 A new skull of an early diverging rorqual (Balaenopteridae, Mysticeti, Cetacea) from the late Miocene to early Pliocene of Yamagata, northeastern Japan
A new skull of an early diverging rorqual (Balaenopteridae, Mysticeti, Cetacea) from the late Miocene to early Pliocene of Yamagata, northeastern Japan
Article number: 23(1):a12
https://doi.org/10.26879/1002
Copyright Society for Vertebrate Paleontology, March 2020
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Submission: 25 May 2019. Acceptance: 20 February 2020.
ABSTRACT
The family of rorquals and humpback whales, Balaenopteridae includes the largest living animal on Earth, the blue whale Balaenoptera musculus. Many new taxa have been named, but not many from the western Pacific, except Miobalaenoptera numataensis from Japan. Here we describe an early balaenopterid, cf. M. numataensis from a late Miocene to early Pliocene sediment in Yamagata Prefecture, northeastern Japan. The species has a straight and sharp lateral ridge of the fovea epitubaria at the ventral surface of the periotic, and a dorsoventrally thin pars cochlearis. The new specimen provides knowledge of supposed ontogenetic variation and periotic morphology in poorly known fossil balaenopterids.
Yoshihiro Tanaka. Osaka Museum of Natural History, Nagai Park 1-23, Higashi-Sumiyoshi-ku, Osaka, 546-0034, Japan. tanaka@mus-nh.city.osaka.jp
and Hokkaido University Museum, Kita 10, Nishi 8, Kita-ku, Sapporo, Hokkaido 060-0810 Japan, Numata Fossil Museum, 2-7-49, Minami 1, Numata town, Hokkaido 078-2225 Japan
Kazuo Nagasawa. Yamagata Prefectural Touohgakkan Junior and Senior High School. 1-7-1 Chuo-Minami, Higashine City, Yamagata Prefecture, Japan 999-3730. snagasawak@pref-yamagata.ed.jp
Yojiro Taketani. Aizuwakamatsu City, Fukushima Prefecture, Japan. yojirotak.11-22@sa2.so-net.ne.jp
Keywords: rorquals; Balaenopteridae; Noguchi Formation; Furukuchi Formation; Miobalaenoptera numataensis; ontogenetic variation
Final citation: Tanaka, Yoshihiro, Nagasawa, Kazuo, and Taketani, Yojiro. 2020. A new skull of an early diverging rorqual (Balaenopteridae, Mysticeti, Cetacea) from the late Miocene to early Pliocene of Yamagata, northeastern Japan. Palaeontologia Electronica, 23(1):a12. https://doi.org/10.26879/1002
palaeo-electronica.org/content/2020/2943-fossil-rorqual-from-japan
Copyright: March 2020 Society of Vertebrate Paleontology.
This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
creativecommons.org/licenses/by/4.0
INTRODUCTION
The family of rorquals and humpback whales, Balaenopteridae includes the largest living animal on Earth, the blue whale Balaenoptera musculus. Many new taxa have been named from Europe (Van Beneden, 1880; Strobel, 1881; Sacco, 1890; Bisconti, 2007a, 2007b, 2010; Bosselaers and Post, 2010; Bisconti and Bosselaers, 2016) and the East Coast of the U.S. (Whitmore and Kaltenbach, 2008). The Pacific also provides many of nominal species (Cope, 1872; Kellogg, 1922; Hanna and McLellan, 1924; Dathe, 1983; Deméré, 1986; Pilleri, 1989; Zeigler et al., 1997; Boessenecker, 2013; Marx and Kohno, 2016), but most of them are known from the eastern Pacific, except Miobalaenoptera numataensis from Japan (Tanaka and Watanabe, 2019a). From Japan, some unnamed balaenopterids were published, such as several Balaenopteridae indet. from Miocene sediments (Nagasawa, 1995; Hasegawa et al., 2002; Kohno et al., 2007; Sato et al., 2010; Kimura et al., 2015), and several Burtinopsis sp. (Hatai et al., 1963; Oishi et al., 1985; Oishi, 1988, see also Oishi and Hasegawa, 1995a), aff. Balaenoptera borealis (Hasegawa et al., 1988), two “Megaptera” sp. (Oishi and Hasegawa, 1995b, Nagasawa, 1999, see also Ichishima, 2005), Balaenoptera sp. (Oishi, 1994), Sibbaldus sp. (Oishi, 1997) and several Balaenopteridae indet. (Oishi, 1987; Iwaki Educational and Cultural Corporation. 1989; Oishi and Hasegawa, 1995b; Kimura and Kanaya, 1996; Nagasawa, 1999) from Neogene sediments.
Our knowledge on the ontogenetic variation among fossil cetaceans is still limited (Tanaka and Watanabe, 2019b). There are many extant cetacean ontogeny studies (Kasuya, 1973; Perrin, 1975; Ito and Miyazaki, 1990; Bisconti, 2001; Galatius and Kinze, 2003; de Buffrénil et al., 2004; Kurihara and Oda, 2009; Chen et al., 2011; Galatius et al., 2011; Walsh and Berta, 2011; Cozzi et al., 2012, 2015; Nakamura et al., 2012; Nakamura and Kato, 2014; Tsai and Fordyce, 2014; Hampe et al., 2015; Moran et al., 2015). But, the number of reports on fossil cetacean ontogeny is still limited (Uhen, 2004; Gutstein et al., 2009; El Adli et al., 2014; Boessenecker and Fordyce, 2015; Tanaka, 2016; Tanaka and Ichishima, 2016; Tanaka and Watanabe, 2019b). Here we describe an early member of the family Balaenopteridae: cf. Miobalaenoptera numataensis from a late Miocene to early Pliocene sediment in Yamagata Prefecture, Japan providing additional insight into the ontogenetic variation and periotic morphology of extinct balaenopterids.
MATERIAL AND METHODS
We follow the anatomical terms of Mead and Fordyce (2009) for the skull and periotic.
Abbreviations. NFL, Numata Fossil Museum, Numata Town, Hokkaido, Japan; USNM, Department of Paleobiology, National Museum of Natural History, Smithsonian Institution, Washington, D.C., USA; YPM, Yamagata Prefectural Museum, Japan.
Referred specimen. YPM 11851 includes the skull (parietal, alisphenoid, pterygoid, squamosal, exoccipital, basioccipital, supraoccipital, fused presphenoid and basisphenoid) and periotics in situ. The specimen was collected by Sadao Ito and Kazuo Nagasawa in 1992.
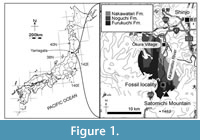 Locality. YPM 11851 was found as a float at the Akamatsu River in Ōkura Village, Yamagata Prefecture, Japan. The site is about 10 km south from the main residential area of the village (Figure 1): latitude 38°35′13.8″N, longitude 140°13′03.2″E.
