 Internal anatomy of a brachyuran crab from a Late Cretaceous methane seep and an overview of internal soft tissues in fossil decapod crustaceans
Internal anatomy of a brachyuran crab from a Late Cretaceous methane seep and an overview of internal soft tissues in fossil decapod crustaceans
Article number: 26.3.a44
https://doi.org/10.26879/1277
Copyright Paleontological Society, November 2023
Proceedings of the 8th Symposium on Fossil Decapod Crustaceans
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Submission: 1 March 2023. Acceptance: 23 September 2023.
ABSTRACT
Cretaceous-aged methane seep carbonates in the Western Interior Seaway, USA, have yielded a relatively high diversity and concentration of fossils, including decapod crustaceans, compared to their surrounding, contemporaneous sediments. With technological advances in non-destructive imaging techniques, the internal anatomy of decapods can be studied in remarkable detail. Here, we present a brachyuran crab specimen from an upper Campanian methane seep carbonate in South Dakota, USA. While the external morphology of the crab is insufficient for species identification (Secretanella sp.), remarkable details of the internal morphology are preserved including soft tissues. Four phyllobranchiate gills are visible due to a broken cuticle. A µCT-scan revealed parts of the digestive tract including the esophagus and the cardiac stomach (foregut), and possible anterior gastric muscles. An esophagus has not been reported previously in a fossil decapod. We also found mandibles and their apodemes. Because of soft tissue preservation, the South Dakotan locality may be a candidate Konservat-Lagerstätte. To our knowledge, the studied crab contains the first preserved animal soft tissue reported from an ancient methane seep. Our new overview of internal soft tissues preserved in fossil decapod crustaceans shows that, besides muscles reported previously, gills and remains of the digestive tract are most often reported (8 and 13 occurrences, respectively). Reproductive organs, nerve chords, and the heart are only known from one or two occurrences. Although reports of soft internal anatomy preservation remain relatively rare, continued application of µCT-scans to fossil decapods has the potential to illuminate our knowledge of the evolution of internal decapod anatomy.
Adiël A. Klompmaker. Department of Museum Research and Collections & Alabama Museum of Natural History, University of Alabama, Box 870340, Tuscaloosa, Alabama 35487, USA. adielklompmaker@gmail.com
Orcid: 0000-0002-6645-6970
Peter A. Kloess. Department of Biology, Whittier College, Whittier, California 90602, USA. & Department of Integrative Biology and Museum of Paleontology, University of California, Berkeley, California, 94720, USA. pkloess@whittier.edu pakloess@berkeley.edu.
Orcid: 0000-0003-1377-135X
Clément Jauvion. Formerly: Centre de Recherche en Paléontologie - Paris (CR2P), CNRS, Muséum national d'Histoire naturelle, Sorbonne Université, 57 rue Cuvier, 75231 Paris CEDEX 05, France. clement.jauvion@gmail.com
Orcid: 0000-0002-3245-8222
Jamie Brezina. South Dakota School of Mines and Technology, Rapid City, South Dakota 57701, USA. rezinagate@hotmail.com
Neil H. Landman. Division of Paleontology (Invertebrates), American Museum of Natural History, New York, New York 10024, USA. landman@amnh.org
Keywords: Brachyura; cold seep; Cretaceous; exceptional preservation; methane seep; soft tissue
Final citation: Klompmaker, Adiël A., Kloess, Peter A., Jauvion, Clément, Brezina, Jamie, and Landman, Neil H. 2023. Internal anatomy of a brachyuran crab from a Late Cretaceous methane seep and an overview of internal soft tissues in fossil decapod crustaceans. Palaeontologia Electronica, 26(3):a44.
https://doi.org/10.26879/1277
palaeo-electronica.org/content/2023/3973-soft-tissues-in-fossil-crab
Copyright: November 2023 Paleontological Society.
This is an open access article distributed under the terms of Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0), which permits users to copy and redistribute the material in any medium or format, provided it is not used for commercial purposes and the original author and source are credited, with indications if any changes are made.
creativecommons.org/licenses/by-nc-sa/4.0/
INTRODUCTION
The internal anatomy of fossil arthropods has been increasingly studied over the last decades, leading to new insights about their biology and evolutionary relationships. Some previous studies have relied on exposed internal anatomy of specimens, i.e., through erosion, or mechanical preparation, including destructive sampling (e.g., Secrétan, 1985; Butterfield, 2002; Siveter et al., 2013; Zacaï et al., 2016; Jauvion et al., 2020a; Ortega-Hernández et al., 2022). Technological advances in the last decade have made it possible to non-destructively scan and illustrate the external and internal anatomy of arthropod fossils in great detail, including, among others, eurypterids (Lamsdell et al., 2020), ostracods (Wang et al., 2020), euarthropods (Liu et al., 2020; Laville et al., 2023), spiders (Downen and Selden, 2021), wasps (Szabó et al., 2022), and scorpions (de Carvalho et al., 2022). Decapod crustaceans, major contributors to marine ecosystems since the Mesozoic Decapod Revolution (Klompmaker et al., 2013, 2015; Schweitzer and Feldmann, 2015), have also been studied using newer techniques such as microCT (µCT) to uncover soft tissues (Jauvion et al., 2016, 2020b; Xing et al., 2021; Luque et al., 2021). Specimens with preserved soft tissue are generally derived from classic Konservat-Lagerstätten, from which exceptional preservation is well-known across multiple taxa. Here we report on a Late Cretaceous brachyuran crab from South Dakota, USA, from a deposit that has not been associated with exceptional preservation previously. While the external morphology of the crab is not very well preserved, we reveal a part of the internal anatomy.
GEOLOGICAL SETTING
Upper Cretaceous carbonates associated with cold seeps such as methane seeps in the Western Interior Seaway of the USA have received increased attention over the last decade (e.g., Landman et al., 2022). They range in age from the middle Campanian to the earliest Maastrichtian and are found in Montana, Wyoming, South Dakota, Nebraska, Colorado, and Kansas thus far (Landman et al., 2022). Seep carbonates vary in size and shape and can form mounds. Jointly, these carbonates contain a diverse fauna of invertebrates, vertebrates, and trace fossils (e.g., Bishop and Williams, 2000; Landman et al., 2012, 2022; Hunter et al., 2016; Rowe et al., 2020; Klompmaker and Landman, 2021; Brezina et al., 2022) at a higher concentration than in the adjacent, contemporaneous rocks. These hotspots of diversity in the Western Interior Seaway may have acted as refuges during times of ash fall and as sources for repopulating other parts of the seaway (Brophy et al., 2022).
