 Mediolus, a new genus of Arcellacea (Testate Lobose Amoebae)
Mediolus, a new genus of Arcellacea (Testate Lobose Amoebae)
Article number: 17.2.28A
https://doi.org/10.26879/440
Copyright Paleontological Society, July 2014
Author biography
Plain-language and multi-lingual abstracts
PDF version
Submission: 15 November 2013. Acceptance: 19 June 2014
{flike id=782}
ABSTRACT
Mediolus, a new arcellacean genus of the Difflugidae (informally known as thecamoebia, testate rhizopods, or testate lobose amoebae) differs from other genera of the family in having distinctive tooth-like inward oriented apertural crenulations and tests generally characterized by a variable number of hollow basal spines.
R. Timothy Patterson. Ottawa-Carleton Geoscience Centre and Department of Earth Sciences, Carleton University, Ottawa, Ontario, K1S 5B6, Canada. Tim.Patterson@carleton.ca
Keywords: Arcellacea; thecamoebian; testate lobose amoebae; Quaternary; new genus
Final citation: Patterson, R. Timothy. 2014. Mediolus, a new genus of Arcellacea (Testate Lobose Amoebae). Palaeontologia Electronica Vol. 17, Issue 2;28A; 8p. https://doi.org/10.26879/440
palaeo-electronica.org/content/2014/782-mediolus-new-genus
www.zoobank.org/E43633B7-2017-48CB-B887-849D86D3F230
PE Note: See In Memoriam: Dr. Franco Medioli
INTRODUCTION
Arcellacea (also informally known as thecamoebians, testate amoebae, testate rhizopods, or testate lobose amoebae; Patterson et al., 2012) are a diverse group of unicellular testate rhizopods that occur in a wide array of aquatic and terrestrial environments (Patterson and Kumar, 2002) from the tropics to poles (Dalby et al., 2000). Although most common in Quaternary sediments fossil arcellaceans have been found preserved in sediments deposited under freshwater and brackish conditions spanning the Phanerozoic and into the Neoproterozoic (Porter and Knoll, 2000; Van Hengstum, 2007).
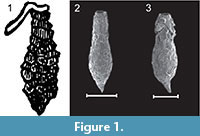 Within the Arcellacea the genus Difflugia Leclerc, 1815 is the most species rich, being comprised of more than 300 species and 200 subspecies (Meisterfeld and Mitchell, 2008). At the species level, especially for fossil material, the most important characteristics for systematic placement, has generally been the shape and size of the simple unilocular test (e.g., Bonnet, 1975; Medioli et al., 1987, 1990; Beyens and Meisterfeld, 2001), although some researchers have placed more emphasis on test composition (e.g., Anderson, 1988). The proliferation of species descriptions within the Difflugia, often based on subtle test differences, has resulted in considerable taxonomic confusion (Patterson and Kumar, 2002). Researchers have often described new species based on regional interest, often with little consideration of the previous literature or the systematic value of distinguishing characters (see discussions in Medioli and Scott, 1983; Medioli et al., 1987; Ogden and Hedley, 1980; Tolonen, 1986; Bobrov et al., 1999; Charman et al., 2000). On the other hand recent research suggests that many morphologically indistinguishable species may actually be cryptospecies (Mitchel and Meisterfeld, 2005; Alizon et al., 2008; Heger et al., 2013; Oliverio et al., 2014). Species ascribed to Difflugia are characterized by a wide variety of morphologies, some bearing little resemblance to the type species Difflugia protaeiformis Lamarck, 1816 (lectotype designated by Loeblich and Tappan, 1964, p. C35, as the specimen illustrated in Lamarck, 1816, pl. 17, figure. 5), which is characterized by an amphora-shaped test with narrow aperture and tapering at the base to a point (Figure 1.1-3).