Locality. YPM 11851 was found as a float at the Akamatsu River in Ōkura Village, Yamagata Prefecture, Japan. The site is about 10 km south from the main residential area of the village (Figure 1): latitude 38°35′13.8″N, longitude 140°13′03.2″E.
Horizon and age. The matrix of YPM 11851 is light gray tuffaceous sandstone with sea shell fragments. At the locality of YPM 11851 and the upper stream of the Akamatsu River, two sedimentary rocks (the Noguchi and Furukuchi Formations) are exposed (Tamiya and Applied Geological Society of Yamagata, 2016). Depositional facies of these formations are varied among each formation, and it is difficult to identify the original formation based on facies. Thus, both the Noguchi and Furukuchi Formations could be the original horizon for YPM 11851. Microfossils (planktonic foraminiferas and radiolarians) were searched from the matrix of YPM 11851 for the age assignment, but they could not be found. The ages of the Noguchi and Furukuchi Formations are from the late Miocene to early Pliocene (Moriya et al., 2008; Tamiya and Applied Geological Society of Yamagata, 2016). From the Akamatsu River near the locality, whale mandibles and a rib (Nagasawa et al., 2002), a vertebra (Nagasawa, 1993), and a sperm whale tooth (Nagasawa et al., 2009) were reported.
SYSTEMATIC PALEONTOLOGY
Order CETACEA Brisson, 1762
Unranked taxon NEOCETI Fordyce and de Muizon, 2001
Suborder MYSTICETI Gray, 1864
Family BALAENOPTERIDAE Gray, 1864
cf. Miobalaenoptera numataensis Tanaka and Watanabe, 2019
(Figure 2, Figure 3, Figure 4, Figure 5, Table 1 and Table 2)
Remarks. YPM 11851 is comparable to Miobalaenoptera numataensis by having similarities such as a straight and sharp lateral lateral ridge of the fovea epitubaria (Character 273 in this study, see discussion), a dorsoventrally thin pars cochlearis, a straight and horizontally expanded subtemporal crest of the squamosal, and strongly posterolaterally diverged basioccipital crest, which were used as diagnoses for M. numataensis in Tanaka and Watanabe (2019a). In this study, YPM 11851 is identified as cf. M. numataensis preliminary, because there are several differences from the holotype of M. numataensis (see discussion and also Table 3). It cannot be identified as the species because of limited knowledge on ontogenetic and other variations of the species.
Emended diagnoses for Miobalaenoptera numataensis. Minor emendation including an identification of the lateral ridge of the fovea epitubaria and adding measurements is made here. Among the Balaenopteridae, Miobalaenoptera numataensis uniquely has the lateral ridge of the fovea epitubaria, which is straight and sharp, running to the anterior pedicle of the ventral surface of the periotic (see discussion). The ridge is located more anterior than that of the modern balaenopterids. M. numataensis also uniquely has a dorsoventrally thin pars cochleais (especially medial edge). The medial edge of the pars cochlearis is thinner than the diameter of the internal acoustic meatus (9.1 mm thick and 12.1 mm in diameter, respectively, on the holotype). Tanaka and Watanabe (2019a) mentioned differences with other balaenopterids as below. M. numataensis is different from other balaenopterids except "Balaenoptera" ryani by a feature, an incipient lateral tuberosities of the periotic. M. numataensis is different from other balaenopterids except “Megaptera” miocaena, Protororquals cuvieri, and Megaptera novaeangliae by features, a slender zygomatic process, and a strongly excavated lateral margin of the orbit in dorsoventral view. M. numataensis is different from other balaenopterids except Fragilicetus velponi by a feature, strongly diverged basioccipital crests. M. numataensis is different from the other extinct balaenopterids except “Megaptera” hubachi by a feature, a broadly or more or less horizontally expanded subtemporal crest. M. numataensis is different from the extant taxa and “Megaptera” miocaena by a feature, a deep suprameatal fossa of the periotic. M. numataensis is different from the extinct taxa except Fragilicetus velponi by a feature, a long caudal tympanic process of the periotic. Here, we add differences between M. numataensis and supposed closely related Incakujira anillodefuego. M. numataensis is different from I. anillodefuego and also Diunatans luctoratemergo by some features, higher basioccipital crests, and straight subtemporal crest in ventral view. M. numataensis is different from I. anillodefuego by several features such as, a more anteriorly located foramen pseudovale, a strongly excavated orbit in dorsoventral view, more robust zygomatic process, larger postorbital process in lateral view, straight ventral margin of the anterior tip of the zygomatic process, a more slender caudal process of the periotic, and deeper area lateral from the fenestra rotunda and dorsal from the posteroventral margin of the pars cochlearis. M. numataensis is different from D. luctoratemergo by several features such as, more laterally projected basioccipital crest, a blunt anterior tip of the anterior process of the periotic in mediolateral view, dorsoventrally thinner and more flat pars cochlearis.
GENERAL DESCRIPTION
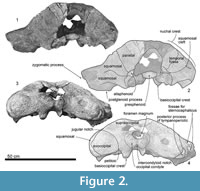 Ontogeny. YPM 11851 shows clear sutures between the squamosal/parietal, squamosal/pterygoid, and exoccipital/squamosal. The suture between squamosal/pterygoid is closed, but still visible. The joint between the basioccipital/exoccipital, and exoccipital/supraoccipital are closed completely. The basioccipital and basisphenoid border is closed, but still visible. The presphenoid and basisphenoid border is closed completely. All of extant mysticete show the same sequence of ossification from the joints of the basioccipital/exoccipital firstly, basioccipital/basisphenoid secondly, and the supraoccipital/exoccipital finally (Walsh and Berta, 2011). The ontogenetic stage of YPM 11851 is not certain, but is thought to be juvenile based on opened joints as mentioned above, and not a calf based on the fused supraoccipital/exoccipital joint.
Ontogeny. YPM 11851 shows clear sutures between the squamosal/parietal, squamosal/pterygoid, and exoccipital/squamosal. The suture between squamosal/pterygoid is closed, but still visible. The joint between the basioccipital/exoccipital, and exoccipital/supraoccipital are closed completely. The basioccipital and basisphenoid border is closed, but still visible. The presphenoid and basisphenoid border is closed completely. All of extant mysticete show the same sequence of ossification from the joints of the basioccipital/exoccipital firstly, basioccipital/basisphenoid secondly, and the supraoccipital/exoccipital finally (Walsh and Berta, 2011). The ontogenetic stage of YPM 11851 is not certain, but is thought to be juvenile based on opened joints as mentioned above, and not a calf based on the fused supraoccipital/exoccipital joint.
Parietal. The parietal forms the anteromedial part of the temporal fossa (Figure 2 and Figure 4), which is flat and vertical. The parietal/squamosal suture is opened.
