 A new Oligo-Miocene dolphin from New Zealand: Otekaikea huata expands diversity of the early Platanistoidea
A new Oligo-Miocene dolphin from New Zealand: Otekaikea huata expands diversity of the early Platanistoidea
Article number: 18.2.23A
https://doi.org/10.26879/518
Copyright Palaeontological Association, May 2015
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Submission: 16 October 2014. Acceptance: 31 March 2015
{flike id=1161}
ABSTRACT
The New Zealand fossil dolphin Otekaikea huata (latest Oligocene to earliest Miocene, in the range 22.28 to 24.61 Ma) is here identified as an early new species in the clade Platanistoidea, which includes the endangered Ganges River dolphin ( Platanista gangetica). Otekaikea huata is known only from the holotype, which comprises a partial skeleton from the marine Otekaike Limestone of the Hakataramea Valley, South Island. Otekaikea huata has multiple procumbent tusks passing back to otherwise near-homodont and polydont teeth, and an elevated face for the nasofacial muscles implicated in production of echolocation sounds. The skull vertex is asymmetrical and strongly left-skewed. Phylogenetic analyses based on morphological features place Otekaikea huata in the clade Platanistoidea. The new species adds to the diversity of the superfamily Platanistoidea near the Oligocene-Miocene boundary.
Yoshihiro Tanaka. Department of Geology, University of Otago, 360 Leith Walk, PO Box 56, Dunedin 9054, New Zealand. yoshihiro.tanaka@otago.ac.nz
R. Ewan Fordyce. Department of Geology, University of Otago, 360 Leith Walk, PO Box 56, Dunedin 9054, New Zealand ewan.fordyce@otago.ac.nz
Keywords: fossil; dolphin; stratigraphy; morphology; feeding
Final citation: Tanaka, Yoshihiro and Fordyce, R. Ewan 2015. A new Oligo-Miocene dolphin from New Zealand: Otekaikea huata expands diversity of the early Platanistoidea. Palaeontologia Electronica 18.2.23A: 1-71. https://doi.org/10.26879/518
palaeo-electronica.org/content/2015/1161-fossil-platanistoid-dolphin
http://zoobank.org/F8462823-1EC9-4C8C-8A43-1EEED202F89C
INTRODUCTION
Fossil cetaceans of Oligocene age reveal the dramatic early radiation of the Neoceti (Whitmore and Sanders, 1977; Fordyce, 2009). In New Zealand, one of the key productive cetacean-bearing units is the Otekaike Limestone, a late Oligocene to earliest Miocene bioclastic grainstone that was deposited largely below storm wave base in the southern Canterbury Basin. Odontocetes from the Otekaike Limestone include Waipatia maerewhenua, Microcetus hectori and Otekaikea marplesi, as well as undescribed squalodontids and smaller platanistoid dolphins. Such fossils, and described Oligocene odontocetes from elsewhere in the world, show disparate morphologies (Abel, 1914; Allen, 1921; Fordyce, 1981; Fordyce, 2002) that are consistent with a major early radiation of odontocetes during the Oligocene. The reported fossils have led to quite different phylogenetic hypotheses (Fordyce, 1994; Geisler and Sanders, 2003; Uhen, 2008; Steeman et al., 2009; Geisler et al., 2011; Murakami et al., 2012b; Aguirre-Fernández and Fordyce, 2014; Geisler et al., 2014) with implications for the origin of modern families, taxonomy above genus level, and understanding of ecological patterns amongst clades. Most of the named Oligocene odontocetes listed by Uhen (2008, Table 1) are based on fragmentary specimens that give little insight into body form and function or, in some cases, phylogenetic relations. The description of associated partial skeletons, as below, will improve understanding of the early history of odontocetes (Tanaka and Fordyce, 2014).
Recently, another New Zealand species, “ Prosqualodon” marplesi Dickson, was placed in a new genus as Otekaikea marplesi (Tanaka and Fordyce, 2014). Here, a second species of Otekaikea is described from a naturally associated skull, ear bones and forelimb materials. Such preservation is not common for early odontocetes. This informative specimen expands the taxonomic and structural diversity of early odontocetes, with implications for ecology and resource partitioning.
MATERIAL AND METHODS
Material was prepared by A. Grebneff using pneumatic chisels and hand tools, and finished by Y. Tanaka and R.E. Fordyce under a Zeiss SR binocular microscope. The positions of most sutures were confirmed using the binocular microscope. Dilute (5%) acetic acid was used to free the articulated periotic from the squamosal and to remove concretionary carbonate. Bone was glued with cyanoacrylate and/or polyvinyl butyral solution, the latter also used as a consolidant. Photographs were taken with a Nikon D700 DSLR camera and a 105 mm micro lens. Most views show the specimen coated with sublimed ammonium chloride, with lighting from the upper left.
No permits were required for the described study, which complied with all relevant New Zealand regulations.
Acronyms
AMNH - American Museum of Natural History, New York, USA; OU - Geology Museum, University of Otago, Dunedin, New Zealand; USNM - Department of Paleobiology, National Museum of Natural History, Smithsonian Institution, Washington, DC, USA. All these institutions are accessible, permanent repositories.
Geological Setting
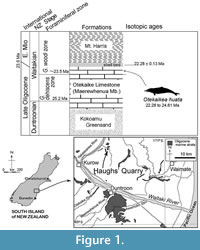 The fossil dolphin OU 22306 was collected in a single plaster jacket from the upper part of the Otekaike Limestone at the Hakataramea Lime Quarry, informally known as Haughs’ Quarry, in the Hakataramea Valley, South Canterbury, around 16 km northeast of the nearest town of Kurow, North Otago (Figure 1). Latitude 44°39'39.82"S, longitude 170°38'57.22"E. The Geosciences Society of New Zealand fossil record number is I40/f0219B.
The fossil dolphin OU 22306 was collected in a single plaster jacket from the upper part of the Otekaike Limestone at the Hakataramea Lime Quarry, informally known as Haughs’ Quarry, in the Hakataramea Valley, South Canterbury, around 16 km northeast of the nearest town of Kurow, North Otago (Figure 1). Latitude 44°39'39.82"S, longitude 170°38'57.22"E. The Geosciences Society of New Zealand fossil record number is I40/f0219B.
The sequence at Haughs’ Quarry includes three formations, from base to top: the Kokoamu Greensand, the Otekaike Limestone, and the Mt. Harris Formation. The Kokoamu Greensand (Gage, 1957) is a bioturbated massive (lower, and top) to diffusely dm-bedded (middle) fossiliferous calcareous greensand. The Kokoamu Greensand grades up into the Otekaike Limestone (Gage, 1957), which is a massive, fossiliferous, mostly soft grainstone, yellow-brown bioclastic limestone with fine glaucony grains and sometimes-abundant shell fragments. In the Waitaki Valley region in general, there are three members of Otekaike Limestone, from bottom to top: Maerewhenua Member (soft to cemented massive, glauconitic limestone), Miller Member (yellow or brown calcareous silt or bioclastic sandstone, sometimes with large low-angle cross-beds), and Waitoura Marl (massive, calcareous silt) (Gage, 1957; Ayress, 1993; Graham et al., 2000). At Hakataramea Lime Quarry, only the Maerewhenua Member is present. A diffuse shellbed with conspicuous Protula, about 20 cm thick, lies in upper part of the Maerewhenua Member, 2-3 m below the gradational boundary with the more-glauconitic and muddy Mt. Harris Formation.
The matrix of OU 22306 is yellow shelly glauconitic bioclastic silty-muddy sandstone with occasional calcareous concretions. The horizon is a few cm below the diffusely-bounded Protula -dominated shellbed in the upper Maerewhenua Member. A strontium isotope (87 Sr/86 Sr) determination from a smooth-shelled scallop Lentipecten hochstetteri, recovered by Fordyce from the top of the Protula shellbed, is 0.708294 (±2SE 0.000013) (D.A. Teagle, personal commun., 2005), equivalent to 22.28 ± 0.13 (22.41 to 22.15) Ma (lookup table of McArthur et al., 2012). Given that Vandenberghe et al. (2012) cited the Chattian-Aquitanian boundary as 23.03 ± 0.1 Ma, the Sr/Sr date puts the upper limit for O. huata in the lowermost Aquitanian, earliest Miocene.
Foraminifera in matrix taken from the fossil dolphin braincase include rare small specimens of the planktic foraminiferan Globoturborotalita woodi, indicating the G. woodi woodi planktic foraminiferal zone of Jenkins (1965), in the middle of the New Zealand Waitakian stage. The next younger zonal species, Globoturborotalita connecta, was not found. For the Otiake Trig Z section 18 km to the south southwest, Graham, Morgans et al. (2000) identified the incoming of G. woodi woodi in the range 23.99 to 24.61 Ma, earlier than the Chattian-Aquitanian boundary. Thus, the age from strontium isotope and planktic foraminifera is in the range 22.28 to 24.61 Ma, straddling the Oligocene/Miocene boundary.
The locality is an important vertebrate fossil source in New Zealand (Fordyce and Maxwell, 2003; Fordyce, 2014). Two Waitakian penguins (Fordyce and Jones, 1990) and a Duntroonian billfish (Gottfried et al., 2012) from the Otekaike Limestone have been reported from the quarry. Several undescribed mysticetes also are known (Fordyce, 2009). The paleoenvironment is probably below storm wave base, in a sheltered middle shelf setting, with warm paleotemperature indicated by bivalve taxa (Fordyce and Maxwell, 2003). Rich mollusc-dominated invertebrate assemblages are also known from the locality.