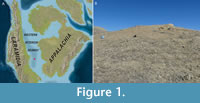 The crab specimen studied herein was found by Jamie Brezina between 2012-2018 at the American Museum of Natural History Invertebrate Paleontology (AMNH IP) locality 3529 on the eastern edge of the Black Hills in Pennington County, South Dakota, USA (43.9061°, -102.9075°), which used to be part of the Western Interior Seaway (Figure 1). This seep is within the upper Campanian Didymoceras cheyennense ammonite Zone in the Pierre Shale, 74.67±0.15 m.y.a. (Landman et al., 2018a, 2022). The fauna recently reported from this locality includes ammonites, bivalves, gastropods, sea stars, crinoids, decapod crustaceans, bryozoans, corals, serpulid annelids, and fish remains (Cochran et al., 2015; Blake et al., 2018; Brezina et al., 2022; Klompmaker et al., 2022; Landman et al., 2022). Decapod crustaceans found here, but not scanned for internal anatomy, include Protocallianassa? cheyennensis (Rathbun, 1930), Latheticocarcinus punctatus (Rathbun, 1917), Dakoticancer overanus (Rathbun, 1917), Bournelyreidus oaheensis (Bishop, 1978), and Secretanella spinosa (Bishop, 1991) (see also Klompmaker et al., 2022, table 5.1). The specimen herein was discovered as surface float on the southern side of the seep hill, down slope rather than strictly in situ. Based on the carbonate mineralogy of the crab specimen, the specimen must have been derived from the in situ carbonates making up the hill.
The crab specimen studied herein was found by Jamie Brezina between 2012-2018 at the American Museum of Natural History Invertebrate Paleontology (AMNH IP) locality 3529 on the eastern edge of the Black Hills in Pennington County, South Dakota, USA (43.9061°, -102.9075°), which used to be part of the Western Interior Seaway (Figure 1). This seep is within the upper Campanian Didymoceras cheyennense ammonite Zone in the Pierre Shale, 74.67±0.15 m.y.a. (Landman et al., 2018a, 2022). The fauna recently reported from this locality includes ammonites, bivalves, gastropods, sea stars, crinoids, decapod crustaceans, bryozoans, corals, serpulid annelids, and fish remains (Cochran et al., 2015; Blake et al., 2018; Brezina et al., 2022; Klompmaker et al., 2022; Landman et al., 2022). Decapod crustaceans found here, but not scanned for internal anatomy, include Protocallianassa? cheyennensis (Rathbun, 1930), Latheticocarcinus punctatus (Rathbun, 1917), Dakoticancer overanus (Rathbun, 1917), Bournelyreidus oaheensis (Bishop, 1978), and Secretanella spinosa (Bishop, 1991) (see also Klompmaker et al., 2022, table 5.1). The specimen herein was discovered as surface float on the southern side of the seep hill, down slope rather than strictly in situ. Based on the carbonate mineralogy of the crab specimen, the specimen must have been derived from the in situ carbonates making up the hill.
MATERIALS AND METHODS
High-resolution X-ray computed tomography (µCT) scans of specimen ALMNH:Paleo:6522 were obtained at the Berkeley Preclinical Imaging Facility (UC Berkeley, California, USA) in July 2019 using a GE Healthcare eXplore Locus Micro CT Scanner. The specimen was scanned using a conebeam energy of 80 kV, a current of 450 µA, 2,000 ms exposure time, and no filter, resulting in a voxel resolution of 20.523 µm. Visualization and three-dimensional reconstruction of the resulting µCT data were performed using the open-source software 3D Slicer v.4.13.0 (Fedorov et al., 2012). The µCT-scan data, stored as a 1.28 GB stack of DICOM images, and an.stl 3D-model of the outer surface of the crab are available at Zenodo [https://doi.org/10.5281/zenodo.7686211, https://doi.org/10.5281/zenodo.7686121].
A standard procedure to evaluate whether anaerobic oxidation of methane took place at suspected methane seep deposits, such as AMNH IP loc. 3529, is to analyze the carbon (δ13C) and oxygen (δ18O) isotopes of the seep carbonates (e.g., Landman et al., 2012). The specimen studied herein (ALMNH:Paleo:6522) did not have sufficient sedimentary or concretionary matrix surrounding the specimen, so the matrix attached to two other decapods from the same locality and carbonate deposit, both benthic decapods (ALMNH:Paleo:20359, Bournelyreidus oaheensis; ALMNH:Paleo:20360a, Protocallianassa cheyennensis), were analyzed at the UC Santa Cruz Stable Isotope Laboratory (California, USA) using the Thermo Fisher Scientific Kiel IV-MAT 253 device. The methodology is described in detail in Landman et al. (2018b).
The cited specimens are reposited in the Alabama Museum of Natural History paleontology collection, University of Alabama, Tuscaloosa, Alabama, USA (ALMNH:Paleo). The locality is abbreviated by an American Museum of Natural History, New York, New York, USA, invertebrate paleontology number (AMNH IP).
RESULTS
Taxon Identification
Specimen ALMNH:Paleo:6522 represents a female individual given the wide abdomen and is identified as Secretanella sp. Two species of Secretanella Guinot and Tavares, 2001, are known from the Pierre Shale: (1) S. occidentalis (Bishop, 1985) from the lower Campanian Gammon Ferruginous Member within the Baculites sp. smooth Zone above the Groat Sandstone and below bentonite H in Butte County, South Dakota (see Landman et al., 2014), and (2) S. spinosa from the upper Campanian Baculites cuneatus Zone of Grand County, Colorado, and from the same locality that ALMNH:Paleo:6522 originates from in South Dakota (Klompmaker et al., 2022). Key carapace features of ALMNH:Paleo:6522 used to distinguish between these species such as the course of the cervical groove, the degree of ornamentation, and the number of spines on the anterolateral margin (Bishop, 1991; Schweitzer et al., 2012) are poorly preserved in this 21 mm wide specimen to the point that species identification is not feasible. Moreover, possible ontogenetic changes in carapace morphology for these two species are insufficiently known due to the low number of available specimens.
Carbon and Oxygen Isotopes
Isotope analyses of carbonate matrix surrounding a specimen of Bournelyreidus oaheensis (ALMNH:Paleo:20359) yielded a δ13C value of -26.03‰ VPDB and a δ18O value of -4.97‰ VPDB. Matrix around a specimen of Protocallianassa? cheyennensis (ALMNH:Paleo:20360a) resulted in a δ13C value of -26.66‰ VPDB and a δ18O value of -5.58‰ VPDB. Similar values were reported by Gao et al. (2021) for this site: a δ13C value of -23.36±0.01‰ and a δ18O value of -5.37±0.09‰. These low δ13C values alone are insufficient to support that AMNH IP loc. 3529 is a methane seep, but the abundant presence of carbonates in the shale in conjunction with typical seep fauna such as lucinid bivalves at this site, in accordance with Landman et al. (2022) support a methane seep hypothesis.
Internal Anatomy
 A part of the outside of the replaced carapace cuticle is missing and reveals clear three-dimensionally preserved gill structures including four visible phyllobranchiate gills (Figure 2, Figure 3).