Alisphenoid. The alisphenoid is exposed ventral to the parietal, anterior to the squamosal and dorsal to the pterygoid (Figure 2.2), and is anteroposteriorly long (45.0 mm+ long and 32.0 mm high) even though its anterior part is broken away. A ventral exposure of the alisphenoid can be seen in the pterygoid sinus fossa as Tanaka and Taruno (2017) described it on a specimen of Balaenoptera edeni. The alisphenoid places posterior to the squamosal and lateral to the pterygoid.
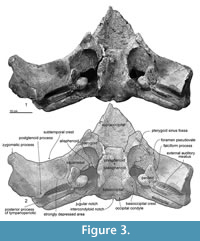 Pterygoid. The pterygoid is located anteromedial to the squamosal, and forms the anteromedial part of the pterygoid sinus fossa (Figure 3). Posteriorly, the pterygoid forms for the foramen pseudovale (Marx et al., 2016, Figure 3.4) with the squamosal. The ventral surface of the pterygoid is damaged.
Pterygoid. The pterygoid is located anteromedial to the squamosal, and forms the anteromedial part of the pterygoid sinus fossa (Figure 3). Posteriorly, the pterygoid forms for the foramen pseudovale (Marx et al., 2016, Figure 3.4) with the squamosal. The ventral surface of the pterygoid is damaged.
Squamosal. The squamosal has a broken base of the zygomatic process is thick (Figure 2, Figure 3, Figure 4). The lateral surface of the zygomatic process has two anteroposteriorly long triangular shallow fossae, which are for the sternocephalicus. Medial to the zygomatic process, the squamosal has a transversely long sigmoidal squamosal cleft (about 170.0 mm wide), running from the alisphenoid, squamosal and pterygoid junction (Figure 4.4).
In ventral view (Figure 3), the squamosal has a sharp falciform process, which runs anteroposteriorly just lateral to the periotic. The anterior end of the falciform process forms the lateral border of the foramen pseudovale (about 260.0 mm wide). Medial to the foramen pseudovale, the pterygoid sinus fossa shows the squamosal/pterygoid suture is almost closed but is visible. An area, lateral to the falciform process and posterior to the subtemporal crest, is strongly depressed. The posterior end of the squamosal forms a dorsoventrally deep external auditory meatus (about 25.0 mm long, 220.0+ mm wide, and 600.0+ mm maximum depth) (Figure 4.6). The squamosal connects to the anterior, posterior and dorsal surfaces of the posterior process of the tympanoperiotic (Figure 4.4, 4.6).
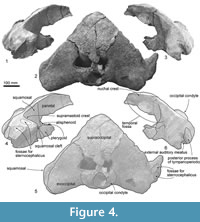 Exoccipital. The exoccipital is fused to the supraoccipital and basioccipital completely, and the joints are obliterated. The lateral end of the exoccipital is rounded (Figure 2.3, 2.4). The posterolateral end of the exoccipital projects posterolaterally in dorsal view (Figure 4.2, 4.5). In ventral view (Figure 3), the anterior and posterior borders of the exoccipital are parallel at the medial portion and anteroposteriorly shorter at the lateral end.
Exoccipital. The exoccipital is fused to the supraoccipital and basioccipital completely, and the joints are obliterated. The lateral end of the exoccipital is rounded (Figure 2.3, 2.4). The posterolateral end of the exoccipital projects posterolaterally in dorsal view (Figure 4.2, 4.5). In ventral view (Figure 3), the anterior and posterior borders of the exoccipital are parallel at the medial portion and anteroposteriorly shorter at the lateral end.
Supraoccipital. The supraoccipital is a wide triangle (Figure 4), and its lateral border is a very weakly curved nuchal crest. The dorsal surface of the supraoccipital is depressed medially.
Presphenoid and basisphenoid. The presphenoid and basisphenoid are fused, and lie anterior to the basioccipital and medial to the pterygoid (Figure 3). The anterior border of the presphenoid shows an open joint with the vomer. The basisphenoid is an anteriorly wider trapezoidal. The lateral borders are the pharyngeal crests.
Basioccipital. The basioccipital is located posterior to the basisphenoid with a fused but visible joint, and is posteriorly wide in ventral view (Figure 3). The basioccipital crest is transversely wide (42.0 mm thick, 95.0 mm long, and 65.0 mm wide). The basioccipital crests widen posteriorly.
 Exoccipital. The occipital condyle does not protrude posteriorly much and does not have a clear lateral border. The foramen magnum is dorsoventrally higher oval. Lateral to the occipital condyle, a wide jugular notch (about 20.0 mm wide) runs anteromedial to posterolateral.
Exoccipital. The occipital condyle does not protrude posteriorly much and does not have a clear lateral border. The foramen magnum is dorsoventrally higher oval. Lateral to the occipital condyle, a wide jugular notch (about 20.0 mm wide) runs anteromedial to posterolateral.
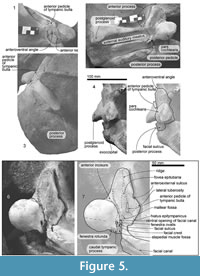
Periotic. Both periotics are in situ (Figure 5, Table 2). The periotic has a short, narrow, and dorsoventrally high anterior process, which is triangular in ventral view, and blunt square in medial view. A dorsoventrally shallow pars cochlearis (16.5 mm deep) is longer than the anterior process. A robust posterior process has an anteroposteriorly long lateral end.
The anterior process projects anteriorly and has an anteroposteriorly long ridge on the ventral surface, which is the lateral margin of the fovea epitubaria. Posterior to the ridge, there is an anteroposteriorly long anterior pedicle of the tympanic bulla (16.5 mm long). The lateral tuberosity projects laterally and is located lateral to the anterior pedicle of the tympanic bulla. Anterior to the lateral tuberosity and medial to the falciform process, there is an excavation, which is a deeply excavated anteroexternal sulcus (about 40.0 mm posterior from the anterior end of the anterior process). A deep and anteriorly opened crescent shaped mallear fossa (6.5 mm wide) is present posteromedial to the anterior pedicle.
An anteroposteriorly long pars cochlearis (52.0 mm long except the caudal tympanic process, 53.5+ mm wide) has a flat ventral surface with a transversely long blunt ridge at the level of the fenestra ovalis and posterior end of the lateral tuberosity. The medial margin of the pars cochlearis is sigmoidal in ventral view. A broken base of the caudal tympanic process is located posterior to the fenestra ovalis, and the preserved part is robust. Dorsomedial to the caudal tympanic process, a small fenestra rotunda opens posteriorly (7.1 mm in diameter). The medial surface of the pars cochlearis is covered by the matrix.
A small ventral opening of the facial canal (2.5 mm in diameter) opens and continues to a shallow facial sulcus (9.0 mm long). An anteroposteriorly long elliptical fenestra ovalis (5.5 mm in length and 1.5 mm in width) is located posteromedial to the ventral opening of the facial canal. The facial crest is mediolaterally thick and is separated from the facial canal and fenestra ovalis.
