 Investigation of a claim of a late-surviving pterosaur and exposure of a taxidermic hoax: the case of Cornelius Meyer's dragon
Investigation of a claim of a late-surviving pterosaur and exposure of a taxidermic hoax: the case of Cornelius Meyer's dragon
Article number: 16.1.6A
https://doi.org/10.26879/346
Copyright Palaeontological Association, January 2013
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Submission: 21 August 2012. Acceptance: 19 January 2013.
{flike id=384}
ABSTRACT
Here we investigate a claim that pterosaurs survived into the seventeenth century in Italy. In 1696 Dutch civil engineer Cornelius Meyer published an engraving of the skeleton of an alleged dragon from near Rome. Some recent young-Earth creationist authors have used the engraving as evidence against the separation of humans and pterosaurs by millions of years, claiming that the skeleton is that of a pterosaur that was alive in the seventeenth century. The engraving is detailed enough to identify the skeleton as a composite of bones from various extant animal species. Until now, however, no one has attempted such identification. Here we identify the specific animals that were used in the construction of this taxidermic hoax. The skull of Meyer's dragon is that of a domestic dog. The mandible is that of a second, smaller domestic dog. The "hindlimb" is the forelimb of a bear. The ribs are from a large fish. Ostensible skin hides the junctions between the parts of different animals. The tail is a sculpted fake. The wings are fake and lack diagnostic traits of bat wings and pterosaur wings. No part of the skeleton resembles its counterpart in pterosaurs. This piece of young-Earth creationist "evidence" therefore now joins the ranks of other discredited "evidence" for human-pterosaur coexistence and against the existence of the passage of millions of years. Also, a three-century-old hoax is finally unveiled, the mystery of its construction is solved, and an interesting and bizarre episode in Renaissance Italian history is elucidated.
Phil Senter. Department of Biological Sciences, Fayetteville State University, 1200 Murchison Road, Fayetteville, North Carolina 28301, U.S.A
Pondanesa D. Wilkins. Department of Biological Sciences, Fayetteville State University, 1200 Murchison Road, Fayetteville, North Carolina 28301, U.S.A
Key words: Pterosauria; Scaphognathus; young-Earth creationism; Cornelius Meyer; Cornelio Meyer; taxidermy; Renaissance; hoax
Final citation: Senter, Phil and Wilkins, Pondanesa D.. 2013. Investigation of a claim of a late-surviving pterosaur and exposure of a taxidemic hoax: the case of Cornelius Meyer’s dragon, Palaeontologia Electronica Vol. 16, Issue 1; 6A 11p. https://doi.org/10.26879/346
palaeo-electronica.org/content/2013/384-late-surviving-pterosaur
INTRODUCTION
Here we investigate the curious claim that a skeleton displayed in Rome in 1691 was that of a recently-killed pterosaur. This claim was made by young-Earth creationist authors John Goertzen (1998) and David Woetzel (2006). They used it to cast doubt on the separation of humans and pterosaurs by millions of years and, by extension, to cast doubt on evolutionary theory.
 The claim is based on an engraving published in a 1696 book by Dutch civil engineer Cornelius Meyer (1696). The book is written in Italian and describes various dike construction projects that occurred near Rome in the late seventeenth century. The last page of Part One of the two-part book sports an engraving of a mounted skeleton on a pedestal (Figure 1.1). On the pedestal is the inscription "Drago come si ritrova nelle mani dell' Ingegniero Cornelio Meyer" ("Dragon as it was recovered in the hands of the engineer Cornelius Meyer"). Above that engraving is a fleshed-out reconstruction of the dragon with the caption "Drago come è stato morto" ("Dragon like the one that is now dead"). On the cover of the book is a second fleshed-out engraving of the dragon, with the caption "Drago come viveva il primo di Decembre 1691 nelle paludi fuori di Roma" ("Dragon as alive on the first of December, 1691, in the marshes outside Rome") (Figure 1.2).