SYSTEMATIC PALEONTOLOGY
Order CETACEA Brisson, 1762
Unranked taxon NEOCETI Fordyce and de Muizon, 2001
Suborder ODONTOCETI Flower, 1867
Superfamily PLATANISTOIDEA Gray, 1863
Family not specified
Genus OTEKAIKEA Tanaka and Fordyce, 2014
Type species. Prosqualodon marplesi Dickson, 1964 = Otekaikea marplesi (Dickson, 1964) sensu Tanaka and Fordyce (2014).
Included species. O. marplesi (Dickson, 1964) and O. huata n. sp.
Diagnosis. Genus Otekaikea includes longirostral odontocetes with: condylobasal length around 790+ mm; procumbent tusks; presence of an intra-premaxillary foramina in each premaxilla; large and rounded carotid foramen; and a deep voluminous facial fossa. Otekaikea has the following synapomorphies: ventromedial edge of the internal opening of infraorbital foramen is formed by both the maxilla and palatine; both nasals have a point on the midline and gap with the premaxillae; nasals are relatively narrow; and accessory ossicle is fused on the periotic. Otekaikea differs from Waipatia in skull features that include: rostrum is relatively longer; premaxillary sac fossa is more elevated; posterolateral plate and posteromedial splint of the premaxilla are more distinct, with an infra-premaxillary foramen on the right or bilaterally; vertex is less symmetrical, with nasals subrhomboidal rather than transversely oval; ascending process of the maxilla is less-rounded; frontal on the vertex is anteroposteriorly shorter; postglenoid process is tapered rather than squared in lateral view; zygomatic process is more robust in ventral view; and paroccipital process has a medially-developed tongue-like articulation for the stylohyal. The teeth are near-homodont, and the cheekteeth mostly lack denticles; the mandibles have a fused symphysis. The periotic differs from Waipatia in that: anterior keel is less distinct; accessory ossicle is fused; a less-strongly projecting lateral tuberosity; a wider and more-circular internal acoustic meatus; posterior process is longer and subrectangular rather than elongate comma-shaped, and angled relative to the axis of the periotic from ventral or dorsal views. The posterior process of the bulla has less prominent sutures for the periotic, posterior meatal ridge, and post-tympanic process.
Otekaikea differs from Squalodontidae and Prosqualodon in lacking large robust heterodont cheekteeth. Differs from Notocetus , Squalodelphis, Huaridelphis, and Platanistidae in lacking: orbital fossae for extensions of pterygoid sinuses; thickened maxilla or elevated maxillary crest over orbit.
Otekaikea differs from other archaic Odontoceti including Xenorophidae, Simocetus, Agorophius, Patriocetus, Prosqualodon, Squalodontidae, and Waipatia, in the unique combination of apomorphies involving: broad dished face; nasals elevated, nodular, and subrhomboidal; frontals elevated; premaxillary sac fossae smooth-surfaced, without prominent premaxillary sulci developed posteriorly; premaxillae bifurcated posteriorly, each with an intra-premaxillary foramin at the junction between posterolateral plate and posteromedial splint, and elevated crests on the maxillae; periotic with long, slender, parallel-sided posterior process. Otekaikea differs from crown odontocetes other than Platanistoidea in lacking: the supracranial basin of Physeteriidae and Kogiidae; an enlarged hamular fossa of Ziphiidae; medially located aperture for the cochlear aqueduct of Eurhinodelphinidae; and the orbital fossae for extensions of pterygoid sinuses, parabullary ridge of periotic, and saddle-shaped involucrum of the bulla of Delphinida.
Comment on higher taxonomy. Gray (1863) was the first to use the family-group name Platanistidae. The content of the family varied in later years, to include species of Platanista, and also Inia and Pontoporia, and finally Lipotes. Simpson (1945) used a new rank in the family group, Superfamily Platanistoidea, to encompass a single family, Platanistidae Gray, 1863. He justified the superfamily rank in general terms with the comment “The number of genera and families [amongst Odontoceti] has become so large that I have introduced superfamilies for the association of families that appear to be specially related” (Simpson, 1945, p. 215). According to the International Code of Zoological Nomenclature (1999), Article 50.3, Gray (1863) remains the author. This authorship for Platanistoidea follows Fordyce and de Muizon (2001).
Otekaikea huata, new species
Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, Figure 9, Figure 10, Figure 11, Figure 12, Figure 13, Figure 14, Figure 15, Figure 16, Figure 17, Figure 18, Figure 19, Figure 20, Table 1, Table 2, Table 3
zoobank.org/CFD7D54E-00B2-40F1-830E-E60644595463
 Diagnosis. Otekaikea huata is an odontocete with: a skull of medium size (bizygomatic width 265 mm, cranial length 222+ mm, likely condylobasal length 790+ mm); apical teeth procumbent with a large tusk (197+ mm in length, not including broken crown); more-posterior teeth near-homodont and polydont, with subconical crowns and at least one tooth with vestigial denticles; supraoccipital depressed; condyles prominent; zygomatic processes robust; and cervical vertebrae unfused. Otekaikea huata is slightly larger than the holotype of O. marplesi in terms of bizygomatic width (8 mm larger), maximum anteroposterior length of the periotic parallel to dorsal margin (7 mm larger) and also all measurements of preserved postcranial elements (each several mm larger). The one cheek-tooth known for O. marplesi has double roots, whereas the cheekteeth in O. huata are all single-rooted, including at least one with a vestigial longitudinal groove as if separating two fused roots. Otekaikea huata also differs from O. marplesi in: ascending process of the premaxilla is reduced, stopping at the level of the nasal (character 58); lateral expansion of the maxillae roofs the temporal fossa (character 101); nasal has an anterolateral process; fenestra rotunda of the periotic is very narrow and slit-like (character 174); caudal tympanic process of the periotic is posteroventrally projecting (character 178); and a prominent fold separates the infraspinous fossa and teres fossa on the scapula (character 236).
Diagnosis. Otekaikea huata is an odontocete with: a skull of medium size (bizygomatic width 265 mm, cranial length 222+ mm, likely condylobasal length 790+ mm); apical teeth procumbent with a large tusk (197+ mm in length, not including broken crown); more-posterior teeth near-homodont and polydont, with subconical crowns and at least one tooth with vestigial denticles; supraoccipital depressed; condyles prominent; zygomatic processes robust; and cervical vertebrae unfused. Otekaikea huata is slightly larger than the holotype of O. marplesi in terms of bizygomatic width (8 mm larger), maximum anteroposterior length of the periotic parallel to dorsal margin (7 mm larger) and also all measurements of preserved postcranial elements (each several mm larger). The one cheek-tooth known for O. marplesi has double roots, whereas the cheekteeth in O. huata are all single-rooted, including at least one with a vestigial longitudinal groove as if separating two fused roots. Otekaikea huata also differs from O. marplesi in: ascending process of the premaxilla is reduced, stopping at the level of the nasal (character 58); lateral expansion of the maxillae roofs the temporal fossa (character 101); nasal has an anterolateral process; fenestra rotunda of the periotic is very narrow and slit-like (character 174); caudal tympanic process of the periotic is posteroventrally projecting (character 178); and a prominent fold separates the infraspinous fossa and teres fossa on the scapula (character 236).
Etymology. From the Maori language: huata, a spear, alluding to the long tusk of the holotype specimen.
 Material. Holotype only: OU 22306; the skull, right periotic and sigmoid process of left bulla, fused mandibles, 60 isolated conical-crowned teeth, atlas, axis, three other cervical vertebrae, 12 thoracic vertebrae, two lumbar vertebrae, eight ribs, scapulae, humeri, radii, an ulna, a carpal and nine digits. Collected by R.E. Fordyce, A. Grebneff, B.V.N. Black, and O. Sarll, 15 January and 12-13 February 1996; field number REF12-2-96-2.
Material. Holotype only: OU 22306; the skull, right periotic and sigmoid process of left bulla, fused mandibles, 60 isolated conical-crowned teeth, atlas, axis, three other cervical vertebrae, 12 thoracic vertebrae, two lumbar vertebrae, eight ribs, scapulae, humeri, radii, an ulna, a carpal and nine digits. Collected by R.E. Fordyce, A. Grebneff, B.V.N. Black, and O. Sarll, 15 January and 12-13 February 1996; field number REF12-2-96-2.
Type locality. Informally-named Haughs’ Quarry, Hakataramea Valley, South Canterbury, New Zealand (Figure 1). See details above.
Horizon. A few centimeters below the diffuse Protula shellbed in the upper part of the Maerewhenua Member, Otekaike Limestone (Figure 1). See details above.
Age. Waitakian stage, Oligo/Miocene boundary, in the range 22.28 to 24.61 Ma. See details above.
General Description
 Descriptions are based on the right or left side, whichever is more informative, with differences between them mentioned only if directional asymmetry is evident. Morphological terms follow Mead and Fordyce (2009) for the skull; postcranial terms mainly follow Flower (1885a), and Evans and de Lahunta (2013). Surface detail is lost in places from bioerosion, as noted when major. Some adherent concretionary carbonate remains.
Descriptions are based on the right or left side, whichever is more informative, with differences between them mentioned only if directional asymmetry is evident. Morphological terms follow Mead and Fordyce (2009) for the skull; postcranial terms mainly follow Flower (1885a), and Evans and de Lahunta (2013). Surface detail is lost in places from bioerosion, as noted when major. Some adherent concretionary carbonate remains.
