 Canidae (Carnivora, Mammalia) from Niedźwiedzia Cave (Silesia, southwest Poland)
Canidae (Carnivora, Mammalia) from Niedźwiedzia Cave (Silesia, southwest Poland)
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Appendix
Supplementry Material
Submission: 7 November 2024. Acceptance: 20 March 2025.
ABSTRACT
Abundant fossil material from Niedźwiedzia Cave documented the presence of three canid species: Canis lupus spelaeus, Vulpes vulpes, and Vulpes lagopus. Dated on MIS 3, the canid assemblage is represented by large individuals of robust posture, typical for the cold phases of the Late Pleistocene. Canis lupus spelaeus is a characteristic element of Late Pleistocene steppe-tundra faunas; a short legged, very large wolf with a powerful dentition. The conducted analysis showed that wolves from Niedźwiedzia Cave are among the largest ever found. Because of the great variability of the postcranial material, any of the morphological features cannot be used as a clearly defined factor to distinguish between Vulpes vulpes and Vulpes lagopus. Accordingly, only metric characters allow for a clear determination of taxonomic affiliation.
Adrian Marciszak. Department of Paleozoology, University of Wrocław, Sienkiewicza 21, 50-335 Wrocław, Poland. adrian.marciszak@uwr.edu.pl
Aleksandra Kropczyk. Department of Paleozoology, University of Wrocław, Sienkiewicza 21, 50-335 Wrocław, Poland (corresponding author). aleksandra.kropczyk2@uwr.edu.pl
Keywords: Canis; Vulpes; biostratigraphy; carnassials; long bones
Final citation: Marciszak, Adrian & Kropczyk, Aleksandra. 2025. Canidae (Carnivora, Mammalia) from Niedźwiedzia Cave (Silesia, southwest Poland). Palaeontologia Electronica, 28(1):a15.
https://doi.org/10.26879/1510
palaeo-electronica.org/content/2025/5489-canids-from-niedzwiedzia-cave
Copyright: April 2025 Society of Vertebrate Paleontology.
This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
creativecommons.org/licenses/by/4.0
INTRODUCTION
After the disappearance of Lycaon lycaonoides (Kretzoi, 1938) during MIS 11, the core of canids in Europe consisted of four species: Canis lupus Linnaeus, 1758, Cuon alpinus (Pallas, 1811), Vulpes vulpes (Linnaeus, 1758), and Vulpes lagopus (Linnaeus, 1758) (Marciszak et al., 2023a, 2023b). And this state has remained basically to the recent times, with the exception of two more serious events that most likely took place in the Holocene. These are the extinction of C. alpinus (Ripoll et al., 2010; Marciszak et al., 2021) and the appearance of Canis aureus Linnaeus, 1758 (Kurtén, 1965; Lapini, 2003; Sommer and Benecke, 2005).
Contrary to other carnivores such as ursids and felids, and despite that their remains are commonly found across Poland, Middle-Late Pleistocene canids only rarely have been an object of detailed scientific interest (Römer, 1883; Kiernik, 1913a, 1913b; Bigaj, 1963; Lipecki and Wojtal, 2015; Marciszak et al., 2021). Partially it is because their remains are often fragmentary, and most of these sites have yielded a low number of canid bones. Specimens usually consist of isolated cranial remains, mainly teeth and some postcranial elements identified by size. Among Late Pleistocene canids of Poland, only Canis lupus and more recently C. alpinus has received attention (Römer, 1883; Kiernik, 1913a, 1913b; Bigaj, 1963; Lipecki and Wojtal, 2015; Marciszak et al., 2021, 2023a, 2023b). In case of foxes, the lack of detailed studies is basically because of the difficulties of correctly identifying postcranial material due to their morphological similarity to each other (Monchot and Gendron, 2010).
Despite the Polish territory usually being considered an area that yielded few canid remains, there are some localities standing in clear contrast to this statement. One of them is the Sudeten site Niedźwiedzia Cave, where remains of three canids have been found. Canid fossils from this locality have never been properly studied, which led to the appearance of numerous mistakes and incorrect identifications (Wiszniowska, 1967, 1970, 1986, 1989; Bieroński et al., 2007, 2009). This resulted in the failure to demonstrate the presence of Vulpes lagopus and the classification of few large individuals of Canis lupus as Crocuta crocuta spelaea Goldfuss, 1823, whose presence was not confirmed at this site (Marciszak et al., 2016, 2020).
MATERIAL AND METHODS
All studied material of canids from Niedźwiedzia Cave is stored in the Department of Paleozoology, University of Wrocław (Appendix 1). The identification of canid remains and their taxonomic classification into individual forms were performed using traditional morphometric analysis (Appendix 1). Measurements were taken point to point, with an electronic calliper, to the nearest 0.01 mm. All measurements are expressed in millimetres (Appendix 1). The scheme of measurements was taken and modified from von den Driesch (1976) and showed on the Supplementary Material (Figure S1, Figure S2, Figure S3), while morphological terminology follows Boudadi‐Maligne (2010). Throughout the text, upper teeth are represented by uppercase letters (e.g., P4), while lowercase letters (e.g., p4) are used for lower teeth. Marine Isotope Stage (MIS) boundaries are used after Lisiecki and Raymo (2005).
For comparison, we used canid fossils from various European sites. Part of these remains were measured by authors, while others were taken from adequate sources: Silesia (southern Poland, killed between 1840-1995), Předmostí and Pavlov (Czech Republic, MIS 3-2), Zoolithenhöhle (Germany, MIS 3), Jaurens Cave (France, MIS 3; Ballesio, 1979), Kents Cavern, Durdham Down (Great Britain, MIS 3; Turner, 1981), Tornewton Cave (lower stratum -- MIS 5e, upper stratum -- MIS 3; Turner, 1981), Biśnik Cave (Poland, layer 14 -- MIS, 6, layer 13 -- MIS 5e, layers 10-9 -- MIS 5a, layers 7-5 -- MIS 3), Banwell Bone Cave (Great Britain, MIS 5a; Turner, 1981), Dziadowa Skała, layers 3-4 (Poland, MIS 5e), Wschodnia Cave (Poland, MIS 6), and Romain-la-Roche (France, MIS 6; Argant, 2009, 2010).
 Site
Site
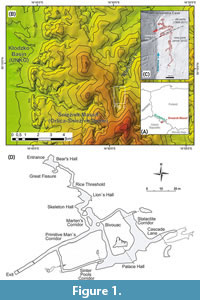
Niedźwiedzia Cave (55°68’70” N, 36°31’65” E; 807-800 m a.s.l.) is located in the Sudety Mts, Silesia region, southwest Poland (Figure 1). Since its discovery on 14 October 1966 during marble mining, the exploration of this cave has continued until present. Its corridors are entirely or partly filled by allochthonous and autochthonous deposits like rock debris, collapsed chamber scree deposits, fluvial gravels, sands, silts, and dripstones (Sobczyk et al., 2016). The deposits contain numerous animal remains, among which the older fauna was dated back to MIS 3-2 and consists of 27 species. The younger (MIS 1) faunal assemblage is represented by 24 species (Marciszak et al., 2020, 2024 and references therein). The presence of Canis lupus spelaeus at the late MIS 3 is confirmed by obtained AMS date from the costae. The date (Poz-143821, 10% Cm 1.8% N) was inferred from the costae and gave an age of 26.640 ± 290 (31.078-30.858 call. BP).
SYSTEMATIC PALEONTOLOGY
Class MAMMALIA Linnaeus, 1758
Order CARNIVORA Bowdich, 1821
Suborder CANIFORMIA Kretzoi, 1943
Family CANIDAE Fischer de Waldheim, 1817
Genus CANIS Linnaeus, 1758
Canis lupus Linnaeus, 1758
Canis lupus spelaeus Goldfuss, 1823
(Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8; Table 1, Table 2, Table 3; Appendix 1)
Referred material. The material of Canis lupus spelaeus, the most abundant canid from Niedźwiedzia Cave, is represented by almost all skeletal elements (NISP 479, MNI 18; left/right bones are given in brackets): two crania fr., nine maxillae fr. (6/3), one I2 (0/1), six I3 (3/3), 16 C1 (5/11), three P1 (1/2), nine P2 (3/6), eight P3 (5/3), 20 P4 (12/8), 15 M1 (8/7), one M2 (1/0), 32 mandibles (14/18), two i1 (1/1), nine i2 (4/5), two i3 (1/1), 18 c1 (7/11), three p1 (1/2), one p2 (0/1), 11 p3 (5/6), seven p4 (5/2), 18 m1 (10/8), two scapulae (2/0), 20 cervical vertebrae, four thoracic vertebrae, seven lumbar vertebrae, two caudal vertebrae, ten costae, 16 humeri (7/9), eight radii (4/4), three ulnae (2/1), one scapholunar (0/1), two pisiformes (1/1), two cuboides (0/2), ten mc 2 (5/5), nine mc 3 (4/5), eight mc 4 (6/2), 16 mc 5 (9/7), 11 femora (9/2), 15 tibiae (7/8), three naviculares (2/1), 18 calcanei (9/9), 15 talii (7/8), 15 mt 2 (9/6), 18 mt 3 (10/8), eight mt 4 (4/4), ten mt 5 (4/6), 33 phalanges 1, 10 phalanges 2, and 10 phalanges 3.
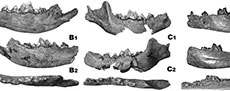
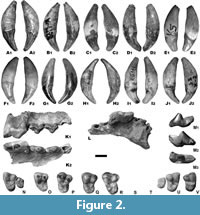 Description. The upper tooth row is curved distally and the teeth are arranged tightly. The broad and curved incisor row is moderately extended forward (Figure 2). The simply built I1 is short mesio-distally and moderately developed bucco-lingually. The apex of the crown is oriented mesio-buccally and forms an asymmetric triangle. The mesial edge is larger than the distal one. The mesial valley between them is V-shaped, shallow, and both edges are connected to each other. The I2 has a massive and compact crown, the top of which is directed distally and slightly buccally. The buccal part of the crown is concave and separated from the convex lingual side by a thin and V-shaped cingulum, almost evenly developed mesially and distally. The large, canine-like I3 has a crown placed at an angle of 40o to the root axis. The crown, ovoid, or oval in cross-section, is also flattened on both sides, especially on the lingual side. The top of the crown is twisted disto-lingually. Two edges extend from it, mesial and distal, which at the base of the crown connect with the moderately developed lingual cingulum, forming a thin ridge around the crown. The robust C1 are flattened laterally and elongated mesio-distally. Weak mesial and distal crests run from their apex to the base.
Description. The upper tooth row is curved distally and the teeth are arranged tightly. The broad and curved incisor row is moderately extended forward (Figure 2). The simply built I1 is short mesio-distally and moderately developed bucco-lingually. The apex of the crown is oriented mesio-buccally and forms an asymmetric triangle. The mesial edge is larger than the distal one. The mesial valley between them is V-shaped, shallow, and both edges are connected to each other. The I2 has a massive and compact crown, the top of which is directed distally and slightly buccally. The buccal part of the crown is concave and separated from the convex lingual side by a thin and V-shaped cingulum, almost evenly developed mesially and distally. The large, canine-like I3 has a crown placed at an angle of 40o to the root axis. The crown, ovoid, or oval in cross-section, is also flattened on both sides, especially on the lingual side. The top of the crown is twisted disto-lingually. Two edges extend from it, mesial and distal, which at the base of the crown connect with the moderately developed lingual cingulum, forming a thin ridge around the crown. The robust C1 are flattened laterally and elongated mesio-distally. Weak mesial and distal crests run from their apex to the base.
The oval-shaped P1 is a small, monocuspid, single-rooted tooth. Buccal and lingual margins are convex, and the lingual one is more strongly developed. The P2 is an elongated tooth, with a strong distal cingulum (Figure 2). The buccal margin is straight and the lingual margin is gently convex in its middle part. The mesial and distal margins are blunt. The protoconid is situated more mesially. Two thin edges run from the protoconid apex, the mesial one in mesio-lingual direction. The distal edge ends exactly medially and is connected with the distal cingulum. The P3 is elongated and robust (Table 1). The crown bears an elongated distal cingular projection. The small mesio-buccal prominence of the cingulum forms a faint mesial crest to the apex of the protoconid. This protoconid is located medially and slightly mesially. Two edges running from the protoconid apex are thick and sharp. The distal one is not connected with the distal cingulum, and its end forms a small, swelling-like bulge. The cingulum is weakly developed on the lingual side. The mesial and distal margins are blunt or rounded, while the buccal margin bears a moderately developed, median concavity. At the same level, but on the lingual margin, is located a moderate convexity (Figure 2).