The posterior process of the tympanoperiotic is mediolaterally wide and anteroposteriorly long (Table 2). The posterior process at the lateral end is dorsoventrally higher elliptical and forms the lateral surface of the skull. The posterior process is surrounded by the squamosal except on the ventral side. There is a long and shallow facial canal, running transversely on the ventral surface of the posterior process. At the anterior end of the posterior process, anterior to the facial sulcus, there is a mediolaterally wide posterior pedicle of the tympanic bulla. A small fenestra rotunda opens posteriorly.
PHYLOGENETIC ANALYSIS
The phylogenetic position of YPM 11851 is analyzed using the datamatrix of Tanaka and Watanabe (2019a), which was derived from the one of Marx et al. (2017). This study adds a new character and specimen (YPM 11851) to the previous datamatrix, containing 273 morphological characters and 99 taxa (see Appendix 1, Appendix 2, Appendix 3) with minor modifications (see Appendix 4). Percentages of coded data of YPM 11851 are 17.5 % (including soft tissue characters) and 18.0 % (excluding soft tissue).
The matrix was managed with Mesquite 2.75 (Maddison and Maddison, 2011), then analysis was performed using TNT version 1.5 (Goloboff and Catalano, 2016). All of the characters were treated as unordered and unweighted, with backbone constraint of extant taxa, based on a topology of the molecular tree by McGowen et al. (2009). The analysis used New Technology Search, with recover minimum length trees = 1000 times.
RESULTS
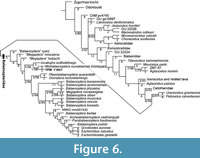 The phylogenetic analysis shows 1994 shortest trees of 1329 steps each. The 50% majority rule consensus tree (Figure 6 and Appendix 5) places YPM 11851 in a clade with the holotype of Miobalaenoptera numataensis (NFL 18) among the Balaenopteridae. The topology among the Balaenopteridae is more or less the same as that of Tanaka and Watanabe (2019a), except for forming a clade of Incakujira anillodefuego and (M. numataensis + YPM 11851). A higher level branching pattern of this study is gained as (((Balaenopteridae + a clade of Uranocetus gramensis and Pelocetus calvertensis) Cetotheriidae) a clade of Isanacetus, Parietobalaena and related taxa). The pattern is different from that of Tanaka and Watanabe (2019a) in terms of branching order of the Cetotheriidae and the clade of Isanacetus, Parietobalaena and related taxa.
The phylogenetic analysis shows 1994 shortest trees of 1329 steps each. The 50% majority rule consensus tree (Figure 6 and Appendix 5) places YPM 11851 in a clade with the holotype of Miobalaenoptera numataensis (NFL 18) among the Balaenopteridae. The topology among the Balaenopteridae is more or less the same as that of Tanaka and Watanabe (2019a), except for forming a clade of Incakujira anillodefuego and (M. numataensis + YPM 11851). A higher level branching pattern of this study is gained as (((Balaenopteridae + a clade of Uranocetus gramensis and Pelocetus calvertensis) Cetotheriidae) a clade of Isanacetus, Parietobalaena and related taxa). The pattern is different from that of Tanaka and Watanabe (2019a) in terms of branching order of the Cetotheriidae and the clade of Isanacetus, Parietobalaena and related taxa.
DISCUSSION
Lateral Ridge of the Fovea Epitubaria
The holotype of Miobalaenoptera numataensis (NFL 18) and cf. M. numataensis (YPM 11851) have similarities such a dorsoventrally thin pars cochlearis, a straight and horizontally expanded subtemporal crest of the squamosal, strongly posterolaterally diverged basioccipital crest, and a sharp anteroposteriorly long straight ridge (the lateral ridge of the fovea epitubaria) at the ventral surface of the anterior process of the periotic (Character 273 in this study). The ridge is a lateral margin of the fovea epitubaria. Therefore, a more basal species, such as Whakakai waipata (Tsai and Fordyce, 2016, figure 3) shows the same relationships of structures with NFL 18 and YPM 11851 (there is an anteriorly narrow triangular fovea epitubaria, medial to the ridge and anterior to the mallear fossa), and this result allows the consideration of the homology of the ridge of NFL 18, YPM 11851, and other species.
The condition of the lateral ridge of the fovea epitubaria on Miobalaenoptera numataensis (NFL 18) and YPM 11851 is straight and sharp, running to the anterior pedicle and is located more anterior to the modern Balaenoptera spp. This newly added character shows mosaic states distribution among the Mysticeti. For example, among the Cetotheriidae, the state of having a straight sharp ridge on the center of the anterior process can be seen on that of Tiucetus rosae (Marx et al., 2017). Other members of the Cetotheriidae, such as Herpetocetus morrowi (see El Adli et al., 2014) and Herpetocetus sp. (USNM 299652 of Geisler and Luo, 1996, and NFL 2083 of Tanaka and Watanabe, 2019b) also show a sharp ridge, but one that is located lateral to the central axis of the anterior process. Other members of the Cetotheriidae, such as Brandtcetus chongulek (Gol’din and Startsev, 2014) and Piscobalaena nana (Bouetel and de Muizon, 2006) show a sharp ridge, but a curved one. On the other hand, among the Cetotheriidae, some members show the ridge as weak and curved, such as Mithridatocetus eichwaldi (Gol’din and Startsev, 2017).
The extant balaenopterids show blunt lateral ridges of the fovea epitubaria, not sharp one of M. numataensis. The modern balaenopterid ridges exist only in the posterior portion of the anterior process of the periotic. On the other hand, the lateral ridge of M. numataensis runs almost the entire part of the anterior process in ventral view.
Among the fossil balaenopterids, “Megaptera” miocaena of Kellogg (1922), Parabalaenoptera baulinensis of Zeigler et al. (1997), Fragilicetus velponi of Bisconti and Bosselaers (2016), and Diunatans luctoretemergo of Bosselaers and Post (2010), show weak or blunt lateral ridges of the fovea epitubaria. Plesiobalaenoptera quarantellii of Bisconti (2010), shows a ridge with the same condition as the Miobalaenoptera numataensis (sharp and continues to the anterior pedicle), but can be distinguished from M. numataensis by having a larger lateral tuberosity and wider anterior process. Incakujira anillodefuego of Marx and Kohno (2016), shows no ridge anterior to the broken base of the anterior pedicle (Marx and Kohno, 2016, figure 10). “Balaenoptera” ryani of Hanna and McLellan (1924) also shows no ridge.
The functional meaning of having the sharp, straight and long lateral ridges of the fovea epitubaria is unknown. The lateral ridge of the fovea epitubaria is related to the position, size, and/or shape of the accessory ossicle, which suggests that the variations of the ridge are possibly relate to hearing.