Body size. The body size of Otekaikea huata can be inferred by the Pyenson and Sponberg (2011) formula for stem Platanistoidea, Log(L)=0.92* (log(BIZYG)-1.51)+2.49. BIZYG (bizygomatic width) of O. huata is 26.5 cm, giving a reconstructed body length of 2.57 m. As with O. marplesi, this is a similar body length to modern Platanista gangetica: 2.6 m in adult female, 2.2 m in adult male (Jefferson et al., 2008). The gender of the type specimen of O. huata is uncertain. Closely related fossil species have comparable sizes: W. maerewhenua, bizygomatic width 24.4 cm (= length 2.39 m); O. marplesi, bizygomatic width 25.7 cm (= length around 2.5 m (Tanaka and Fordyce, 2014). For O. huata, the minimum condylobasal length of 79.0+ cm would comprise around 30 % of the BIZYG-predicted body size of 2.57 m, implying a long rostrum and large cranium with a short trunk. Not enough postcranial material is known to provide independent evidence of body length.
 Ontogenetic age. Skull sutures are mostly closed but distinct. All preserved vertebral epiphyses (in the atlas, axis, three cervical, 12 thoracic, and two lumbar vertebrae) are fused. These features suggest that O. huata is equivalent in ontogenetic age to stage VI, physically mature adult, in Stenella attenuata as proposed by Perrin (1975, p. 42). The skull lacks obvious roughened or eroded bone that might indicate a gerontic age. Otekaikea huata also has an ossified carpus, which supports an adult age class based on a flipper radiograph study on Phocoena sinus by Mellor et al. (2009).
Ontogenetic age. Skull sutures are mostly closed but distinct. All preserved vertebral epiphyses (in the atlas, axis, three cervical, 12 thoracic, and two lumbar vertebrae) are fused. These features suggest that O. huata is equivalent in ontogenetic age to stage VI, physically mature adult, in Stenella attenuata as proposed by Perrin (1975, p. 42). The skull lacks obvious roughened or eroded bone that might indicate a gerontic age. Otekaikea huata also has an ossified carpus, which supports an adult age class based on a flipper radiograph study on Phocoena sinus by Mellor et al. (2009).
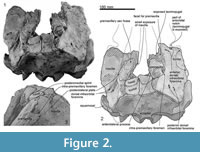
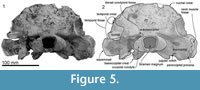
Skull topography. The cranium is moderately complete dorsally (Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, Figure 9), while the separated rostrum is broken and represented by dorsal parts (porcelanous portions) of the isolated right and left premaxillae, the posterior end of the right maxilla, and anterior of the vomer with the attached posterior end of the left maxilla (Figure 7). The separated rostral elements can be placed close to original articulation with the cranium, although minor burial-related distortion prevents exact matches that would allow full reassembly. The incomplete rostral left maxilla, which is sutured with the vomer and can be articulated with the cranium, preserves an elongate, flat, medially-dipping suture that matches a corresponding suture on the ventral surface of the premaxilla; a comparable suture is on the right. 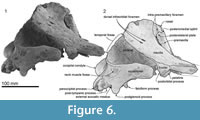 Both premaxillae (porcelanous parts) can be articulated with the flat sutures on the maxillae to indicate approximate life position on the rostrum (below). To reconstruct the skull, the right premaxilla was positioned to match the left; there was no independent evidence of bilateral asymmetry in the rostral sections of premaxilla. The face has a wide hexagonal shape in dorsal view, is flat anteriorly and rises steeply posteriorly. Compression slightly distorts the skull; additionally, the vertex is naturally skewed to left, as seen clearly in a posterior view, which shows the right nasal and frontal in the midline. Natural directional asymmetry affects the nasals, frontals, premaxillae, and maxillae. In lateral view (Figure 6 and Figure 8), the posterior part of the face has very steep medial and posterior walls involving both maxilla and premaxilla, and these walls form a strongly elevated but anteroposteriorly short vertex comprised of the maxillae, premaxillae, nasals, and frontals. The temporal fossa is mostly invisible in dorsal view because of the laterally expanded temporal crest. The orbit is long and weakly arched, with relatively thin antorbital and postorbital processes, above which the frontal is almost fully overlapped by the maxilla. The anteriorly broken external nares open from a subvertical narial passage about level with the postorbital processes. The nuchal crest is not developed. The pterygoids and most of the palatines are missing.
Both premaxillae (porcelanous parts) can be articulated with the flat sutures on the maxillae to indicate approximate life position on the rostrum (below). To reconstruct the skull, the right premaxilla was positioned to match the left; there was no independent evidence of bilateral asymmetry in the rostral sections of premaxilla. The face has a wide hexagonal shape in dorsal view, is flat anteriorly and rises steeply posteriorly. Compression slightly distorts the skull; additionally, the vertex is naturally skewed to left, as seen clearly in a posterior view, which shows the right nasal and frontal in the midline. Natural directional asymmetry affects the nasals, frontals, premaxillae, and maxillae. In lateral view (Figure 6 and Figure 8), the posterior part of the face has very steep medial and posterior walls involving both maxilla and premaxilla, and these walls form a strongly elevated but anteroposteriorly short vertex comprised of the maxillae, premaxillae, nasals, and frontals. The temporal fossa is mostly invisible in dorsal view because of the laterally expanded temporal crest. The orbit is long and weakly arched, with relatively thin antorbital and postorbital processes, above which the frontal is almost fully overlapped by the maxilla. The anteriorly broken external nares open from a subvertical narial passage about level with the postorbital processes. The nuchal crest is not developed. The pterygoids and most of the palatines are missing.
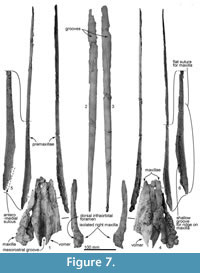 Rostrum. Most of the anterior part of the rostrum, comprising the maxillae and ventral parts of the premaxillae, is not preserved. The long, straight dorsal remnants of premaxillae are reconstructed based on posterior connections with the maxillae (see below). This reconstruction is consistent with a narrow, parallel-sided and deep mesorostral groove, while the broken margins of the mandible suggest a narrow elongate Y-shaped lower jaw and, by implication, a narrow rostrum. The thick posterior part of the vomer (Figure 7) sits medially, with sutured remnants of the maxillae indicating a posteriorly wide rostrum. Bone is damaged at the right antorbital notch, but the profiles of the lacrimojugal and adjacent frontal suggest an open notch with a little-projecting lateral antorbital process.
Rostrum. Most of the anterior part of the rostrum, comprising the maxillae and ventral parts of the premaxillae, is not preserved. The long, straight dorsal remnants of premaxillae are reconstructed based on posterior connections with the maxillae (see below). This reconstruction is consistent with a narrow, parallel-sided and deep mesorostral groove, while the broken margins of the mandible suggest a narrow elongate Y-shaped lower jaw and, by implication, a narrow rostrum. The thick posterior part of the vomer (Figure 7) sits medially, with sutured remnants of the maxillae indicating a posteriorly wide rostrum. Bone is damaged at the right antorbital notch, but the profiles of the lacrimojugal and adjacent frontal suggest an open notch with a little-projecting lateral antorbital process.
The rostral porcelanous part of each premaxilla is incomplete posteriorly; this is interpreted as postmortem break when the cranium and rostrum separated before final burial. The elongate oblique suture on the maxilla is consistent with the premaxilla passing back to merge with the cranial parts of the premaxillary sac fossae that rise steeply to the vertex. Surficial structures include the rounded dorsal surface of the premaxilla, the remant of a shallow premaxillary sulcus at the posterior end of the rostrum, and a steep-walled mesorostral groove (see Figure 8).
Premaxilla. The rostral and cranial parts of the premaxillae are present (Figure 7), but separated post-mortem. Each bone is represented by the long, thin, posteriorly-narrowing, faintly bowed (barely concave laterally) porcelanous part of the rostral surface, each found separate from the cranium; more-ventral rostral parts of the premaxilla are lost. In life, the straight medial margin of each premaxilla would have partly roofed the mesorostral groove, converging toward each other anteriorly, as reconstructed in Figure 8. The left premaxilla is distorted slightly outwards and down toward the rostral apex, probably a result of burial. Both ends are incomplete for each premaxilla, and bone is missing between the posterior of the rostral part and the premaxillary sac fossa on the cranium. Anteriorly, the rounded external surface on the rostral left premaxilla (511.5+ mm long) rises steeply, facing obliquely outwards. Posteriorly, the external surface becomes narrower and more abruptly rounded, and rotates gradually to face obliquely medially at the posterior apex of the porcelanous part. The posterior ~200 mm of the ventral surface of premaxilla is smooth at the suture for the underlying flat dorsal face of the maxilla; this flat suture on the maxilla is horizontal anteriorly on the rostrum but posteriorly it rotates to become steeply oblique by the level of the antorbital notch, facing medially and slightly dorsally. Here, each premaxilla narrows posteriorly, and is deflected slightly outwards. A broken narrow medial shelf on the left premaxilla, barely visible in dorsal view, is presumed to be part of the anteromedial sulcus, originally arising further posteriorly from the now-missing premaxillary foramen.
