| |
INTRODUCTION
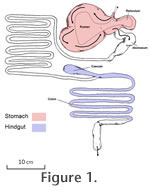 A challenge common to all herbivores is the processing of tough plant tissues, notably high in lignin and cellulose. Cellulose (a polysaccharide) is the major structural component of the plant cell wall, and it cannot be hydrolysed by the endogenous enzymes of vertebrates (Stevens and Hume 1995). It is only after symbiosis with cellulase-producing micro-organisms that sufficient nutritional content can be gained from this food source. Herbivorous mammals all utilise fermentation chambers in some portion of the gastrointestinal tract to maximise exposure of fibrous foods to these digestive bacterial agents. The fermentation chamber may be situated in the foregut area of the stomach (e.g., ruminating artiodactyls, hippos, colobine monkeys, sloths, muroid rodents, kangaroos, koalas) or in the hindgut area of the caecum and/or colon (e.g., perissodactyls, hyraxes, proboscideans, ateline monkeys, caviomorph rodents, rabbits, wombats). This study is concerned primarily with the differences between ruminating artiodactyls (antelope, cattle, deer, giraffe, camels, etc.), which are the only foregut fermenters to regurgitate their food (i.e., "chew the cud"), and perissodactyls (horses, rhinos, and tapirs) (Figure 1). A challenge common to all herbivores is the processing of tough plant tissues, notably high in lignin and cellulose. Cellulose (a polysaccharide) is the major structural component of the plant cell wall, and it cannot be hydrolysed by the endogenous enzymes of vertebrates (Stevens and Hume 1995). It is only after symbiosis with cellulase-producing micro-organisms that sufficient nutritional content can be gained from this food source. Herbivorous mammals all utilise fermentation chambers in some portion of the gastrointestinal tract to maximise exposure of fibrous foods to these digestive bacterial agents. The fermentation chamber may be situated in the foregut area of the stomach (e.g., ruminating artiodactyls, hippos, colobine monkeys, sloths, muroid rodents, kangaroos, koalas) or in the hindgut area of the caecum and/or colon (e.g., perissodactyls, hyraxes, proboscideans, ateline monkeys, caviomorph rodents, rabbits, wombats). This study is concerned primarily with the differences between ruminating artiodactyls (antelope, cattle, deer, giraffe, camels, etc.), which are the only foregut fermenters to regurgitate their food (i.e., "chew the cud"), and perissodactyls (horses, rhinos, and tapirs) (Figure 1).
Note: the opportunity for a confusion of terminology: the term "ruminant" can either refer to physiology, that of foregut fermentation combined with cud-chewing, or to a phylogenetic grouping of artiodactyls [subfamily Ruminantia] that excludes camelids (camels and lamas, in the artiodactyl subfamily Tylopoda). We will use the term "ruminant" in the physiological sense here, to include camelids.
Ruminating artiodactyls possess three non-absorptive chambers of the stomach (two in camelids) where food is stored and processed, followed by a digestive chamber called the abomasum, the equivalent of the true stomach in other mammals (Figure 1.1). The first (and largest) chamber is the rumen, which serves as the main fermentation "vat", where the huge numbers of bacteria and protozoans that break down cellulose are cultured. The products of fermentation (volatile fatty acids) account for the majority of the animal's nutritional requirement (up to 70% in cattle) (Schmidt-Nielson 1997). The acids are buffered by large volumes of saliva, containing dilute sodium bicarbonate (100-190 litres a day in cattle), and this also aids in maintaining an appropriate growing medium for the digestive microorganisms (Schmidt-Nielson 1997). This system is extremely efficient as the cellulose is fermented prior to the site of absorption in the small intestine, and additionally this breakdown of the plant cell wall means that the cell contents are released prior to the site of absorption. Ruminants also engage in a process termed "nitrogen cycling" whereby the ammonia produced by protein fermentation in the rumen is transported via the blood system to the liver, returned to the rumen as urea and then used for further bacterial growth. The overspill of bacteria into the abomasum then provides the animal with microbial protein as its protein source. As a result, ruminants can afford to be specialist feeders, given that all essential amino acids and many vitamins are synthesised by the bacteria (Schmidt-Nielson 1997). The process of thorough fermentation in the rumen, although allowing for a high degree of cellulose digestion, entails a long retention time of the digesta, and as a result food intake may be more limited than in nonruminants (Clauss et al. 2003).
