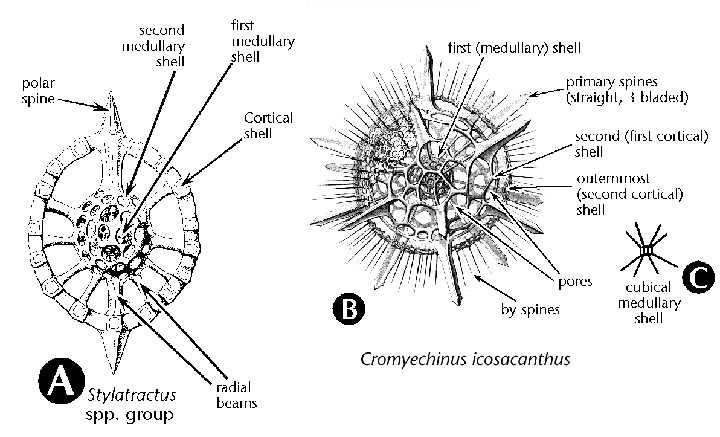
| Figure 2ABC. Characters used for the identification of spumellarian radiolarians. A, B, from Haeckel (1887); C: original. |
 |
| Figure 2D, E. Characters used for the identification of spumellarian radiolarians. From Haeckel (1887). |
 |
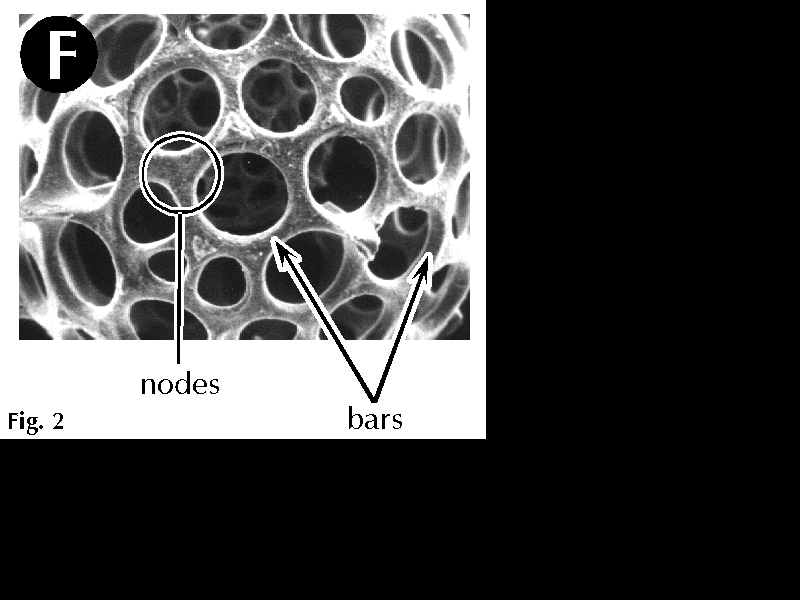
| Figure 2F. Characters used for the identification of spumellarian radiolarians. Original. |
 |
| Figure 2G. Characters used for the identification of spumellarian radiolarians. From Haeckel (1887). |
 |
| Figure 2H, I. Characters used for the identification of spumellarian radiolarians. H: from Haeckel (1887); I: from Riedel (1958). |
 |
| Figure 2J. Characters used for the identification of spumellarian radiolarians. Original. |
 |
| Figure 2K. Characters used for the identification of spumellarian radiolarians. From Haeckel (1887). |
 |
| Figure 2L, M. Characters used for the identification of spumellarian radiolarians. Original. |
 |
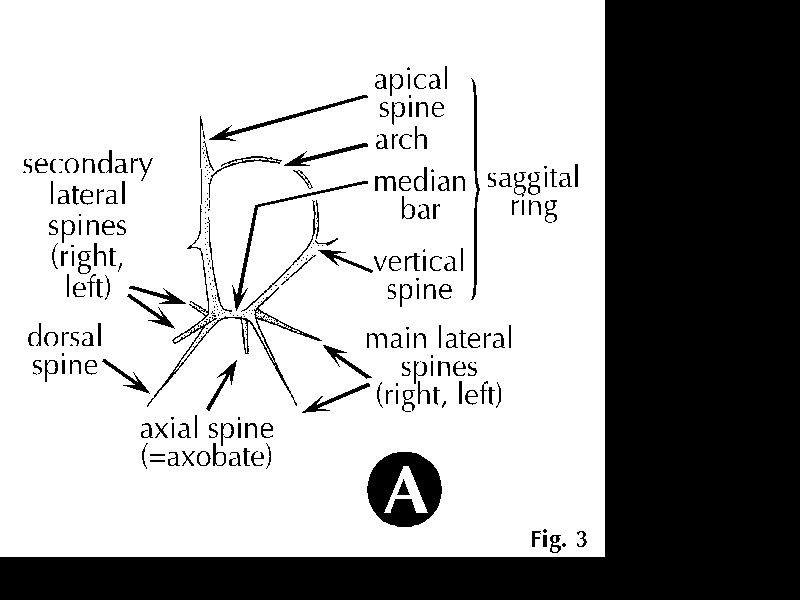
| Figure 3A. Characters used for the identification of nassellarian radiolarians. From Petrushevskaya (1981). |
 |
| Figure 3B, C. Characters used for the identification of nassellarian radiolarians. B: from Riedel (1958); C: from Petrushevskaya (1971a). |
 |
| Figure 3D, E. Characters used for the identification of nassellarian radiolarians. From Petrushevskaya (1971a). |
 |
| Figure 3F. Characters used for the identification of nassellarian radiolarians. From Haeckel (1887). |
 |
| Figure 3G. Characters used for the identification of nassellarian radiolarians. From Petrushevskaya (1971a). |