The long and robust P4 has straight buccal and lingual margins of the talon (Figure 2). The mesial and distal margins are blunt or rounded. The moderately high paracone bears a thin crest across the mesial border from the apex to the base of the crown. It is separated from the moderately long and low protocone, whose mesial margin is aligned with that of the paracone. Its length is on average smaller than the distal breadth of the crown (Table 2). The metacone is separated from the paracone by a deep valley. The cingulum is more strongly developed on the lingual margin of the metacone. The M1 is large; its breadth is smaller than the P4 length (Table 3; Figure 2). The trigon is moderately wide and short, with a moderate and abrupt concavity of the buccal margin. The paracone and the metacone are elongated and well developed, where paracone is larger. They are well separated by a deep, narrow, V-shaped valley. Apexes of both main cusps are connected by a thin, long crest. The talon is shorter than the trigon and separated by a deep and broad depression running through the middle part of the crown. The protocone is low and long. It is divided by a shallow and wide valley into two parts of similar length and height. The reduced metaconule is not connected with any other cusp or crest, and is a low, elongated cuspid situated in the middle part of the crown. A long and thin crest corresponding to the buccal cingulum margin runs in parallel along the whole talon length. The well-developed lingual cingulum forms a thick crest (Figure 2).
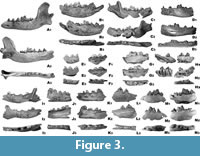 The mandibular body is long and high, and its height measured behind the m1 is larger than the m1 length (Figure 3). The height of the mandibular body gradually increases distally. Two rounded mental foramens are moderately spaced and similar in size. The mesial one is situated under the p2, while the distal mental foramen is located under the distal root of p3, slightly lower than the mesial one (Figure 3). The masseteric fossa is moderately deep and its rounded, mesial edge reaches the m1/m2 boundary. The mesial part narrows dorso-ventrally and its ventral margin only slightly exceeds the midline of the mandibular body in dorso-ventral direction. The lower mandibular body margin forms a gently curved arch, uniformly domed mesially and distally, with the strongest curvature under m1. The symphysis part is massive and elongated.
The mandibular body is long and high, and its height measured behind the m1 is larger than the m1 length (Figure 3). The height of the mandibular body gradually increases distally. Two rounded mental foramens are moderately spaced and similar in size. The mesial one is situated under the p2, while the distal mental foramen is located under the distal root of p3, slightly lower than the mesial one (Figure 3). The masseteric fossa is moderately deep and its rounded, mesial edge reaches the m1/m2 boundary. The mesial part narrows dorso-ventrally and its ventral margin only slightly exceeds the midline of the mandibular body in dorso-ventral direction. The lower mandibular body margin forms a gently curved arch, uniformly domed mesially and distally, with the strongest curvature under m1. The symphysis part is massive and elongated.
The series of cheek teeth is almost straight and the premolars are located more buccally in relation to the molars. The premolars are tightly arranged and all teeth are situated at a similar level. The triangular crown of the i3 is double cusped, with the main protoconid occupying the larger surface. It is slightly asymmetrical, being more developed on the lingual side. Its vertical top is massive and rather blunt. The c1 is long and robust, with a proportionally elongated and hook-shaped crown (Figure 3). Two longitudinal grooves run on the buccal and lingual sides of the crown.
The relatively large p1 is an oval, small, and one-rooted tooth. It is tightly squeezed between c1 and p2. The two-rooted p2 is low-crowned, with the protoconid strongly displaced mesially. Its occlusal outline is almost rectangular, with an elongated distal part. Two thin crests run in the mesial and distal direction from the protoconid top. The distal cingulum forms a thin ridge, collaring the smooth area in the distal part. The mesial part of the crown bears an elongated distal cingular projection (Figure 3). The larger p3 has a similar outline in occlusal view, with straight buccal and lingual margins. The mesial and distal margins are blunt. The protoconid is also displaced mesio-medially, but less so than in p2. An elongated distal, cingular projection is oriented slightly disto-buccally. Two thin edges running from the protoconid apex. On the distal edge, a small tubercular convexity is present just behind the top. The mesial and distal cingulum are relatively strongly developed. The two-rooted p4 is relatively high-crowned and has the protoconid placed almost exactly centrally and pushed slightly mesially. A relatively large hypoconid is present after the protoconid. It is associated with the distal crest, running distally from the protoconid apex. The mesial ridge is thinner than the distal one. The mesial and distal halves of the tooth are equal in length. The mesial part is narrower than the distal one. The crown is slightly broadened in the distal direction. A gentle lingual convexity occurs in the middle part of the crown. The lingual margin is straight. The mesial margin is blunt, while the distal one is rounded. The mesial and distal cingulum are relatively strongly developed. The distal, cingular projection is less elongated compared to the rest of the crown. Collared by a thick cingulum, the inner surface of this projection is crescent-shaped and shallow (Figure 3).
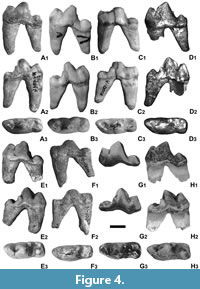 The large m1 has a massive and high trigonid (Figure 4). The short and low talonid is narrower than the trigonid. The small metaconid is connected with the protoconid. A thin, longitudinal ridge, surrounding the talonid field, ending on the metaconid base, runs from the elongated and low hypoconid. The paraconid edge is weakly developed. The mesial margin is rounded, while the distal one is blunt. The buccal margin is almost straight, with gentle concavity on the transition between the trigonid and talonid. The lingual margin of the paraconid and half of the protoconid is straight, while more distally a moderate convexity is present.
The large m1 has a massive and high trigonid (Figure 4). The short and low talonid is narrower than the trigonid. The small metaconid is connected with the protoconid. A thin, longitudinal ridge, surrounding the talonid field, ending on the metaconid base, runs from the elongated and low hypoconid. The paraconid edge is weakly developed. The mesial margin is rounded, while the distal one is blunt. The buccal margin is almost straight, with gentle concavity on the transition between the trigonid and talonid. The lingual margin of the paraconid and half of the protoconid is straight, while more distally a moderate convexity is present. 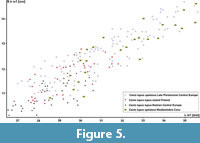 The cingulum is moderately developed. The m2 is moderately reduced. The crown has a slightly irregular, rounded occlusal outline. The large and low paraconid and protoconid are located on the trigonid. The larger paraconid is located mesio-buccaly, while the smaller, but not lower protoconid is situated medially and lingually. The talonid is narrower, with a conical and low hypoconid. The moderately developed cingulum is stronger only on the distal margin (Figure 4-Figure 5).
The cingulum is moderately developed. The m2 is moderately reduced. The crown has a slightly irregular, rounded occlusal outline. The large and low paraconid and protoconid are located on the trigonid. The larger paraconid is located mesio-buccaly, while the smaller, but not lower protoconid is situated medially and lingually. The talonid is narrower, with a conical and low hypoconid. The moderately developed cingulum is stronger only on the distal margin (Figure 4-Figure 5).
The long and massive humeri have a flat and robust head (Figure 6). The greater tubercle extends distally along the side of the articulation surface. The distally directed neck and the smaller tubercle are well-developed. The lateral epicondylar crest is very prominent towards the lateral epicondyle from the last distal fourth. The medial epicondylar crest is prominent and has a narrow supracondylar foramen. The prominent rugose area occurs on the inner surface of the shaft for the attachment of the inner humeral head of the triceps muscle. The strong medial condyle is located slightly back, while the lateral condyle is pushed deeper in mesio-distal direction. A rounded and deep coronoid fossa does not connect with the wide and deep olecranon pit (Figure 6).
The radius is robust, flattened dorso-ventrally, and has a slightly concave ventral surface (Figure 6). The large head has a shallow and rounded articular surface. It is mesially collared by a prominent coronoid process. The articulation surface for the lesser sigmoid cavity is large. The distal end is widened laterally, with a well-marked elliptical glenoid cavity, framed by a rough condyle on ventral and medial sides. The facet for the ulna and the styloid process are well developed. The grooves for extensor muscle tendons, extensors carpi radialis longior and brevior muscles are broad and deep. The scapholunar articulation area is relatively short and broad (Figure 6).
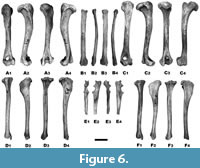 The ulna is elongated and narrow, with a strong but short proximal end (Figure 6). The surface for attachment of the scapular head of the triceps muscle is broad and strongly rugose. The olecranon has a quadrilateral shape with its apex hollowed out in part of a small cavity bordered by two fairly prominent tubercles. The shaft is curved in the lateral and medial view and massive in its longitudinal extent. The articulating surface for the radius is large, while the articulating surface for the pisiform is quite narrow. The distal end is characterised by a significant protrusion of the ulnar styloid process (Figure 6).
The ulna is elongated and narrow, with a strong but short proximal end (Figure 6). The surface for attachment of the scapular head of the triceps muscle is broad and strongly rugose. The olecranon has a quadrilateral shape with its apex hollowed out in part of a small cavity bordered by two fairly prominent tubercles. The shaft is curved in the lateral and medial view and massive in its longitudinal extent. The articulating surface for the radius is large, while the articulating surface for the pisiform is quite narrow. The distal end is characterised by a significant protrusion of the ulnar styloid process (Figure 6).
Long, cylindrical femora have a spherical head with a prominent neck (Figure 6). The trochanteric fossa is broad and deep. The lesser trochanter is a slight conical prominence. The shaft is slightly bowed in its longitudinal extent. The greater trochanter rises distinctly above the head level and is obliquely truncated on its lateral side. The surface of the trochanter extends further down the proximal extremity of the femur. The tibial articulations are nearly equal in size. At the distal end, the condyles are separated by a broad and deep groove. The lateral condyle is slightly more developed as the medial one (Figure 6).
The body of long and massive tibiae are triangular in its proximal third, slightly convex medially, with a large tibial fossa from the lateral side (Figure 6). The distal surface below the head is narrow and shallow. The tibial crest is well marked but relatively short. At the distal end, the articular surface consists of two separate grooves. The medial throat is deeper than the lateral one. The grooves for tendons of the flexor longus digitorum and tibialis posticus muscles are well defined at the distal epiphysis. The notch incising the mesial border of the distal surface is quite small (Figure 6).
 The calcanei are long and robust, with strongly developed tuber calcanei. The coracoid process is prominent and distinct from the sustentaculum tali. The medial part of the bone is flat and wide. Its proximal articular surface is concave whereas the distal articular surface is flat. The distal end of a tali has a well-marked neck and head. The body is flattened in the dorsoplantar direction. The trochlear margins are slightly asymmetrical. The lateral lip is slightly more prominent but shorter than the medial one.
The calcanei are long and robust, with strongly developed tuber calcanei. The coracoid process is prominent and distinct from the sustentaculum tali. The medial part of the bone is flat and wide. Its proximal articular surface is concave whereas the distal articular surface is flat. The distal end of a tali has a well-marked neck and head. The body is flattened in the dorsoplantar direction. The trochlear margins are slightly asymmetrical. The lateral lip is slightly more prominent but shorter than the medial one.
Comparison. The comparison of various European populations, dated to the last 130 kya, showed considerable size variation within this period. Some significant differences were found in tooth proportions regarding the proportions of the length and width of the trigonid, but those from cold periods have more massive m1 overall (Figure 1, Figure 2, Figure 3). From the Eemian, the length of m1 rarely exceeds 30 mm, and the specimens are characterised by moderate dimensions and stature. Since these wolves are metrically and morphologically indistinguishable from the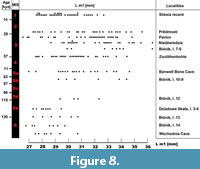 Central European populations, they were classified as Canis lupus lupus. A sharp increasing of the size is observed since MIS 5d, with the first cold period. The period between MIS 5d-5c is poorly represented in Central European sites, but few specimens from dated on MIS 5d layer 12 of Biśnik Cave showed presence of robust form. Interesting similarity was found between British site Banwell Bone Cave and layers 10-9 of Biśnik Cave, dated on MIS 5b-5a. In this cold period, large wolves were found in both localities. The largest specimens were documented from the MIS 3, which is the best represented by the whole analysed time period. Later in the Holocene (MIS 1), a decrease in size is observed, and wolves from that period are comparable to that of MIS 5e (Figure 8). The numerous French or Italian sites with abundant wolf material were not used for comparison, because wolves from contemporary Central European localities are significantly larger than those from the southern or western part of the continent.