Within the Arcellacea the genus Difflugia Leclerc, 1815 is the most species rich, being comprised of more than 300 species and 200 subspecies (Meisterfeld and Mitchell, 2008). At the species level, especially for fossil material, the most important characteristics for systematic placement, has generally been the shape and size of the simple unilocular test (e.g., Bonnet, 1975; Medioli et al., 1987, 1990; Beyens and Meisterfeld, 2001), although some researchers have placed more emphasis on test composition (e.g., Anderson, 1988). The proliferation of species descriptions within the Difflugia, often based on subtle test differences, has resulted in considerable taxonomic confusion (Patterson and Kumar, 2002). Researchers have often described new species based on regional interest, often with little consideration of the previous literature or the systematic value of distinguishing characters (see discussions in Medioli and Scott, 1983; Medioli et al., 1987; Ogden and Hedley, 1980; Tolonen, 1986; Bobrov et al., 1999; Charman et al., 2000). On the other hand recent research suggests that many morphologically indistinguishable species may actually be cryptospecies (Mitchel and Meisterfeld, 2005; Alizon et al., 2008; Heger et al., 2013; Oliverio et al., 2014). Species ascribed to Difflugia are characterized by a wide variety of morphologies, some bearing little resemblance to the type species Difflugia protaeiformis Lamarck, 1816 (lectotype designated by Loeblich and Tappan, 1964, p. C35, as the specimen illustrated in Lamarck, 1816, pl. 17, figure. 5), which is characterized by an amphora-shaped test with narrow aperture and tapering at the base to a point (Figure 1.1-3).
There have been various attempts to subdivide this large genus into more manageable subgroups. For example, Gauthier-Lièvre and Thomas (1958) divided the genus into 10 artificial groups based on morphologic test variability (e.g., elongate, globose, ovoid, urceolate, pyriform, and compressed tests; lobate, collared, and toothed apertures; presence or absence of spiked processes). Mazei and Warren (2012) carried out a similar survey of Difflugia species, informally subdividing the genus into groupings based on morphology. The newest available systematic tool to potentially study the systematics of Difflugia is the application of molecular systematics. Unfortunately, due to the difficulty of sequencing protists with agglutinated tests, only a handful of studies have been carried out on thecamoebian taxa (e.g., Nikolaev et al., 2005; Lara et al., 2008; Kudryavtsev et al., 2009), and only a single study has been carried out on Difflugia (Gomaa et al., 2012). In an analysis of five taxa (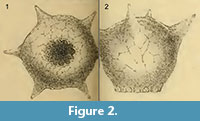 Difflugia bacillariarum Perty, 1849, D. hiraethogii Ogden, 1983, D. acuminata Ehrenberg, 1838; D. lanceolata Penard, 1890; D. achlora Penard, 1902) Gomaa et al. (2012) determined that Difflugia is not monophyletic, and as suggested in previous morphologic studies, shell shape, and not test composition, most closely correlates with genetic clades. Genetic analysis of Arcellacea is in its infancy, and it will be many years before such analysis is routine. It is obvious though, that due to the morphological plasticity within lineages across evolutionary time scales, it will be a combination of morphometric and molecular analysis that will eventually provide a more complete understanding of arcellacean biodiversity (Oliverio et al., 2014). New advances in morphometric analysis techniques may also provide guidance as to the validity of at least morphologically distinct taxa.
Difflugia bacillariarum Perty, 1849, D. hiraethogii Ogden, 1983, D. acuminata Ehrenberg, 1838; D. lanceolata Penard, 1890; D. achlora Penard, 1902) Gomaa et al. (2012) determined that Difflugia is not monophyletic, and as suggested in previous morphologic studies, shell shape, and not test composition, most closely correlates with genetic clades. Genetic analysis of Arcellacea is in its infancy, and it will be many years before such analysis is routine. It is obvious though, that due to the morphological plasticity within lineages across evolutionary time scales, it will be a combination of morphometric and molecular analysis that will eventually provide a more complete understanding of arcellacean biodiversity (Oliverio et al., 2014). New advances in morphometric analysis techniques may also provide guidance as to the validity of at least morphologically distinct taxa.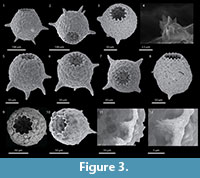 For example, Macumber, et al. (2014) employed a novel statistical approach to determine that two strains of Arcellacea, Difflugia protaeiformis Lamarck strain ‘acuminata’ and Difflugia protaeiformis Lamarck strain ‘claviformis’ most likely represent distinct species.