The claim is based on an engraving published in a 1696 book by Dutch civil engineer Cornelius Meyer (1696). The book is written in Italian and describes various dike construction projects that occurred near Rome in the late seventeenth century. The last page of Part One of the two-part book sports an engraving of a mounted skeleton on a pedestal (Figure 1.1). On the pedestal is the inscription "Drago come si ritrova nelle mani dell' Ingegniero Cornelio Meyer" ("Dragon as it was recovered in the hands of the engineer Cornelius Meyer"). Above that engraving is a fleshed-out reconstruction of the dragon with the caption "Drago come è stato morto" ("Dragon like the one that is now dead"). On the cover of the book is a second fleshed-out engraving of the dragon, with the caption "Drago come viveva il primo di Decembre 1691 nelle paludi fuori di Roma" ("Dragon as alive on the first of December, 1691, in the marshes outside Rome") (Figure 1.2).
Nowhere else in the book is the dragon mentioned, so further details of its story cannot be directly gleaned from the text but must be inferred from available clues. First, before the dikes were built the vicinity regularly experienced damage wrought by floodwaters and other problems caused by rivers (Meyer, 1696). Second, in medieval and Renaissance Europe, floods and other problems caused by water were often blamed on serpentine monsters that were said to inhabit the depths (Meurger and Gagnon, 1988). Dragons were often thought to dwell under water (Westwood, 1985; Ryan, 1993; Amann, 2011) and were sometimes identified with flood-causing lake monsters. Entries on dragons in medieval and Renaissance encyclopedic works show that even the learned considered their existence plausible if not definite (Gesner, 1589; Topsell, 1608; Aldrovandi, 1640). If the engraving of the skeleton is accurate, then an ostensible dragon skeleton was at one time displayed with an inscription that claimed that Meyer had recovered it. The dragon was said to be dead but had supposedly been alive in the marshes near Rome in 1691, in the same year that one of the dike construction projects occurred (Meyer, 1696). A rumor of a dragon that had been killed near Rome three decades previously was circulating in 1691, according to German author George Kirchmeyer. In the preface to his 1691 book On the Dragon he wrote: "On the 16th of October of this year [1691], news was brought from Rome of a flying serpent that had been killed by a hunter after a severe and dangerous struggle. This story, which appeared more like some fable than real truth, was a subject of discussion among the learned. The circumstance was denied by many, believed by others, and left in doubt by several." Elsewhere in the book he wrote: "On the 27th of October, 1660, the following circumstance was described as occurring at Rome: —A winged dragon appeared in the Campagna [countryside], which, after wounding a hunter, was killed. Yet the hunter himself eight days after (doubtless by the influence of the poison) expired, and his whole body became of a green hue" (Kirchmeyer, 1691).
When we put these clues together the picture that emerges is that in 1691, while a dike construction project occurred near Rome, a rumor began that a dragon killed in 1660 was alive again. Given this, Meyer's display of the skeleton was most likely an attempt to allay local fears of revenge by a monster whose home was being destroyed by the construction project. Available clues do not reveal whether Meyer did this simply to convince his laborers that it was safe to work, or whether rumors that the project itself had resurrected the beast were circulating and Meyer needed a dragon corpse to quiet local objections to the project. Either way, the figure captions and the inscription on the pedestal show that Meyer chose not to invite opposition by expressing skepticism about the local rumor that the dragon existed and had returned to life. Instead, he wisely chose to avoid resistance by humoring the locals, accomplishing this by embracing the local rumor and providing visual evidence that their source of concern had been vanquished.
After being all but forgotten for nearly 300 years, the illustration of Meyer's dragon skeleton resurfaced when a reproduction of his figure was published in a 1979 book on dragons (Allen and Griffiths, 1979). Goertzen (1998) and Woetzel (2006) brought the illustration to the attention of the young-Earth creationist community, and Goertzen specifically identified the alleged pterosaur as Scaphognathus crassirostris because the dragon has a long tail and a horn, which he identified as a crest. According to Goertzen, Scaphognathus crassirostris is the only known long-tailed pterosaur with a crest.
The story of Meyer's dragon is particularly interesting not only because of its having inspired recent claims of late-surviving pterosaurs but also because the engraving is detailed enough to test the pterosaur hypothesis and to simultaneously test the competing hypothesis that the skeleton is a composite of parts from different animals. However, until now no one has tested these hypotheses. Here, we present a study in which we use comparative osteology to do just that.