Central European populations, they were classified as Canis lupus lupus. A sharp increasing of the size is observed since MIS 5d, with the first cold period. The period between MIS 5d-5c is poorly represented in Central European sites, but few specimens from dated on MIS 5d layer 12 of Biśnik Cave showed presence of robust form. Interesting similarity was found between British site Banwell Bone Cave and layers 10-9 of Biśnik Cave, dated on MIS 5b-5a. In this cold period, large wolves were found in both localities. The largest specimens were documented from the MIS 3, which is the best represented by the whole analysed time period. Later in the Holocene (MIS 1), a decrease in size is observed, and wolves from that period are comparable to that of MIS 5e (Figure 8). The numerous French or Italian sites with abundant wolf material were not used for comparison, because wolves from contemporary Central European localities are significantly larger than those from the southern or western part of the continent.
Genus VULPES Garsault, 1764
Vulpes vulpes (Linnaeus, 1758)
(Figure 9-Figure 10; Table 4; Appendix 1)
 Referred material. The material (NISP 35, MNI 5; left/right bones are given in brackets) includes one C1 (0/1), one P4 (1/0), two M1 (1/1), one M2 (0/1), eight mandibles (5/3), three c1 (3/0), three m1 (2/1), one scapula (1/0), one atlas, two humeri (1/1), one radius (0/1), two ulnae (1/1), two femora (2/0), one tibia (0/1), three calcanei (2/1), one mt 4 (0/1), and one phalanx 2.
Referred material. The material (NISP 35, MNI 5; left/right bones are given in brackets) includes one C1 (0/1), one P4 (1/0), two M1 (1/1), one M2 (0/1), eight mandibles (5/3), three c1 (3/0), three m1 (2/1), one scapula (1/0), one atlas, two humeri (1/1), one radius (0/1), two ulnae (1/1), two femora (2/0), one tibia (0/1), three calcanei (2/1), one mt 4 (0/1), and one phalanx 2.
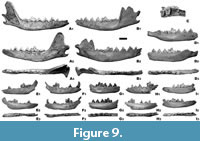
Description. The elongated and robust P4 has a high and prominent paracone and a mesio-distally elongated metastyle. The moderately developed protocone is pushed more mesially than the mesial crown margin. The mesio-buccal cingulum is absent, and a small but sharply pointed parastyle is present. The disto-lingual cingulum is strongly developed. The M1 trigon is broadly expanded mesio-distally. The trigon basin is round and wide, and as deep as the talon basin, which is weakly compressed and semi-circular. There is a prominent shelf-like buccal cingulum that extends from the mesial wall of the paracone to the distal wall of the metacone. The paracone and metacone are similar in size, but the paracone is slightly higher. On the mesio-buccal side, there is a prominent parastyle. The well-developed and high protocone and the hypocone are separated from the lingual cingulum by a marked furrow. The metaconule is moderately high and prominent, the protoconule is low and small. The M2 trigon is mesio-distally less expanded than that in M1. The paracone is higher and larger than the metacone. The protocone is well developed and a small but well recognised metaconule is situated distally (Figure 9).
The mandibular corpus is deep and stout, becoming medio-laterally thicker ventrally. Its height gradually increases distally. Two mental foramina are located below the p1 (mesial, larger) and below the mesial root of the p3 (smaller, distal) that emerges more dorsally than the former (Figure 9). The rounded mesial edge of the deep masseteric fossa reaches the m3. The mesial part narrows dorso-ventrally and its ventral margin only slightly exceeds the midline of the mandibular body in dorso-ventral direction. The lower mandibular body margin forms a gently curved arch, with the strongest curvature under m1. The symphysis part is elongated and moderately robust. The cheek teeth row is almost straight, and the premolars are located more buccally in relation to the molars. The premolars are loosely arranged and separated by small diastemas.
The c1 is long and robust, with a proportionally elongated and hook-shaped crown (Figure 9). Two longitudinal grooves run on the buccal and lingual sides of the crown. The relatively large p1 is an elongated, oval, small, and one-rooted tooth. The two-rooted p2 is high-crowned, with the protoconid strongly displaced mesially. The crown is elongated and narrow, gently convex buccally and with weak median convexity. It has an elongated distal cingular projection. After the protoconid is located slightly disto-buccally a minute cuspid, which in some specimens is weakly marked only as a ridge. The elongated and narrow p3 gently widens distally. It has straight buccal and lingual margins, while the mesial and distal margins are blunt. The protoconid is also displaced mesio-medially, however less than in p2. An elongated distal cingular projection is oriented more disto-buccally. On the distal edge, a small tubercular convexity is present just behind the top. The mesial and distal cingulum is weakly developed. The high-crowned p4 has the protoconid strongly displaced mesio-medially. A prominent cuspid is present after the protoconid. It is associated with the distal crest, running distally from the protoconid apex. The crown is slightly broadened in the distal direction, with straight buccal and lingual margins. The mesial and distal margins are blunt. The distal cingular projection is less elongated in comparison to the p2-p3 and collared by a thick cingulum. The inner surface of this projection is crescent-shaped and shallow.
The elongated and robust m1 has a massive and high trigonid and proportionally long and low talonid, slightly narrower than the trigonid. The paraconid is low and short. The large and trapezoidal metaconid is moderately distinct from the high and prominent protoconid. The mesial margin is rounded to triangular, and the distal one is blunt. The lingual margin is straight, while the buccal one holds a strong concavity on the transition between the trigonid and talonid. The cingulum is moderately developed. The hypoconid is large and high, while the entoconid is lower and smaller. The m1 is bucco-lingually stout and high, especially at the level of the trigonid, whereas the protoconid is stout and large. The talonid basin is round, smooth, and wide, partially enclosed lingually by the entoconulid. Distally, there is a prominent cingulid (Figure 9).
The broad and large m2 has a slightly irregular, rounded occlusal outline. On the trigonid are located mesio-buccaly a larger and higher protoconid and medio-lingually a lower and smaller metaconid. The talonid is narrower, with a conical and low hypoconid and a distinctly lower and smaller entoconid. A small but well-recognised mesoconid is also present. Before the entoconid, which developed an inner edge, is also present a small proentoconid. The moderately developed cingulum is stronger only on the distal margin. The relatively weakly reduced m3 has oval or rounded outline and bears two equally sized cusps, the protoconid and the metaconid. The talonid is preserved as a semi-circular ridge (Figure 9).
Comparison. The analysed material of Vulpes vulpes from Niedźwiedzia Cave is a good example of great metrical and morphological variability of this species (Lucenti and Madurell-Malapeira, 2020). The single P4 (JN.2.5) has a narrow mesial margin of the paracone, typical for the species. However, the morphology of this structure is highly variable, and this part can be reduced or, on the contrary, expanded. The enlarged and separated protocone dominated in V. vulpes. Nevertheless, in some P4, it is reduced or fairly prominent and partially associated to the paracone, like in JN.2.5. The mesial embayment of P4 (JN.2.5) is moderately developed, and strongly vary in V. vulpes, where it can be narrow, wide, or even absent. Vulpes vulpes generally does not possess cingulum on the mesio-buccal side of the P4, but specimens with fairly to well-developed cingula are not uncommon, like that in JN.2.5. The occurrence of a distinct parastyle is rare in V. vulpes and V. corsac and more common in V. lagopus (Gimranov, 2017; Lucenti and Madurell-Malapeira, 2020). It is also absent in P4 from Niedźwiedzia Cave. Some P4 of V. vulpes possesses an accessory cuspule on the preparacrista, located just above the mesial embayment. Such structure is also present in JN.2.5 in a form of minute, but well-recognised ridge.
Characteristic for Vulpes vulpes is that the medial protocone crest is fused to the preparacrista, but in some specimens it is incomplete or curved distally not reaching it. Generally, the morphology of the medial protocone crest and its relation to the preparacrista is highly variable in this canid (Lucenti and Madurell-Malapeira, 2020). In JN.2.5, this structure is straight, projecting medially toward the lower third of the preparacrista, and this type of morphology dominated in V. vulpes. Much less commonly occurs an arched medial protocone crest, with parabola-like shape, which extends linguo-medially and then ventrally toward the apex of the crown. Typical for V. vulpes, which is also observed in JN.2.5 tooth, is the protocone collared by a prominent cingulum, which is sometimes incomplete and rarely absent. The P4 paracone is longer than the metastyle, like in JN.2.5, although in some specimens they are comparable in length. The disto-lingual cingulum in JN.2.5 is moderately marked. The stage of development of this feature is highly variable, from nearly absent to particularly strong.
The occlusal outline of the M1 is highly variable, especially the proportion and stage of development between the trigon and talon. In JN.2.5, the M1 has a particularly broad and expanded trigon, T-shaped in the occlusal view, with proportionally narrow and short talon. The M1 embayment on the buccal cingulum is strongly marked, while in Vulpes vulpes varies from reduced to strongly developed. The JN.2.5 has an expanded buccal cingulum on the metacone, where the hypocone lobe connects mesially to the mesial cingulum. The bean-shaped M2 has a moderately expanded trigon and possesses a distinctly marked notch, situated lingually to the metacone. A strong median notch is also located in the buccal margin.
All p4 from Niedźwiedzia Cave have the distal portion slightly elongated and enlarged distally, with a rounded outline, typical for the species. Of the 10 m1 from Niedźwiedzia Cave, eight are stout and enlarged in bucco-lingual direction. Two other teeth are bucco-lingually compressed, narrow, and smaller. The morphology of the m1 from Niedźwiedzia Cave is highly variable, similar to that of the extant Vulpes vulpes. Of the 10 m1 from Niedźwiedzia Cave, seven have reduced or weakly developed inflexion on its lingual side of the protoconid and the area is nearly convex. In another three teeth, this structure is stronger developed. The metaconid is well developed, prominent, and well separated from the protoconid in nine m1 from Niedźwiedzia Cave, while only a single specimen possesses a more reduced and less separated metaconid. The presence of a transverse cristid is common for the extant V. vulpes, as in the material from Niedźwiedzia Cave, where this structure is present in all 10 teeth analysed. In six m1, this feature is strongly marked, while in four others is weakly to moderately developed. Very variable is also a constellation of accessory lingual cuspulids, located mesially to the m1 entoconid on the m1 talonid. In eight teeth, the presence of a reduced (n = 5) or an enlarged (n = 3) entoconulid was found, while in two others the entoconulid was present with the addition of a mesial accessory cuspulid. No m1 from Niedźwiedzia Cave with the absence of cuspulids mesial to the entoconid have been found. Another highly variable feature is the morphology of the distal margin of the m1. In three specimens, the margin is a simple cristid bounding the distal margin, while in three others distal accessory cuspulids are located in place of a cingulid. In single specimens this structure is developed into a form of a distal cristid arising from an evident hypoconulid, as an enlarged hypoconulid with no distal cristid, a strongly reduced one, or a reduced distal cristid or no cristid.
In three m1 from Niedźwiedzia Cave, the m2 has a more ovoid shape, with moderately to strongly marked hypoflexid and buccal median concavity. This structure is not present in the other two teeth, and their occlusal outline is more bean-like. Similarly variable is the morphology of a buccal cingulid situated on the mesio-buccal side of the protoconid. In four m2 from Niedźwiedzia Cave, it is reduced to moderately developed, limited to the mesial part of the protoconid. Only a sole specimen possesses an enlarged buccal cingulid that prominently extends distally on the buccal side of the hypoconid. In single specimens, the m2 possess the entoconid as a single cuspid or a very reduced one, while the rest of the specimens show a large entoconid and a mesial accessory cuspulid (Figure 9). All three m3 from Niedźwiedzia Cave are small and rounded in occlusal view, while in the extant V. vulpes this tooth is often large and rounded or large and oval (Lucenti and Madurell-Malapeira, 2020). The buccal protoconid and the lingual metaconid are generally subequal in size, while the protoconid can be larger than the metaconid. In some specimens of the extant V. vulpes the metaconid is absent. Instead, those teeth have a single buccal cuspid and a lingual cristid or a single central cuspid (Lucenti and Madurell-Malapeira, 2020).
Despite the great variability of Vulpes vulpes from Niedźwiedzia Cave, we did not find any differences between the fossil material from Niedźwiedzia Cave and extant Polish populations. While no particular morphological features distinguishing the Niedźwiedzia Cave material and the extant V. vulpes have been found, there are some interesting metric patterns. Firstly, the red fox from this locality is characterised by considerable size, exceeding the average size of the extant Silesian population, even if their ranges of variation overlap. It is well documented, when compared the L m1 of the material from Niedźwiedzia Cave (17.19 mm, 16.31-17.88 mm, n = 6 in males and 14.75 mm, 14.22-15.22 mm, n = 4 in females) with the extant Silesian V. vulpes (16.32 mm, 14.78-18.16 mm, n = 114 in males and 14.48 mm, 12.66-15.56 mm, n = 108 in females). Documented sexual dimorphism is the second factor, typical for the carnivores (Dayan et al., 1989). The result of this pattern is a separation of food niches of particular species and a consistent trend for females to consume smaller preys than males (Pimm and Gittleman, 1990). Across carnivores there is a general tendency for carnassials to be less dimorphic than canines. Canids display less specialised killing behaviour than, for example, mustelids, the canines are 8-10% larger in males, while the m1 is larger by 5-6% (Gittleman and Van Valkenburgh, 1997; Szuma, 2000, 2004).