For example, Macumber, et al. (2014) employed a novel statistical approach to determine that two strains of Arcellacea, Difflugia protaeiformis Lamarck strain ‘acuminata’ and Difflugia protaeiformis Lamarck strain ‘claviformis’ most likely represent distinct species.
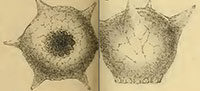
One of the most distinctive and easily recognizable arcellacean taxa is D. corona, Wallich, 1864, which is characterized by an agglutinated subspheroidal test, a fundus covered with a variable number of processes and a round aperture bordered by numerous crenulations or teeth (Figure 2.1-2; Figure 3.1-12). Aside from both being unilocular and characterized by an agglutinated test this species bears no resemblance to the Difflugia type species, D. protaeiformis (Figure 1.1-3). In an attempt to make a start at formally breaking Difflugia into more systematically realistic groups it is proposed that D. corona be recognized as the type species of the new genus Mediolus n. gen.
TAXONOMY
Subkingdom PROTOZOA
Phylum AMOEBOZOA Lühe, 1913
Class LOBOSIA Carpenter, 1861
Order ARCELLINIDA Kent, 1880
Superfamily ARCELLACEA Ehrenberg, 1830
Family DIFFLUGIDAE Stein, 1859
Genus Mediolus n. gen.
www.zoobank.org/E43633B7-2017-48CB-B887-849D86D3F230
Type species.Difflugia proteiformis (sic) (Ehrenberg) subspecies D. globularis (Dujardin) var. D. corona (Wallich) WALLICH 1864 p. 244, pl. 16, figures 19, 20 (reillustrated here as Figure 2.1-2).
Diagnosis. A genus of Difflugidae characterized by a symmetrical globular test, a test most typically comprised of agglutinated particles from the environment, round aperture with multiple inward-oriented angular tooth-line crenulations, and a test typically characterized by a variable number of spines extending outward on the test.
Description. Test free, unilocular; ovoid to subspherical to spheroid; circular in section, about the vertical aperture through the fundus; test wall comprised of agglutinating particles variously composed of xenogenous mineral grains and/or organic material derived from the ambient environment; agglutinating particles attached together with an organic cement; circular aperture characterized by thin collar of secreted cement with variable number of inward-oriented angular crenulations also composed of cement; delicate spines may be present, spines long and narrow, hollow, and composed of very fine agglutinating particles.
Types and occurrence. Hypotype (Canadian Museum of Nature (CANA 87186), recent from “Lake 10” near Yellowknife, Northwest Territories, Canada (62°30.924’N, 113°46.817’W); Hypotype (CANA 91905), recent from Mew Lake, Algonquin Park, Ontario (45°34.575’N, 78°31.087’W); Hypotype (CANA 87197), recent from Bell’s Lake, municipality of King (Greater Toronto Area), Ontario, Canada (43°N56.597, 79°39.762W); Hypotypes (CANA 87198 and CANA 87199), Holocene from Laguna de Quistococha, Iquitos, Peruvian Amazon (3°49.746’N, 73°19.157’W).
Dimensions. 100-400µm; mean ~ 150 µm (Medioli and Scott, 1983; Hu et al., 1997; Yang et al., 2004; Lahr and Lopes, 2006).
Remarks. The genus is readily distinguished from Protocurbitella Gauthier-Liévre and Thomas, 1960, as Mediolus lacks the distinctive diaphragm around the peristome. The peristomal diaphragm of Protocurbitella is similar to a similar structure characterizing Cuburbitella Penard, 1902. In addition, the pronounced lobed apertural collar of Cucurbitella is also very distinct from the apertural crenulations characterizing Mediolus. Cucurbitella is distinct from most other arcellaceans with regard to its distinctive relationship with some algal species. As the spring and summer growing season progresses and algae begins to grow into the water column Cucurbitella individuals go with it. In the absence of xenogenous grains to construct their test, late season specimens of this genus have the ability to produce idiosomic particles, which they use to construct their shell (Schönborn, 1984; Medioli et al., 1987). Mediolus differs from Loboforamina Jung, 1942 in that the latter is characterized by a lobate apertural region similar to that found in Cucurbitella.