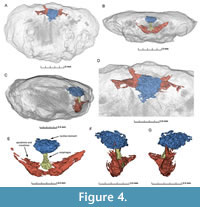
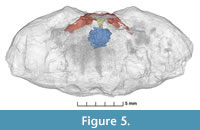
A part of the outside of the replaced carapace cuticle is missing and reveals clear three-dimensionally preserved gill structures including four visible phyllobranchiate gills (Figure 2, Figure 3).  We interpret that the fourth partly overlays the third gill. The individual branchial lamellae are visible as dark, vertical bands with calcium carbonate layers in between them (Figure 2, Figure 3). A possible partial afferent vessel may be preserved in the second gill (Figure 3). Despite the incomplete preservation, the individual gills appear similar in overall structure to those in fossil and extant brachyurans (e.g., Robin et al., 2018; Luque et al., 2021). More gills may be present, but they are not exposed. Comparison with gills from extant representatives within the same group (Etyoida) is not possible because all species of this clade are extinct. The subsequent µCT-scan we carried out did not reveal additional gills, but it uncovered other structures not visible to the naked eye. These structures include the esophagus (≤ 1 mm in cross section), cardiac stomach (foregut) (~2.5 mm wide), the mandibles and apodemes (~7 mm wide), and possible anterior gastric muscles (~1.5 mm wide for each) (Figure 4, Figure 5; Appendix 1, Figures S1-S4). The esophagus is positioned below the cardiac stomach to which it connects, which is known from various extant decapods such as brachyurans, anomurans, dendrobranchiatans, and astacideans (e.g., McLaughlin, 1980; Keiler et al., 2016; Castejón et al., 2018; Smolowitz, 2021). The esophageal walls in conjunction with muscles help transport the ingested food into the cardiac stomach for further processing (Davie et al., 2015; Castejón et al., 2018). The apodemes and in particular the attached mandibles are very close to the ventrally located opening of the esophagus. The remaining portions of the digestive tract, gills, and other internal anatomy could not be resolved by µCT-scanning.
We interpret that the fourth partly overlays the third gill. The individual branchial lamellae are visible as dark, vertical bands with calcium carbonate layers in between them (Figure 2, Figure 3). A possible partial afferent vessel may be preserved in the second gill (Figure 3). Despite the incomplete preservation, the individual gills appear similar in overall structure to those in fossil and extant brachyurans (e.g., Robin et al., 2018; Luque et al., 2021). More gills may be present, but they are not exposed. Comparison with gills from extant representatives within the same group (Etyoida) is not possible because all species of this clade are extinct. The subsequent µCT-scan we carried out did not reveal additional gills, but it uncovered other structures not visible to the naked eye. These structures include the esophagus (≤ 1 mm in cross section), cardiac stomach (foregut) (~2.5 mm wide), the mandibles and apodemes (~7 mm wide), and possible anterior gastric muscles (~1.5 mm wide for each) (Figure 4, Figure 5; Appendix 1, Figures S1-S4). The esophagus is positioned below the cardiac stomach to which it connects, which is known from various extant decapods such as brachyurans, anomurans, dendrobranchiatans, and astacideans (e.g., McLaughlin, 1980; Keiler et al., 2016; Castejón et al., 2018; Smolowitz, 2021). The esophageal walls in conjunction with muscles help transport the ingested food into the cardiac stomach for further processing (Davie et al., 2015; Castejón et al., 2018). The apodemes and in particular the attached mandibles are very close to the ventrally located opening of the esophagus. The remaining portions of the digestive tract, gills, and other internal anatomy could not be resolved by µCT-scanning.
DISCUSSION
Konservat-Lagerstätte and Preservation
 According to its original definition, the presence of fossilized soft parts is what defines a Konservat-Lagerstätte (Seilacher, 1970). Given the recognition of soft parts within a single crab, AMNH IP loc. 3529 may be considered a candidate Konservat-Lagerstätte. Additional fossils with soft tissue preservation from this site would strengthen the case to refer to this site as a Konservat-Lagerstätte, because it is generally understood that multiple species with soft tissue preservation at a site constitute a Konservat-Lagerstätte (e.g., Allison and Briggs, 1993; Muscente et al., 2017). The development of imaging methods within the context of fossil specimens has aided the identification of preserved fine morphological details, and has facilitated the recognition of originally soft parts from localities where soft tissue preservation would be less likely to be recognized without these techniques. As a result, many more Konservat-Lagerstätten and perhaps more types of Konservat-Lagerstätten may be reported in the future, but the proportion of sites with exceptional preservation of mineralized soft tissues among all fossil sites is likely to remain low.
According to its original definition, the presence of fossilized soft parts is what defines a Konservat-Lagerstätte (Seilacher, 1970). Given the recognition of soft parts within a single crab, AMNH IP loc. 3529 may be considered a candidate Konservat-Lagerstätte. Additional fossils with soft tissue preservation from this site would strengthen the case to refer to this site as a Konservat-Lagerstätte, because it is generally understood that multiple species with soft tissue preservation at a site constitute a Konservat-Lagerstätte (e.g., Allison and Briggs, 1993; Muscente et al., 2017). The development of imaging methods within the context of fossil specimens has aided the identification of preserved fine morphological details, and has facilitated the recognition of originally soft parts from localities where soft tissue preservation would be less likely to be recognized without these techniques. As a result, many more Konservat-Lagerstätten and perhaps more types of Konservat-Lagerstätten may be reported in the future, but the proportion of sites with exceptional preservation of mineralized soft tissues among all fossil sites is likely to remain low.
 The cause for the preservation of these crab soft tissues may differ from classic Konservat-Lagerstätten because of its probable link to methane seep carbonate formation. It is likely that the specimen did not live exclusively in this seep environment (see Mode of life), but methane seepage leading to elevated levels of poisonous hydrogen sulfide could have been the reason for the crab’s death (cf. Rowe et al., 2020, for ammonites). Experiments have shown that decapod soft tissues decay relatively quickly (Plotnick, 1986; Krause et al., 2011; Klompmaker et al., 2017) and, therefore, a preservation process would be required to geologically stabilize the soft tissues. At this site, microbially-induced carbonate precipitation and sulfate reduction may have occurred (Peckmann and Thiel, 2004; Pierre and Fouquet, 2007; Case et al., 2015). Microbial methanogenesis and sulfate reduction are linked to seep carbonate precipitation (Beauchamp et al., 1989; Capozzi et al., 2012). The combination of such physio-chemical conditions and microbial activity seems very favorable for rapid mineralization of soft tissue (Jauvion et al., 2020a, and references therein). Rapid mineralization of echinoid muscular soft tissue by amorphous calcium carbonate precipitation was shown experimentally in a marine brine seep setting, suggested to have been aided by sulphide-oxidizing bacteria (Deline and Parsons-Hubbard, 2013).
