| |
RESULTS AND DESCRIPTION
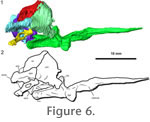
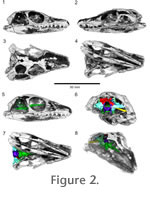 Figure 2 presents a voltex rendering of the HRXCT scan data. From this rendering it is clear that successful separation between bone and matrix was achieved. However, the skull roof has been extensively damaged during preparation, particularly in the rostrum and mandible. The braincase, however, is preserved in its entirety. It is shown in context in the voltex rendering and is described below. Figure 2 presents a voltex rendering of the HRXCT scan data. From this rendering it is clear that successful separation between bone and matrix was achieved. However, the skull roof has been extensively damaged during preparation, particularly in the rostrum and mandible. The braincase, however, is preserved in its entirety. It is shown in context in the voltex rendering and is described below.
Anatomy of the Braincase
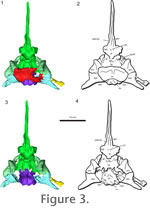 Supraoccipital. The supraoccipital is a broad, large bone that contacts both the prootic and opisthotic bones on its lateral edges and the exoccipital caudally (Figure 3.1-2,
Figure 4). It connects the braincase dorsally to the rest of the skull through contact with the parietals. The supraoccipital bears facets for the postparietals and parietals, but does not show the prominent median or lateral ascending processes that are found in Captorhinus (Price 1935) and in placodonts (Rieppel 1995), or the prominent median process found in Proganochelys (Gaffney 1990) and Kayentachelys (Gaffney and Jenkins 2010). It forms a small portion of the dorsal border of the foramen magnum and covers the caudal portion of the brain dorsally. The right rostrolateral portion of the supraoccipital is broken in AMNH 5561, but the supraoccipital is complete UC 1528. Each lateral portion of the supraoccipital encloses a small part of the caudal semicircular canal. Supraoccipital. The supraoccipital is a broad, large bone that contacts both the prootic and opisthotic bones on its lateral edges and the exoccipital caudally (Figure 3.1-2,
Figure 4). It connects the braincase dorsally to the rest of the skull through contact with the parietals. The supraoccipital bears facets for the postparietals and parietals, but does not show the prominent median or lateral ascending processes that are found in Captorhinus (Price 1935) and in placodonts (Rieppel 1995), or the prominent median process found in Proganochelys (Gaffney 1990) and Kayentachelys (Gaffney and Jenkins 2010). It forms a small portion of the dorsal border of the foramen magnum and covers the caudal portion of the brain dorsally. The right rostrolateral portion of the supraoccipital is broken in AMNH 5561, but the supraoccipital is complete UC 1528. Each lateral portion of the supraoccipital encloses a small part of the caudal semicircular canal.
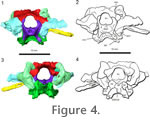 Exoccipital. The exoccipitals form the lateral border of the foramen magnum (Figure 4). Dorsally they extend as triangular processes that approach each other but do not meet medially. The exit for the hypoglossal nerve (CN XII) is present lateral to the foramen magnum within the exoccipital body. The suture between the ventral portion of the exoccipital is distinct from the occipital condyle in UC 1528 and is triangular in shape in dorsocaudal view. There is no distinct suture between the exoccipital and the occipital condyle in AMNH 5561. The exoccipital contributed significantly to the dorsolateral corner of the occipital condyle in Youngina. Exoccipital. The exoccipitals form the lateral border of the foramen magnum (Figure 4). Dorsally they extend as triangular processes that approach each other but do not meet medially. The exit for the hypoglossal nerve (CN XII) is present lateral to the foramen magnum within the exoccipital body. The suture between the ventral portion of the exoccipital is distinct from the occipital condyle in UC 1528 and is triangular in shape in dorsocaudal view. There is no distinct suture between the exoccipital and the occipital condyle in AMNH 5561. The exoccipital contributed significantly to the dorsolateral corner of the occipital condyle in Youngina.