METHODS
We compared the parts of Meyer's dragon skeleton to their counterparts in the skeletons of Scaphognathus crassirostris, other pterosaurs, and extant vertebrates. We supplemented personal observations of vertebrate specimens with observations of specimens illustrated in the literature and of specimen photos and X-ray images that museum staff prepared and shared with us.
RESULTS AND CONCLUSIONS
 The Skull
The Skull
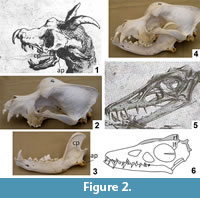
The skull of Meyer's dragon (Figure 2.1) has large, forward-facing orbits. The ventral margin of the cheek arches up to a level considerably higher (more dorsal) than the tooth row. The dorsal margin of the skull is sigmoidally curved such that there is a sudden rise in the height of the skull at the orbits, so that the dorsal surface of the postorbital area is at a much higher (more dorsal) level than the snout. On each side a large, anterior fang is followed by a diastema (a gap between teeth), which is followed by a small, peglike tooth. Meyer's illustration shows both the left and right fang and the left and right peglike tooth. Posterior to the peglike tooth is another diastema, followed by an anteroposteriorly short set of molariform cheek teeth. The posterior end of the skull is covered by ostensible skin that includes a pair of pointed ear flaps and a midline horn that curves posteriorly. A downcurved hook, not part of the skull itself, is attached to the tip of the snout.
The skull of Scaphognathus crassirostris differs from this skull in nearly every respect (Figure 2.5). The orbits do not face as prominently forward. The ventral margin of the cheek is level with the tooth row. The dorsal margin of the snout gradually rises to the level of the supraorbital margin instead of having a sudden rise at the orbits. The postorbital part of the skull is much shorter relative to the snout than is that of Meyer's dragon (Wellnhofer, 1975). In fact, these observations are true for pterosaurs in general (Arthaber, 1919). As in most other toothed pterosaurs (Arthaber, 1919), the dentition of Scaphognathus crassirostris consists of uniform, conical teeth with no differentiation into different tooth types, no peglike teeth, and no molariform teeth. Furthermore, there is no horn or crest in S. crassirostris. Goertzen's (1998) claim of a crest is understandable, because it is based on a line drawing of a juvenile skeleton of S. crassirostris—published in Wellnhofer (1991)—in which it looks like the artist drew a crest. However, a published photo of the same specimen (Wellnhofer, 1991) shows that the apparent crest is actually a dorsally displaced (and crestless) right frontal bone (Figure 2.6).
In Meyer's dragon the morphology of the skull and the dentition match those of the domestic dog, Canis familiaris (Figure 2.2). The dentition of Meyer's dragon is markedly different from any extant non-mammal or from any extant mammal of Europe, North Africa, and the Middle East outside the dog family, Canidae (MacDonald and Barrett, 2001; Aulagnier, 2009). Within Canidae, the sudden rise in skull height at the orbits is consistent only with a domestic dog, and falsifies the hypothesis that the skull is from a wild canid species (MacDonald and Barrett, 2001; Aulagnier, 2009).
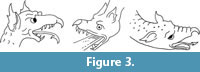 Because dogs have neither horns nor beaks, the cranial horn and the hooked proboscis must be fake. The hooked proboscis is a feature found on no pterosaur snout. It is, however, commonly found in Renaissance-era European depictions of dragons (Figure 3) and is therefore consistent with contemporaneous conceptions of the appearance of a dragon.
Because dogs have neither horns nor beaks, the cranial horn and the hooked proboscis must be fake. The hooked proboscis is a feature found on no pterosaur snout. It is, however, commonly found in Renaissance-era European depictions of dragons (Figure 3) and is therefore consistent with contemporaneous conceptions of the appearance of a dragon.
The Mandible
The mandible of Meyer's dragon exhibits a high, anteroposteriorly wide coronoid process and a pronounced angular process. The coronoid process exhibits a distinct masseteric fossa, bounded anteriorly and posteriorly by a vertical ridge at the margin of the bone (Figure 2.1). The dentition includes a pair of large anterior fangs, followed by a series of closely-spaced teeth with smaller crown heights than the fangs. The teeth immediately posterior to the fangs have the smallest crown heights.