 Posteriorly, each premaxilla thickens, widens, and rises upward. In dorsal view, the lateral edge of the premaxillae is widest (125.1 mm) at the level of the nares. In spite of some compaction crushing, there is some directional asymmetry in the cranial parts of the premaxillae: 1, the right premaxilla is wider, lies further toward the midline, with a more-laterally flanged posterolateral plate than the left; 2, the right intra-premaxillary foramen, which opens dorsally between the maxilla and the posterolateral plate, is larger (elliptical; 10.6 mm long, 4.8 mm wide), and the lip of the foramen is formed mostly by the premaxilla, with maxilla contributing posteromedially. The left intra-premaxillary foramen opens almost the same level with the right, and it is smaller (also elliptical; 3.0 mm long, 1.2 mm wide; occluded by cemented matrix) and formed by the premaxilla; 3, the blunt right posteromedial splint reaches half way back along the nasal, bounded laterally by a slightly raised crest and vertical face of maxilla, whereas the left splint appears not to reach the nasofrontal suture and the adjacent maxilla is lower and more-nodular. The intra-premaxillary foramen is probably one of many foramina from the infraorbital complex, as discussed below.
Posteriorly, each premaxilla thickens, widens, and rises upward. In dorsal view, the lateral edge of the premaxillae is widest (125.1 mm) at the level of the nares. In spite of some compaction crushing, there is some directional asymmetry in the cranial parts of the premaxillae: 1, the right premaxilla is wider, lies further toward the midline, with a more-laterally flanged posterolateral plate than the left; 2, the right intra-premaxillary foramen, which opens dorsally between the maxilla and the posterolateral plate, is larger (elliptical; 10.6 mm long, 4.8 mm wide), and the lip of the foramen is formed mostly by the premaxilla, with maxilla contributing posteromedially. The left intra-premaxillary foramen opens almost the same level with the right, and it is smaller (also elliptical; 3.0 mm long, 1.2 mm wide; occluded by cemented matrix) and formed by the premaxilla; 3, the blunt right posteromedial splint reaches half way back along the nasal, bounded laterally by a slightly raised crest and vertical face of maxilla, whereas the left splint appears not to reach the nasofrontal suture and the adjacent maxilla is lower and more-nodular. The intra-premaxillary foramen is probably one of many foramina from the infraorbital complex, as discussed below.
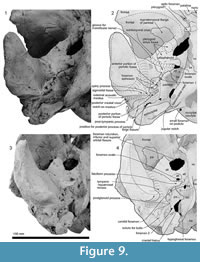 Maxilla. The rostral part of the left maxilla is firmly sutured with the vomer (possibly cemented by matrix); the two bones can be placed close to their original positions against the cranium, with the maxilla close to the antorbital notch. The rostral part of the right maxilla, not attached to the vomer (but originally sutured), can be mirrored against the left. Dorsally, each maxilla has an elongate planar suture dipping obliquely ventromedially, formed by dense rather than cancellous bone, for the premaxillae (Figure 7 and Figure 8); the sutural face can be traced back onto the cranium. A small (2.6 mm diameter) dorsal infraorbital foramen opens bilaterally, facing outwards, toward the posterior of the rostral maxilla. Ventrally, the maxillae form a medially arched palate with flattened lateral margins at the base of the rostrum.
Maxilla. The rostral part of the left maxilla is firmly sutured with the vomer (possibly cemented by matrix); the two bones can be placed close to their original positions against the cranium, with the maxilla close to the antorbital notch. The rostral part of the right maxilla, not attached to the vomer (but originally sutured), can be mirrored against the left. Dorsally, each maxilla has an elongate planar suture dipping obliquely ventromedially, formed by dense rather than cancellous bone, for the premaxillae (Figure 7 and Figure 8); the sutural face can be traced back onto the cranium. A small (2.6 mm diameter) dorsal infraorbital foramen opens bilaterally, facing outwards, toward the posterior of the rostral maxilla. Ventrally, the maxillae form a medially arched palate with flattened lateral margins at the base of the rostrum.
Posteriorly, the cranial part of the maxilla is overlaid by the premaxilla, with clear sutures with the frontal and lacrimal. In places (e.g., left near the position of the maxillary-premaxillary suture), the maxillary surface is dense and porcelanous; adjacent irregular cancellous surface is interpreted as (bio) eroded. About 25 mm medial to the right antorbital notch, the maxilla has an anteroposteriorly long, wide and smooth trough, presumably partly for the premaxilla, extending ~15 mm anterior and ~40 mm posterior to the level of the antorbital notch (Figure 2.2, Figure 7.4). This trough has a steep lateral face that passes sharply at an elongate fold onto the flat supraorbital process of the maxilla.
On the braincase, dorsally, the cranial part of the maxilla covers most of the frontal except the lateral part of the postorbital process (right side; Figure 2). The maxilla on the supraorbital process is flat and smooth; it is not obviously thickened and it lacks a pneumatic maxillary crest. At the level of the right lacrimojugal, behind the antorbital notch, a small anterior dorsal infraorbital foramen (1.4 mm diameter) opens into a short oblique sulcus (Figure 2.2). The region is damaged on the left. The posteromedial surface of the maxilla is very steep; at the level of the nasal/frontal border, two posterior dorsal infraorbital foramina open on each side, which are small and rounded (right side diameters: anterior, 3.4 mm; posterior, 4.6 mm), opening into sulci that run toward the posterolateral edge of the maxilla. The squared apex of each maxilla meets the frontal medially and the supraoccipital posteriorly. Just lateral to the nasal and frontal, the medial border of the weakly curved crest on the right maxilla rises abruptly. At the anterior end of the nares, a small area of the maxilla is exposed medially; whether this was originally covered by now-lost premaxilla, or exposed to form a maxillary intrusion (sensu Arnold and Heinsohn, 1996) is uncertain.
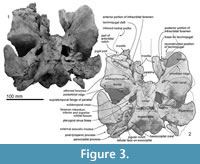
 In ventral view (Figure 3), the better-preserved right maxilla forms the borders for two infraorbital foramina: a larger and more posteromedial foramen in the common position for odontocetes anterior to the antorbital ridge, and a smaller foramen a little anterolateral. The larger posterior foramen is bounded by frontal, palatine and maxilla, and opens anteriorly into a sulcus in the maxilla. The smaller anterior foramen is bounded mainly by maxilla and lacrimojugal, plus a small contribution of frontal; the lacrimojugal cleft is immediately lateral. It is likely that this smaller foramen was originally covered by maxilla that was lost postmortem, with the sulcus representing the infraorbital canal. On the type of Otekaikea marplesi, the single visible ventral infraorbital foramen is equivalent to the posterior foramen in O. huata.
In ventral view (Figure 3), the better-preserved right maxilla forms the borders for two infraorbital foramina: a larger and more posteromedial foramen in the common position for odontocetes anterior to the antorbital ridge, and a smaller foramen a little anterolateral. The larger posterior foramen is bounded by frontal, palatine and maxilla, and opens anteriorly into a sulcus in the maxilla. The smaller anterior foramen is bounded mainly by maxilla and lacrimojugal, plus a small contribution of frontal; the lacrimojugal cleft is immediately lateral. It is likely that this smaller foramen was originally covered by maxilla that was lost postmortem, with the sulcus representing the infraorbital canal. On the type of Otekaikea marplesi, the single visible ventral infraorbital foramen is equivalent to the posterior foramen in O. huata.
Palatine. Ventrally, a vertical plate-like robust fragment of palatine underlies the maxilla (Figure 7), slightly posteromedial to the infraorbital foramen, and connected with the frontal posteriorly. The palatine has a smooth lateral wall and, medially, a grooved vertical surface that contributes to the lateral wall of the nares.
Pterygoid. A fragment of pterygoid is lateral to the posterior end of the vomer, posterior to the nares. Otherwise the bone is lost. A facet for the medial lamina of the pterygoid covers the anterior two thirds of the basioccipital crest.
 Nasal. The smooth, flat, asymmetrical nasals lie just behind, rather than roofing, the nares (Figure 2). In anterior view, the nasals are dorsoventrally thick and nodular. The smaller left nasal is sub-oblong, transversely wide and anteroposteriorly narrow; the lateral and posterior margins are rounded, the anterior, straight. The sagittally-placed larger right nasal is a rounded irregular triangle, transversely wide and anteroposteriorly longer than the left, and projected forward more than the left. Both have a prominently rounded medial corner and a semicircular posterior margin. The anterior edge of each nasal lies far posterior to the antorbital process, almost level with the posterior end of the postorbital process. The anteromedial angle projects strongly, while the posteromedial angle is displaced by the narial process of the frontal. Each nasal has a plate-like anterolateral process of the nasal [new term] laterally, ~ 20 mm long and ~4 mm wide, projecting forward to meet the posteromedial margin of the premaxilla, and separated from the main part of the nasal (anterior view) by a deep groove; the anterior margins of the two nasals form a W-shape. A slightly similar condition of the groove on the anterior surface of the nasal is seen on some kentriodontids (Pithanodelphis cornutus Abel, 1905, Delphinodon dividum True, 1912 and Kentriodon pernix Kellogg, 1927), but the grooves are much shallower and the anterolateral process less marked than in Otekaikea huata . An undescribed platanistoid OU 22126, from the Otekaike Limestone of Waitaki district (Fordyce, 2003), shows an anteroposteriorly thin nasal without a groove on the anterior surface, but the platelike anterolateral process of the nasal projects well anteriorly. The internarial, nasofrontal, nasopremaxillary, and nasomaxillary sutures are deep and wide. The internarial suture is invaded by narrow fingers of frontal, forming the narial process. In anterior view, the nasal is medially very thick but laterally thin, resulting in a triangular profile.