Such great metric and morphological variability is characteristic for Vulpes vulpes (Szuma, 2000, 2003, 2004, 2007, 2008a, 2008b), while is less pronounced in other foxes (Szuma, 2008c, 2011; Gimranov et al., 2015; Gimranov, 2017; Lucenti and Madurell-Malapeira, 2020). This variability results from the combination of biological, climatic, and geographic factors. Among them latitude, habitat productivity, differential food availability, intraguild, intrafamily or intraspecific competition, character displacement, genetic diversity, and population density are the most important (Szuma, 2008b; Szuma and Germonpré, 2020). Different Vulpes species are susceptible to different combinations of these proxies, with resulting different variability and features (Szuma, 2011). For V. vulpes, latitude, mean annual temperature, and longitude significantly affect the distribution of morphotypes (Szuma, 2007). Instead, geographic and climatic factors are less important for V. lagopus, which is more influenced by its interspecific competition with V. vulpes and food source variability and accessibility (Szuma, 2011; Szuma and Germonpré, 2020). Additionally, the geographic range of the extant V. vulpes is enormously larger and more diverse in terms of habitat conditions and types, compared to that of V. lagopus, which affects the importance of the above-mentioned factors (Szuma 2007, 2008b, 2011; Lucenti and Madurell-Malapeira, 2020).
Vulpes lagopus (Linnaeus, 1758)
(Figure 9-Figure 10; Table 4; Appendix 1)
Material. The material (NISP 22, MNI 4; left/right bones are given in brackets) includes three maxillae fr. (2/1), one M1 (0/1), one M2 (0/1), seven mandibles (4/3), three humeri (1/2), two ulnae (1/1), two femora (2/0), and three tibiae (2/1).
Description. The premolar teeth row is straight, and the teeth are set loosely. The elongated and narrow P2 and P3 bear an elongated distal cingular projection. The prominent protocone is situated almost exactly in the middle axis of the crown and is pushed more mesially. The small mesio-lingual prominence of the cingulum forms a faint mesial crest to the apex of the protocone of the P3. The cingulum is weakly developed on the lingual side. The relatively long and narrow P4 has almost straight buccal and lingual margins. The paracone is high and well separated from the proportionally short and low protocone. Its length is smaller than the distal breadth of the crown. The cingulum is strongly developed on the lingual margin of the metacone. The triangular M1 has a broad, moderately expanded trigon and a long and wide talon. The paracone is large and high, exceeding the metacone slightly in size and height. The small and low protocone is connected by a thin and sharp crest with the small entocone. A small protoconule is situated on the mesio-lingual part of the talon. The low and elongated hypocone is a strong crest-like structure. The main basin between the trigon and the talon and between the protocone and hypocone are deep. There is a well-developed mesio-buccal cingulum. The M2 is more strongly reduced compared to the size of M1, with a wide talon oriented strongly disto-lingually. All main cusps are low with the paracone being larger and higher than the metacone. There is no connection between the protocone and the metacone. The trigon fossa communicates with the talon fossa by a narrow, V-shaped valley (Figure 9).
The mandibular corpus is deep and stout, becoming medio-laterally thicker ventrally and its height gradually increases distally. Two mental foramina are located below the mesial roots of p2 and p3 (Figure 9). The rounded mesial edge of the deep masseteric fossa reaches the m3. The lower mandibular body margin is straight, only in one specimen forms a gently curved arch, with the maximum under the m1. The symphysis part is elongated and narrow. The cheek teeth row is straight, and the premolars are located more buccally in relation to the molars and are tightly arranged.
The c1 is long and robust, with an elongated and hook-shaped crown (Figure 9). The p1 is an elongated, oval, small, and one-rooted tooth. The p2 is high-crowned, with the protoconid strongly displaced mesially. The crown is elongated and narrow, gently convex buccally and with a weak median convexity. It has an elongated distal cingular projection. The elongated and narrow p3 gently widens distally. The protoconid is also displaced mesio-medially, but less so than in p2. The high-crowned p4 has a protoconid strongly displaced mesio-medially. A prominent cuspid is present after the protoconid. The crown is slightly broadened in the distal direction, with straight buccal and lingual margins. The elongated and narrow m1 has a massive and high trigonid and a proportionally long and low talonid, slightly narrower than the trigonid. The large and trapezoidal metaconid is moderately distinct from the protoconid. The cingulum is moderately developed. The hypoconid is large and high, while the entoconid is lower and smaller. The talonid basin is round, smooth, and wide, partially enclosed lingually by the entoconulid (Figure 9). The elongated and narrow m2 has a slightly irregular, rounded occlusal outline. On the trigonid are located a larger and higher protoconid mesio-buccally and a lower and smaller metaconid medio-lingually. The talonid is narrower, with a conical and low hypoconid and a rudimentary entoconid. The moderately developed cingulum is stronger only on the distal margin. The m3 has an oval outline and bears two equally sized cusps, the protoconid, and the metaconid.
Comparison. The dentognathic material from Niedźwiedzia Cave metrically and, above all, morphologically corresponds to Vulpes lagopus. Numerous features that distinguish the dental material of both foxes from Niedźwiedzia Cave have been found.
Compared to Vulpes vulpes, V. lagopus possesses:
(1) more tightly set teeth;
(2) less elongated and expanded crowns of I1-I3 with weak lingual cingulum;
(3) shorter and less curved C1 with weak lingual cingulum;
(4) narrower and more reduced P1;
(5) P2 relatively short and low, not widened distally;
(6) broader and more compact P4, with short protocone that does not protrude far mesio-lingually, less projected metastyle, and without lingual cingulum;
(7) M1 with distinctly less expanded trigon, more reduced metacone and protocone, and a mesial cingulum that is not connected to the hypocone forearm;
(8) M2 with distinctly narrower trigon, a completely interrupted protocone-metacone connection, and a trigon fossa that is connected to the talon fossa;
(9) mandible with straight lower margin and lower and narrower mandibular body in its mesial part;
(10) narrower and less curved c1;
(11) more compact and broader p2-p4, with less-developed distal accessory cuspulids;
(12) narrower m1 with less positioned metaconid and no connection between the entoconid and hypoconid; and
(13) narrower m2.
Postcranial material. In addition to the described dentognathic remains, the postcranial material of foxes from Niedźwiedzia Cave is also abundant. It is represented by long bones and calcanei, mainly well preserved and often complete. However, most authors concluded that there are no substantial differences between Vulpes vulpes and V. lagopus in the morphology of the postcranial bones (Mostecký, 1969; Beneš, 1975). The variability of particular features is so high that it is impossible to establish any characteristics for the particular species. In this context, the only reliable criteria for distinguishing the postcranial elements of Vulpes species are dimensions. A diagnosis of the postcranial skeleton usually has not been given. Gromova (1950) pointed out that a “distinction is possible on the basis of measurement only, the possibility of confusion of extreme values being considerable.” Additionally, Beneš (1975) summarised this pattern as follows: “The great uniformity of canids leads either to underestimation or overestimation of morphological or metric differences. This results either in the endeavour to concentrate all Pleistocene foxes into one or two recent species, or the creation of a great number of new species based on subordinate features only.”
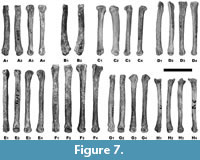
Apart from the uniform morphology and great variability, there is also the problem of the often-fragmentary nature of the analysed postcranial material. However, this problem can be overcome by estimating the total length of a given bone based on the preserved fragment. Analysis of the extant series of Vulpes vulpes from Silesia showed that the mutual proportions of length and width of a given skeletal element show a relatively small range of variability and are quite constant. Using the obtained constants, it is possible to estimate the total length with statistically significant probability and compare it to the range of variability of a given species. Body size and sexual dimorphism is marked enough in foxes to separate V. vulpes and V. lagopus and to separate males and females based on long bones (Monchot and Gendron, 2010). Metrically, there is some overlap between V. vulpes and V. lagopus, but it is possible to distinguish the long bones of these two species. The bones of V. lagopus are always smaller than those of V. vulpes of the same sex. No overlap occurs in the ranges of measurements between the two species and sexes, except for a very few parameters, and even that overlap is rather little (Monchot and Gendron, 2010).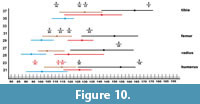 Long bones are recognisable and measurable markers for species determination (Figure 10). Foxes are characterised by moderate sexual dimorphism, with male being 10-15% larger than females (Monchot and Gendron, 2010). The metric analysis of sexes of V. vulpes and V. lagopus from Niedźwiedzia Cave is based on the differences in size between males and females of extant foxes (Table 4). These differences were shown in all long bones, and the most important was the greatest length (GL). This value was lower in females than in males in all analysed bones (Figure 10).
Long bones are recognisable and measurable markers for species determination (Figure 10). Foxes are characterised by moderate sexual dimorphism, with male being 10-15% larger than females (Monchot and Gendron, 2010). The metric analysis of sexes of V. vulpes and V. lagopus from Niedźwiedzia Cave is based on the differences in size between males and females of extant foxes (Table 4). These differences were shown in all long bones, and the most important was the greatest length (GL). This value was lower in females than in males in all analysed bones (Figure 10).
For the humerus, the two main indexes are the ratio of the length of the proximal epiphysis to the greatest length (pL/GL) and the breadth of the distal epiphysis to the greatest length (dB/GL) (Table 4). The first ratio (pL/GL) is 20.4 (18.7-22.3, n = 68), while the second index (dB/GL) is 16.6 (14.8-18.2, n = 68). Of the five fox humeri from Niedźwiedzia Cave, two were identified as Vulpes vulpes, such as the huge complete humerus (JN.2.10) with a GL of 166.14 mm, which even slightly exceeds the size of the extant Silesian V. vulpes, and belongs to a male (Table 4). The second specimen (JN.2.24) is smaller, with an estimated GL of 135 mm (125-145 mm), which may indicate both a small male and a large female, but rather closer to a male. Three humeri were assigned to V. lagopus, two of which have similar dimensions: JN.3.6 with a GL of 112.76 mm and JN.3.21 belonged to males, whereas the distinctly smaller third bone (JN.3.22) was a female.
The morphological differences are much less pronounced. Beneš (1975) found that the tuberculus major on the proximal epiphysis is larger and more expanded in relation to the surface in Vulpes vulpes than in V. lagopus. Simultaneously, all humerus edges of V. lagopus are more sharply developed and marked than those of V. vulpes. Our observations showed that both features, however, are quite variable, and can be traced only with difficulty. With regard to the metric differences, confusion of the humerus between V. vulpes and V. lagopus is little probable.
All three ulnae from Niedźwiedzia Cave are incomplete and are represented only by proximal parts. The two larger (JN.2.2 and JN.2.26) were assigned to Vulpes vulpes, while the smaller one (JN.3.4) to V. lagopus. As in the case of the humerus, there is a clear distinctness in the GL and pL between both fox species. Gromova (1950) found that the ulna of V. lagopus can be morphologically distinguished from that of V. vulpes on the basis of two main criteria. The central part of the diaphysis of V. lagopus is mostly strongly flattened, so that the largest anterio-posterior diameter rarely attains less than 140% of the transverse section. The posterior part of the olecranon processes in V. lagopus is raised only slightly. In V. vulpes, the dorsal points are highly raised, to the same levels as that or the olecranon itself, and between the posterior points and the olecranon there is a saddle-like depression (Beneš, 1975). Our observations did not confirm this distinctness with certainty and those features showed some degree of variation. Additionally, they are not regarded as very useful because the olecranon processes are often mechanically damaged. In this context, the metric differences are much more indicative (Table 4).
The best preserved among all long bones of foxes are the femora represented by four complete specimens. The analysis of their taxonomic affiliation well illustrates the complexity of correct identification of the fox material. The largest bone (JN.2.22), with a GL of 133.82 mm, falls into the size variability of Vulpes vulpes as a small male or a large female. The two smaller femora, JN.3.19 with a GL of 114.16 mm and JN.3.20 with a GL of 105.64 mm, were identified as V. lagopus. The identification of the specimen JN.2.23 with a GL of 119.72 mm is problematic, which falls in the range of variability of V. vulpes females and V. lagopus males. The value is close to the mean of V. vulpes females and was therefore determined as such. However, it cannot be ruled out that this particular specimen is, in fact, V. lagopus, since no substantial morphological differences have been found between the two foxes (Gromova, 1950; Mostecký, 1969; Beneš, 1975).