The mandible of Scaphognathus crassirostris is very different from that of Meyer's dragon. The length-to-height ratio of the mandibular ramus is much greater in S. crassirostris. The mandible of S. crassirostris has no coronoid process, and the dentition consists of six widely-spaced, conical teeth (Wellnhofer, 1975) (Figure 2.5).
The high coronoid process, the masseteric fossa, and the angular process diagnose the mandible of Meyer's dragon as that of a mammal. The morphology and dentition of the mandible are consistent with those of a domestic dog (Figure 2.3) and inconsistent with those of any wild mammal of Europe, North Africa, or the Middle East (MacDonald and Barrett, 2001; Aulagnier, 2009). However, in dogs and in mammals generally, the lower canines are further anterior than the upper canines, whereas the reverse is the case in Meyer's dragon (Figure 2.1). The mandible of Meyer's dragon is therefore from a smaller dog than the one from which the skull came.
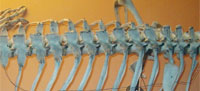
The Rib Cage
Meyer's dragon has 12 pairs of ribs. The anteriormost ribs are the shortest, and they increase in length until the sixth pair of ribs. The sixth, seventh, and eighth pairs of ribs are the longest, and the posterior pairs decrease in length so that the twelfth pair is approximately the same length as the second pair. Each rib is very slender and tapers to a fine point.
In Scaphognathus crassirostris and in pterosaurs generally, the longest pair of ribs is more anterior than the sixth pair (Arthaber, 1919; Wellnhofer, 1975). The rib cage proportions of Meyer's dragon are therefore inconsistent with a pterosaur.
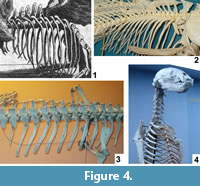 The length of the ribs of Meyer's dragon and their tapering to a fine point diagnoses them as those of a bony fish (Figure 4.2). In contrast, the ribs of chondrichthyan fishes (sharks and kin) and amphibians are very short, and those of large amniotes are squared-off at the tips (Figure 4.3-4). Several species of fish large enough to have provided the ribs of Meyer's dragon inhabit the Mediterranean Sea and the lakes and rivers of Europe (Lythgoe and Lythgoe, 1991; Maitland and Linsell, 2001). It is difficult to determine which species of fish was used in this case.
The length of the ribs of Meyer's dragon and their tapering to a fine point diagnoses them as those of a bony fish (Figure 4.2). In contrast, the ribs of chondrichthyan fishes (sharks and kin) and amphibians are very short, and those of large amniotes are squared-off at the tips (Figure 4.3-4). Several species of fish large enough to have provided the ribs of Meyer's dragon inhabit the Mediterranean Sea and the lakes and rivers of Europe (Lythgoe and Lythgoe, 1991; Maitland and Linsell, 2001). It is difficult to determine which species of fish was used in this case.
In bony fishes the ribs articulate with the lateral or lateroventral surface of the centrum of the thoracic vertebrae (Figure 4.2), whereas the ribs on Meyer's dragon are mounted in articulation with short transverse processes at the bases of the neural arches. The short transverse process is particularly visible on the vertebra that articulates with the sixth visible left rib. The presence of transverse processes indicates that the vertebrae are those of a tetrapod and not a fish. There is a pair of indentations on the posterior margin of the neural spine of each thoracic vertebra of Meyer's dragon (Figure 4.1). Such a shape is not found in the thoracic neural spines of any vertebrate. We therefore conclude that a fake extension was added to the tip of each neural spine. This obscuration of vertebral morphology makes it difficult to identify the animal from which the vertebrae came. If they are thoracic vertebrae, as they appear to be, their large size and the shortness of the transverse processes are inconsistent with any reptile but a large monitor lizard (Varanus). There are too many vertebrae in the series for them to have come from a bird. A combination of size and the shortness of the neural spines of the anterior thoracic vertebrae eliminates most wild and domestic mammals of Europe, North Africa, and the Middle East. Potential remaining mammalian candidates include only pangolins (order Pholidota) and the European beaver (Castor fiber). It is difficult to eliminate with certainty Varanus, Pholidota, or C. fiber from consideration. However, on geographical grounds, the beaver seems most likely, because the other two are not native European fauna and would therefore have been more difficult to acquire.