Nasal. The smooth, flat, asymmetrical nasals lie just behind, rather than roofing, the nares (Figure 2). In anterior view, the nasals are dorsoventrally thick and nodular. The smaller left nasal is sub-oblong, transversely wide and anteroposteriorly narrow; the lateral and posterior margins are rounded, the anterior, straight. The sagittally-placed larger right nasal is a rounded irregular triangle, transversely wide and anteroposteriorly longer than the left, and projected forward more than the left. Both have a prominently rounded medial corner and a semicircular posterior margin. The anterior edge of each nasal lies far posterior to the antorbital process, almost level with the posterior end of the postorbital process. The anteromedial angle projects strongly, while the posteromedial angle is displaced by the narial process of the frontal. Each nasal has a plate-like anterolateral process of the nasal [new term] laterally, ~ 20 mm long and ~4 mm wide, projecting forward to meet the posteromedial margin of the premaxilla, and separated from the main part of the nasal (anterior view) by a deep groove; the anterior margins of the two nasals form a W-shape. A slightly similar condition of the groove on the anterior surface of the nasal is seen on some kentriodontids (Pithanodelphis cornutus Abel, 1905, Delphinodon dividum True, 1912 and Kentriodon pernix Kellogg, 1927), but the grooves are much shallower and the anterolateral process less marked than in Otekaikea huata . An undescribed platanistoid OU 22126, from the Otekaike Limestone of Waitaki district (Fordyce, 2003), shows an anteroposteriorly thin nasal without a groove on the anterior surface, but the platelike anterolateral process of the nasal projects well anteriorly. The internarial, nasofrontal, nasopremaxillary, and nasomaxillary sutures are deep and wide. The internarial suture is invaded by narrow fingers of frontal, forming the narial process. In anterior view, the nasal is medially very thick but laterally thin, resulting in a triangular profile.
Ethmoid. The ethmoid is used in the sense of Mead and Fordyce (2009), but note that Ichishima (2011) suggested that the mesethmoid might be absent in odontocetes. Morphological terms of the ethmoid follow Godfrey (2013).
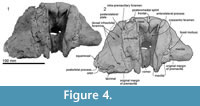
 The profile of the ethmoid is uncertain but in the mesethmoid ridge (area for attachment of the mesorostral cartilage) of the vomer, there is a spongy bone with a foramen at the center (Figure 4). The large void dorsal to the foramen may be for the missing or undeveloped area of the cribriform plate of the mesethmoid. On the posterodorsal part, the cribriform plate has a pair of large and strongly curved crescentic foramina. Between the foramina, there is a tubercle, which is assumed to be the mesethmoid.
The profile of the ethmoid is uncertain but in the mesethmoid ridge (area for attachment of the mesorostral cartilage) of the vomer, there is a spongy bone with a foramen at the center (Figure 4). The large void dorsal to the foramen may be for the missing or undeveloped area of the cribriform plate of the mesethmoid. On the posterodorsal part, the cribriform plate has a pair of large and strongly curved crescentic foramina. Between the foramina, there is a tubercle, which is assumed to be the mesethmoid.
Vomer. The robust rostral part of the vomer is broken and isolated from the cranium (Figure 7). The mesorostral groove is anteriorly shallow and U-shaped, and posteriorly deep and more V-shaped. Its lateral wall is unexpectedly thick, thinning ventrally; the greatest bilateral width is 75.4 mm, with one side attaining a thickness of 27.6 mm, and the depth is around 30 mm. From the widest point, the vomer narrows rapidly, both anteriorly and posteriorly. Both ventrally and laterally, the vomer is covered by the maxilla cemented by matrix; the sutures are not fused. In ventral view (Figure 7 and Figure 8), the vomer carries two flat areas, which might be sutures for the maxilla (this feature occurs in an unfused young individual of Cephalorhynchus hectori and in Otekaikea marplesi). Vomer is present below the olfactory region, as shown in broken section in anterior view. Posteriorly, the vomer covers the basisphenoid.
Lacrimojugal. The right lacrimal and jugal are fused without evident suture. There is no obvious lacrimal foramen or groove. A transversely long (36.3+ mm), thin (6.8 mm), and bar-like anterior margin of lacrimojugal contributes to the right antorbital notch (Figure 3.2), and is separated from the rest of the bone by a lacrimojugal cleft. There is no evident base for the styloid process of the jugal. Medially, the anterior margin enlarges to become subspherical, sutured with the maxilla in a fossa (seen ventrally on the left maxilla) close to a prominent anterior infraorbital foramen. The lacrimojugal does not, however, reach the larger more-posterior infraorbital foramen. More posteriorly, the lacrimojugal is plate-like, as also implied by the slightly ridged planar suture for the missing lacrimojugal under the left antorbital process of the frontal.
 Frontal. Each frontal contributes a large surface to the orbit and a small exposure at the vertex (Figure 2). At the vertex, the frontals are bounded by the nasals, maxillae, and supraoccipital; there is a clear but irregular median interfrontal suture, not to be confused with a mollusc shell imprint (limpet homing mark?) on the right nasal. Each frontal is trapezoidal and has a long finger-like medial projection which forms a narial process. The frontals are asymmetrical: wider on the left and longer on the right. Each frontal contacts the parietal laterally. On the face, the left ascending process of the maxilla is lost and shows the profile of the postorbital process; the right process, which is almost covered by maxilla, appears not to project as far as the left, judging from the profile of the bar-like jugal (above).
Frontal. Each frontal contributes a large surface to the orbit and a small exposure at the vertex (Figure 2). At the vertex, the frontals are bounded by the nasals, maxillae, and supraoccipital; there is a clear but irregular median interfrontal suture, not to be confused with a mollusc shell imprint (limpet homing mark?) on the right nasal. Each frontal is trapezoidal and has a long finger-like medial projection which forms a narial process. The frontals are asymmetrical: wider on the left and longer on the right. Each frontal contacts the parietal laterally. On the face, the left ascending process of the maxilla is lost and shows the profile of the postorbital process; the right process, which is almost covered by maxilla, appears not to project as far as the left, judging from the profile of the bar-like jugal (above).
Ventrally, the frontal forms the anterodorsal wall of the braincase and most of the shallow and long orbit (Figure 3). A weak preorbital ridge (Fordyce, 1994) is indistinct at the antorbital process laterally, but becomes elevated and narrow toward the posterior margin of the narial passage. The frontal contributes the posterior and lateral margins of the infraorbital foramen. Medially, an oval presumed optic foramen (3.0 mm diameter) opens anterolaterally. The strongly posteriorly-concave postorbital ridge is sharp, high, and meets the postorbital process, which is a blunt, ventrally-projecting triangular shape as seen in lateral view. Between these ridges, the triangular roof of the orbit is anteroposteriorly long and dorsoventrally shallow. Foramina for the diploic vein are absent.
Orbitosphenoid. The orbitosphenoid (Figure 3) is identified by the presumed ethmoid foramen, which opens 61-62 mm medial to the orbital margin, lateral and anterior relative to the region of the optic foramen. The foramen is surrounded by a smooth bone surface, presumed orbitosphenoid, in the roof of the orbit toward the optic foramen. No sutures are evident.
 Parietal. The parietal forms the lateral wall of the braincase (Figure 6) at the temporal fossa, which is strongly excavated medially and anteroposteriorly shorter than in Archaeoceti and stem Odontoceti, which have dorsally more open temporal fossa. Anteriorly, the parietal has a roughly rectangular thin supratemporal flange [new term], directed obliquely forward to encroach the frontal in the roof of the right temporal fossa. (The flange is lost on the left, but an exposed suture is apparent.) Such a flange is also clear in the undescribed specimen OU 22540 and is uncertainly present (the bone surface is damaged) in Otekaikea marplesi. Within the temporal fossa, the parietosquamosal suture is not clear in O. huata, and there is no obvious postparietal foramen. Parietal forms a ventral exposure between the basioccipital and squamosal, separating the foramen ovale from the cranial hiatus as in Waipatia maerewhenua and O. marplesi. The parietal-alisphenoid suture is clear on the left, between the foramen ovale and the base of the falciform process. "Foramen 1" sensu Fordyce (1994) is seen in O. huata further suggesting the position of parietal (see ventral details of the squamosal, below). The clear parietal-alisphenoid suture has foramen 1 on its junction with the squamosal, but otherwise the relationships of the foramen and sutures are obscure. The parietal contacts "foramen 2" sensu Fordyce (1994) posteriorly and the cranial hiatus medially.
Parietal. The parietal forms the lateral wall of the braincase (Figure 6) at the temporal fossa, which is strongly excavated medially and anteroposteriorly shorter than in Archaeoceti and stem Odontoceti, which have dorsally more open temporal fossa. Anteriorly, the parietal has a roughly rectangular thin supratemporal flange [new term], directed obliquely forward to encroach the frontal in the roof of the right temporal fossa. (The flange is lost on the left, but an exposed suture is apparent.) Such a flange is also clear in the undescribed specimen OU 22540 and is uncertainly present (the bone surface is damaged) in Otekaikea marplesi. Within the temporal fossa, the parietosquamosal suture is not clear in O. huata, and there is no obvious postparietal foramen. Parietal forms a ventral exposure between the basioccipital and squamosal, separating the foramen ovale from the cranial hiatus as in Waipatia maerewhenua and O. marplesi. The parietal-alisphenoid suture is clear on the left, between the foramen ovale and the base of the falciform process. "Foramen 1" sensu Fordyce (1994) is seen in O. huata further suggesting the position of parietal (see ventral details of the squamosal, below). The clear parietal-alisphenoid suture has foramen 1 on its junction with the squamosal, but otherwise the relationships of the foramen and sutures are obscure. The parietal contacts "foramen 2" sensu Fordyce (1994) posteriorly and the cranial hiatus medially.