The cause for the preservation of these crab soft tissues may differ from classic Konservat-Lagerstätten because of its probable link to methane seep carbonate formation. It is likely that the specimen did not live exclusively in this seep environment (see Mode of life), but methane seepage leading to elevated levels of poisonous hydrogen sulfide could have been the reason for the crab’s death (cf. Rowe et al., 2020, for ammonites). Experiments have shown that decapod soft tissues decay relatively quickly (Plotnick, 1986; Krause et al., 2011; Klompmaker et al., 2017) and, therefore, a preservation process would be required to geologically stabilize the soft tissues. At this site, microbially-induced carbonate precipitation and sulfate reduction may have occurred (Peckmann and Thiel, 2004; Pierre and Fouquet, 2007; Case et al., 2015). Microbial methanogenesis and sulfate reduction are linked to seep carbonate precipitation (Beauchamp et al., 1989; Capozzi et al., 2012). The combination of such physio-chemical conditions and microbial activity seems very favorable for rapid mineralization of soft tissue (Jauvion et al., 2020a, and references therein). Rapid mineralization of echinoid muscular soft tissue by amorphous calcium carbonate precipitation was shown experimentally in a marine brine seep setting, suggested to have been aided by sulphide-oxidizing bacteria (Deline and Parsons-Hubbard, 2013).
Several factors may have played a role in the preservation of decapods in seeps including the rate of carbonate precipitation and where crabs were located relative to the highest concentration of seepage. Some seeps may not produce enough carbonate near the surface to allow rapid precipitation of carbonates to preserve soft parts. If crabs are directly on top of the seep field, then carbonate precipitation might have rapidly encased them. If specimens were living on the outer perimeter of the seep, then carbonate precipitation might have been slower or non-existent, allowing predators and scavengers to feed upon animals not rapidly buried. For this particular crab, carbonate precipitation occurred rapidly, and it is likely the crab was at or very near the highest concentration of seepage upon its death. The same scenario may apply to some articulated sea stars (Blake et al., 2018) and a fish with many 3D-preserved, articulated scales (J. B., pers. obs.) from the same site.
Although the gills are visible from the outside of the specimen, they were not visible in the µCT-scan. It was also the case for the two reconstructed polychelidan lobsters from La Voulte-sur-Rhône (Jauvion et al., 2016, 2020a), whereas thylacocephalans (Secrétan, 1985; Wilby et al., 1996; Jauvion et al., 2020b) and dendrobranchiate shrimps (Jauvion, 2020) from the same locality display exquisite gill preservation when cut and prepared as thin sections. Decapod and thylacocephalan specimens from La Voulte preserved within carbonate concretions display an identical mineralogical composition (Jauvion, 2020). The lack of X-ray contrast of the fossilized gills compared to the fossil matrix might explain why they do not render well with µCT-scans despite their presence. Other internal organs are missing, which may be due to several factors: insufficient µCT-scan resolution, insufficient density contrast compared to the fossil matrix, decay prior to mineralization (e.g., Clements et al., 2022), or, simply, absence of mineral replication of the structures.
Mode of Life
Although only the carapace of this specimen is preserved, we suggest this specimen was capable of swimming because of the relatively low carapace length/width ratio of approximately 0.6, relatively low height (~25% of width), and the flattened morphology of the dorsal carapace (e.g., Hartnoll, 1971; Bishop, 1991; Fraaye, 1996). The mode of life allowed this crab to visit seeps as a migrant rather than being an obligatory inhabitant of seeps. In support of this hypothesis, other specimens of this genus have also been found in non-seep sites in the Pierre Shale (Bishop, 1985, 1991).
Internal Soft Tissue in Fossil Decapod Crustaceans
Although soft tissue preservation in decapod crustaceans remains a rare phenomenon in general, an increasing number of specimens with such preservation are being reported. Muscle tissues in decapod crustaceans have been reported in over 30 species from primarily classic Jurassic and Cretaceous Konservat-Lagerstätten (Klompmaker et al., 2019: table 1) out of well over 3,650 fossil decapod species (Schweitzer and Feldmann, 2016). Other internal structures are even rarer and are also mostly reported from the Jurassic and Cretaceous. Herein (Table 1), we show that gills are known for only eight fossil species, and at least part of the digestive tract has been mentioned for 13 species. Other internal soft tissues interpreted to be preserved in fossil decapods include ovaries, nerve chords, and a heart (Table 1). The specimen reported here is unique in that an esophagus is reported for the first time in a fossil decapod and it is the first decapod from an ancient methane seep reported to preserve soft tissues.
As more finds become available, a more thorough assessment of the evolution of soft tissues within and across decapod clades may become possible. Specimens that do not show soft tissues externally may also contain soft tissues, however, and as imaging techniques become more widely available and cheaper, scanning of fossil decapod specimens will allow the proportion of specimens with soft tissue preserved across deposits and across decapod clades to be evaluated. Our find from a methane seep may serve as a reminder that soft tissue preservation is not restricted to classic Konservat-Lagerstätten but can be encountered at any place where the geochemical conditions are suitable for early diagenetic mineralization.
To our knowledge, no mineralized soft tissue has ever been found in any organismal group from ancient methane seeps until this paper, but fossils from these deposits typically have not been investigated using CT-scanning. Thus, to what extent soft tissue is preserved among seep animals is unknown. Non-soft tissue organic material has been reported from organic-walled fossil elasmobranch egg capsules from ancient seeps (Kiel et al., 2013; Treude, 2022) and fossil organic-walled annelids from fossil seeps and hydrothermal vents (Georgieva et al., 2019; Georgieva and Little, 2022). True soft tissue remains of the digestive tract of pholadoidean bivalves were reported from another ancient chemosynthetic community, a late Oligocene-early Miocene wood fall from Washington State, USA (Kiel et al., 2012).
CONCLUSIONS
The crab (Secretanella sp.) we studied from a Campanian methane seep in South Dakota, USA, shows four phyllobranchiate gills in the left branchial chamber. These gills are visible because the carapace cuticle is broken there. A µCT-scan of this crab reveals more details of its internal anatomy, including the esophagus, cardiac stomach (foregut), and possible anterior gastric muscles. The mandibles and their apodemes were also identified. The esophagus is reported for the first time in fossil decapod crustaceans. This crab represents the first animal from a fossil methane seep to have fossil soft tissues identified. Hence, the site can be considered a candidate for a Konservat-Lagerstätte.
Our new overview of internal soft tissues preserved in fossil decapods shows that, besides muscles reported on previously, gills and remains of the digestive tract are most often reported. Despite an increase in reports on fossil decapods with internal soft tissues preserved over the last decade, soft internal anatomy preservation remains relatively rare.