As illustrated in this paper living representatives of Mediolus are found from the tropics to the poles. It is relatively common in mesotrophic to eutrophic lakes where it is often found attached to water plants (Medioli and Scott, 1983; Roe et al., 2010; Patterson et al., 2012; Roe and Patterson, 2014). It is less common in ponds and ditches and has also been reported from sphagnum bogs. Individuals are heterotrophic using its lobose pseudopods to engulf organic material or smaller prey (e.g., diatoms and bacteria) (Medioli and Scott, 1983).
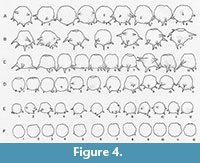 The crenulated aperture typical of the type species D. corona is much more diagnostic of the genus that the spines. Using broods of D. corona, Jennings (1916) carried out what is now recognized as a classic experiment on hereditary in uniparental organisms. He found that although clones differed greatly in overall size and the number of spines produced through many generations, there was an almost perfect correlation between the number of apertural teeth produced within brood lineages. As the spines would sometimes disappear entirely in some broods it is the apertural teeth that are the single most distinguishing characteristic of Mediolus (Figure 4). The seasonally planktic species Difflugia tuberspinifera Hu, Shen, Gu, and Gong, 1997, a species endemic to east Asia, is morphologically similar to D. corona, differing in that the toothed aperture in D. tuberspinifera tends to be contained within a protruding apertural flange, and that the conical spines are restricted to the upper equatorial region (Yang et al., 2004; Han et al., 2008). Yu et al. (2014) have recognized that, as also observed in D. corona, spineless strains also exist. Based on the general similarity of the apertural crenulations in both D. corona and D. tuberspinifera, the latter species is tentatively assigned to Mediolus.
The crenulated aperture typical of the type species D. corona is much more diagnostic of the genus that the spines. Using broods of D. corona, Jennings (1916) carried out what is now recognized as a classic experiment on hereditary in uniparental organisms. He found that although clones differed greatly in overall size and the number of spines produced through many generations, there was an almost perfect correlation between the number of apertural teeth produced within brood lineages. As the spines would sometimes disappear entirely in some broods it is the apertural teeth that are the single most distinguishing characteristic of Mediolus (Figure 4). The seasonally planktic species Difflugia tuberspinifera Hu, Shen, Gu, and Gong, 1997, a species endemic to east Asia, is morphologically similar to D. corona, differing in that the toothed aperture in D. tuberspinifera tends to be contained within a protruding apertural flange, and that the conical spines are restricted to the upper equatorial region (Yang et al., 2004; Han et al., 2008). Yu et al. (2014) have recognized that, as also observed in D. corona, spineless strains also exist. Based on the general similarity of the apertural crenulations in both D. corona and D. tuberspinifera, the latter species is tentatively assigned to Mediolus.
The extreme range of habitat observed for the type species of Mediolus may provide indirect evidence that it may encompass two or more cryptospecies. A possible non-genetic analysis approach to eventually test this hypothesis has been demonstrated using planktic foraminifera (Morard et al., 2013). Using transfer functions Morard et al. (2013) modeled the temperature dependence of several species of planktic species in the southern hemisphere and determined that several morphologically indistinguishable morphotypes are most likely distinct species.
Etymology. In honor of the late Professor Emeriti Franco Medioli (1935-2014), Department of Earth Sciences, Dalhousie University, in recognition of his extensive research on the ecology, systematics, and biostratigraphy of Arcellacea and related groups (Patterson, 2014).
Stratigraphic Range. Quaternary, but particularly common in Holocene deposits.
ACKNOWLEDGEMENTS
This research was supported by a Natural Sciences and Engineering Research Council (NSERC) Discovery Grant to RTP.
REFERENCES
Alizon, S., Kucera, M., and Janser, V.A.A. 2008. Competition between cryptic species explains variations in rates of lineage evolution. Proceedings of the National Academy of Sciences, 105:12382-12386.