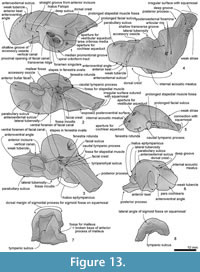
Squamosal. Description is based mainly on the right. In dorsal view (Figure 2), a wide (34.2 mm at the subtemporal crest) and long zygomatic process (90.1 mm) projects slightly laterally, posterior to the orbit. The anterior end is blunt and narrower than the posterior, and reaches to the level of the posterior end of the nasals, still distant from the postorbital process (14.6 mm gap between the processes, possibly widened by postmortem distortion). The temporal fossa is exposed widely in lateral view, is not obvious in dorsal view, and is widely open to posterior view. The temporal fossa has an unclear squamosal-parietal suture on its lateral wall on the right (Figure 6). In lateral view, the anterior end of the squamosal is squared. On the lateral surface, a triangular, depressed neck muscle fossa (34.0 mm long) occupies midpoint of the zygomatic. This depression has been termed sternomastoid fossa in recent literature on fossil Cetacea, but the muscle insertions in extant species are quite variable (Schulte, 1916; Howell, 1927, 1930; Cotten et al., 2008), and the more-general term neck muscle fossa (Fordyce, 1981, p. 1035) makes fewer assumptions about homologies.
Ventrally (Figure 3 and Figure 9), the zygomatic process has a wide and shallow mandibular fossa without an obvious anterior border, a slightly swollen lateral portion, and a strongly curved internal face. The postglenoid process is not thickened anteroposteriorly, nor markedly expanded laterally, and slopes obliquely posteroventrally. The large, prominent tympanosquamosal recess separates the zygomatic process from the falciform process, occupying the squamosal from the subtemporal crest to the anterior meatal crest and spiny process. Posteriorly, the recess descends down the medial margin of the postglenoid process, while posteromedially the recess deepens toward the spiny process. The periotic apposes the falciform process dorsally and anteriorly, separated by a small gap. In the articulated periotic, the position of the lateral tuberosity lies more ventrally than the margin of the falciform process (Figure 11). The apex of the spiny process points forward toward the lateral tuberosity of the periotic, to bridge (pass ventrally to) the path of the middle sinus. An irregular transverse depression lies lateral to the spiny process, immediately in front of the anterior meatal crest and behind a small anterior transverse ridge sensu Fordyce (2002, p. 203); this may include the sigmoid fossa sensu Geisler et al. (2005, p. 16). The long base of the well-developed falciform process arises near the squamosal-alisphenoid suture at the subtemporal crest, and extends to the spiny process. There is no evidence that a lateral lamina was directed forward from the falciform process to bound the pterygoid sinus fossa. The falciform process is skewed medially to underlie the alisphenoid; its ventral tip has a small oblique facet probably for the outer lip of the bulla, while further dorsally the concave posterior face closely parallels the anterior process of the periotic. Laterally, the margin of the falciform process closely matches the anterior process and lateral tuberosity of the periotic; the spiny process fills the hiatus epitympanicus but with the periotic articulated it is apparent that a few mm of the spiny process is missing (there is incomplete contact posteriorly at the hiatus epitympanicus). The walls of the external auditory meatus form a narrow triangle medially, widening a little laterally, where the meatus undercuts the postglenoid process and passes into a groove that rises forward onto the lateral face of the squamosal ventral to the neck muscle fossa. Associated with the latter groove is a small notch at the lateral end of the meatus. The anterior meatal crest is damaged; the posterior crest is arched transversely, with a facet on the posterior face at the suture for the posterior process of the bulla.
The anteroposteriorly long periotic fossa is hidden when the periotic is in situ; it includes anterior and posterior portions that are transversely wide and divided by a strong ridge. In the middle of the anterior portion, the foramen spinosum has two openings, anterior and posterior, which are separated by an elliptical tubercle, as also seen in Otekaikea marplesi. The anterior foramen spinosum is associated with a deep groove (the parietal-squamosal and parietal-alisphenoid sutures), which runs to the foramen ovale. The posterior portion of the periotic fossa is transversely wide (15.9 mm) and deep, and contains the suprameatal pit. When in situ, the periotic sits firmly in a single position in the periotic fossa, with the apex of the posterior process in close contact with the squamosal, medial to the post-tympanic process, but separated a few mm from the exoccipital. A conical space, which opens medially between the periotic and exoccipital, could be for part of the peribullary sinus. The squamosal is fissured, with several foramina, directly above the posterior process of the periotic. The ventrolateral apex of the post-tympanic process lacks obvious grooved sutures for the tip of the posterior process of the tympanic bulla, in contrast to the structure in Waipatia.
Basioccipital. Ventrally (Figure 3), the basioccipital is a trapezoid. Anteriorly, the basisphenoid-basioccipital synchondrosis is fused. Each basioccipital crest gradually widens posteriorly, forms a gently curved ventral profile, and has an indistinct muscular tubercle on the medial surface of the crest. Between the crests, the basioccipital basin is trapezoidal and flat. The lateral border of the basioccipital, which is slightly excavated behind the carotid foramen, contributes to the anterior and medial margins of the cranial hiatus. It is not clear whether the exoccipital contributes to the posterodorsal part of the basioccipital crest medial to the jugular notch.
Supraoccipital and exoccipital. Posteriorly, the supraoccipital is rugose and roughly squared, and constricted by the temporal fossa. The nuchal crest is weakly developed. The dorsal condyloid fossa is deep and wide (60.9 mm) and curves over the condyle. Each occipital condyle has a smooth and small surface, is semicircular in lateral view, is less obviously curved horizontally (dorsal view), is somewhat elliptical in posterior view, and has a very short but distinct pedicle that is slightly narrower than the condyle. The right pedicle has a small foramen (2.7 mm diameter), on the ventral surface. The foramen magnum is a dorsally wider trapezoid, widely open, wider than high (37.9 mm wide and 29.0 mm height), and slightly arched dorsally; the profile may result from dorsoventral crushing. The intercondyloid notch is U-shaped. Laterally, the exoccipital is a thin rectangular plate. There is no distinct posterior sinus fossa here.
In ventral view, the exoccipital extends laterally almost as far as the adjacent post-tympanic process of the squamosal. The ventral surface is a smooth, transversely elongate, narrow, tabular to slightly depressed, tongue-shaped paroccipital process for stylohyal articulation. The rounded tip of the process extends medially to within about 5 mm of the basioccipital crest, and partly underlies the oblique narrow jugular notch (2.9 mm wide, 10.1 mm deep) that opens posterolaterally above the paroccipital process. The small hypoglossal foramen opens in the medial end of the jugular notch.
Alisphenoid. The large alisphenoid forms the posteromedial margin of the subtemporal crest. On the basicranium (Figure 3 and Figure 9), the alisphenoid is posterior to the frontal, anterior to the squamosal and basisphenoid, and lateral to the pharyngeal crest; there are clear sutures with the squamosal and parietal, but the contact with the frontal is broken. A blunt projection of alisphenoid extends back to the base of the falciform process, but does not form a posterolaterally-prolonged wedge. There is no distinct suture with the basisphenoid medially; the latter is indicated by the carotid foramen, but its lateral extent beyond the foramen is uncertain.
Anteriorly, the pterygoid sinus fossa comprises 3 small shallow depressions between the subtemporal crest, the large medially-placed foramen for the optic nerve, foramen rotundum and orbital fissure, and the foramen ovale. The foramen for the optic infundibulum is anteroposteriorly long and elliptical (31.4 mm long and 25.6 mm wide); it is bordered by alisphenoid posteriorly, basisphenoid medially, and frontal (and orbitosphenoid?) anteriorly. Just slightly posterior to the pterygoid sinus, a weak transverse ridge on the alisphenoid marks the groove for the mandibular nerve and associated vessels between the small foramen ovale (8.8 mm wide and 7.6 mm long) and the anterior end of the falciform process. The foramen ovale is complete, formed entirely by alisphenoid, which is apparently sutured with itself posteriorly. The alisphenoid is separated from the cranial hiatus by 25-26 mm of parietal and possibly by basisphenoid. A prominent rod-like projection of alisphenoid underlies the anterior border of the foramen ovale, prolonged toward the basisphenoid.
Basisphenoid. Anteriorly, the basisphenoidal-vomer suture has two foramina, which open posteriorly into anteroposteriorly long shallow grooves (Figure 3). The basisphenoid-basioccipital synchondrosis is fused. Laterally, a large and rounded carotid foramen (5.3 mm diameter) opens, just posteromedial to the foramen ovale. The basisphenoid apparently does not contribute to the margin of the foramen ovale or, anteriorly, to the border of the foramen for the optic infundibulum (the combined openings for the foramen rotundum and infra- and superior orbital fissures).
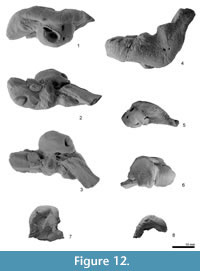
Periotic. The periotic is a structurally complex and phylogenetically informative bone, and deserves detailed description. Figure 11 shows the orientation of the periotic in its original position on the skull. When the periotic is in situ, the ventral foramen of the facial canal can be seen in ventral view, but the fenestra ovalis is partially obscured by the pars cochlearis.