Of the four tibiae from Niedźwiedzia Cave, only the largest one, JN.2.3 with a GL of 172.84 mm, represents a robust male of Vulpes vulpes (Table 4). The other three tibiae (JN.3.16, JN.3.17, and JN.3.18) were assigned to small and medium-sized specimens of Vulpes lagopus. Also, in the case of this bone, no reliable morphological features distinguishing V. vulpes from V. lagopus can be found. Concluding, only metric data are reliable enough to distinguish between the two foxes, while morphological features have little value in taxonomic identification (Mostecký, 1969; Beneš, 1975; Reichstein, 1984; Bisaillon and Deroth, 1979, 1980; Altuna, 2004; Germonpré and Sablin, 2004; Baryshnikov, 2006; Monchot and Gendron, 2010).
DISCUSSION
The first occurrence of Canis lupus, which evolved from Canis mosbachensis Soergel, 1925, is one of the defining bioevents of the faunal turnover that took place during MIS 12-11 (ca. 450-400 kya) (Lucenti et al., 2017; Iurino et al., 2022). The morphological differences between C. mosbachensis and C. lupus have long been vaguely defined, as the remains of early wolves are mainly of poor taxonomic value and frequently share mosbachensis -like characters (Mecozzi et al., 2020). Those small wolves of gracile posture, metrically comparable with small subspecies of the extant C. lupus like the Indian wolf Canis lupus pallipes Sykes, 1831 or the Arabian wolf Canis lupus arabs Pocock, 1934, were traditionally assigned as a chronosubspecies Canis lupus lunellensis Bonifay, 1971, dated to MIS 11 (Bonifay, 1971; Baryshnikov, 2012, 2020). However, the estimated age of the fossil material from Lunel-Viel was based on biochronological data, and new studies are pointing to a younger age of MIS 9 (Brugal et al., 2020).
An increase in dimensions is recorded across the late Middle Pleistocene, with the largest Canis lupus reported from the Late Pleistocene (Flower, 2016; Mecozzi and Lucenti, 2018; Iurino et al., 2022). Since the first description (Goldfuss, 1823), the robust and short-legged C. l. spelaeus from the Late Pleistocene has always been distinguished from the extant C. lupus by its larger size and robustness. This resulted in classification under different names like Canis lupus brevis Kuzmina and Sablin, 1994 or Canis lupus maximus Boudadi-Maligne, 2012 (Kuzmina and Sablin, 1994; Boudadi-Maligne, 2010, 2012). Later revisions regarded both forms as a younger synonym of C. l. spelaeus (Diedrich, 2022; Marciszak et al., 2023b). Features described their independency like 10-20% larger dimensions than in the extant C. lupus, enlarged and more robust dentition (especially m1), with more strongly developed distal cusplets on P2-P3/p2-p4, and shorter legs proportionally to the body size were highlighted by previous authors (Ballesio, 1979; Kuzmina and Sablin, 1994; Boudadi-Maligne, 2010, 2012). All those features characterised also C. l. spelaeus from Niedźwiedzia Cave, as well as various populations of this chronosubspecies across Eurasia, dated to MIS 5-2 (Diedrich, 2022; Marciszak et al., 2023b).
Canis lupis spelaeus is a characteristic element of the Late Pleistocene European palaeoassemblages. Their remains are abundant across Europe at sites dated to MIS 5-2 (Boudadi-Maligne, 2010, 2012; Diedrich, 2014, 2017, 2022). No complete wolf skull is known from the Sudeten sites. However, Langenhan (1904a, 1904b) described a skull of a giant wolf with particularly massive dentition from the Południowa Cave, which is now probably lost. Large-sized wolves with robust dentition and enlarged carnassials seem to be typical of cold phases of the Late Pleistocene (Boudadi-Maligne, 2010, 2012; Flower and Schreve, 2014; Diedrich, 2017). In the Sudeten and Silesia, remains of C. l. spelaeus are commonly found (Frenzel, 1936; Zotz 1939, 1951; Wiszniowska, 1967, 1970, 1986, 1989; Marciszak et al., 2016, 2020, 2023b).
Silesia, like the rest of Europe, was profoundly affected by the glacial/interglacial cycles, and this is reflected in a geographic variation following Bergmann’s ecogeographical rule. Climatic changes produced strong palaeoecological and paleoenvironmental instability that affected the faunal assemblages. However, in the case of the wolf, the problem is more complex, as was noted by Sansalone et al. (2015, p. 45): “Indeed, the carnassial dimensions are not directly correlated with the climate changes, but they are possibly correlated with the changes in faunal composition.” This pattern is also observable in Sudeten wolves, which were the main scavengers and hunters under the absence of Crocuta crocuta spelaea in this region (Marciszak et al., 2020).
The first large Canis lupus was recorded in sediments dated to MIS 8 of the Sudeten sites Naciekowa and Wschodnia Cave, metrically and morphologically comparable with the extant wolves (van der Made et al., 2014). Both records are regarded as one of the oldest large-bodied wolves in Europe, which probably documented an eastern immigration to Central Europe. The Polish wolves from the period of MIS 7-5e, the lowermost horizons of Komarowa and Wierzchowska Górna Caves, Nietoperzowa Cave (layers 18-15), Biśnik Cave (layers 18-14), and Dziadowa Skała Cave (layers 4-3), are slightly smaller than the extant Polish C. lupus and distinctly smaller than C. l. spelaeus (Marciszak et al., 2023b).
The body size of Canis lupus in Europe has followed a steady increase from their first appearance up to the peak of the Last Glacial Maximum (LGM). The dimensions of these wolves are thought to be an adaptation to cold environments (wolf as a species follows Bergmann’s rule) and hunting on large herbivores (Sardella and Palombo, 2007; Meloro and Sansalone, 2022). This event is followed by high taxonomic diversity during the Early to Middle Pleistocene and, later, by a specific lineage with high size variation during the Middle and Late Pleistocene (Brugal and Boudadi-Maligne, 2011). The geographic ranges of canids are particularly wide across the earth, and they demonstrate a high morphological and size (clinal) variability. Such wide distributions cover several distinct climates and vegetation environments. This often implies a morphological, biometrical, and genetic variability for the species, expressed as subspecies or geographical variants in taxonomy. The morphology and body size are among the two main analytical parameters of canids (Brugal and Boudadi-Maligne, 2011).
Morphological changes of Canis lupus during the late Middle-Late Pleistocene might be due to a preference for larger preys, which results in larger individuals. The main differences from the extant C. lupus are the shortened rostrum, considerably stronger development of the temporalis muscle, and distinctly robust premolars and molars (Therrien, 2005). These features were specialised adaptations for the processing of carcass and associated with hunting and scavenging on Late Pleistocene large ungulates, members of the mammoth steppe fauna (Leonard et al. 2007; Tanner et al., 2008; Diedrich, 2014, 2017, 2022). Some of the Late Pleistocene C. lupus show considerably higher (5-14%) percentage of individuals with worn and broken teeth than in the extant populations (1.1-2.6%) (Van Valkenburgh et al., 2019).
A similar pattern was observed in Crocuta crocuta spelaea Goldfuss, 1823 and the extant Crocuta crocuta (Erxleben, 1777), suggesting that enlarged incisor (for gnawing) and carnassial (for cracking) fracture reflects habitual and notorious bone consumption, since bones are gnawed with incisors and subsequently cracked with the cheek teeth (Leonard et al., 2007; Tanner et al., 2008; Diedrich, 2014, 2017, 2022). These higher fracture rates were across all teeth, but those for the canines were the same as in the extant Canis lupus. It can be related not only to hunting larger prey, but also with the increasing competition (Van Valkenburgh and Hertel, 1993; Van Valkenburgh, 2008). When the potential prey availability lowered, competition between carnivores increased, causing them to eat faster and thus consume more bones, leading to tooth breakage and damage (O’Keefe et al., 2014). Additionally, it was also an adaptation for grinding up quickly freezing carcasses. Wolves from populations older than 13-12 kya showed a significantly higher percentage of worn teeth than those younger. It is linked with the disappearance of larger ungulates and the decline and extinction of other competitors like large ursids and felids (Binder et al., 2002; Van Valkenburgh, 2008; Van Valkenburgh et al., 2019).
The ecological plasticity of Canis lupus during the late Middle-Late Pleistocene (MIS 7-1) was well studied in Britain (Flower and Schreve, 2014). Its abilities to cutting and crushing, highlighted in its cranio-dental plasticity, are responses to dietary changes. It showed species-wide dietary shifts, and not just local ecomorphs, in response to climatic and ecological variables in the whole Eurasian context (Flower and Schreve, 2014). Canis lupus spelaeus from Niedźwiedzia Cave and the climatic conditions in the Sudeten during MIS 3 are partially the analogues of Britain’s wolves from the period dated to MIS 5a (82-71 kya). The paleoenvironment was cold, the open tundra with summer temperatures between 11-7°C and winter temperatures between −10 °C and −30 °C was dominated by Equus ferus Boddaert, 1785, Cervus elaphus Linnaeus, 1758, Rangifer tarandus (Linnaeus, 1758), Bison priscus Bojanus, 1825, and Saiga tatarica (Linnaeus, 1766) (Marciszak et al., 2020). The main competitors of C. l. spelaeus in Niedźwiedzia Cave, and generally in the Sudeten, were the giant Ursus arctos taubachensis Rode, 1935 and Panthera spelaea spelaea (Goldfuss, 1810), while Crocuta crocuta spelaea was absent. The robust wolves of MIS 5 were larger than the largest extant C. lupus. They also suffered from harsh climatic conditions, low prey availability and dietary stress leading to a more carnivorous diet, with increased scavenging of frozen carcasses and bone consumption. They developed strong jaws with the highest flesh-slicing ability compared to the other wolves. They exhibited the longest and narrowest P4, which suggests improved slicing ability, and the largest M1-M2 but with reduced breadth and therefore reduced crushing ability, indicating a hypercarnivorous feeding pattern. They have a higher percentage of tooth breakage compared to the other wolves, and may have been using their P4 and m1 to crush bone rather than their M1/m1, leading to a higher frequency of damage (Flower and Schreve, 2014). Metrically, C. l. spelaeus from Niedźwiedzia Cave matches or exceed those wolves from MIS 5a, but differs in having a powerful dentition. Their mandibles are deep and massive, P4 is long and robust, M1-M2 elongated and broad, robust m1 with prominent trigonid. Powerful dentition indicates the ability to hunt and subdue large prey. Simultaneously, the large molars retained a crushing ability.
Characteristic for Canis lupus spelaeus i s also a large and elongated neurocranium, with high sagittal and nuchal crests and strongly furrowed surface. Such morphology is related to an allometric development of cranial bones, observed in many large carnivores (Kitchener et al., 2010). Larger skulls are characterised by narrower neurocranium and high sagittal and nuchal crests, which provide a wider surface for the attachment of jaw muscles like musculus temporalis and musculus masseter. In canids such a condition can be seen, for instance, in Aenocyon dirus (Leidy, 1858), Lycaon falconeri (Forsyth Major, 1877), Lycaon lycaonoides (Kretzoi, 1938), Canis lupus lycaon (Schreber, 1775), or Canis lupus nubilus Say, 1823 (Sardella et al., 2014). Skulls of small- to medium-sized Canis taxa have rounded neurocranium, with low sagittal and nuchal crests, and the muscle attachments are more weakly developed. Among them are taxa such as the smaller subspecies of C. lupus like Canis lupus pallipes Sykes, 1831, Canis lupaster Hemprich and Ehrenberg, 1832, Canis latrans Say, 1823, Canis aureus Linnaeus, 1758, Canis etruscus Forsyth Major, 1877, and Canis mosbachensis (Kitchener et al., 2010; Sardella et al., 2014).
Vulpes vulpes is the second most common canid in Sudeten caves; the red fox was found in the Late Pleistocene and Holocene sediments. Individuals from cooler periods were larger and more massive, while those from warmer periods were smaller and more delicately built (Marciszak et al., 2016, 2020). Late Pleistocene records include, among others, Biały Kamień, Kontaktowa, Miniaturka, Naciekowa, Na Ścianie, Niedźwiedzia, Południowa, Północna, Rogóżka, Duża Obok Wschodniej, and Wschodnia caves (Frenzel, 1936; Marciszak et al., 2016, 2020, 2023b). Localities as Aven w Połomie, Niedźwiedzia (uppermost strata), Radochowska, Small Przy Torach, and Solna Jama caves as well as numerous small rock shelters on Mount Miłek (Małgorzata, Trwoga Paleontologa, Kuny, Cisowe 1 and 2, and Panna) yielded postglacial and Holocene records of the species (Zotz, 1939). In Małgorzata Rock Shelter, the red fox material was especially abundant and included also juveniles, indicating that the site was a breeding place (Frenzel, 1936; Marciszak et al., 2016, 2020, 2023b).