The Wings
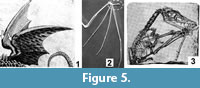 In Meyer's dragon the posterior edges of the wings are scalloped (Figure 5.1), as in a bat and unlike the smooth posterior edges of pterosaur wings. In a bat each point on the scalloped trailing edge is formed by the tip of a finger, and the fingers converge proximally at the palm (Figure 5.2). In contrast, the three visible "fingers" within the wing of Meyer's dragon do not converge. One ends proximally near the axillary region, one ends proximally at the elbow, and the third forms the leading edge of the distal wing.
In Meyer's dragon the posterior edges of the wings are scalloped (Figure 5.1), as in a bat and unlike the smooth posterior edges of pterosaur wings. In a bat each point on the scalloped trailing edge is formed by the tip of a finger, and the fingers converge proximally at the palm (Figure 5.2). In contrast, the three visible "fingers" within the wing of Meyer's dragon do not converge. One ends proximally near the axillary region, one ends proximally at the elbow, and the third forms the leading edge of the distal wing.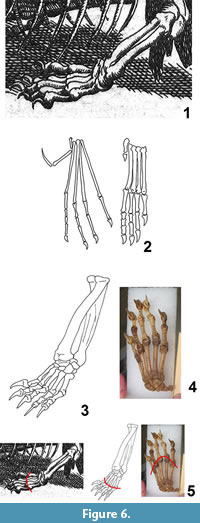 The wings completely lack clawed digits that project from the leading edge. In contrast, bats have one such digit (the thumb) (Figure 5.2) and pterosaurs have three (Figure 5.3). Also, in Meyer's dragon the base of each wing, which would be the shoulder joint in a bat or pterosaur, is located at about the middle of the dorsal surface of the ribcage, while the shoulder joints of bats and pterosaurs are anterior to the rib cage. Given all this, the wings do not match the morphology of any extant or extinct animal and are therefore fake.
The wings completely lack clawed digits that project from the leading edge. In contrast, bats have one such digit (the thumb) (Figure 5.2) and pterosaurs have three (Figure 5.3). Also, in Meyer's dragon the base of each wing, which would be the shoulder joint in a bat or pterosaur, is located at about the middle of the dorsal surface of the ribcage, while the shoulder joints of bats and pterosaurs are anterior to the rib cage. Given all this, the wings do not match the morphology of any extant or extinct animal and are therefore fake.
The "Hind Limb"
The "hind limb" of Meyer's dragon (Figure 6.1) ends in five digits with strongly curved claws. The metapodials are subequal in length, so that a line connecting their tips forms a shallow arc. The third digit is the longest, but the other four are not much shorter. The shafts of the "tibia" and "fibula" are of subequal diameter.
In pterosaurs generally, the fibula is but a small fraction of the diameter of the tibia, and the metapodials are much more slender than those of Meyer's dragon (Arthaber, 1919). In pterosaurs the fifth digit is either vestigial or exhibits an extremely elongated terminal phalanx (Arthaber, 1919) (Figure 6.2). The "hind limb" of Meyer's dragon is therefore not that of a pterosaur.
Digital proportions falsify the hypothesis that the "hind limbs" are those of any extant reptile of sufficient size. In large lizards the fourth toe is the longest, and in crocodylians the fifth toe is absent. In mammals, the diameter of the fibula is much smaller than that of the tibia. The hypothesis that the "hind limb" of Meyer's dragon is a hind limb at all, is therefore falsified. The "tibia" and "fibula" are actually a radius and ulna. In Crocodylia the fifth finger lacks a claw, and the only large enough lizard—the Komodo monitor (Varanus komodoensis)—was unknown to Europeans until the twentieth century. The limb is therefore from a mammal. Size and digital proportions rule out all European, North African, and Middle Eastern mammals but the wolverine (Gulo gulo) and the Eurasian brown bear (Ursus arctos) (MacDonald and Barrett, 2001; Aulagnier, 2009). The metacarpals of the bear are of subequal length, whereas in the wolverine metacarpals I and V are markedly shorter than III and IV, so that in the bear a line connecting the tips of the metacarpals forms a shallow arc, as in Meyer's dragon, whereas in the wolverine a line connecting the tips of the metacarpals forms an acute arc, unlike Meyer's dragon (Figure 6.3 – 5). Phalangeal proportions and general morphology are consistent with the bear. We therefore conclude that the "hind limbs" of Meyer's dragon are the forelimbs of a bear. Given its size in relative to the dog skull used as the skull of the "dragon," the bear was a juvenile.