The right periotic (Figure 12 and Figure 13) has a slender anterior process, slightly smaller posterior process, and a dorsoventrally weakly inflated pars cochlearis between these processes. Both the anterior and posterior processes make a wide angle with the anteroposterior axis when the periotic is placed into original position on the skull.
The anterior process is prolonged anteriorly, with a blunt apex. The anterior process is deep and somewhat laterally widened, as seen in Waipatia maerewhenua. Dorsally, a very weak anteroexternal sulcus runs from just anterior to the lateral tuberosity to the anterior end of the dorsal crest. The parabullary sulcus (Tanaka and Fordyce, 2014) is deep and strongly curved (C-shaped). The anteroventral angle, as seen anteriorly, is a small bump just medial to the anterior bullar facet. There is no obvious anterior keel or anterodorsal angle, and this region of the anterior process is smoothly rounded. A small tubercle on the medial surface of the anterior process has a tiny foramen or vertical canal as seen in W. maerewhenua. On the weak tubercle, a straight groove runs from the anterior incisure. The anterior bullar facet is a shallow groove, about 8.5 mm long, with raised parallel-sided margins (3.7 mm width). The fovea epitubaria is occupied by a slightly damaged and moderate sized accessory ossicle, which is preserved between the anterior bullar facet and the mallear fossa. The ossicle has a thin subrectangular flange on its medial side. The flange has a shallow groove on its margin (see Figure 12 and Figure 13.2) and is also widely grooved anteroposteriorly on its dorsal face, possibly forming a sulcus for the tensor tympani muscle. Just posteromedial to the accessory ossicle, there is a tiny foramen. The dorsal surface of the periotic has a prominent dorsal crest, medial to which is a deep anteroposterior groove (9.1 mm), as seen in Otekaikea marplesi but longer and shallower. The anterior incisure is less distinctly grooved than seen in Waipatia maerewhenua and Otekaikea marplesi.
The pars cochlearis is hemispherical ventrally, transversely compressed, and long relative to width. The smooth anteromedial corner passes back into a straight medial margin then an obtuse posteromedial angle. The internal auditory meatus opens on the center medially. A tiny slit-like foramen for the hiatus Fallopii opens on the anterior incisure. The internal auditory meatus is open, posteriorly wider, and rounded tear-shape (maximum length, 8.8 mm). The meatus contains four foramina or groups of foramina: the proximal opening of the facial canal, the foramen singulare, the spiral cribriform tract, and the area cribrosa media. A very low transverse ridge separates the small, narrow proximal opening for the facial canal (2.6 mm) from the foramen singulare, which is also small and narrow. Between the foramen singulare and the spiral cribriform tract, there is a higher crest that bounds a large (anteroposterior length; 5.6 mm), deep and rounded area, which is shared by a cluster of foramina forming the spiral cribriform tract, and a pit for the area cribrosa media. As with some other odontocetes (Mead and Fordyce, 2009), the area cribrosa media is not clearly perforate. The spiral cribriform tract has a rounded opening and screw structure.
The fenestra rotunda is an anteroposteriorly short compressed reniform opening (transverse diameter 3.3 mm). The aperture for the cochlear aqueduct is subcircular and small (1.3 mm diameter). The small aperture for the vestibular aqueduct is a transversely long ellipse (2.6 mm). Just ventral to the medial margin of the internal auditory meatus, there is a weak groove that might be the median promontorial groove (see Mead and Fordyce, 2009, p. 122).
The posterior extremity of the pars cochlearis is lying lateral to the fenestra rotunda and medial to the fossa for the stapedial muscle, with a small tubercle of the dorsoventrally slightly thin caudal tympanic process, which projects posteroventrally.
On the body of the periotic, ventrally, the mallear fossa is deep, wide, and slightly reniform in shape; the rim of the fossa connects with the accessory ossicle anteriorly. The lateral tuberosity is weakly developed. A short grooved facial crest arises at the base of the mallear fossa and descends posteriorly to the apex of the posterior process. An indistinct depression lateral to the crest at the posterior part of the mallear fossa is probably the fossa incudis. Further posteriorly and lateral to the facial crest, the large hiatus epitympanicus comprises a smooth anterior depression and a rugose grooved posterior part just anterior to and parallel with the face of the posterior process. The narrow groove between the facial crest and the facial sulcus is possibly the tympanohyal sulcus, as seen in Otekaikea marplesi. The fenestra ovalis is rounded, small (2.1 mm), and filled by the footplate of the stapes (the head is missing). The ventral foramen of the facial canal is small, round, and posteriorly directed, located about level with the anterior of the fenestra ovalis; the foramen is slightly covered by the facial crest. The facial sulcus runs along the posteromedial face of the posterior process, close to the posterior bullar facet, becoming indistinct about 3 mm behind the level of the caudal tympanic process (13-14 mm anterior to the tip of the posterior process). The stapedial muscle fossa runs back along the face of the posterior process to within about 10 mm of the tip of the posterior process, separated from the facial sulcus by a faint ridge. Most of the stapedial muscle fossa lies immediately behind the fenestra ovale; the fossa is deep and slightly longer than wide, and forms a subcylindrical depression on the face of the caudal tympanic process.
The posterior process is rectangular and slender (17.6 mm long and 7.8 mm wide). The posterior bullar facet has three sections separated by faint ridges: small anterior and posterior sections, and a larger medial section. The dorsal margin of the tip of the process was originally sutured with the squamosal; the periotic was freed by removing matrix with dilute acetic acid and was found to be unfused with the squamosal.
Posterior to the lateral tuberosity, the distinctive articular rim projects laterally. Medial to the articular rim, there are at least three large presumed posteroexternal foramina. A deep groove runs from anteromedial to posterolateral, at the lateral end of the posterior process, so that the anterolateral part of the posterior process is a thin flange. Posterior to the groove, there are many weak striae on the posterolateral end of the posterior process.
Stapes. The stapes has a wide footplate, in situ in the right periotic. The crura are broken at the dorsal level of the stapedial foramen. The footplate is anteroposteriorly long and sub-elliptical, with slightly perpendicular anterior and medial margins, and slightly rounded posterior and lateral margins. A fine crest spans between the two broken crura; whether the stapedial foramen was open is uncertain. Ventrally, the head provides a small articulation for the incus. Just dorsal to the head, posteromedially, there is a large rounded muscular process. This preservation, broken in the mid-height, is very similar with the stapes of Prosqualodon davidis (Flynn, 1948, figure 7), perhaps reflecting weakness caused by a large stapedial foramen. Conversely, some more-crownward platanistoids (Platanista gangetica and Notocetus vanbenedeni AMNH 29026) reportedly lack the stapedial foramen (Anderson, 1878; de Muizon, 1987). In Tursiops, the foramen is vestigial and non-patent (Mead and Fordyce, 2009).
Bulla. Only the rounded left sigmoid process (Figure 12 and Figure 13) is preserved. The dorsal surface lacks the slightly flattened facet that, in some other odontocetes, matches the sigmoid fossa on the squamosal. Anterior to the sigmoid process, on the ventral surface, there is a depressed area with a few weak grooves. In ventral view, the margins of the sigmoid process are thickened. Inside the sigmoid process, the tympanic sulcus runs from the posteromedial end to anterolateral part of the sigmoid process. A broken fossa for malleus is on the medial base of the sigmoid process.
Mandibles. The fused mandibles preserve the symphysis and the alveoli but lack the anterior and posterior ends. The incomplete mandible is 520+ mm long; judging from premaxillary length, at least 170 mm is missing from the anterior.
The anterior part of the mandible is flat ventrally, with at least five mental foramina directed anteriorly on the ventral surface (Figure 14.7). The most anterior preserved foramen is notably long (131 mm) and deep. The anteroposteriorly long and shallow (20.5 mm height) mandibular symphysis has three foramina on its suture on dorsal surface (Figure 14.4). The damaged part of the symphysis shows internal openings of the mental foramina. Some of the internal areas are filled by cemented matrix, suggesting that the mandible contains canals for the nerves, vessels, and especially the intra-mandibular fat body. Mental foramina are preserved on the ventral surface of the mandible (Figure 14.8). The ventral surface of the mandibular symphysis has a triangular depression between the prominent ventral margins. The dorsal surface is tabular.
Around 28 mm lateral from the posterior end of the mandibular symphysis, the long bilateral alveolar groove opens dorsolaterally, widening posteriorly. Width ranges from 4.5-7.5 mm. Posteriorly, there are slightly elongate alveoli (around 17 mm long and 6 mm width). Here, the body of each mandible swells laterally; the dorsal border rises gradually. The most dorsal part of the coronoid process is broken but the base of the process shows a strong coronoid crest. Medially, each mandibular fossa is long and high. Only the dorsal border is preserved near the coronoid process, indicating a maximum depth of about 95 mm. The profile of the inflated lateral margin suggests that the fossa could have been 150+ mm long.
Teeth. Otekaikea huata is near-homodont and polydont (Figure 10). Fifty-one single-rooted teeth are preserved isolated; in at least one cheek-tooth, the roots are vestigially double but almost completely fused. Tooth positions are identified with reference to other archaic odontocetes, especially Waipatia maerewhenua. The term dimension is used in preference to “length” for overall size, as length is an anteroposterior or mesiodistal measurement, akin to diameter in most of the teeth described below.