Comparison between Incakujira anillodefuego and YPM 11851 + Miobalaenoptera numataensis
The result of phylogenetic analysis supports that (Incakujira anillodefuego [YPM 11851 + the holotype of Miobalaenoptera numataensis]) as a clade. However, there are several differences between them. I. anillodefuego differs from M. numataensis and YPM 11851 by having lower basioccipital crests, anteriorly bulged subtemporal crest in ventral view, and more posteriorly located foramen pseudovale (M. numataensis and YPM 11851 have the foramen pseudovale at the anterior margin of the subtemporal crest). I. anillodefuego differs from YPM 11851 by having a posteriorly suddenly widen nuchal crest, and well posteriorly projected occipital condyle. I. anillodefuego differs from the holotype of M. numataensis in several features such as having a weakly excavated orbit in dorsoventral view, a more slender zygomatic process, smaller postorbital process in lateral view, anteroventrally bent anterior tip of the zygomatic process, longer and robust caudal process of the periotic (YPM 11851 does not preserve the process), and shallow area lateral from the fenestra rotunda and dorsal from the posteroventral margin of the pars cochlearis (M. numataensis and YPM 11851 show deeper areas). Thus, I. anillodefuego is taxonomically different from M. numataensis and YPM 11851.
Differences between YPM 11851 and the holotype of Miobalaenoptera numataensis
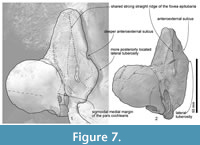 YPM 11851 is identified as cf. M. numataensis preliminary, and cannot be identified as the species, because of limited knowledge on ontogenetic and other variations of the species. Between YPM 11851 and NFL 18, there are some morphological differences as noticed below (see also Table 3). YPM 11851 has a dorsoventrally deeper (or higher) basioccipital crest, a narrower area between the basioccipital crests, a deeper area on the anteromedial portion of the ventral surface of the squamosal (NFL 18 shows that as a flat area), a dorsoventrally deeper anterior process of the periotic (NFL 18 shows that as very shallow), a more posteriorly located lateral tuberosity (Character 157) (NFL 18 shows that much more anterior), a deeply excavated anteroexternal sulcus locating about 40.0 mm posterior from the anterior end of the anterior process (NFL 18 shows the sulcus more anteriorly, about 24.0 mm posterior from the anterior end of the anterior process) (Figure 7), a dorsoventrally thicker pars cochlearis (a thinner pars cochlearis was stated as a diagnosis for NFL 18), and a sigmoidal lateral margin of the pars cochlearis in ventral view (NFL 18 shows a straight medial margin) (Figure 7). In addition, two analyzed characters of the anterior process of the periotic are coded differently between NFL 18 and YPM 11851 (Characters 148 and 151: anterior process shape and length) (see Table 3), because YPM 11851 shows a larger anterior process than that of NFL 18.
YPM 11851 is identified as cf. M. numataensis preliminary, and cannot be identified as the species, because of limited knowledge on ontogenetic and other variations of the species. Between YPM 11851 and NFL 18, there are some morphological differences as noticed below (see also Table 3). YPM 11851 has a dorsoventrally deeper (or higher) basioccipital crest, a narrower area between the basioccipital crests, a deeper area on the anteromedial portion of the ventral surface of the squamosal (NFL 18 shows that as a flat area), a dorsoventrally deeper anterior process of the periotic (NFL 18 shows that as very shallow), a more posteriorly located lateral tuberosity (Character 157) (NFL 18 shows that much more anterior), a deeply excavated anteroexternal sulcus locating about 40.0 mm posterior from the anterior end of the anterior process (NFL 18 shows the sulcus more anteriorly, about 24.0 mm posterior from the anterior end of the anterior process) (Figure 7), a dorsoventrally thicker pars cochlearis (a thinner pars cochlearis was stated as a diagnosis for NFL 18), and a sigmoidal lateral margin of the pars cochlearis in ventral view (NFL 18 shows a straight medial margin) (Figure 7). In addition, two analyzed characters of the anterior process of the periotic are coded differently between NFL 18 and YPM 11851 (Characters 148 and 151: anterior process shape and length) (see Table 3), because YPM 11851 shows a larger anterior process than that of NFL 18.
The width of the squamosal and occipital bones are grown on Balaenoptera acutrostrata (Nakamura and Kato, 2014). Thus some of these differences between YPM 11851 and NFL 18 might cause of ontogenetic variation. The anterior process is known as a part largely changed through growth on an extant balaenopterid study (Bisconti, 2001). Based on these conditions, YPM 11851 possibly shows an older state than NFL 18 as above. However, possible ontogenetic stages of these specimens are confusing. Cranial joints (parietal/squamosal, and basisphenoid/basioccipital joints) suggest that YPM 11851 is younger than NFL 18. Especially the parietal/squamosal of YPM 11851 is opened, but that of NFL 18 is closed and invisible.
Other differences between YPM 11851 and NFL 18 on the basioccipital and also squamosal (Table 3) can be seen on modern Balaenoptera acutorostrata (an adult of 25 years old, 8.4 m total long at Toyohashi Museum of Natural History, and a juvenile at Hokkaido University Botanic Garden). This is a limited examination, but it suggests that morphological variations between YPM 11851 and NFL 18 on the basioccipital and also squamosal are possibly ontogenetic variations.
The number of M. numataensis specimens is simply not enough to consider ontogenetic variations. YPM 11851 has similarities with NFL 18, but cannot identify as the same species in this study, but add morphological information including supposed ontogenetic variation. A third similar specimen will be very informative to picture it.
CONCLUSION
Here we describe an early balaenopterid, cf. Miobalaenoptera numataensis (YPM 11851) from a late Miocene to early Pliocene sediment in Yamagata Prefecture, Japan. The new specimen is comparable to M. numataensis by having a straight and sharp lateral ridge of the fovea epitubaria at the ventral surface of the periotic, and a dorsoventrally thin pars cochlearis. YPM 11851 shows differences from the holotype of M. numataensis such as having a dorsoventrally deeper basioccipital crest, a narrower area between the basioccipital crests, a deeper area on the anteromedial portion of the ventral surface of the squamosal, a dorsoventrally deeper anterior process of the periotic, a more posteriorly located lateral tuberosity, a deeply excavated anteroexternal sulcus locating about 40.0 mm posterior from the anterior end of the anterior process, a dorsoventrally thicker pars cochlearis, and a sigmoidal medial margin of the pars cochlearis in ventral view. Morphological variations among fossil species have been merely reported. Some of these variations on the periotic are possibly ontogenetic variations. The new specimen expands morphological variations, but the third comparable specimen might solve the question on variations.
ACKNOWLEDGEMENTS
We thank S. Ito (Shinjo City, Yamagata Prefecture) for collecting the specimen, students of Yamagata University for preparation. We also thank D. Savage (Touohgakkan Senior High School) for revising our manuscript. Thanks go to T. Kimura (Gunma Museum of Natural History), C-H Tsai (National Taiwan University) and an anonymous reviewer for giving constructive comments. Y. Tanaka thanks M. Kato (Botanic Garden, Hokkaido University) and K. Yasui (Toyohashi Museum of Natural History) for giving access to the comparative materials.