The Tail
As illustrated, the tail of Meyer's dragon (Figure 7.1) has 51 visible vertebrae, each approximately as long as it is tall. Each vertebra exhibits a short anterodorsal process resembling a prezygapophysis that slightly overlaps the preceding vertebra. Each also exhibits a short anteroventral process that slightly overlaps the preceding vertebra and is approximately a mirror-image of the anterodorsal process. Along the side of each vertebra is a pair of slender ridges, each of which ends in a process that overlaps the preceding vertebra; in lateral view this pair of processes resembles a pair of prezygapophyses viewed dorsally. The vertebrae are progressively smaller distally so that the tail gradually tapers to a sharp tip.
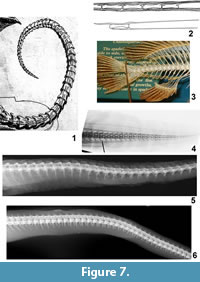 In the tail of Scaphognathus crassirostris and other long-tailed pterosaurs, each vertebral centrum is elongate, which is not the case in Meyer's dragon. Also, in Scaphognathus crassirostris and other long-tailed pterosaurs, each prezygapophysis is elongated into a rod that overlaps several more proximal vertebrae (Wellnhofer, 1975) (Figure 7.2). The resulting set of bony rods is a prominent feature of the tail of long-tailed pterosaurs but is absent in Meyer's dragon.
In the tail of Scaphognathus crassirostris and other long-tailed pterosaurs, each vertebral centrum is elongate, which is not the case in Meyer's dragon. Also, in Scaphognathus crassirostris and other long-tailed pterosaurs, each prezygapophysis is elongated into a rod that overlaps several more proximal vertebrae (Wellnhofer, 1975) (Figure 7.2). The resulting set of bony rods is a prominent feature of the tail of long-tailed pterosaurs but is absent in Meyer's dragon.
No extant vertebrate has tail vertebrae like those of Meyer's dragon. In chondrichthyan fishes, there are no processes like those in the tail of Meyer's dragon, and there are interneural arches, which are absent in Meyer's dragon. In most bony fishes the tail vertebrae do not gradually taper to a point (Figure 7.3). Instead, at the base of the caudal fin there is an abrupt transition from a typical, large caudal vertebra to a short series of tiny vertebrae in the base of the dorsal lobe of the fin. Also, the caudal vertebrae of bony fishes are usually fused to the neural and hemal arches so that an elongate neural spine extends dorsally and an elongate hemal spine extends ventrally from each vertebra. Such spines are absent in the tail of Meyer's dragon. In eels (Anguilliformes) the caudal vertebral series does taper to a point, but the processes that extend from the lateral surface of the caudal vertebrae of Meyer's dragon are absent (Figure 7.4). In salamanders, lizards, crocodylians, and long-tailed mammals, the caudal centra are elongate, the anteroventral process is absent, and there are less than 40 caudal vertebrae; if transverse processes extend from the lateral surface of the vertebra there is only one on each side. In the posterior vertebrae of a snake there is no pair of lateral processes such as are present on the caudal vertebrae of Meyer's dragon (Figure 7.5-6). In snake species in which an anteroventral process is present, e.g., members of Viperidae, it does not mirror the dorsal process (the neural spine) in shape (Figure 7.5). In fact, there is no vertebrate with caudal vertebrae in which a pair of processes extends forward from the lateral surface of the centrum as in Meyer's dragon. We therefore conclude that the vertebrae in the tail of Meyer's dragon are sculpted fakes and not real vertebrae of any animal.
Other Features
It is noteworthy that Meyer's dragon has neither a pectoral girdle nor a pelvic girdle, casting further doubt its authenticity as a single skeleton. Also, patches of ostensible dragon skin hide the junctions between parts of different animals, both at the neck and at the hips (Figure 1.1). It is impossible to know of what material the ostensible dragon hide was composed. However, its lack of scales shows that it was not reptile skin, and its lack of fur or feathers shows that if it was mammal or bird skin, then the fur or feathers had been removed.