Anderson, O.R. 1988. Testate Amoebae (Classes: Lobosea and Filosea), p. 63-65. In Anderson, O.R. (ed.), Comparative Protozoology: Ecology, Physiology, Life History. Springer-Verlag, Berlin, New York.
Beyens, L.D. and Meistefeld, R. 2001. Testate protozoa, p. 121-153. In Smol, J.P., Birks, H.J.B., and Last, W.M. (eds.), Tracking Environmental Change Using Lake Sediments: Biological Techniques and Indicators. v. II, Kluwer Academic Press.
Bobrov, A.A., Charman, D.J., and Warner, B.G. 1999. Paleoecology of testate amoebae (Protozoa: Rhizopoda) on peatlands in western Russia with special attention to niche separation in closely related taxa. Protist, 150:125-136.
Bonnet, L. 1975. Types morphologiques, écologie et évolution de la thèque chez les thècamoebiens. Protistologica, 11:363-378.
Carpenter, W.B. 1861. On the systematic arrangement of the Rhizopoda. Natural History Review New Series, 1:456-472.
Charman, D.J., Hendon, D., and Woodland, W. 2000. The identification of peatland testate amoebae. Quaternary Research Association Technical Guide no. 9, London, 147 pp.
Dalby, A.P., Kumar, A., Moore, J.M., and Patterson, R.T. 2000. Utility of arcellaceans (thecamoebians) as paleolimnological indicators in tropical settings: Lake Sentani, Irian Jaya, Indonesia. J ournal of Foraminiferal Research, 30:135-142.
Ehrenberg, C.G. 1830. Organisation, Systematik und geographisches Verhältnis der Infusionsthierchen. Druckerei der Königliche Akademie der Wissenschaften, Berlin.
Ehrenberg, G.C. 1838. Die Infusionthierchen als Vollkommene Organismen. Verlag von Leopold Voss, Leipzig.
Gauthier-Lièvre, L. and Thomas, R. 1958. Les genres Difflugia, Pentagonia, Maghrebia et Hoogenraadia (Rhizopodes testaces) en Afrique. Archiv Protistenkunde, 103:241-370.
Gauthier-Lièvre, L. and Thomas, R. 1960. Le genre Cucurbitella Penard. Archiv Protistenkunde, 104:569-602.
Gomaa, F., Todorov, M., Heger, T.J., Mitchell, E.A.D., and Lara, E. 2012. SSU rRNA phylogeny of Arcellinida (Amoebozoa) reveals that the largest Arcellinid genus, Difflugia Leclerc 1815, is not monophyletic. Protist, 163:389-399.
Han, P.-P., Wang, T., Lin, Q.-Q., and Dumont, H.J. 2008. Carnivory and active hunting by the planktonic testate amoebae Difflugia tuberspinifera. Hydrobiologia, 596:197-201.
Heger, T.J., Mitchell, E.A., and Leander, B.S. 2013. Holarctic phylogeography of the testate amoeba Hyalosphenia papilio (Amoebozoa: Arcellinida) reveals extensive genetic diversity explained more by environment than dispersal limitation. Molecular Ecology, 22:5172-5184.
Hu, D.L., Shen, Y.F., Gu, M.R., and Gong, X.J. 1997. New species and new records of protozoa from Wuling Mountains Area p. 40-72. In Song, D.X. (ed.), Invertebrates of Wuling Mountains Area, Southwestern China. Science Press, Beijing (in Chinese).
Jennings, H.S. 1916. Heredity, variation and the results of selection in the uniparental reproduction of Difflugia corona. Genetics, 1:407-534.
Jung, W. 1942. Südchilenische Thekamöben (aus dem südchilenischen Küstengebiet, Beitrag 10). Archiv Protistenkunde, 95:253-356.
Kent, W.S. 1880. A manual of the Infusoria: including a description of all known Flagellate, Ciliate, and Tentaculiferous Protozoa, British and foreign, and an account of the organization and affinities of sponges, Vol. 1. Bogue, London.
Kudryavtsev, A., Pawlowski, J., and Hausmann, K. 2009. Description and phylogenetic relationships of S pumochlamys perforate n. sp and Spumochlamys bryora n. sp (Amoebozoa, Arcellinida). Journal Eukaryotic Microbiology, 56:495-503.