REFERENCES
Bisconti, M. 2001. Morphology and postnatal growth trajectory of rorqual petrosal. Italian Journal of Zoology, 68:87-93. https://doi.org/10.1080/11250000109356390
Bisconti, M. 2007a. A new basal balaenopterid whale from the Pliocene of northern Italy. Palaeontology, 50:1103-1122. https://doi.org/10.1111/j.1475-4983.2007.00696.x
Bisconti, M. 2007b. Taxonomic revision and phylogenetic relationships of the rorqual-like mysticete from the Pliocene of Mount Pulgnasco, northern Italy (Mammalia, Cetacea, Mysticeti). Palaeontographia Italica, 91:85-108.
Bisconti, M. 2010. A new balaenopterid whale from the Late Miocene of the Stirone River, northern Italy (Mammalia, Cetacea, Mysticeti). Journal of Vertebrate Paleontology, 30:943-958. https://doi.org/10.1080/02724631003762922
Bisconti, M. and Bosselaers, M. 2016. Fragilicetus velponi: a new mysticete genus and species and its implications for the origin of Balaenopteridae (Mammalia, Cetacea, Mysticeti). Zoological Journal of the Linnean Society, 177:450-474. https://doi.org/10.1111/zoj.12370
Boessenecker, R.W. 2013. A new marine vertebrate assemblage from the Late Neogene Purisima Formation in Central California, part II: pinnipeds and cetaceans. Geodiversitas, 35:815-940. https://doi.org/10.5252/g2013n4a5
Boessenecker, R.W. and Fordyce, R.E. 2015. Anatomy, feeding ecology, and ontogeny of a transitional baleen whale: a new genus and species of Eomysticetidae (Mammalia: Cetacea) from the Oligocene of New Zealand. PeerJ, 3:e1129. https://doi.org/10.7717/peerj.1129
Bosselaers, M. and Post, K. 2010. A new fossil rorqual (Mammalia, Cetacea, Balaenopteridae) from the Early Pliocene of the North Sea, with a review of the rorqual species described by Owen and Van Beneden. Geodiversitas, 32:331-363. https://doi.org/10.5252/g2010n2a6
Bouetel, V. and de Muizon, C. 2006. The anatomy and relationships of Piscobalaena nana (Cetacea, Mysticeti), a Cetotheriidae s. s. from the early Pliocene of Peru. Geodiversitas, 28:319-395.
de Buffrénil, V., Dabin, W., and Zylberberg, L. 2004. Histology and growth of the cetacean petro-tympanic bone complex. Journal of Zoology, 262:371-381. https://doi.org/10.1017/s0952836903004758
Chen, I., Chou, L.-S., Chen, Y.-J., and Watson, A. 2011. The maturation of skulls in postnatal Risso’s dolphins (Grampus griseus) from Taiwanese waters. Taiwania, 56:177-185.
Cope, E.D. 1872. On an extinct whale from California. Proceedings of the Academy of Natural Sciences of Philadelphia, 24:29-30.
Cozzi, B., Podestà, M., Mazzariol, S., and Zotti, A. 2012. Fetal and early post-natal mineralization of the tympanic bulla in fin whales may reveal a hitherto undiscovered evolutionary trait. PLoS ONE, 7:e37110. https://doi.org/10.1371/journal.pone.0037110
Cozzi, B., Podestà, M., Vaccaro, C., Poggi, R., Mazzariol, S., Huggenberger, S., and Zotti, A. 2015. Precocious ossification of the tympanoperiotic bone in fetal and newborn dolphins: an evolutionary adaptation to the aquatic environment? The Anatomical Record, 298:1294-1300. https://doi.org/10.1002/ar.23120
Dathe, F. 1983. Megaptera hubachi n. sp., ein fossiler Bartenwal aus marinen Sandsteinschichten des tietferen Pliozäns Chiles. Zeitschrift fur geologische wissenschaften, 11:813-848.
Deméré, T.A. 1986. The fossil whale, Balaenoptera davidsonii (Cope 1872), with a review of other Neogene species of Balaenoptera (Cetacea: Mysticeti). Marine Mammal Science, 2:277-298. https://doi.org/10.1111/j.1748-7692.1986.tb00136.x
El Adli, J.J., Deméré, T.A., and Boessenecker, R.W. 2014. Herpetocetus morrowi (Cetacea: Mysticeti), a new species of diminutive baleen whale from the Upper Pliocene (Piacenzian) of California, USA, with observations on the evolution and relationships of the Cetotheriidae. Zoological Journal of the Linnean Society, 170:400-466. https://doi.org/10.1111/zoj.12108
Fordyce, R.E. and de Muizon, C. 2001. Evolutionary history of whales: a review, p. 169–234. In Mazin, J.-M. and de Buffrenil, V. (eds.), Secondary Adaptation of Tetrapods to Life in Water. Pfeil, München, Germany.
Galatius, A., Berta, A., Frandsen, M.S., and Goodall, R.N.P. 2011. Interspecific variation of ontogeny and skull shape among porpoises (Phocoenidae). Journal of Morphology, 272:136-148. https://doi.org/10.1002/jmor.10900
Galatius, A. and Kinze, C.C. 2003. Ankylosis patterns in the postcranial skeleton and hyoid bones of the harbour porpoise (Phocoena phocoena) in the Baltic and North Sea. Canadian Journal of Zoology, 81:1851-1861. https://doi.org/10.1139/z03-181
Geisler, J.H. and Luo, Z. 1996. The petrosal and inner ear of Herpetocetus sp. (Mammalia: Cetacea) and their implications for the phylogeny and hearing of archaic mysticetes. Journal of Paleontology, 70:1045-1066. https://doi.org/10.1017/S0022336000038749
Gol’din, P. and Startsev, D. 2014. Brandtocetus, a new genus of baleen whales (Cetacea, Cetotheriidae) from the late Miocene of Crimea, Ukraine. Journal of Vertebrate Paleontology, 34:419-433. https://doi.org/10.1080/02724634.2013.799482
Gol’din, P. and Startsev, D. 2017. A systematic review of cetothere baleen whales (Cetacea, Cetotheriidae) from the Late Miocene of Crimea and Caucasus, with a new genus. Papers in Palaeontology, 3:49-68. https://doi.org/10.1002/spp2.1066
Goloboff, P.A. and Catalano, S.A. 2016. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics, 32:221-238. https://doi.org/10.1111/cla.12160
Gray, J.E. 1864. On the Cetacea which have been observed in the seas surrounding the British Island. Proceedings of the Zoological Society of London, 2:195–248.