As illustrated, the lengths of the bear radius and ulna used in the dragon's "hind limb" are subequal to that of the paw. However, the full length of the radius of a brown bear is half-again that of the paw, and the ulna is longer. The proximal ends of the radius and ulna had therefore been sawed off. Not surprisingly, the proximal ends of these two bones were covered in "dragon" hide in the skeletal mount (Figure 1.1), concealing their incompleteness.
Summary
The skull of Meyer's dragon is that of a dog. The mandible is that of a second, smaller dog. The ribs are those of a large fish. The thoracic vertebrae probably are those of a beaver. The "hind limbs" are the forelimbs of a juvenile bear. The wings, tail, beak, and cranial horn are fake.
DISCUSSION
There are several important things to note about the case of Meyer's dragon. First, every part of its anatomy is inconsistent with that of a pterosaur, Scaphognathus crassirostris or otherwise. This piece of "evidence" for the coexistence of pterosaurs and humans is therefore falsified. It now joins the ranks of other examples of falsified "evidence" for the coexistence of humans and pterosaurs that have been proffered by young-Earth creationist authors, such as the alleged ancient pterosaur painting in Black Dragon Canyon, Utah, and the alleged pterosaur rock paintings near Alton, Illinois (Senter, 2012). Several recent young-earth creationist authors claim that live pterosaurs currently roam the United States, Cuba, Africa, and Papua New Guinea ( e.g., Gibbons and Hovind, 1999; Woetzel, 2006; Stuckwish, 2009; Whitcomb, 2010), but to date no one has produced a pterosaur body, nor footage or a photo of a live pterosaur.
Second, Meyer's dragon is not unique but is but one of a plethora of composite monsters that taxidermic hoaxers created in Renaissance and post-Renaissance Europe. A taxidermically-created hydra with seven heads was given to a French king in the sixteenth century, a fifteen-headed hydra was displayed in Rome in the seventeenth century, and another seven-headed hydra passed through several hands in the seventeenth and eighteenth centuries (Dance, 1975). Several fake mermaids made their way to Europe in the eighteenth and nineteenth centuries (Bondeson, 1999). Numerous fake dragons made of carved rays (Rajiformes) were made in Europe in the sixteenth through eighteenth centuries (Dance, 1975), and fake dragons composed of parts of terrestrial animals were made in sixteenth and seventeenth-century Europe (Findlen, 1996).
In the case of this taxidermic fakery, Meyer himself was in on the ruse. This is demonstrated by his having claimed, via the inscription upon the pedestal of the mount, that he had recovered the corpse of the beast, which he knew to be a false statement. This suggests that it was Meyer himself who commissioned the creation of the dragon mount.
The solving of the mystery of the zoological composition of Meyer's dragon puts to rest the notion that Italians encountered live pterosaurs in the seventeenth century. It also sheds light on a strange and little-remembered episode in Italian history. The case involves superstition, rumor, political intrigue, shady dealings, mighty feats of engineering, the impressive talent of an artisan savvy enough to combine two dogs and a bear and a fish and make it work, and the sagacity of an engineer who risked his career to turn a potentially job-wrecking superstition to his advantage. Such an episode deserves to be counted among one of the greatest zoological hoaxes of Renaissance Europe.
ACKNOWLEDGMENTS
We would like to thank the following people for their contributions to this project. J. Allen provided contact information for J. Griffiths, who showed PS where to find an online copy of Meyer's book. K. Tighe and J. Jacobs (United States National Museum) provided X-rays of snakes. A. Williston and K. Hartel (Harvard University) provided X-rays of the Mediterranean moray eel, Muraena helena. D. Smith (USNM) located a published picture of the M. helena caudal skeleton. C. Bennett shared a copy of Wellnhofer's 1975 pterosaur article. Fayetteville State University student J. Moch provided photos of bear skeletons. S. Peurach (USNM) provided photos of the hand skeletons of wolverines and bears. W. Starnes and G. Hogue (North Carolina State Museum of Natural Sciences) provided a loan of eel skeletons. F. King and D. Okunbor provided funding through the Institutes of Undergraduate Research program. S. Allen (FSU) served as coordinator for BIOL 430, the student research course under the auspices of which this project began. Two anonymous reviewers provided helpful suggestions that improved this manuscript.