ACKNOWLEDGMENTS
This study is a part of a project supported by a 2018 American Museum of Natural History Lerner-Gray grant and a 2020 Western Interior Paleontological Society Karl Hirsch Memorial grant to A.A.K. We thank S. Kiel (Swedish Museum of Natural History) and K. Hryniewicz (Polish Academy of Sciences) for providing items of literature and information, N.L. Larson (Larson Paleontology Unlimited) for reading the paper via J.B., and the USDA Forest Service Department for permits to collect and preserve. Finally, we thank reviewers M. Andrada (Universidad de Buenos Aires) and T. Clements (Friedrich-Alexander-University of Erlangen-Nürnberg) as well as the editor for very useful comments that improved this paper.
REFERENCES
Allison, P.A. and Briggs, D.E.G. 1993. Exceptional fossil record: distribution of soft-tissue preservation through the Phanerozoic. Geology, 21:527-530.
https://doi.org/10.1130/0091-7613(1993)021<0527:EFRDOS>2.3.CO;2
Audo, D. and Charbonnier, S. 2013. Late Cretaceous crest-bearing shrimps from the Sahel Alma Lagerstätte of Lebanon. Acta Palaeontologica Polonica, 58:335-349.
https://doi.org/10.4202/app.2011.0056
Audo, D., Schweigert, G., Martin, J.-P.S., and Charbonnier, S. 2014. High biodiversity in Polychelida crustaceans from the Jurassic La Voulte-sur-Rhône Lagerstätte. Geodiversitas, 36:489-525.
https://doi.org/10.5252/g2014n4a1
Beauchamp, B., Harrison, J.C., Nassichuk, W.W., Krouse, H.R., and Eliuk, L.S. 1989. Cretaceous cold-seep communities and methane-derived carbonates in the Canadian Arctic. Science, 244:53-56.
https://doi.org/10.1126/science.244.4900.53
Bishop, G.A. 1978. Two new crabs, Sodakus tatankayotankaensis n. gen., n. sp. and Raninella oaheensis n. sp. (Crustacea, Decapoda), from the Upper Cretaceous Pierre Shale of South Dakota. Journal of Paleontology, 52:608-617.
Bishop, G.A. 1985. Fossil decapod crustaceans from the Gammon Ferruginous Member, Pierre Shale (Early Campanian), Black Hills, South Dakota. Journal of Paleontology, 59:605-624.
Bishop, G.A. 1991. Xanthosia occidentalis Bishop, 1985, and Xanthosia spinosa, new species, two Late Cretaceous crabs from the Pierre Shale of the Western Interior. Journal of Crustacean Biology, 11:305-314.
https://doi.org/10.1163/193724091X00112
Bishop, G.A. and Williams, A.B. 2000. Fossil crabs from tepee buttes, submarine seeps of the Late Cretaceous Pierre Shale, South Dakota and Colorado, U.S.A. Journal of Crustacean Biology, 20:286-300.
https://doi.org/10.1163/1937240X-90000031
Blake, D.B., Halligan, W.K., and Larson, N.L. 2018. A new species of the asteroid genus Betelgeusia (Echinodermata) from methane seep settings, Late Cretaceous of South Dakota. Journal of Paleontology, 92:196-206.
https://doi.org/10.1017/jpa.2017.96
Brezina, J., Larson, N.L., and Landman, N.H. 2022. Echinoderms at ancient hydrocarbon seeps and cognate communities, p. 407-418. In Kaim, A., Cochran, J.K., and Landman, N.H. (eds.), Ancient Hydrocarbon Seeps. Topics in Geobiology 53. Springer International Publishing, Cham.
https://doi.org/10.1007/978-3-031-05623-9_13
Brocchi, P. 1875. Note sur une nouvelle espèce de Crustacé fossile (Penaeus libanensis). Bulletin de la Societé Geologique de France, Paris, 3:609-610.
Brophy, S.K., Garb, M.P., Naujokaityte, J., Witts, J.D., Landman, N.H., Cochran, J.K., and Brezina, J. 2022. Methane seeps as refugia during ash falls in the Late Cretaceous Western Interior Seaway of North America. Geology, 50:442-447.
https://doi.org/10.1130/G49372.1
Butterfield, N.J. 2002. Leanchoilia guts and the interpretation of three-dimensional structures in Burgess Shale-type fossils. Paleobiology, 28:155-171.
https://doi.org/10.1666/0094-8373(2002)028<0155:LGATIO>2.0.CO;2
Capozzi, R., Guido, F.L., Oppo, D., and Gabbianelli, G. 2012. Methane-Derived Authigenic Carbonates (MDAC) in northern-central Adriatic Sea: relationships between reservoir and methane seepages. Marine Geology, 332-334:174-188.
https://doi.org/10.1016/j.margeo.2012.06.006
Case, D.H., Pasulka, A.L., Marlow, J.J., Grupe, B.M., Levin, L.A., and Orphan, V.J. 2015. Methane seep carbonates host distinct, diverse, and dynamic microbial assemblages. mBio, 6:e01348-15.
https://doi.org/10.1128/mBio.01348-15
Castejón, D., Rotllant, G., Ribes, E., Durfort, M., and Guerao, G. 2018. Morphology and ultrastructure of the esophagus during the ontogeny of the spider crab Maja brachydactyla (Decapoda, Brachyura, Majidae). Journal of Morphology, 279:710-723.
https://doi.org/10.1002/jmor.20805
Clements, T., Purnell, M.A., and Gabbott, S. 2022. Experimental analysis of organ decay and pH gradients within a carcass and the implications for phosphatization of soft tissues. Palaeontology, 65:e12617.
https://doi.org/10.1111/pala.12617
Cochran, J.K., Landman, N.H., Larson, N.L., Meehan, K.C., Garb, M., and Brezina, J. 2015. Geochemical evidence (C and Sr isotopes) for methane seeps as ammonite habitats in the Late Cretaceous (Campanian) Western Interior Seaway. Swiss Journal of Palaeontology, 134:153-165.
https://doi.org/10.1007/s13358-015-0087-9
Cushman, J.A. 1905. Fossil crabs of the Gay Head Miocene. The American Naturalist, 39:381-390.