Gutstein, C.S., Cozzuol, M.A., Vargas, A.O., Suárez, M.E., Schultz, C.L., and Rubilar-Rogers, D. 2009. Patterns of skull variation of Brachydelphis (Cetacea, Odontoceti) from the Neogene of the Southeastern Pacific. Journal of Mammalogy, 90:504-519. https://doi.org/10.1644/07-MAMM-A-081.1
Hampe, O., Franke, H., Hipsley, C.A., Kardjilov, N. and Müller, J. 2015. Prenatal cranial ossification of the humpback whale (Megaptera novaeangliae). Journal of Morphology, 276:564-582. https://doi.org/10.1002/jmor.20367
Hanna, G.D. and McLellan, M.E. 1924. A new species of fin whale from the type locality of the Monterey Group. Proceedings of the California Academy of Sciences, 13:237-241.
Hasegawa, Y., Koda, Y., Yanagisawa, Y., Sato, Y., and Omori, S. 1988. On a fossil cetacean off the coast of Oarai-machi, Ibaraki Prefecture, p. 48-50. In Hasegawa, Y. (ed.), Studies on the Fossil Marine Mammals from Japan. The Grant-in-Aid for Scientific Research from the Ministry of Education, Science and Culture in the 62nd fiscal year of Showa.
Hasegawa, Y., Koizumi, A., Matushima, Y., Imanaga, I., and Hirata, D. 1991. Fossil remains from the Nakatsu Group, p. 1-98. In Hasegawa, Y. (ed.), Fossil Remains from the Nakatsu Group, Kanagawa Prefectural Museum, Yokohama.
Hasegawa, Y., Takakuwa, Y., and Nakajima, H. 2002. A balaenopterid whale from the Haraichi Formation (Middle Miocene), Tomioka Group, Annaka City, Gunma Prefecture, Japan. Bulletin of Gunma Museum of Natural History, 6:39-44.
Hatai, K., Hayasaka, S., and Masuda, K. 1963. Some fossil tympanics from the Mizuho period of northern Japan. Saito Ho-on Kai Museum of Natural History, Research Bulletin, 32:5-17.
Ichishima, H. 2005. A re-evaluation of some Japanese cetacean fossils. Memoir of the Fukui Prefectural Dinosaur Museum, 4:1-20.
Ito, H. and Miyazaki, N. 1990. Skeletal development of the striped dolphin (Stenella coeruleoalba) in Japanese waters. Journal of the Mammalogical Society of Japan, 14:79-96. https://doi.org/10.11238/jmammsocjapan1987.14.79
Iwaki Educational and Cultural Corporation. 1989. Excavation Report of Fossil Whales from Yotsukura-cho, Iwaki City. Board of Education, Iwaki City, Fukushima Prefecture, Japan.
Kasuya, T. 1973. Systematic consideration of recent toothed whales based on the morphology of tympano-periotic bone. Scientific Reports of the Whales Research Institute Tokyo, 25:1-103.
Kellogg, R. 1922. Description of the skull of Megaptera miocaena, a fossil humpback whale from the Miocene diatomaceous earth of Lompoc, California. Proceedings of the United States National Museum, 61:1-18. https://doi.org/10.5479/si.00963801.61-2435.1
Kimura, M. and Kanaya, K. 1996. On the fossil cetacea from the Haboro-cyo, Hokkaido, Japan. Kyoudo to Kagaku, 109:23-32.
Kimura, T., Adaniya, A., Oishi, M., Marx, F., Hasegawa, Y., and Kohno, N. 2015. A Late Miocene balaenopterid (“Shimajiri-kujira”) from the Okamishima Formation, Shimajiri Group, Miyako Island, Okinawa, Japan. Bulletin of Gunma Museum of Natural History, 19:39-48.
Kohno, N., Koike, H., and Narita, K. 2007. Outline of fossil marine mammals from the Middle Miocene Bessho and Aoki Formations, Nagano Prefecture, Japan. Research Report of the Shinshushinmachi Fossil Museum, 10:1-45.
Kurihara, N. and Oda, S. 2009. Effects of size on the skull shape of the bottlenose dolphin (Tursiops truncatus). Mammal Study, 34:19-32. https://doi.org/10.3106/041.034.0104
Maddison, W.P. and Maddison, D.R. 2011. Mesquite: a modular system for evolutionary analysis. Available at http://mesquiteproject.org.
Marx, F.G. and Kohno, N. 2016. A new Miocene baleen whale from the Peruvian desert. Royal Society Open Science, 3:1-27. https://doi.org/10.1098/rsos.160542
Marx, F. G., Lambert, O., and Uhen, M. D. 2016. Cetacean Paleobiology. John Wiley and Sons, New York.
Marx, F.G., Lambert, O., and de Muizon, C. 2017. A new Miocene baleen whale from Peru deciphers the dawn of cetotheriids. Royal Society Open Science, 4:1-22. https://doi.org/10.1098/rsos.170560
McGowen, M.R., Spaulding, M., and Gatesy, J. 2009. Divergence date estimation and a comprehensive molecular tree of extant cetaceans. Molecular Phylogenetics and Evolution, 53:891-906. https://doi.org/10.1016/j.ympev.2009.08.018
Mead, J.G. and Fordyce, R.E. 2009. The therian skull: a lexicon with emphasis on the odontocetes. Smithsonian Contributions to Zoology, 627:1-248. https://doi.org/10.5479/si.00810282.627
Moran, M.M., Bajpai, S., George, J.C., Suydam, R., Usip, S., and Thewissen, J.G.M. 2015. Intervertebral and epiphyseal fusion in the postnatal ontogeny of cetaceans and terrestrial mammals. Journal of Mammalian Evolution, 22:93-109. https://doi.org/10.1007/s10914-014-9256-7
Moriya, S., Danhara, T., Iwano, H., Yamashita, T., Nakajima, T., and Chinzei, K. 2008. Fission-track ages of the Pliocene strata in the Shinjo Basin, Yamagata Prefecture. The Journal of the Geological Society of Japan, 114:1-15. https://doi.org/10.5575/geosoc.114.1
Nagasawa, K. 1993. A fossil caudal vertebra of baleen whale from the Pliocene Noguchi Formation, Okura-mura, Yamagata Prefecture, Northeast Japan. Bulletin of the Yamagata Prefectural Museum, 14:15-22.
Nagasawa, K. 1995. Reconsideration of fossil mandibles of baleen whale from the Upper Miocene, Oe-machi, Yamagata Prefecture, Northeast Japan. Bulletin of the Yamagata Prefectural Museum, 16:31-38.
Nagasawa, K. 1999. Fossil cetacea from the Pliocene, Noguchi Formation of Mamurogawa Town, Yamagata Prefecture, p. 11-52. In Yamagata Prefectural Museum (ed), Report on Fossil Cetacea from the Pliocene, Noguchi Formation of Mamurogawa Town, Yamagata Prefecture. Yamagata Prefectural Museum, Yamagata.
Nagasawa, K., Ooba, S., Abe, R., and Abe, H. 2002. Cetacean fossils from the Neogene, the Akamatsu River, Okura Village, Mogami District, Yamagata Prefecture. Applied Geology of Yamagata, 22:52-57.
Nagasawa, K., Ooba, S., Abe, R., Abe, H., and Ogasawara, K. 2009. A physeteriid tooth from the Akamatsu River, Okura Village, Yamagata Prefecture. Applied Geology of Yamagata, 29:57-60.