REFERENCES
Aldrovandi, U. 1640. Serpentum et Draconu Historiae Libri Duo. M. Antony Berm, Bologna.
Allen, J. and Griffiths, J. 1979. The Book of the Dragon. Chartwell, Secaucus.
Amann, D. 2011. La Tarasque. Un Dragon en Provence. Gaussen, Marseille.
Arthaber, G. 1919. Studien über Flugsaurier auf Grund der Bearbeitung des wiener Exemplars von Dorygnathus banthensis Theod. sp. Denkschriften der königlichen Akademie der Wissenschaften. Mathematisch-Naturwissenschaftlichen Klasse 97:391-464.
Aulagnier, S. 2009. Mammals of Europe, North Africa, and the Middle East. A & C Black, London.
Bondeson, J. 1999. The Feejee Mermaid and Other Essays in Natural and Unnatural History. Cornell University Press, Ithaca.
Dance, P. 1975. Animal Fakes and Frauds. Sampson Low, Maidenhead, United Kingdom.
Findlen, P. 1996. Possessing Nature: Museums, Collecting, and Scientific Culture in Early Modern Italy. University of California Press, Berkeley.
Gesner, K. 1589. Schlangenbuch. Froschow, Zurich.
Gibbons, W.J. and Hovind, K. 1999. Claws, Jaws & Dinosaurs. CSE, Pensacola.
Goertzen, J. 1998. The rhamphorhynchoid pterosaur Scaphognathus crassirostris: a living fossil until the seventeenth century? pp. 253-269. In Walsh, R.E. (ed.), Proceedings of the Fourth International Conference on Creationism. Creation Science Fellowship, Pittsburgh.
Goldfuss, G.A. 1831. Beiträge zur Kentniss verschiedener Reptilien der Vorwelt. Breslau Universität, Bonn.
Hogarth, P. and Clery, V. 1979. Dragons. Penguin, New York.
Huxley, F. 1979. The Dragon. Nature of Spirit, Spirit of Nature. Thames and Hudson, London.
Kirchmeyer, G.C. 1691. [1886 translation] On the dragon. pp. 15-36. In Goldsmid, E. (ed.), Un-natural History, or Myths of Ancient Science, Volume 3. Privately printed, Edinburgh.
Lythgoe, J. and Lythgoe, G. 1991. Fishes of the Sea: the North Atlantic and Mediterranean. MIT Press, Cambridge, Massachusetts.
MacDonald, D.W. and Barrett, P. 2001. Mammals of Europe. Princeton University Press, Princeton.
Maitland, P. and Linsell, K. 2001. Guide to Freshwater Fish of Britain and Europe. Hamlyn, London.
Meurger, M. and Gagnon, C. 1988. Lake Monster Traditions. A Cross-cultural Analysis. Fortean Times, London.
Meyer, C. 1696. Nuovi ritrovamenti Divisi in Due Parti. Printing House of Gio, Rome.
Ryan, W.G. 1993. The Golden Legend. Readings on the Saints. Princeton University Press, Princeton.
Senter, P. 2012. More "dinosaur" and "pterosaur" rock art that isn't. Palaeontologia Electronica 15(2.22A):1-14.
Stuckwish, D. 2009. Biblical Cryptozoology: Revealed Cryptids of the Bible. Xlibris, Bloomington.
Topsell, E. 1608. The Historie of Serpents. William Taggard, London.
Wellnhofer, P. 1975. Die Rhamphorhynchoidea (Pterosauria) der Oberjura-Plattenkalke Süddeutschlands. Palaeontographica Abteilung A 148:1-33.
Wellnhofer, P. 1991. The Illustrated Encyclopedia of Pterosaurs. Crescent, New York.
Westwood, J. 1985. Albion. A Guide to Legendary Britain. Paladin, London.
Whitcomb, J.D. 2010. Live Pterosaurs in America. Self-published.
Woetzel, D. 2006. The fiery flying serpent. Creation Research Society Quarterly 42:241-251.