Lahr, D.J.G. and Lopes, S.G.B.C. 2006. Morphology, biometry, ecology and biogeography of five species of Difflugia Leclerc, 1815 (Arcellinida: Difflugiidae), from Tiete River, Brazil. Acta Protozoologica, 45:77-90.
Lamarck, J.B. 1816. Histoire Naturelle des Animaux sans Vèrtèbres. Verdièie, Paris, tome 2, p. 1-568.
Lara, E., Heger, T.J., Ekelund, F., Lamentowicz, M., and Mitchell, E.A.D. 2008. Ribosomal RNA genes challenge the monophyly of the Hyalospheniidae (Amoebozoa: Arcellinida). Protist, 159:165-176.
Leclerc, L. 1815. Note sur la Difflugie, nouveau genre de polype amorphe. Mémoires du Muséum d’Histoire Naturelle 2, 474-478.
Loeblich, A.R., Jr., and Tappan, H. 1964. Sarcodina, chiefly “Thecamoebians” and Foraminiferida, p. C1-900. In Moore, R.C. (ed.), Treatise on Invertebrate Paleontology, Part C, Protista 2, Geological Society of America and University of Kansas Press, Boulder, Colorado and Lawrence, Kansas.
Lühe, M. 1913. Faunistische Untersuchung der Moore Ostpreußens. Schriften Physikalisch-ökonomischen Gesellschaft Königsberg 54:84-86.
Macumber, A.L., Patterson, R.T., Roe, H.M., Reinhardt, E.G., Neville, L.A., and Swindles, G.T. 2014. Autecological approaches to resolve subjective taxonomic divisions within Arcellacea. Protist, 165:305-316.
Mazei, Y. and Warren, A., 2012. A survey of the testate amoeba genus Difflugia Leclerc, 1815 based on specimens in the E. Penard and C.G. Ogden collections of the Natural History Museum, London. Part 1: Species with shells that are pointed aborally and/or have aboral protuberances. Protistology, 7:121-171.
Medioli, F.S. and Scott, D.B. 1983. Holocene Arcellacea (Thecamoebians) from eastern Canada. Cushman Foundation For Foraminiferal Research Special Publication 21.
Medioli, F.S., Scott, D.B., and Abbott, B.H. 1987. A case study of protozoan interclonal variability: taxonomic implications. Journal of Foraminiferal Research, 17:28-47.
Medioli, F.S., Scott, D.B., Collins, E.S., and McCarthy, F.M.G. 1990. Fossil thecamoebians: present status and prospects for the future, p. 813-840. In Hemleben, C., Kaminski, M.A., Kuhnt, W., and Scott, D.B. (eds.), Paleoecology, Biostratigraphy, Paleoceanography and Taxonomy of Agglutinated Foraminifera. NATO Advanced Study Institute Series, Series C, Mathematical and Physical Sciences 327.
Meisterfeld, R. and Mitchell, E.A.D. 2008. Difflugia, Leclerc 1815 Version 02 September 2008. The Tree of Life Web Project, tolweb.org/Difflugia/124487/2008.09.02. Retrieved 4 Nov. 2013.
Mitchell, E.A.D. and Meisterfeld, R. 2005. Taxonomic confusion blurs the debate on cosmopolitanism versus local endemism of free-living protists. Protist, 156:263-267.
Morard, R., Quillévéré, F., Escarguel, G., de Garidel-Thoron, T., de Vargas, C., and Kucera, M. 2013. Ecological modeling of the temperature dependence of cryptic species of planktonic foraminifera in the southern hemisphere. Palaeogeography, Palaeoclimatology, Palaeoecology, 391:13-33.
Nikolaev, S.I., Mitchell, E.A.D., Petrov, N.B., Berney, C., Fahrni, J., and Pawlowski, J. 2005. The testate lobose amoebae (order Arcellinida Kent, 1880) finally find their home within Amoebozoa. Protist, 156:191-202.