 Basioccipital. The basioccipital forms the ventral border of the foramen magnum, and has a stout main body that supports the brain caudoventrally (Figure 3,
Figure 5). The basioccipital has weakly developed basal tubera that lack an extensive bony contact with the parasphenoid caudal processes. The contact between these two elements was likely cartilaginous. The exoccipitals and basioccipital are fused together in AMNH 5561, but a suture is present between the elements in UC 1528. The occipital condyle has a deep notochordal pit. Basioccipital. The basioccipital forms the ventral border of the foramen magnum, and has a stout main body that supports the brain caudoventrally (Figure 3,
Figure 5). The basioccipital has weakly developed basal tubera that lack an extensive bony contact with the parasphenoid caudal processes. The contact between these two elements was likely cartilaginous. The exoccipitals and basioccipital are fused together in AMNH 5561, but a suture is present between the elements in UC 1528. The occipital condyle has a deep notochordal pit.
Basisphenoid. The basisphenoid is an endochondral element that supports the brain ventrally (Romer 1956) (Figure 3.3-4,
Figure 4.3-4). In AMNH 5561, the basisphenoid shows no significant differences from the descriptions of TM 3603 made by
Evans (1987). The basisphenoid bears small paired foramina for the internal carotid arteries rostral to the sella turcica (or hypophyseal fossa), close to the base of the parasphenoid cultriform process which are not observed in Hovasaurus (Currie 1981). The width of the sella turcica is one third that of the rostral portion of the basisphenoid body. The dorsum sellae separates the sella turcica from the caudal region of the basisphenoid. There is no evidence of an ossified pila antotica, although stout, short clinoid processes are present. The groove for the abducens nerve (CN VI) appears to be present on the lateral surface of the dorsum sellae as described by
Evans (1987), similar to Prolacerta and other early archosauromorphs (Evans 1986). In captorhinids, Proganochelys and Sphenodon, the abducens nerve pierces the dorsum sellae rather than laying in an open groove. Lateral to the dorsum sellae, the clinoid process extends dorsally and contacts the prootic bone ventrally but not the parietal, as in Proganochelys (Gaffney 1990). The basipterygoid has large rostroventral processes (i.e., basipterygoid processes) that form a broad palatobasal articulation with the pterygoid. These structures are also visible ventrally in UC 1528 (Olsen 1936).
 Parasphenoid. The parasphenoid is a dermal element that covers the basisphenoid ventrally and has an elongate cultriform process rostrally that supports ossified trabeculae cranii, producing a V-shaped cross section (Figure 3,
Figure 5, and
Figure 6). The suture between the parasphenoid and basisphenoid is largely indistinguishable in the HRXCT data, and the two elements have been left the same color in the model. The cultriform process is directed only slightly rostrodorsally, less than 15 degrees from the horizontal plane of the skull. There are small lateral crests on the rostral body of the parasphenoid, which partially enclose the vidian canals (path of the internal carotid artery and palatine ramus of the facial nerve).
Evans (1987) reported that the large cultriform process she observed in UC 1528 (as we see in AMNH 5561 via HRXCT data) was not present in TM 3603 despite its large size. She suggested it might not be ossified, which could imply that its ossification might be variable within Youngina as reported for varanids and other lepidosaurs (Bever et al. 2005). The cultriform process extends rostrally to the rostral margin of the orbit as in Hovasaurus and Tangasaurus. There are no teeth present on the parasphenoid, unlike more primitive reptiles such as Petrolacosaurus (Reisz 1981) or kuehneosaurids (Colbert 1970;
Evans 2009). The general morphology of the parasphenoid of AMNH 5561 is similar to that of UC 1528 (Evans 1987). The caudal portion of the parasphenoid divides into paired crista ventrolateralis processes (or posterior alar wings) and the caudal margin is deeply concave as in Petrolacosaurus (Reisz 1981). These processes do not appear to have contacted the basioccipital tubers through osseous contact; a cartilagenous contact may have been present. Parasphenoid. The parasphenoid is a dermal element that covers the basisphenoid ventrally and has an elongate cultriform process rostrally that supports ossified trabeculae cranii, producing a V-shaped cross section (Figure 3,
Figure 5, and
Figure 6). The suture between the parasphenoid and basisphenoid is largely indistinguishable in the HRXCT data, and the two elements have been left the same color in the model. The cultriform process is directed only slightly rostrodorsally, less than 15 degrees from the horizontal plane of the skull. There are small lateral crests on the rostral body of the parasphenoid, which partially enclose the vidian canals (path of the internal carotid artery and palatine ramus of the facial nerve).