Nakamura, G. and Kato, H. 2014. Developmental changes in the skull morphology of common minke whales Balaenoptera acutorostrata. Journal of Morphology, 275:1113-1121. https://doi.org/10.1002/jmor.20288
Nakamura, G., Kato, H., and Fujise, Y. 2012. Relative growth of the skull of the common minke whale Balaenoptera acutorostrata from the North Pacific in comparison to other Balaenoptera species. Mammal Study, 37:105-112. https://doi.org/10.3106/041.037.0201
Oishi, M. 1994. Cervical vertebrae of a fossil balaenopterid whale from the lower Pliocene strata in Ichinoseki City, Iwate Prefecture, Northeast Japan. Monograph of the Association of Geological Collaborators Japan, 43:111-122.
Oishi, M. 1987. Pliocene baleen whales and large sea lion from Ichinoseki and Hiraizumi area, Iwate Prefecture, Japan. Bulletin of the Iwate Prefectural Museum, 5:85-98.
Oishi, M. 1988. Fossil mysticetes from the Pliocene strata in the southernpart of Iwate Prefecture, p. 35-41. In Hasegawa, Y. (ed), Studies on the Fossil Marine Mammals from Japan. The Grant-in-Aid for Scientific Research from the Ministry of Education, Science and Culture, Yokohama.
Oishi, M. 1997. A dentary of Sibbaldus sp. from the lower Pliocene of Hiraizumi, Iwate Prefecture, northeast Japan. Bulletin of the Iwate Prefectural Museum, 15:1-10.
Oishi, M. and Hasegawa, Y. 1995a. A list of fossil cetaceans in Japan. Island Arc, 3(4):493-505.
Oishi, M. and Hasegawa, Y. 1995b. Diversity of Pliocene mysticetes from eastern Japan. Island Arc, 3(4):436-452.
Oishi, M., Ono, K., Kawakami, T., Sato, J., Nokariya, H., and Hasegawa, Y. 1985. Pliocene baleen whales and bony-toothed bird from Iwate Prefecture, Japan (Parts 1–6). Bulletin of the Iwate Prefectural Museum, 3:143-162.
Perrin, W.F. 1975. Variation of spotted and spinner porpoise (Genus, Stenella) in the eastern Pacific and Hawaii. Bulletin of the Scripps Institution of Oceanography, 21:1-206.
Pilleri, G. 1989. Balaenoptera siberi, ein neuer spatmiozaner bartenwal aus der Pisco-formation Perus, p. 65–84. In Pilleri, G. (ed), Beitrage zur Palaontologie der Cetaceen Perus. Bern.
Sacco, F. 1890. Sopra una mandibola di balaenoptera dell’Astigiana. Carlo Clausen.
Sato, T., Sato, M., Kobayashi, S., and Aizu Fossil Research Group. 2010. A fossil balaenopterid whale from the Upper Miocene Shiotsubo Formation in Takasato, Kitakata City, Fukushima Prefecture, northeast Japan. Earth Science (Chikyu Kagaku), 64:23-28. https://doi.org/10.15080/agcjchikyukagaku.64.1_23
Strobel, P. 1881. Iconografia comparata delle ossa fossili del gabinetto di storia naturale dell’Università di Parma. L. Battei. https://doi.org/0.5962/bhl.title.60846
Tamiya, R. and Applied Geological Society of Yamagata. 2016. Geological Map of Yamagata Prefecture, 1:100000, the Mogami District. Yamagata University Press, Prefecture.
Tanaka, Y. 2016. A new and ontogenetically younger specimen of Numataphocoena yamashitai from the lower Pliocene, the upper part of the Horokaoshirarika Formation, Numata, Hokkaido, Japan. Paleontological Research, 20:105-115. https://doi.org/10.2517/2015PR026
Tanaka, Y. and Ichishima, H. 2016. A new skull of the fossil porpoise Numataphocoena yamashitai (Cetacea: Phocoenidae) from the upper part of the Horokaoshirarika Formation (lower Pliocene), Numata Town, Hokkaido, Japan, and its phylogenetic position. Palaeontologia Electronica, 19.3.49A:1-28. https://doi.org/10.26879/663
palaeo-electronica.org/content/2016/1663-a-new-skull-of-numataphocoena
Tanaka, Y. and Taruno, H. 2017. Balaenoptera edeni skull from the Holocene (Quaternary) of Osaka City, Japan. Palaeontologia Electronica, 20.3.50A:1-13. https://doi.org/10.26879/785
palaeo-electronica.org/content/2017/2025-a-japanese-holocene-whale
Tanaka, Y. and Watanabe, M. 2019a. An early and new member of the Balaenopteridae from the upper Miocene of Hokkaido, Japan. Journal of Systematic Palaeontology, 17(16):1417-1431. https://doi.org/10.1080/14772019.2018.1532968
Tanaka, Y. and Watanabe, M. 2019b. A geologically old and ontogenetically young Herpetocetus sp. from the late Miocene of Hokkaido, Japan. Journal of Vertebrate Paleontology, 38:1-10. https://doi.org/10.1080/02724634.2018.1478842
Tsai, C.H. and Fordyce, R.E. 2014. Juvenile morphology in baleen whale phylogeny. Naturwissenschaften, 101:765-769. https://doi.org/10.1007/s00114-014-1216-9
Tsai, C.H. and Fordyce, R.E. 2016. Archaic baleen whale from the Kokoamu Greensand: earbones distinguish a new late Oligocene mysticete (Cetacea: Mysticeti) from New Zealand. Journal of the Royal Society of New Zealand, 46:1-22. https://doi.org/10.1080/03036758.2016.1156552
Uhen, M.D. 2004. Form, function, and anatomy of Dorudon atrox (Mammalia, Cetacea): an archaeocete from the middle to late Eocene of Egypt. University of Michigan Papers on Paleontology, 34:1-222.
Van Beneden, P.J. 1880. Les mysticetes a courts fanons des sables des environs d’anvers. Bulletins de L’Academie Royale des Sciences, des Lettres et des Beaux-Arts de Belgique, 2:11-27.
Walsh, B.M. and Berta, A. 2011. Occipital ossification of balaenopteroid mysticetes. The Anatomical Record, 294:391-398. https://doi.org/10.1002/ar.21340
Whitmore, F.C. and Kaltenbach, J.A. 2008. Neogene Cetacea of the Lee Creek Phosphate Mine, North Carolina. Virginia Museum of Natural History Special Publication, 14:181-269.
Zeigler, C.V., Chan, G.L., and Barnes, L.G. 1997. A new Late Miocene balaenopterid whale (Cetacea: Mysticeti), Parabalaenoptera baulinensis (new genus and species) from the Santa Cruz Mudstone, Point Reyes Peninsula, California. Proceedings of the California Academy of Sciences, 50:115-138.