Humans did not have to be involved in the accumulation of Vulpes vulpes remains in Sudeten sites (Marciszak et al. 2020, 2023b). A high number of V. vulpes bones was found in many localities from Polish Jura regarded as hyena dens (Wojtal, 2007). Vulpes vulpes could have been hunted by Crocuta crocuta spelaea or could have lived as its commensal. It might have also used the cave as a den when hyenas were not around. Vulpes vulpes may also have been brought to the sites by other carnivores. In such cases, these bones should then show evidence of bite marks, as, for example, in the case of archaeologically sterile layers in Niedźwiedzia Cave. In Radochowska Cave, V. vulpes remains were deposited naturally (Marciszak et al., 2023b). In numerous caves of Mount Połom, some of the V. vulpes remains were deposited or scavenged by non-human carnivores as evidenced by puncture marks, bite marks, and gnawing traces on the bones (Marciszak et al., 2023b).
Palaeolithic hunters considered Vulpes vulpes to be sources of raw material such as fur, teeth, and probably also food resources (Conard et al., 2013; Camarós et al., 2016; Baumann et al., 2020). Few specimens from the Sudeten caves hold some delicate signs that suggest they used foxes for their fur. However, it should be kept in mind that cut marks are often found on small skeletal elements, such as carpals, tarsals, or phalanges. Due to their small size, such bones are usually only recovered using more detailed methods of excavation, such as wet-sieving (Charles, 1997; Baumann et al., 2020). As most of the Sudeten sites were excavated without wet-sieving, this may affect the results. Additionally, not every cut leaves a mark and experienced hunters or butchers hardly leave any marks on bones during the butchering process (Andersson and Paulsson, 1993; Binford, 1981; Charles, 1997). Such direct evidence of V. vulpes exploitation by humans are well documented in numerous sites from Polish Jura like Biśnik Cave, Mamutowa or Komarowa Caves, while are very rare in Sudeten caves and still are not well recognised.
There can be no general statement as to the agent of accumulation for Vulpes vulpes remains in Sudeten caves in general. Palaeolithic human occupation was less intense and not so regular in the Sudeten than in the Polish Jura (Marciszak et al., 2020). This indicates that the majority of V. vulpes remains were likely deposited in the cave sites naturally and that humans were only sporadically involved. It is also possible that there was a kind of commensal relationship between V. vulpes and those Palaeolithic hunters. Commensalism could explain the relatively high number of V. vulpes remains in the Sudeten during some periods, as was noted already in Levantine (Yeshurun et al., 2009) and in Alaska (West and Yeshurun, 2019). For V. vulpes, commensal behaviour is an adaptation to a human-influenced environment (Lord et al., 2019; Baumann et al., 2020). Commensal, food-habituated individuals may have been hunted more often with traps, alongside non-commensal specimens that were trapped or occasionally hunted as single prey items (Marciszak et al., 2023b).
In comparison with Vulpes vulpes, V. lagopus is a rare element of the Sudeten faunas, recorded only from Niedźwiedzia, Radochowska, and Wschodnia caves (Marciszak et al., 2016, 2020, 2023b). The species was not recorded before from any of the Sudeten localities (Frenzel, 1936; Zotz, 1939, 1951; Marciszak et al., 2016, 2020). Much more numerous accumulations of bones of V. lagopus are known from sites located on Polish Jura (Wojtal, 2007; Lipecki and Wojtal, 2015). Even more numerous localities with accumulations of bones going in the hundreds are open-air sites like Dolní Věstonice, Předmostí, or Kraków Spadzista (Lipecki and Wojtal, 2015). The large accumulation of V. lagopus remains there is a result of mainly human exploitation of this carnivore. A large number of V. lagopus small bones, such as carpals, tarsals, phalanges, vertebrates, and sesamoids, and the presence of primarily complete skeletons, along with a large number of flint artefacts and the remains of camp fires indirectly confirm that hunter-gatherers are responsible for the creation of the bone assemblage of this species (Lipecki and Wojtal, 2015).
The permanent teeth of Vulpes lagopus from these open-air sites are in various stages of wear, indicating the presence of animals ranging in age from pre-adult to old adult. Simultaneously, no deciduous teeth or bones with unfused epiphyses of immature individuals were found at the site. The lack of young or immature individuals suggests that V. lagopus was exploited by Palaeolithic hunters most probably in the colder period of the year when the fox fur was of the highest quality. Vulpes lagopus had a great value because its fur is thick and soft, with long guard hairs and dense under-fur. Its winter coat has the best insulative properties of all mammals. Humans could also have used fox carcasses as a source of protein during periods of food scarcity. Contrary to that, the accumulation of fox bones in Sudeten caves was rather incidental, correlated with abiotic factors or carnivore activity rather than human exploitation (Tannerfeldt, 1997; Lipecki and Wojtal, 2015).
The smallest postcranial elements from Niedźwiedzia Cave could not belong to Vulpes lagopus, but might represent Vulpes corsac Linnaeus, 1768, which was in the past occasionally reported from the Late Pleistocene of Europe (Nehring, 1889; Kafka, 1900; Boule, 1919; Stehlin, 1933; Musil, 1962). Some of these reports are rather problematic finds of uncertain taxonomic position and need revision (Mostecký, 1969; Beneš, 1975). Jánossy (1955) considered the small fox from Istállóskő to be V. vulpes and explained the size differences only as a result of sexual dimorphism. Musil (1965) also reported the potential presence of V. corsac in Pod Hradem Cave. Beneš (1975) suggested that small foxes from the interglacial and interstadial periods represented rather V. corsac than V. lagopus. So far, V. corsac was not recorded in any of the Sudeten caves nor from Poland, but its presence is highly probable.
ACKNOWLEDGEMENTS
The research was financed by subsidies for the activities of the University of Wrocław, subsidy 2024 - research activities (501), MPK 2599280000 ZP, discipline 73 - biological sciences. It was also financed by an internal grant no. BPIDUB.4610.6.2021.KP.A. “The Middle Pleistocene Revolution -- How the modern theriofauna of Eurasia was developed,” University of Wrocław.
REFERENCES
Altuna, J. 2004. Estudio biométrico de Vulpes vulpes L. y Alopex lagopus L. Contribucion a su differenciation en los yacimientos paleoliticos cantábricos. Munibe, 56:45-59.
Andersson, A. and Paulsson, T. 1993. Jägarens skinn och hudar. Bokförlaget Settern, Örkelljung.
Argant, A. 2009. Biochronologie et grands mammifères au Pléistocène moyen et supérieur en Europe occidentale: l’apport des Canidés, des Ursidés et des Carnivores en general. Quaternaire, 20(4):467-480.
https://doi.org/10.4000/quaternaire.5291
Argant, A. 2010. Carnivores (Canidae, Felidae et Ursidae) de Romain-la-Roche (Doubs, France). Revue de Paléobiologie, 29:495-601.
Ballesio, R. 1979. Le gisement pléistocène supérieur de la grotte de Jaurens à Nespouls, Corrèze, France: les carnivores (Mammalia, Carnivora). I. Canidae et Hyaenidae. Nouvelles archives du Muséum d’histoire Naturelle de Lyon, 17:25-55.
Baryshnikov, G. 2006. Late Pleistocene arctic fox (Alopex lagopus) from Crimea, Ukraine. Quaternary International, 142-143:208-217.
https://doi.org/10.1016/j.quaint.2005.03.018
Baryshnikov, G.F. 2012. Pleistocene Canidae (Mammalia, Carnivora) from the Palaeolithic Kudaro caves in the Caucasus. Russian Journal of Theriology, 11(2):77-120.
https://doi.org/10.15298/rusjtheriol.11.2.01
Baryshnikov, G.F. 2020. Krupnye mlekopitayushchie Kudarskoy pleystotsenovoy fauny Kavkaza. Proceedings of the Zoological Institute of the Russian Academy of Sciences, 9:1-400.
Baumann, C., Wong, G.L., Starkovich, B.M., Münzel, S.C., and Conard, N.J. 2020. The role of foxes in the Palaeolithic economies of the Swabian Jura (Germany). Archaeological and Anthropological Sciences, 12(9):208.
https://doi.org/10.1007/s12520-020-01173-4
Beneš, J. 1975. The Würmian foxes of Bohemian and Moravian Karst. Acta Musei Nationalis Pragae, 31B(3-5):149-210.
Bieroński, J., Socha, P., and Stefaniak, K. 2007. Deposits and fauna of the Sudeten caves - the state of research. Studies of the Faculty of Earth Sciences University of Silesia, 45:183-201.
Bieroński, J., Stefaniak, K., Hercman, H., Socha, P., and Nadachowski, A. 2009. Palaeogeographical and palaeoecological studies of sediments of the Niedźwiedzia (Bear) cave in Kletno. Studies of the Faculty of Earth Sciences University of Silesia, 56:401-422.
Bigaj, J. 1963. Szczątki Canidae z plejstocenu Polski. Folia Quaternaria, 13:1-18.
Binder, W.J., Thompson, E.N., and Van Valkenburgh, B. 2002. Temporal variation in tooth fracture among Rancho La Brea dire wolves. Journal of Vertebrate Palaeontology, 22(2):423-428.
https://doi.org/10.1671/0272-4634(2002)022[0423:TVITFA]2.0.CO;2
Binford, LR. 1981. Bones: ancient men and modern myths. Academic Press, New York.
Bisaillon, A. and Deroth, L. 1979. Morphology and morphometry of the appendicular skeleton of the red fox (Vulpes vulpes). Canadian Journal of Zoology, 57(11):2089-2099.
https://doi.org/10.1139/z79-276
Bisaillon, A. and DeRoth, L. 1980. Cranial measurements in the arctic fox (Alopex lagopus). Revue de Canadienne de Biologie 39:81-84.
Boddaert, P. 1785. Elenchus Animalium. Volumen I. Sistens quadrupedia huc usque nota, eorumque varietates. C.R. Hake, Rotterdam.
Bojanus, L.H. 1825. De uro nostrato ejusque sceleto commentatio, Bovis primigenii sceleto aucta. Palaeontology Library of Muséum National d’Histoire Naturelle, Paris.
Bonifay, M.-F. 1971. Carnivores quaternaires du sud-est de la France. Mémoires du Museum National d’histoire naturelle, Série C Géologie, 21(2):1-377.
Boudadi-Maligne, M. 2010. Les Canis pléistocènes du Sud de la France: approche biosystématique, évolutive et biochronologique. PhD thesis. Université Bordeaux, Bordeaux.
Boudadi-Maligne, M. 2012. Une nouvelle sous-espèce de loup (Canis lupus maximus nov. subsp.) dans le Pléistocène supérieur d’Europe occidentale. Comptes Rendus Palevol, 11(7):475-484.
https://doi.org/10.1016/j.crpv.2012.04.003
Boule, M. 1919. Les Grottes de Grimaldi (Baoussé-Roussé). Tome I. Fasc. IV: Géologie et Paléontologie. Villeneuve, Monaco.
Bowdich, T.E. 1821. An analysis of the natural classifications of Mammalia for the use of students and travellers. J. Smith, Paris.