Evans (1987) reported that the large cultriform process she observed in UC 1528 (as we see in AMNH 5561 via HRXCT data) was not present in TM 3603 despite its large size. She suggested it might not be ossified, which could imply that its ossification might be variable within Youngina as reported for varanids and other lepidosaurs (Bever et al. 2005). The cultriform process extends rostrally to the rostral margin of the orbit as in Hovasaurus and Tangasaurus. There are no teeth present on the parasphenoid, unlike more primitive reptiles such as Petrolacosaurus (Reisz 1981) or kuehneosaurids (Colbert 1970;
Evans 2009). The general morphology of the parasphenoid of AMNH 5561 is similar to that of UC 1528 (Evans 1987). The caudal portion of the parasphenoid divides into paired crista ventrolateralis processes (or posterior alar wings) and the caudal margin is deeply concave as in Petrolacosaurus (Reisz 1981). These processes do not appear to have contacted the basioccipital tubers through osseous contact; a cartilagenous contact may have been present.
Opisthotic. The opisthotic and prootic enclose most of the vestibular system laterally, but remain unossified medially (Figure 3,
Figure 4, and
Figure 6). The opisthotic and exoccipitals form the lateral borders of the metotic fissure, the common exit of the jugular vein, the glossopharyngeal nerve (CN IX) and the vagus nerve (CN X) (Rieppel 1993). The margins of the fenestra ovalis are poorly ossified, with the opisthotic and prootic forming the dorsal portion of the window.
Gow (1975) reported that the paroccipital process of the opisthotic articulated with the quadrate in a pocket formed by the supratemporal, similar to the condition in Proganochelys (Gaffney 1990). We could not confirm the presence of this pocket; however, the paroccipital processes do contact the quadrate extensively, unlike in some tangasaurids (Evans 1987). The right opisthotic was damaged slightly in its dorsomedial portion by preparation, whereas the left opisthotic is partly damaged by a crack resulting from taphonomic processes. The opisthotic clearly shows a large ventral ramus that contributes to the ossified wall of the braincase.
Prootic. Our HRXCT data confirms many of the characters described by
Evans (1987) for the prootic
(Figure 3,
Figure 4.3-4, and
Figure 6). On the mediodorsal surface, there is a large subarcuate fossa. However, there does appear to be a small rostral inferior process on the prootic dorsal to rostroventral contact with the basisphenoid in AMNH 5561.
Evans (1987) reported this process as being absent in TM 3603. The dorsal surface of the prootic lacks an extensive contact with the parietal, unlike the condition in turtles. There appears to be a weak ridge ventrally on the rostral ramus contacting the basisphenoid, but there is no prominent expanded crest (as in derived lepidosaurs and archosauromorphs).