Davie, P.J.F., Guinot, D., and Ng, P.K.L. 2015. Anatomy and functional morphology of Brachyura, p. 11-163. In Castro, P., Davie, P.J.F., Guinot, D., Schram, F.R., and von Vaupel Klein, J.C. (eds.), Treatise on Zoology - Anatomy, Taxonomy, Biology. The Crustacea, Volume 9 Part C (2 vols). Brill, Leiden.
https://doi.org/10.1163/9789004190832_004
de Carvalho, M.G.P., Maisey, J.G., Mendes, I.D., and de Souza Carvalho, I. 2022. Micro-tomographic analysis of a scorpion fossil from the Aptian Crato Formation of Northeastern Brazil. Cretaceous Research, 105454. https://doi.org/10.1016/j.cretres.2022.105454
Deline, B. and Parsons-Hubbard, K.M. 2013. Experimentally observed soft-tissue preservation near a marine brine seep. Palaeontology, 56:893-900.
https://doi.org/10.1111/pala.12023
de Mazancourt, V., Wappler, T., and Wedmann, S. 2022. Exceptional preservation of internal organs in a new fossil species of freshwater shrimp (Caridea: Palaemonoidea) from the Eocene of Messel (Germany). Scientific Reports, 12:18114.
https://doi.org/10.1038/s41598-022-23125-9
Downen, M.R. and Selden, P.A. 2021. The earliest palpimanid spider (Araneae: Palpimanidae), from the Crato Fossil-Lagerstätte (Cretaceous, Brazil). Journal of Arachnology, 49:91-97.
https://doi.org/10.1636/JoA-S-19-059
Fedorov, A., Beichel, R., Kalpathy-Cramer, J., Finet, J., Fillion-Robin, J.-C., Pujol, S., Bauer, C., Jennings, D., Fennessy, F., Sonka, M., Buatti, J., Aylward, S., Miller, J.V., Pieper, S., and Kikinis, R. 2012. 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magnetic Resonance Imaging, 30:1323-1341.
https://doi.org/10.1016/j.mri.2012.05.001
Feldmann, R.M., Schweitzer, C.E., and Leahy, J. 2011. New Eocene crayfish from the Mcabee Beds in British Columbia: first record of Parastacoidea in the Northern Hemisphere. Journal of Crustacean Biology, 31:320-331.
https://doi.org/10.1651/10-3399.1
Fraaye, R.H.B. 1996. Late Cretaceous swimming crabs: radiation, migration, competition, and extinction. Acta Geologica Polonica, 46:269-278.
Franţescu, O.D., Feldmann, R.M., and Schweitzer, C.E. 2016. Cretaceous fossil Raninoida De Haan, 1839 (Crustacea, Decapoda, Brachyura) from northeast Texas. Journal of Paleontology, 90:1118-1132.
https://doi.org/10.1017/jpa.2016.106
Gao, Y., Henkes, G.A., Cochran, J.K., and Landman, N.H. 2021. Temperatures of Late Cretaceous (Campanian) methane-derived authigenic carbonates from the Western Interior Seaway, South Dakota, USA, using clumped isotopes. GSA Bulletin, 133:2524-2534.
https://doi.org/10.1130/B35846.1
Garassino, A., Pasini, G., and Dutheil, D.B. 2006. Cretapenaeus berberus n. gen., n. sp. (Crustacea, Decapoda, Penaeidae) from the Late Cretaceous (Cenomanian) of southeastern Morocco. Atti della Società italiana di scienze naturali e del museo civico di storia naturale di Milano, 147:3-17.
Garassino, A., De Angeli, A., and Pasini, G. 2009. A new hermit crab (Crustacea, Anomura, Paguroidea) from the Late Cretaceous (Cenomanian) of Lebanon. Atti della Società italiana di Scienze naturali e del Museo civico di Storia naturale in Milano, 150:215-228.
Georgieva, M.N. and Little, C.T.S. 2022. Non-calcareous tubeworms in ancient hydrocarbon seeps, p. 201-214. In Kaim, A., Cochran, J.K., and Landman, N.H. (eds.), Ancient Hydrocarbon Seeps. Topics in Geobiology 53. Springer International Publishing, Cham.
https://doi.org/10.1007/978-3-031-05623-9_6
Georgieva, M.N., Little, C.T.S., Watson, J.S., Sephton, M.A., Ball, A.D., and Glover, A.G. 2019. Identification of fossil worm tubes from Phanerozoic hydrothermal vents and cold seeps. Journal of Systematic Palaeontology, 17:287-329.
https://doi.org/10.1080/14772019.2017.1412362
Gueriau, P., Mocuta, C., Dutheil, D.B., Cohen, S.X., Thiaudière, D., The OT1 Consortium, Charbonnier, S., Clément, G., and Bertrand, L. 2014. Trace elemental imaging of rare earth elements discriminates tissues at microscale in flat fossils. PLoS ONE, 9:e86946.
https://doi.org/10.1371/journal.pone.0086946
Guinot, D. and Tavares, M. 2001. Une nouvelle famille de crabes du Crétacé, et la notion de Podotremata Guinot, 1977 (Crustacea, Decapoda, Brachyura). Zoosystema, 23:507-546.
Hartnoll, R.G. 1971. The occurrence, methods and significance of swimming in the Brachyura. Animal Behaviour, 19:34-50.
https://doi.org/10.1016/S0003-3472(71)80132-X
Hunter, A.W., Larson, N.L., Landman, N.H., and Oji, T. 2016. Lakotacrinus brezinai n. gen. n. sp., a new stalked crinoid from cold methane seeps in the Upper Cretaceous (Campanian) Pierre Shale, South Dakota, United States. Journal of Paleontology, 90:506-524.
https://doi.org/10.1017/jpa.2016.21
Jauvion, C. 2020. De la vie à la pierre: préservation exceptionnelle d'arthropodes marins fossiles. Dissertation, Muséum national d’Histoire naturelle, Paris.
Jauvion, C., Audo, D., Charbonnier, S., and Vannier, J. 2016. Virtual dissection and lifestyle of a 165-million-year-old female polychelidan lobster. Arthropod Structure & Development, 45:122-132.
https://doi.org/10.1016/j.asd.2015.10.004
Jauvion, C., Bernard, S., Gueriau, P., Mocuta, C., Pont, S., Benzerara, K., and Charbonnier, S. 2020a. Exceptional preservation requires fast biodegradation: thylacocephalan specimens from La Voulte-sur-Rhône (Callovian, Jurassic, France). Palaeontology, 63:395-413.
https://doi.org/10.1111/pala.12456
Jauvion, C., Audo, D., Bernard, S., Vannier, J., Daley, A.C., and Charbonnier, S. 2020b. A new polychelidan lobster preserved with its eggs in a 165 Ma nodule. Scientific Reports, 10:3574.
https://doi.org/10.1038/s41598-020-60282-1
Keiler, J., Richter, S., and Wirkner, C.S. 2016. Revealing their innermost secrets: an evolutionary perspective on the disparity of the organ systems in anomuran crabs (Crustacea: Decapoda: Anomura). Contributions to Zoology, 85:361-386.
https://doi.org/10.1163/18759866-08504001
Kiel, S., Götz, S., Pascual-Cebrian, E., and Hennhöfer, D.K. 2012. Fossilized digestive systems in 23 million-year-old wood-boring bivalves. Journal of Molluscan Studies, 78:349-356.
https://doi.org/10.1093/mollus/eys021
Kiel, S., Peckmann, J., and Simon, K. 2013. Catshark egg capsules from a Late Eocene deep-water methane-seep deposit in western Washington State, USA. Acta Palaeontologica Polonica, 58:77-84.