Brugal, J.P. and Boudadi-Maligne, M. 2011. Quaternary small to large canids in Europe: taxonomic status and biochronological contribution. Quaternary International, 243:171-182.
https://doi.org/10.1016/j.quaint.2011.01.046
Brugal, J.-B., Argant, A., Boudadi-Maligne, M., Crégut-Bonnoure, E., Croitor, R., Fernandez, P., Fourvel, J.-B., Fosse, P., Guadelli, J.-L., Labe, B., Magniez, P., and Uzunidis, A. 2020. Pleistocene herbivores and carnivores from France: An updated overview of the literature, sites and taxonomy. Annales de Paléontologie, 106(2):102384.
https://doi.org/10.1016/j.annpal.2019.102384
Camarós, E., Münzel, S.C., Cueto, M., Rivals, F., and Conard, N.J. 2016. The evolution of Palaeolithic hominin-carnivore interaction written in teeth: stories from the Swabian Jura (Germany). Journal of Archaeological Science: Reports, 6:798-809.
https://doi.org/10.1016/j.jasrep.2015.11.010
Charles, R. 1997. The exploitation of carnivores and other fur-bearing mammals during the north-western European ate and per Palaeolithic and Mesolithic. Oxford Journal of Archaeology, 16(3):253-277.
https://doi.org/10.1111/1468-0092.00040
Conard, N.J., Kitagawa, K., Krönneck, P., Böhme, M., and Münzel, S.C. 2013. The importance of fish, fowl and small mammals in the Palaeolithic diet of the Swabian Jura, south-western Germany. In Clark, J.L. and Speth, J.D. (eds.), Zooarchaeology and modern human origins: human hunting behaviour during the Later Pleistocene. Paleobiology and Palaeoanthropology Vertebrates, 2013:173-190.
https://doi.org/10.1007/978-94-007-6766-9_11
Dayan, Т., Tchernov, E., Yom-Tov, Y., and Simberloff, D. 1989. Ecological character displacement in Saharo-Arabian Vulpes: outfoxing Bergmann’s rule. Oikos, 55(2):263-272.
https://doi.org/10.2307/3565430
Diedrich, C.G. 2014. Holotype skulls, stratigraphy, bone taphonomy and excavation history in the Zoolithen Cave and new theory about Esper's “great deluge”. Journal of Quaternary Science, 63(1):78-98.
https://doi.org/10.3285/eg.63.1.05
Diedrich, C.G. 2017. Famous planet Earth caves: Hermann’s cave (Germany) -- a Late Pleistocene cave bear den, vol. 2. Bentham Books, London.
https://doi.org/10.2174/97816810853021170201
Diedrich, C.G. 2022. Eurasian grey and white wolf ancestors -- 800,000 years evolution, adaptation, pathologies and European dog origins. Acta Zoologica, 105(1):25-37.
https://doi.org/10.1111/azo.12451
Driesch, A.v.d. 1976. A guide to the measurement of animal bone from archaeological sites. Peabody Museum Bulletin, 1:1-138.
Erxleben, J.C.P. 1777. Systema regni animalis per classes, ordines, genera, species, varietates cvm synonymia et historia animalivm. Classis I. Mammalia. Impensis Weygandianis, Lipsiae.
Fischer de Waldheim, G. 1817. Adversaria zoologica. Mémoires de la Société Impériale des Naturalistes de Moscou, 5:357-472.
Flower, L.O.H. 2016. New body mass estimates of British Pleistocene wolves: palaeoenvironmental implications and competitive interactions. Quaternary Science Reviews, 149:230-247.
https://doi.org/10.1016/j.quascirev.2016.07.023
Flower, L.O.H. and Schreve, D.C. 2014. An investigation of palaeodietary variability in European Pleistocene canids. Quaternary Science Reviews, 96:188-203.
https://doi.org/10.1016/j.quascirev.2014.04.015
Forsyth Major, C.I. 1877. Considerazioni sulla fauna dei mammiferi pliocenici e postpliocenici della Toscana. Atti della Società toscana di scienze naturali. Memorie, Serie B, 1:207-227.
Frenzel, J. 1936. Knochenfunde in der Reyersdorfer Tropsteinhöhle. Beiträge zur Biologie des Glatzer Schneeberges, 2:121-134.
Garsault, F.A.P.d. 1746. Le nouveau parfait maréchal, ou la connoissance générale et universelle du cheval, divisé en sept traités (2nd edition). Huart, Paris.
Germonpré, M. and Sablin, M.V. 2004. Systematics and osteometry of Late Glacial foxes from Belgium. Bulletin de l'Institut Royal des Sciences Naturelles de Belqique, Sciences de la Terre, 74:175-188.
Gimranov, D.O. 2017. Species diagnostics of the corsac (Vulpes corsac), fox (Vulpes vulpes) and Arctic fox (Vulpes lagopus, Carnivora, Canidae) using the upper teeth. Zoological Journal, 96(6):684-697.
https://doi.org/10.7868/S0044513417060046
Gimranov, D.O., Kosintsev, P.A., and Gasilin, V.V. 2015. Species diagnostics of corsac (Vulpes corsac), fox (Vulpes vulpes), and arctic fox (Vulpes lagopus) according to ontological characteristics of mandible teeth. Zoological Journal, 94(11):1338-1338.
https://doi.org/10.13140/RG.2.1.2518.7923
Gittleman, J.L. and Van Valkenburgh, B. 1997. Sexual dimorphism in the canines and skulls of carnivores: effects of size, phylogeny, and behavioural ecology. Journal of Zoology, 242(1):97-117.
https://doi.org/10.1111/j.1469-7998.1997.tb02932.x
Goldfuss, G.A. 1810. Die Umgebungen von Muggensdorf. Johann Jakob Palm, Erlangen.
Goldfuss, G.A. 1823. Ueber den Höhlenwolf (Canis spelaeus). Osteologische Beiträge zur Kenntniss verschiedener Säugethiere der Vorwelt. Nova Acta Physico‐Medica Academiea Caesarae Leopoldino‐Carolinae Naturae Curiosorum, 3:451-455.
Gromova, V.I. 1950. Opredelitel mlekopitajuscich SSSR po kostjam skeleta. Trudy Komissii po izučeniû četvertičnogo perioda, 9(1):1-107.
Hemprich, W. and Ehrenberg, Ch.G. 1833. Symbolae Physicae seu Icones et Descriptiones Mammalium quae ex itinere per Africam Borealem et Asiam Occidentalem. Cz. 1. Berolini: Ex Officina Academica, venditur a Mittlero.
Iurino, D.A., Mecozzi, B., Iannucci, A., Moscarella, A., Strani, F., Bona, F., Gaeta, M., and Sardella, R. 2022. A Middle Pleistocene wolf from central Italy provides insights on the first occurrence of Canis lupus in Europe. Scientific Reports, 12(1):2882.
https://doi.org/10.1038/s41598-022-06812-5
Jánossy, D. 1955. Die Vogel- und Säugetierreste der spätpleistozänen Schichten der Höhle von Istalloskö. Acta Archaeologica Academie Scientiarum Hungaricae, 5:149-181
Kafka, J. 1900. Šelmy (Carnivora) země české, žijící i fossilní. Archiv pro přírodovědecké prozkoumání Čech, 10(6):1-101.
Kiernik, E. 1913a. Materiały do paleozoologii dyluwialnych ssaków ziem polskich. Cz. 5. Szczątki wilka z dyluwium warstw ziem polskich. Rozprawy Wydziału Matematyczno-Przyrodniczego Polskiej Akademii Umijętności, seria 3, 13:491-541.
Kiernik, E. 1913b. Materiały do paleozoologii dyluwialnych ssaków ziem polskich. Cz. 5. Familia: Canidae; Sectio: Lupinae. Spraw. Sprawozdania z czynności i posiedzeń Polskiej Akademii Umiejętności w Krakowie, 18:15.
Kitchener, A.C., Van Valkenburgh, B., and Yamaguchi, N. 2010. Felid form and function, pp. 83-106. In Macdonald, D.W. and Sillero-Zubiri, C. (eds.), The Biology and Conservation of Wild Canids. Oxford University Press, New York.
Kretzoi, M. 1938. Die Raubtiere von Gombaszög nebst einer Übersicht der Gesamtfauna (Ein Beitrag zur Stratigraphie des Altquartärs). Annales Musei Nationalis Hungarici, pars Mineralogica, Geologica, Paleontologica, 31:88-157.
Kretzoi, M. 1943. Kochitis centenii n. g. n. sp., ein altertümlicher Creodonte aus dem Oberoligozän Siebenbürgens. Földtani Közlöny, 73:10-195.
Kurtén, B. 1965. The Carnivora of the Palestine caves. Acta Zoologica Fennica, 107:1-74.
Kuzmina, I.E. and Sablin, M.V. 1994. Wolf Canis lupus L. from the Late Palaeolithic sites Kostenki on the Don River. Trudy Zoologicheskogo Instituta, 256:44-58.
Langenhan, A. 1904a. Ueber fossile funde am Kitzelberg. Zeitschrift der Deutschen Geologischen Gesellschaft, 56:5-7.
Langenhan, A. 1904b. Schädel eines Höhlenwolfs im Kitzelberg bei Kauffung im Katzbachtal. Monatsschriften für Mineralien-, Gesteins- und Petrefaktensammler, 2:24-25.
Lapini, L. 2003. Canis aureus (Linnaeus, 1758). In Boitani, L., Lovari, S., and Vigna Taglianti, A. (eds.). Mammalia 3: Carnivora -- Artiodactyla. Fauna d’Italia, 38:47-58.
Leidy, J. 1858. Notice of remains of extinct vertebrata, from the Valley of the Niobrara River, collected during the Exploring Expedition of 1857, in Nebraska, under the command of Lieut. G. K. Warren, U.S. Top. Eng., by Dr. F.V. Hayden, Geologist to the Expedition. Proceedings of the Academy of Natural Sciences of Philadelphia, 10:20-29.
Leonard, J.A., Vilà, C., Fox-Dobbs, K., Koch, P.L., Wayne, R.K., and Van Valkenburgh, B. 2007. Megafaunal extinctions and the disappearance of a specialized wolf ecomorph. Current Biology, 17(13):1146-1150.
https://doi.org/10.1016/j.cub.2007.05.072
Linnaeus, C.v. 1758. Caroli Linnaei... Systema naturae per regna tria naturae:secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. Holmiae, Impensis Direct. Laurentii Salvii.
Linnaeus, C. 1766. Systema naturae per regna tria naturae, secundum classes, ordines, genera, species, cum characteribus, differentiis synonymis, locis. Vol. 1. Regnum Animale. pt. 1. Laurentii Salvii, Stockholm.
Lipecki, G. and Wojtal, P. 2015. Carnivores from the open-air Gravettian site Kraków Spadzista, pp. 117-157. In Wilczyński, J., Wojtal, P., and Haynes, G. (eds.), A Gravettian site in southern Poland: Kraków Spadzista. Institute of Systematics and Evolution of Animals of the Polish Academy of Sciences, Kraków.
Lisiecki, L.E. and Raymo, M.E. 2005. A Pliocene-Pleistocene stack of 57 globally distributed benthic 18O records. Paleoceanography and Paleoclimatology, 20(1):PA1003.
https://doi.org/10.1029/2004PA001071
Lord, K.A., Larson, G., Coppinger, R.P., and Karlsson, E.K. 2019. The history of farm foxes undermines the animal domestication syndrome. Trends in Ecology & Evolution, 35(2):125-136.
https://doi.org/10.1016/j.tree.2019.10.011
Lucenti, S.B., Alba, D.M., Rook, L., Moyà-Solà, S., and Madurell-Malapeira, J. 2017. Latest Early Pleistocene wolf-like canids from the Iberian Peninsula. Quaternary Science Reviews, 162:12-25.
https://doi.org/10.1016/j.quascirev.2017.02.023
Lucenti, S.B. and Madurell-Malapeira, J. 2020. Unravelling the fossil record of foxes: An updated review on the Plio-Pleistocene Vulpes spp. from Europe. Quaternary Science Reviews, 236:106296.
https://doi.org/10.1016/j.quascirev.2020.106296
Made, v.d.J., Stefaniak, K., and Marciszak, A. 2014. The Polish fossil record of the wolf Canis and the deer Alces, Capreolus, Megaloceros, Dama and Cervus in an evolutionary perspective. Quaternary International, 326-327:406-430.
https://doi.org/10.1016/j.quaint.2013.11.015
Marciszak, A., Stefaniak, K., and Gornig, W. 2016. Fossil theriofauna from the Sudety Mts (SW Poland). The state of research. Cranium, 33(1):31-41.