Anatomy of the Osseous Labyrinth
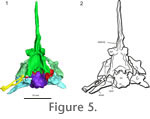
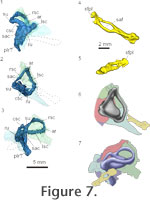 Using our HRXCT data, we examined the shape of the osseous labyrinth and stapes in Youngina (Figure 7.1-3). It shares a common chamber, with little separation of the tracts of the semicircular canals or the otolithic organs within the prootic and opisthotic. The sacculus likely occupied much of the chamber encased in the opisthotic and prootic, though it is difficult to fully assess the rostral extent of the saccular maculae in the HRXCT data. The caudal portion of chamber is filled by dense metallic precipitates that obscure the precise details of the shape of the saccular maculae. There appears to have been a lagenar (or possibly a postlagenar) recess that extended ventrally along the ventral ramus of the opisthotic. The rostral and lateral semicircular canals meet rostrally within the ampullary recess of the prootic. The caudal and lateral semicircular canals meet caudomedially within the recessus utriculi between the supraoccipital and the opisthotic. The rostral and caudal semicircular canals meet dorsal to the sacculus within the superior utriculus. The endolymphatic duct extends medially under the supraoccipital from the main body of the sacculus. Using our HRXCT data, we examined the shape of the osseous labyrinth and stapes in Youngina (Figure 7.1-3). It shares a common chamber, with little separation of the tracts of the semicircular canals or the otolithic organs within the prootic and opisthotic. The sacculus likely occupied much of the chamber encased in the opisthotic and prootic, though it is difficult to fully assess the rostral extent of the saccular maculae in the HRXCT data. The caudal portion of chamber is filled by dense metallic precipitates that obscure the precise details of the shape of the saccular maculae. There appears to have been a lagenar (or possibly a postlagenar) recess that extended ventrally along the ventral ramus of the opisthotic. The rostral and lateral semicircular canals meet rostrally within the ampullary recess of the prootic. The caudal and lateral semicircular canals meet caudomedially within the recessus utriculi between the supraoccipital and the opisthotic. The rostral and caudal semicircular canals meet dorsal to the sacculus within the superior utriculus. The endolymphatic duct extends medially under the supraoccipital from the main body of the sacculus.
The lateral semicircular canal has the greatest path diameter compared to the other two canals (6.77 mm; rostral semicircular canal: 4.79 mm, caudal semicircular canal: 4.35 mm). However, it does not trend as far laterally as the anterior canal in that the ratio of the length of the path diameter of the canal versus the greatest path radii of the canal is 3.62 compared to 2.01 in the rostral semicircular canal and 2.33 in the caudal semicircular canal. The lateral semicircular canal has a wider diameter than the rostral semicircular canal (1.12 mm compared to 1.05 mm), and the widest preserved portion of the caudal semicircular canal is 1.12 mm. The lateral and caudal canals are at right angles to each other, while the rostral and caudal canals are oriented at 84 degrees to each other, and the lateral and rostral canals are oriented at 66 degrees to each other.
Anatomy of the Columellar Apparatus
Our HRXCT data set allows examination of the complete stapes (or columella) in Youngina for the first time (Figure 7.4-6). In most modern reptiles, the stapes is ossified, while the extrastapes (or extracolumella) is cartilaginous. In Youngina, both appear ossified, as in Sphenodon and more primitive reptiles. In TM 3603, the left stapes is fragmented, and the right stapes preserves the footplate only (Evans 1987). In AMNH 5561, the right stapes is reasonably complete and appears to be attached to a lateral ossification, which we interpret as the ossified extrastapes. No suture was identified between these elements. The left stapes could not be identified.
Evans (1987),
Gow (1975) and
Olsen (1936) interpreted the stapes in Youngina as being slender and rodlike, but Carroll (1981) correctly interpreted it as being a massive rod, presumably retaining a support function for the dermal skull elements as the stapes contacts the suspensorium medially. There is no evidence of a dorsal process on the stapes, unlike the condition in captorhinids. There is a large stapedial foramen through which the stapedial artery would have passed, unlike the condition in most modern reptiles in which the stapedial artery passes around the stapes. This is similar to the primitive condition found in captorhinids (Heaton
1979).
Gow (1975) described this foramen as medially positioned in the stapes and "bounded by an extremely thin bridge which is bowed slightly outward." Our data support Gow's description of the stapedial foramen and the nature of its surrounding bone. It appears that the stapes and an ossified extrastapes were closely articulated without a definite joint, as in Sphenodon (Gans and Wever 1976). The extrastapedial portion appears to be pierced laterally by a foramen which could be homologous to Huxley's foramen, as present in crown-group diapsids (Laurin and Gauthier 2000).
Three-Dimensional Movies
In order to aid visualization of the braincase elements and their manner or articulation, we also present three Quicktime movies that allow rotation of the entire structure in lateral roll (Figure 8) and rostral-caudal yaw (Figure 9). A Quicktime movie of the osseous labyrinth with its surrounding bones depicted transparently is presented in
Figure 10.
|