Hindgut fermentation takes place mainly in the colon in perissodactyls and additionally in the enlarged caecum
(Figure 1.2), which act like fermentation chambers in much the same way as the
rumen does in ruminants. This arrangement presents the problem that the cellulose is not fermented until this point, and the volatile fatty acids must be absorbed in the colon, rather than in the small intestine. Hindgut fermenters have a shorter passage time than ruminants, and hence are less efficient in cellulose digestion, for which they compensate with a higher intake of food (Clauss et al. 2003, 2007, 2009b). Note that an additional problem for hindgut fermenters is that they must access the cell contents of the herbage prior to the fermentation of the cellulose in the hindgut. Although the products of cellulose fermentation can be absorbed in the colon, the enzyme-producing glands for the digestion of the sugars, fats and proteins of the cell contents are located in the small intestine. Some non-ungulate hindgut fermenters, such as rabbits and certain rodents, circumvent this problem by refection (eating the initially-produced faeces):
however, refection is not practiced by any ungulate (nor by hyraxes and elephants). Thus it must be the case, for hindgut fermenting ungulates, that the initial mastication of the food is sufficient to fracture the plant cell walls to release the cell contents prior to the site of cellulose fermentation (Janis et al. 2010).
Thus, hindgut fermenters face two functional problems with food comminution in which they differ from ruminants. Not only must they consume more food per day than a ruminant of similar size and diet, but they must also ensure that the cell walls are ruptured on initial food ingestion (while a ruminant can rely on fermentation to break down the cell walls). One would therefore predict that initial food mastication would be more prolonged and intensive in hindgut fermenters than in ruminants. Even though ruminants later regurgitate their food and chew it as cud, at this point the food has been softened by fermentation processes and may present a reduced load on the masticatory system (Fortelius 1985). Morphological studies do appear to show that hindgut fermenters have deeper jaws, larger areas for the insertion of masticatory muscles and greater cheek tooth occlusal area than ruminants (e.g.,
Turnbull 1970;
Janis 1990a;
Mendoza et al. 2002), which would accord with the hypothesis that hindgut fermenters experience a greater load on the masticatory system than ruminants. However, these observations have not been subjected to rigorous biomechanical analysis.
With regards to food ingestion, and digestive physiology in general, the usual comparison between hindgut fermenters and ruminants is between horses and cattle both medium to large-sized ungulates (~300 kg) with a similar diet of grass. Both animals have been the subject of many agricultural studies, and while other ruminants have also been studied in this fashion (sheep, deer, llamas, etc.) studies of other extant hindgut fermenting ungulates (rhinos, tapirs, hyraxes, elephants) are few in number. Direct comparisons of food intake behaviour between horses and cows are rare, although horses do seem to show longer grazing times and/or higher food intake than cattle (Arnold 1984;
Duncan et al. 1990;
Menard et al. 2002). In addition, it has been shown that while ruminants initially swallow large particles, which are later reduced in size via rumination (Clauss et al. 2009a), the faecal particle size in horses is relatively small, despite the fact that they only chew their food once (Fritz et al. 2009). One pilot study has directly compared the ingestion behaviour of horses and cows (Janis et al. 2010). This study suggests, although it cannot statistically be demonstrated (because of small sample sizes), that horses chew their food longer than cows on initial ingestion, and that this difference is more pronounced with forage of increasing fibre content.
Here we test the hypothesis that, as hindgut fermenters most likely chew their food more on initial ingestion than ruminants, and also must process more food per day, then hindgut fermenters should possess features that increase mandibular robustness relative to ruminants to deal with prolonged, cyclical stress and strain produced during mastication. As mentioned above, morphological observations and measurements suggest that this case holds true, but has not been tested within a biomechanical framework. For this study it is assumed that a robust jaw would exhibit less deformation under a set load than a more gracile one, and thus robustness is defined here by the stress observed in the jaw when experiencing quasi-feeding loads. More robust jaws would be expected to experience lower stresses. In this study we estimate Von Mises stress, which is a function of the three principle stress directions formed under loading conditions that distort a material.
|