https://doi.org/10.4202/app.2011.0077
Klompmaker, A.A., Schweitzer, C.E., Feldmann, R.M., and Kowalewski, M. 2013. The influence of reefs on the rise of Mesozoic marine crustaceans. Geology, 41:1179-1182.
https://doi.org/10.1130/G34768.1
Klompmaker, A.A., Schweitzer, C.E., Feldmann, R.M., and Kowalewski, M. 2015. Environmental and scale-dependent evolutionary trends in the body size of crustaceans. Proceedings of the Royal Society B: Biological Sciences, 282:20150440.
https://doi.org/10.1098/rspb.2015.0440
Klompmaker, A.A., Portell, R.W., and Frick, M.G. 2017. Comparative experimental taphonomy of eight marine arthropods indicates distinct differences in preservation potential. Palaeontology, 60:773-794.
https://doi.org/10.1111/pala.12314
Klompmaker, A.A., Hyžný, M., Portell, R.W., Jauvion, C., Charbonnier, S., Fussell, S.S., Klier, A.T., Tejera, R., and Jakobsen, S.L. 2019. Muscles and muscle scars in fossil malacostracan crustaceans. Earth-Science Reviews, 194:306-326.
https://doi.org/10.1016/j.earscirev.2019.04.012
Klompmaker, A.A. and Landman, N.H. 2021. Octopodoidea as predators near the end of the Mesozoic Marine Revolution. Biological Journal of the Linnean Society, 132:894-899.
https://doi.org/10.1093/biolinnean/blab001
Klompmaker, A.A., Nyborg, T., Brezina, J., and Ando, Y. 2022. Crustaceans in cold seep ecosystems: fossil record, geographic distribution, taxonomic composition, and biology, p. 123-200. In Kaim, A., Cochran, J.K., and Landman, N.H. (eds.), Ancient Hydrocarbon Seeps. Topics in Geobiology 53. Springer International Publishing, Cham.
https://doi.org/10.1007/978-3-031-05623-9_5
Krause, R.A., Parsons-Hubbard, K., and Walker, S.E. 2011. Experimental taphonomy of a decapod crustacean: long-term data and their implications. Palaeogeography, Palaeoclimatology, Palaeoecology, 312:350-362.
https://doi.org/10.1016/j.palaeo.2011.03.020
Lamsdell, J.C., McCoy, V.E., Perron-Feller, O.A., and Hopkins, M.J. 2020. Air breathing in an exceptionally preserved 340-million-year-old sea scorpion. Current Biology, 30:4316-4321.
https://doi.org/10.1016/j.cub.2020.08.034
Landman, N.H., Cochran, J.K., Larson, N.L., Brezina, J., Garb, M.P., and Harries, P.J. 2012. Methane seeps as ammonite habitats in the U.S. Western Interior Seaway revealed by isotopic analyses of well-preserved shell material. Geology, 40:507-510.
https://doi.org/10.1130/G32782.1
Landman, N.H., Fraaije, R.H.B., Klofak, S.M., Larson, N.L., Bishop, G.A., and Kruta, I. 2014. Inquilinism of a baculite by a dynomenid crab from the Upper Cretaceous of South Dakota. American Museum Novitates, 3818:1-16.
https://doi.org/10.1206/3818.1
Landman, N.H., Jicha, B.R., Cochran, J.K., Garb, M.P., Brophy, S.K., Larson, N.L., and Brezina, J. 2018a. 40Ar/39Ar date of a bentonite associated with a methane seep deposit in the upper Campanian Baculites compressus Zone, Pierre Shale, South Dakota. Cretaceous Research, 90:90-96.
https://doi.org/10.1016/j.cretres.2018.03.024
Landman, N.H., Cochran, J.K., Slovacek, M., Larson, N.L., Garb, M.P., Brezina, J., and Witts, J.D. 2018b. Isotope sclerochronology of ammonites (Baculites compressus) from methane seep and non-seep sites in the Late Cretaceous Western Interior Seaway, USA: Implications for ammonite habitat and mode of life. American Journal of Science, 318:603-639.
https://doi.org/10.2475/06.2018.01
Landman, N.H., Cochran, J.K., Brezina, J., Larson, N.L., Garb, M.P., Meehan, K.C., and Myers, C. 2022. Methane seeps in the Late Cretaceous Western Interior Seaway, USA, p. 425-477. In Kaim, A., Cochran, J.K., and Landman, N.H. (eds.), Ancient Hydrocarbon Seeps. Topics in Geobiology 53. Springer International Publishing, Cham.
https://doi.org/10.1007/978-3-031-05623-9_15
Laville, T., Hegna, T.H., Forel, M.-B., Darroch, S., and Charbonnier, S. 2023. New look at Concavicaris woodfordi (Euarthropoda: Pancrustacea?) using micro-computed tomography. Palaeontologia Electronica, 26:a1.
https://doi.org/10.26879/1218
Liu, Y., Ortega-Hernández, J., Zhai, D., and Hou, X. 2020. A reduced labrum in a Cambrian great-appendage euarthropod. Current Biology, 30:3057-3061.
https://doi.org/10.1016/j.cub.2020.05.085
Luque, J., Xing, L., Briggs, D.E.G., Clark, E.G., Duque, A., Hui, J., Mai, H., and McKellar, R.C. 2021. Crab in amber reveals an early colonization of nonmarine environments during the Cretaceous. Science Advances, 7:eabj5689.
https://doi.org/10.1126/sciadv.abj5689
McLaughlin, P.A. 1980. Comparative morphology of recent Crustacea. W.H. Freeman and Company, San Francisco.
Moreau, J.-D., Vullo, R., Charbonnier, S., Jattiot, R., Trincal, V., Néraudeau, D., Fara, E., Baret, L., Garassino, A., Gand, G., and Lafaurie, G. 2022. Konservat-Lagerstätten from the Upper Jurassic lithographic limestone of the Causse Méjean (Lozère, southern France): palaeontological and palaeoenvironmental synthesis. Geological Magazine, 159:761-781.
https://doi.org/10.1017/S0016756821001382
Muscente, A.D., Schiffbauer, J.D., Broce, J., Laflamme, M., O'Donnell, K., Boag, T.H., Meyer, M., Hawkins, A.D., Huntley, J.W., McNamara, M., and MacKenzie, L.A. 2017. Exceptionally preserved fossil assemblages through geologic time and space. Gondwana Research, 48:164-188.
https://doi.org/10.1016/j.gr.2017.04.020
Ortega-Hernández, J., Lerosey-Aubril, R., Losso, S.R., and Weaver, J.C. 2022. Neuroanatomy in a middle Cambrian mollisoniid and the ancestral nervous system organization of chelicerates. Nature Communications, 13:410.