 |
| Figure 3H-J. Characters used for the identification of nassellarian radiolarians. H: original; I, J: from Petrushevskaya (1971a). |
 |
| Figure 3K, L. Characters used for the identification of nassellarian radiolarians. K: from Petrushevskaya (1971a); L: from Riedel (1957). |
 |
| Figure 3M, N. Characters used for the identification of nassellarian radiolarians. M: from Petrushevskaya (1971a); N: from Petrushevskaya (1981). |
 |
| Figure 3O, P. Characters used for the identification of nassellarian radiolarians. O: from Petrushevskaya (1971a); P: from Paverd (1995). |
 |
| Figure 3Q, R. Characters used for the identification of nassellarian radiolarians. From Petrushevskaya (1971a). |
 |
| Figure 3S-W. Characters used for the identification of nassellarian radiolarians. S, T, U, V: original; W: from Petrushevskaya (1971a). |
 |
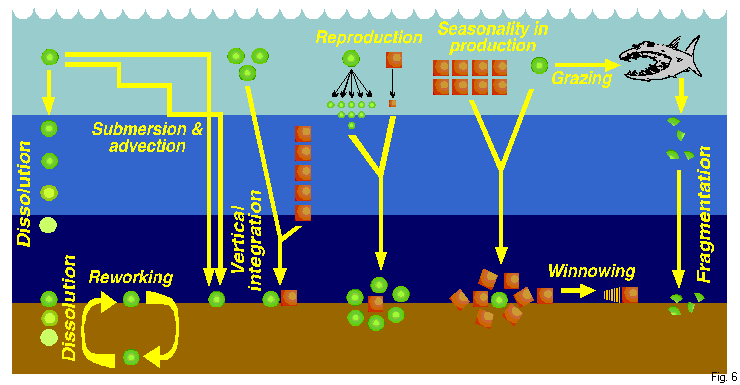
| Figure 6. Schematic diagram of the mechanisms that can distort the sedimentary imprint of the planktonic pattern of fossilizable microplankton in general, and of polycystine radiolarians in particular (see text for detailed explanation). From Boltovskoy 1995. |
 |
| Figure 7. Quantitative radiolarian distribution in the surface sedimentary layer of the South Atlantic. Redrawn from Goll and Bjørklund (1974). |
 |
| Figure 12H. Assumed proportions of live polycystine cells at various depths in the water column. From data in Kling and Boltovskoy (1995). |
 |
| Figure 14. Successive stages of growth of polycystine skeletons. From Petrushevskaya (1962, 1967). |
 |