Marciszak, A., Sobczyk, A., Kasprzak, M., Gornig, W., Ratajczak, U., Wiśniewski, A., and Stefaniak, K. 2020. Taphonomic and paleoecological aspects of large mammals from Sudety Mts (Silesia, SW Poland), with particular interest to the carnivores. Quaternary International, 546:42-63.
https://doi.org/10.1016/j.quaint.2019.11.009
Marciszak, A., Kropczyk, A., and Lipecki, G. 2021. The first record of Cuon alpinus (Pallas, 1811) from Poland and the possible impact of other large canids on the evolution of the species. Journal of Quaternary Science, 36(6):1101-1121.
https://doi.org/10.1002/jqs.3340
Marciszak, A., Gornig, W., and Szynkiewicz, A. 2023a. Carnivores from Draby 3 (central Poland): The latest record of Lycaon lycaonoides (Kretzoi, 1938) and the final accord in the long history of ancient faunas. Quaternary International, 674-675:62-86.
https://doi.org/10.1016/j.quaint.2023.03.012
Marciszak, A., Kropczyk, A., Gornig, W., Kot, M., Nadachowski, A., and Lipecki, G. 2023b. History of Polish Canidae (Carnivora, Mammalia) and their biochronological implications on the Eurasian background. Genes, 14(3):539.
https://doi.org/10.3390/genes14030539
Marciszak, A., Mackiewicz, P., Borówka, R.K., Capalbo, C., Chibowski, P., Gąsiorowski, M., Hercman, H., Cedro, B., Kropczyk, A., Gornig, W., Moska, P., Nowakowski, D., Ratajczak-Skrzatek, U., Sobczyk, A., Sykut, M.T., Zarzecka-Szubińska, K., Kovalchuk, O., Barkaszi, Z., Stefaniak, K., and Mazza, P.P.A. 2024. Fate and preservation of the Late Pleistocene cave bears from Niedźwiedzia Cave in Poland, through taphonomy, pathology, and geochemistry. Scientific Reports, 14:9775.
https://doi.org/10.1038/s41598-024-60222-3
Mecozzi, B. and Lucenti, S.B. 2018. The Late Pleistocene Canis lupus (Canidae, Mammalia) from Avetrana (Apulia, Italy): reappraisal and new insights on the European glacial wolves. Italian Journal of Geosciences, 137(1):138-150.
https://doi.org/10.3301/IJG.2017.22
Mecozzi, B., Iurino, D.A., Profico, A., Rosa, C., and Sardella, R. 2020. The wolf from the Middle Pleistocene site of Ostiense (Rome): the last occurrence of Canis mosbachensis (Canidae, Mammalia) in Italy. Historical Biology, 33(10): 2031-2042.
https://doi.org/10.1080/08912963.2020.1769090
Meloro, C. and Sansalone, G. 2022. Palaeoecological significance of the “wolf event” as revealed by skull ecometrics of the canid guilds. Quaternary Science Reviews, 281:107419. https://doi.org/10.1016/j.quascirev.2022.107419
Monchot, H. and Gendron, D. 2010. Disentangling long bones of foxes (Vulpes vulpes and Alopex lagopus) from arctic archaeological sites. Journal of Archaeological Science, 37:799-806.
https://doi.org/10.1016/j.jas.2009.11.009
Mostecký, V. 1969. Jungpleiztozäne Säugethiere aus der “Chlupáč-Höhle” auf dem Hügel “Kobyla” bei Koněprusy (Böhmischer Karst). Acta Musei Nationalis Pragae, 25B(1):1-28.
Musil, R. 1962. Die Höhle „Švédův stůl“, ein typischer Höhlenhyänenhorst. Anthropos, 13(N.S.5):97-260.
Musil, R. 1965. Die Bärenhöhle Pod Hradem. Anthropos, 18(N.S.10):7-92.
Nehring, A. 1889. Ueber das fossile Vorkommen von Canis caragan, Canis corsac, Felis manul und Felis chaus im Plistocaen Mitteleuropas. Sitzungsberichte der Gesellschaft Naturforschender Freunde zu Berlin, 1889:109-111.
O’Keefe, F.R., Binder, W.J., Frost, S.R., Sadlier, R.W., and Van Valkenburgh, B. 2014. Cranial morphometrics of the dire wolf, Canis dirus, at Rancho La Brea: temporal variability and its links to nutrient stress and climate. Palaeontologia Electronica, 17(1):1-24.
https://doi.org/10.26879/437
Pimm, S.L. and Gittleman, J.L. 1990. Carnivores and ecologists on the road to Damascus. Trends in Ecology & Evolution, 5(3):70-73.
https://doi.org/10.1016/0169-5347(90)90232-3
Pocock, R.I. 1934. LXVI.-Preliminary diagnoses of some new races of South Arabian mammals. Journal of Natural History, Series 10, 14 (84):635-636.
Reichstein, H. 1984. Beitrag zur Kenntnis frühmittelalterlicher Rotfüchse, Vulpes vulpes aus Schleswig-Holstein. Schriften aus der Archäologisch-Zoologischen Arbeitsgruppe Schleswig-Kiel, 8(1):1-137.
Ripoll, M.P., Morales Pérez, J.V., Sanchis Serra, A., Aura Tortosa, J.E., and Montañana, I.S. 2010. Presence of the genus Cuon in Upper Pleistocene and initial Holocene sites of the Iberian Peninsula: new remains identified in archaeological contexts of the Mediterranean region. Journal of Archaeological Science, 37(3):437-450.
https://doi.org/10.1016/j.jas.2009.10.008
Rode, K. 1935. Untersuchungen uber das Gebiss der Bären. Monographien zur Geologie und Paläontologie, 2(7):1-162.
Römer, F. 1883. Die Knochenhöhlen von Ojców in Polen. Palaeontographica, 29:193-235.
Sansalone, G., Bertè, D.F., Maiorino, L., and Pandolfi, L. 2015. Evolutionary trends and stasis in carnassial teeth of European Pleistocene wolf Canis lupus (Mammalia, Canidae). Quaternary Science Reviews, 110:36-48.
https://doi.org/10.1016/j.quascirev.2014.12.009
Sardella, R. and Palombo, M.P. 2007. The Pliocene-Pleistocene boundary: Which significance for the so called “wolf event”? Evidences from Western Europe. Quaternaire, 18:65-71.
https://doi.org/10.4000/quaternaire.969
Sardella, R., Bertè, D., Iurino, D.A., Cherin, M., and Tagliacozzo, A. 2014. The wolf from Grotta Romanelli (Apulia, Italy) and its implications in the evolutionary history of Canis lupus in the Late Pleistocene of Southern Italy. Quaternary International, 328-329:179-195.
https://doi.org/10.1016/j.quaint.2013.11.016
Say, T. 1823. Account of an expedition from Pittsburgh to the Rocky Mountains, performed in the years 1819 and '20: by order of the Hon. J.C. Calhoun, sec'y of war: under the command of Major Stephen H. Long. From the notes of Major Long, Mr. T. Say, and other gentlemen of the exploring party. H.C. Carey and I. Lea, Philadelphia.
Schreber, J.C.D.v. 1775. Die Säugthiere in Abbildungen nach der Natur mit Beschreibungen, Zweiter Teil. Erlangen.
Sobczyk, A., Kasprzak, M., Marciszak, A., and Stefaniak, K. 2016. Karst phenomena in metamorphic rocks of the Śnieżnik Massif (East Sudetes): state of the art and significance for tracing a Late Cenozoic evolution of the Sudetes. Przegląd Geologiczny, 64(9):426-434.
Soergel, W. 1925. Die Säugetierfauna des altdiluvialen Tonlagers von Jockgrim in der Pfalz. Zeitschrift der Deutschen Geologischen Gesellschaft 77:405-438.
Sommer, R. and Benecke, N. 2005. Late-Pleistocene and early Holocene history of the canid fauna of Europe (Canidae). Mammalian Biology, 70(4):227-241.
https://doi.org/10.1016/j.mambio.2004.12.001
Stehlin, H.G. 1933. Paleotologie des couches paleolithiques. In Dubois, A. and Stehlin, H.G. (eds.), La grotte de Cotencher, station mousterienne. Mémoires de la Société paléontologique Suisse, 52-53:33-178.
Sykes, W.H. 1831. Catalogue of the Mammalia of Dukun (Deccan); with observations on the habits, etc., and characters of new species. Proceedings of the Committee of Science and Correspondence of the Zoological Society of London, 1830-1831:99-106.
Szuma, E. 2000. Variation and correlation patterns in the dentition of the red fox from Poland. Annales Zoologici Fennici, 37(2):113-127.
Szuma, E. 2003. Microevolutionary trends in the dentition of the red fox (Vulpes vulpes). Journal of Zoological Systematics and Evolutionary Research, 41(1):47-56.
https://doi.org/10.1046/j.1439-0469.2003.00196.x
Szuma, E. 2004. Evolutionary implications of morphological variation in the lower carnassial of red fox Vulpes vulpes. Acta Theriologica, 49(4):433-447.
https://doi.org/10.1007/BF03192588
Szuma, E. 2007. Geography of dental polymorphism in the red fox Vulpes vulpes and its evolutionary implications. Biological Journal of the Linnean Society, 90(1):61-84.
https://doi.org/10.1111/j.1095-8312.2007.00712.x
Szuma, E. 2008a. Geography of sexual dimorphism in the tooth size of the red fox Vulpes vulpes (Mammalia, Carnivora). Journal of Zoological Systematics and Evolutionary Research, 46(1):73-81.
https://doi.org/10.1111/j.1439-0469.2007.00418.x
Szuma, E. 2008b. Evolutionary and climatic factors affecting tooth size in the red fox Vulpes vulpes in the Holarctic. Mammal Research, 53(4):289-332.
https://doi.org/10.1007/BF03195193
Szuma, E. 2008c. Geographic variation of tooth and skull sizes in the arctic fox Vulpes (Alopex) lagopus. Annales Zoologici Fennici, 45(3):185-199.
https://doi.org/10.5735/086.045.0304
Szuma, E., 2011. Ecological and evolutionary determinants of dental polymorphism in the arctic fox Vulpes (Alopex) lagopus. Annales Zoologici Fennici, 48(4):191-214.
https://www.jstor.org/stable/23737080
Szuma, E. and Germonpré, M. 2020. Size of the lower carnassial in the arctic and the red fox from Late Pleistocene in Belgium compared to other ancient and extant populations. Mammal Research, 65(1):127-139.
https://doi.org/10.1007/s13364-019-00459-w
Tanner, J.B, Dumont, E., R., Sakai, S.T., Lundrigan, B.L., and Holekamp, K.E. 2008. Of arcs and vaults: the biomechanics of bone-cracking in spotted hyenas (Crocuta crocuta). Biological Journal of the Linnaean Society, 95:246-255.
https://doi.org/10.1111/j.1095-8312.2008.01052.x
Tannerfeldt, M. 1997. Population fluctuations and life history consequences in the Arctic fox. PhD Thesis, Stockholm University, Stockholm.
Therrien, F. 2005. Mandibular force profiles of extant carnivorans and implications for the feeding behaviour of extinct predators. Journal of Zoology, 267(3):249-270.
https://doi.org/10.1017/S0952836905007430
Turner, A. 1981. Aspects of the paleoecology of large predators, including man, during the British Upper Pleistocene, with particular emphasis on predator-prey relationships. University of Sheffield, Sheffield.
Van Valkenburgh, B. 2008. Costs of carnivory: tooth fracture in Pleistocene and recent carnivorans. Biological Journal of the Linnean Society, 96(1):68-81.
https://doi.org/10.1111/j.1095-8312.2008.01108.x
Van Valkenburgh, B. and Hertel, F. 1993. Tough times at La Brea: tooth breakage in large carnivores of the Late Pleistocene. Science, New Series, 261(5120):456-459.
https://doi.org/10.1126/science.261.5120.456
Van Valkenburgh, B., Peterson, R.O., Smith, D.W., Stahler, D.R., and Vucetich, J.A. 2019. Tooth fracture frequency in gray wolves reflects prey availability. eLife, 8:e48628.
https://doi.org/10.7554/eLife.48628
West, C.F. and Yeshurun, R. 2019. Foxes and humans at the Late Holocene Uyak site, Kodiak. Alaska Arctic Anthropology, 56(1):39-51.
https://www.jstor.org/stable/26848873
Wiszniowska, T. 1967. Nowe znalezisko paleontologiczne w Sudetach. Przegląd Zoologiczny, 11:430-433.
Wiszniowska, T. 1970. Wstępne wyniki badań fauny kopalnej w Jaskini Niedźwiedziej. Acta Universitatis Wratislaviensis, 127:45-70.
Wiszniowska, T. 1986. Szczątki fauny w namuliskach jaskiń na Śląsku. Prace Komisji Archeologicznej Polskiej Akademii Nauk Ossolineum, 3:9-19.
Wiszniowska, T. 1989. Kopalne szczątki zwierzęce, pp. 255-279. In Jahn, A., Kozłowski, S., and Wiszniowska, T. (eds.), Jaskinia Niedźwiedzia w Kletnie. Badania i udostępnianie. Zakład Narodowy Imienia Ossolińskich, Wrocław-Warszawa.
Wojtal, P. 2007. Zooarchaeological studies of the Late Pleistocene sites in Poland. Institute of Systematics and Evolution of Animals Polish Academy of Sciences, Cracow.
Yeshurun, R., Bar-Oz, G., and Weinstein-Evron, M. 2009. The role of foxes in the Natufian economy: a view from Mount Carmel. Israel Before Farming, 2009(1):1-15.
https://doi.org/10.3828/bfarm.2009.1.3
Zotz, L.F. 1939. Die Altsteinzeit in Niederschlesien. Kabitsch Verlag, Leipzig.
Zotz, L.F. 1951. Altsteinzeitkunde Mitteleuropas. F. Enke Verlag, Stuttgart.