https://doi.org/10.1038/s41467-022-28054-9
Pasini, G., Poore, G.C.B., and Garassino, A. 2020. A new ghost shrimp (Axiidea, Callianopsidae) from the Late Cretaceous (Cenomanian) of Hadjoula, Lebanon. Neues Jahrbuch für Geologie und Paläontologie - Abhandlungen, 297:217-225.
https://doi.org/10.1127/njgpa/2020/0922
Peckmann, J. and Thiel, V. 2004. Carbon cycling at ancient methane-seeps. Chemical Geology, 205:443-467.
https://doi.org/10.1016/j.chemgeo.2003.12.025
Pierre, C. and Fouquet, Y. 2007. Authigenic carbonates from methane seeps of the Congo deep-sea fan. Geo-Marine Letters, 27:249-257.
https://doi.org/10.1007/s00367-007-0081-3
Plotnick, R.E. 1986. Taphonomy of a modern shrimp: Implications for the arthropod fossil record. PALAIOS, 1:286-293.
https://doi.org/10.2307/3514691
Rathbun, M.J. 1917. New species of South Dakota Cretaceous crabs. Proceedings of the United States National Museum, 52:385-391, pl. 32-33.
Rathbun, M.J. 1930. A new Callianassa from the Cretaceous of South Dakota. Journal of the Washington Academy of Sciences, 20:1-3.
Rathbun, M.J. 1935. Fossil Crustacea of the Atlantic and Gulf Coastal Plain. Geological Society of America, (special paper), 2:i-viii, 1-160.
Robin, N., Van Bakel, B.W.M., Blanc-Valleron, M.-M., Noël, P.Y., Lemaire, L., and Charbonnier, S. 2018. First fossil crabs (Crustacea, Decapoda, Brachyura) from the Kerguelen Islands (Miocene) with exceptionally preserved gills. Journal of Paleontology, 92:466-477.
https://doi.org/10.1017/jpa.2017.129
Rowe, A.J., Landman, N.H., Cochran, J.K., Witts, J.D., and Garb, M.P. 2020. Late Cretaceous methane seeps as habitats for newly hatched ammonites. PALAIOS, 35:151-163.
https://doi.org/10.2110/palo.2019.105
Sampson, S.D., Loewen, M.A., Farke, A.A., Roberts, E.M., Forster, C.A., Smith, J.A., and Titus, A.L. 2010. New horned dinosaurs from Utah provide evidence for intracontinental dinosaur endemism. PLOS ONE, 5(9):e12292.
https://doi.org/10.1371/journal.pone.0012292
Schweitzer, C.E., Feldmann, R.M., Franţescu, O.D., and Klompmaker, A. 2012. Revision of Etyidae Guinot and Tavares, 2001 (Crustacea: Brachyura). Journal of Paleontology, 86:129-155.
https://doi.org/10.1666/11-060.1
Schweitzer, C.E. and Feldmann, R.M. 2015. Faunal turnover and niche stability in marine Decapoda in the Phanerozoic. Journal of Crustacean Biology, 35:633-649.
https://doi.org/10.1163/1937240X-00002359
Schweitzer, C.E. and Feldmann, R.M. 2016. Species of Decapoda (Crustacea) in the fossil record: patterns, problems, and progress, p. 278-300. In Allmon, W.D. and Yacobucci, M.M. (eds.), Species and Speciation in the Fossil Record. University of Chicago Press, Chicago.
Secrétan, S. 1985. Conchyliocarida, a class of fossil crustaceans: relationships to Malacostraca and postulated behaviour. Earth and Environmental Science Transactions of The Royal Society of Edinburgh, 76:381-389.
https://doi.org/10.1017/S0263593300010592
Seilacher, A. 1970. Begriff und Bedeutung der Fossil-Lagerstätten. Neues Jahrbuch für Geologie und Paläontologie Monatshefte, 7:34-39.
Siveter, D.J., Briggs, D.E.G., Siveter, D.J., Sutton, M.D., and Joomun, S.C. 2013. A Silurian myodocope with preserved soft-parts: cautioning the interpretation of the shell-based ostracod record. Proceedings of The Royal Society B, 280:20122664.
https://doi.org/10.1098/rspb.2012.2664
Smolowitz, R. 2021. Arthropoda: Decapoda, 1st ed., p. 277-299. In LaDouceur, E.E.B. (ed.), Invertebrate Histology. Wiley.
https://doi.org/10.1002/9781119507697.ch11
Stimpson, W. 1863. On the fossil crab of Gay Head. Boston Journal of Natural History, 7:583-589, pl. 12.
Szabó, M., Brazidec, M., Perrichot, V., Szenti, I., Kukovecz, Á., and Ősi, A. 2022. A unique record of the Late Cretaceous of East-Central Europe: the first fossil wasps (Hymenoptera: Bethylidae, Spathiopterygidae) from the ajkaite amber (Bakony Mts., western Hungary). Cretaceous Research, 139:105314.
https://doi.org/10.1016/j.cretres.2022.105314
Treude, T. 2022. Vertebrates: skate and shark egg capsules at ancient hydrocarbon seeps, p. 419-421. In Kaim, A., Cochran, J.K., and Landman, N.H. (eds.), Ancient Hydrocarbon Seeps. Topics in Geobiology 53. Springer International Publishing, Cham.
https://doi.org/10.1007/978-3-031-05623-9_14
Van Straelen, V. 1923. Les crustacés décapodes du Callovien de la Voulte-sur-Rhône (Ardèche). Comptes Rendus des séances de l’Académie des Sciences, Paris, 175:982-983.
Wang, H., Matzke-Karasz, R., Horne, D.J., Zhao, X., Cao, M., Zhang, H., and Wang, B. 2020. Exceptional preservation of reproductive organs and giant sperm in Cretaceous ostracods. Proceedings of the Royal Society B: Biological Sciences, 287:20201661.
https://doi.org/10.1098/rspb.2020.1661
Wilby, P.R., Briggs, D.E.G., and Riou, B. 1996. Mineralization of soft-bodied invertebrates in a Jurassic metalliferous deposit. Geology, 24:847-850.
https://doi.org/10.1130/0091-7613(1996)024<0847:MOSBII>2.3.CO;2
Xing, L., Liu, Y., McKellar, R.C., Luque, J., Li, G., Wang, Y., Yi, Q., Sun, R., Wang, E., and Audo, D. 2021. The first shrimp preserved in mid-Cretaceous Kachin amber: systematics, palaeoecology, and taphonomy. Science Bulletin, 66:1723-1726.
https://doi.org/10.1016/j.scib.2021.05.008
Zacaï, A., Vannier, J., and Lerosey-Aubril, R. 2016. Reconstructing the diet of a 505-million-year-old arthropod: Sidneyia inexpectans from the Burgess Shale fauna. Arthropod Structure and Development, 45:200-220.
https://doi.org/10.1016/j.asd.2015.09.003