Ogden C.G. 1983. Observations on the systematics of the genus Difflugia in Britain (Rhizopoda, Protozoa). Bulletin British Museum (Natural History). Zoology, 44:1-73.
Ogden, C.G. and Hedley, R.H., 1980. An Atlas of Fresh Water Testate Amoeba. British Museum (Natural History), Oxford University Press, Oxford.
Oliverio, A.M., Lahr, D.J., Nguyen, T., and Katz, L.A., 2014. Cryptic diversity within morphospecies of testate amoebae (Amoebozoa: Arcellinida) in New England bogs and fens. Protist, 165:196-207.
Patterson, R. Timothy. 2014. IN MEMORIAM: Dr. Franco Medioli, April 1, 1935 – January 31, 2014. Palaeontologia Electronica Vol. 17, Issue 2;4E; 4p;
palaeo-electronica.org/content/2014/783-in-memoriam-dr-franco-medioli
Patterson, R.T. and Kumar, A., 2002. A review of current testate rhizopod (thecamoebian) research in Canada. Palaeogeography, Palaeoclimatology, Palaeoecology, 180:225-251.
Patterson, R.T., Roe, H.M., and Swindles, G.T. 2012. Development of an Arcellacea (testate lobose amoebae) based transfer function for sedimentary phosphorus in lakes. Palaeogeography, Paleoclimatology, Palaeoecology, 348-349:32-44.
Penard, E. 1890. Études sur les Rhizopodes d'eau douce. Mémoires de la Société de Physique et d'Histoire Naturelle de Genève, 31:1-230.
Penard, E. 1902. Faune Rhizopodique du Bassin du Leman. Henry Kundig, Geneve.
Perty, M. 1849. Mikroskopische Organismen der Alpen und der Italienischen Schweiz. Naturforschende Gesellschaft in Bern Mittheilungen, 164-165:153-176.
Porter, S.A., and Knoll, A.H. 2000. Testate amoeba in the Neoproterozoic Era: evidence from vase-shaped microfossils in the Chuar Group, Grand Canyon. Paleobiology, 26:360-385.
Roe, H.M. and Patterson, R.T. 2014. Arcellacea (testate amoebae) as bio-indicators of road salt contamination in lakes. Microbial Ecology, DOI: 10.1007/s00248-104-0408-3.
Roe, H.M., Patterson R.T., and Swindles, G.T. 2010. Controls on the contemporary distribution of lake thecamoebians (testate amoebae) within the Greater Toronto Area and their potential as water quality indicators. J ournal of Paleolimnology, 43:955-975.
Schöeborn, W.E. 1984. Studies on Remains of Testacea in cores of the Great Woryty Lae (NE Poland): Limnologica, 16:185-190.
Stein, S.F.N. 1859. Über die ihm aus eigener Untersuchung bekannt gewordenen Süßwasser-Rhizopoden. Königliche Böhmische Gesellschaft der Wissenschaften Abhandlungen, ser. 5, 10:41-43.
Tolonen, K. 1986. Rhizopod analysis, p. 645-666. In Berglund, B.E. (ed.), Handbook of Holocene Palaeoecology and Palaeohydrology. John Wiley and Sons, New York.
Van Hengstum, P.J., Reinhardt, E.G., Medioli, F.S., and Gröcke, D.R. 2007. Exceptionally preserved Late Albian (Cretaceous) arcellaceans (Thecamoebians) from the Dakota Formation near Lincoln, Nebraska, USA. Journal of Foraminiferal Research, 37:300-308.
Wallich, G.C. 1864. On the extent, and some of the principal causes of structural variation among the difflugian rhizopods. Annals and Magazine of Natural History Series 3, 13:215-245.
Yang, J., Beyens, L., Shen, Y.F., and Feng, W.S. 2004. Redescription of Difflugia tuberspinifera Hu, Shen, Gu et Gong, 1997 (Protozoan: Rhizopoda: Arcellinida: Difflugiidae) from China. Acta Protozoologica, 43:281-289.
Yu, Z., Zhang, W., Liu, L., and Yang, J. 2014. Evidence for two different morphotypes of Difflugia tuberspinifera from China. European Journal of Protistology, 50:205-211.