An extremely long-rooted tusk (Figure 10.1-2) with a worn crown, presumably an incisor, is thick at the crown and the upper part of the root, tapering and becoming slightly flattened toward the root apex. The rough-surfaced root is long (197+ mm) and becomes thickest (16.1 mm widest diameter) slightly below the crown; this root is around 2.5 times longer and 1.6 times the diameter than the largest tooth in Otekaikea marplesi. The crown is naturally worn, leaving the crown base (11.9 mm maximum diameter at the enamelo-dentin junction) with exposed dentin and a pulp cavity infilled by matrix. Of note, the wear pattern around the crown is asymmetric and elliptical; one side is worn more than the other, retaining a small area of enamel. From another view, the worn end is slightly curved like a shovel (Figure 10.2).
Two more tusks (Figure 10.3-4, 93.5 and 93.1 mm, respectively) are shorter than the former tooth. Both are also worn, with exposed dentin. Their diameters are 7.7 mm and 9.6 mm, respectively, narrowing toward the root tip. They have a stronger curve than the large tusk, viewed from the worn surface and are presumed to be posterior incisors (i.e., second or third incisors).
Four conical medium-sized teeth (Figure 10.5-6) are 48-57 mm in total dimension and 7-8 mm in diameter. In the figured tooth, the crown, which is slightly laterally compressed, comprises 40% of the total dimension; it is slightly worn on the apex. A small enamel slit may indicate the posterior face, judging from the single-rooted teeth of Waipatia maerewhenua (Tanaka and Fordyce, 2014). The outer surface (probably labial, rather than buccal) of the conical crown is weakly convex. A little below the crown, the root swells on one face and forms the thickest part on the tooth. Apically (away from the crown), the root becomes thinner and slightly laterally compressed, with a shallow long groove on the presumed labial face.
Thirty-six smaller moderately curved teeth (Figure 10.7-9) are from 31 to 26 mm in total dimension and 5.7-6.7 mm diameter. The crown is about 30% of the total dimension. Loch et al. (2015) examined one tooth from this specimen, and reported the enamel as thin (75-85 microns), with an inner layer of radial enamel and an outer layer of prismless enamel. Some teeth are worn markedly; most show slight wear. They also have a small enamel slit posteriorly.
Eleven buccolingually flattened teeth (Figure 10.10-12) are 21-23 mm long and 6.4-6.7 mm diameter. Around 30% of the total length comprises the crown. There are elongate grooves on the buccal and lingual surfaces of the root.
One tooth, with a large main denticle and two small posterior accessory denticles (Figure 10.13-14), is the only tooth indicating some vestigial heterodonty, in contrast with the homodont teeth of most extant odontocetes (single rooted teeth with a conical crown and no accessory denticles). Dimensions are 23.2 mm from tip of crown to tip of root, 7.0 mm anteroposteriorly and 5.7 mm buccolingually. The crown comprises 30% of the total dimension. Its anterior keel is worn along a narrow (1.0 mm wide) band. The accessory denticles barely project from the posterior keel; the more-apical (more-anterior) denticle is slightly larger and more prominent than the more basal denticle. Both faces on the crown have subvertical long enamel ridges that are more-regularly subparallel buccally. Lingually, a weak entocingulum is developed. The single root has marked grooves on both buccolingual surfaces, as if marking the junction of two confluent roots. Just below the crown base, the root swells markedly posteriorly.
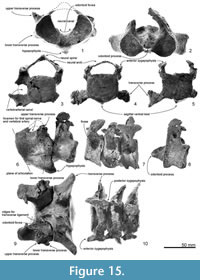 Atlas. The atlas (Figure 15.1, 15.6 and 15.11) is anteroposteriorly thick and not fused to the axis. The epiphyses are fused with the body. Only basal parts of the neural arch are preserved, including part of the margin of the transverse foramen (for the first spinal nerve and vertebral artery; see Flower, 1885b, figure 15). The neural canal and confluent odontoid fovea have a dorsally wide open V-shape. The maximum dorsal width is 41.9 mm, and height (between a line of the both sides of the fragmental neural arch and the ventral border of the neural canal) is 37.5 mm. The upper transverse process is small (25.0+ mm projection beyond the body) and directed strongly dorsolaterally. The lower process is smaller but more robust than the upper transverse process, is wide (9.2 mm long) and low (10.9 mm in thickness), and projects posterolaterally. The condyloid facets (articular surfaces) for the occipital condyles are deep and distinctly separated ventrally with clear ridges. In lateral view, the condyloid facets face down at about 20 degrees to the plane of the tabular articular facets for the axis. The latter facets are separated by the deep fovea for the odontoid process of the axis. On each articular facet, oblique strong tubercles descend forward to delimit the odontoid region; the transverse ligament (see Struthers, 1872) probably arose from the ridge. From the posteroventral margin of the ventral arch, a long robust hypapophysis (ventral tubercle) projects posteroventrally from an elongate base. The process extends 12.9 mm below and 15.7 mm behind the body. Its cross-section is elliptical.
Atlas. The atlas (Figure 15.1, 15.6 and 15.11) is anteroposteriorly thick and not fused to the axis. The epiphyses are fused with the body. Only basal parts of the neural arch are preserved, including part of the margin of the transverse foramen (for the first spinal nerve and vertebral artery; see Flower, 1885b, figure 15). The neural canal and confluent odontoid fovea have a dorsally wide open V-shape. The maximum dorsal width is 41.9 mm, and height (between a line of the both sides of the fragmental neural arch and the ventral border of the neural canal) is 37.5 mm. The upper transverse process is small (25.0+ mm projection beyond the body) and directed strongly dorsolaterally. The lower process is smaller but more robust than the upper transverse process, is wide (9.2 mm long) and low (10.9 mm in thickness), and projects posterolaterally. The condyloid facets (articular surfaces) for the occipital condyles are deep and distinctly separated ventrally with clear ridges. In lateral view, the condyloid facets face down at about 20 degrees to the plane of the tabular articular facets for the axis. The latter facets are separated by the deep fovea for the odontoid process of the axis. On each articular facet, oblique strong tubercles descend forward to delimit the odontoid region; the transverse ligament (see Struthers, 1872) probably arose from the ridge. From the posteroventral margin of the ventral arch, a long robust hypapophysis (ventral tubercle) projects posteroventrally from an elongate base. The process extends 12.9 mm below and 15.7 mm behind the body. Its cross-section is elliptical.
Axis. The axis (Figure 15.2, 15.7, and 15.12) is anteroposteriorly thinner than the atlas and not fused with the adjacent vertebrae. The epiphyses are fused with the body. The neural spine is broken but the transversely robust neural arch curves and rises dorsally. In anterior and posterior view, the neural canal appears ventrally wide and subtriangular, with a prominent median crest that separates bilateral depressions at the base of each pedicle. Each transverse process is thick, high, and strongly projected laterally, with bifurcated extremities. There is no vertebrarterial canal, but the weak depression on the posterior surface of the transverse process is probably the homologous feature.
The wide and flat articulations for the atlas anteriorly merge at the smooth surface under the strongly projecting odontoid process. The elliptical (wide) posterior epiphysis is depressed at the center, has a vertical crease, has an indistinct notochordal pit, and has indistinct circumferential striations for the annular ligament. The ventral margin projects, passing into a weak crest on the ventral surface of the body to the odontoid process.
Cervical vertebrae. Three cervical vertebrae (Figure 15.3-5, 15.7, and 15.10), probably the fourth to sixth, are preserved; all the vertebral epiphyses are ankylosed. Each vertebra has a tiny neural spine, thin neural arches, a wide rounded-pentagonal neural canal, and strongly ventrolaterally projecting and wide transverse processes, each inferred to have formed a vertebrarterial canal. The anteroposteriorly flattened body has a slightly convex anterior face, and has a sagittal ventral keel and a low dorsal sagittal crest separating bilateral dorsal depressions. On the lateral surface of the body, there is a deep and large depression, dorsal to the transverse process. The bodies of the posterior cervical vertebrae are anteroposteriorly thicker than the most anterior one.
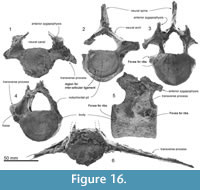 Thoracic vertebrae. Twelve thoracic vertebrae are preserved (Figure 16; not all are figured), all with the anterior epiphyses ankylosed. Each thoracic vertebra has: a high, long, and flat neural spine; a small fovea (synovial articulation) for the rib on the robust transverse process, and a larger fovea on the body; a weakly depressed anterior surface of the body; an anteroposteriorly long body (lengthens progressively posteriorly in the series); and no distinctive ridge on ventral surface of the body. On the dorsal surface of the body, there are paired anteroposteriorly long oval dorsal foramina.
Thoracic vertebrae. Twelve thoracic vertebrae are preserved (Figure 16; not all are figured), all with the anterior epiphyses ankylosed. Each thoracic vertebra has: a high, long, and flat neural spine; a small fovea (synovial articulation) for the rib on the robust transverse process, and a larger fovea on the body; a weakly depressed anterior surface of the body; an anteroposteriorly long body (lengthens progressively posteriorly in the series); and no distinctive ridge on ventral surface of the body. On the dorsal surface of the body, there are paired anteroposteriorly long oval dorsal foramina.
Thoracic vertebra 1 on Figure 16 is assumed to be an anterior thoracic vertebra because the transverse process arises in a lower position. It has a wide and low neural canal, short body and anteroposteriorly shortest neural arch. Two to four thoracic vertebrae are more posterior and have an anteroposteriorly long neural spine, high pentagonal shaped neural canal, and a longer body. The transverse process arises from a high point dorsally on the neural arch. Vertebra 4 shows a short and blunt transverse process from the dorsal part of the body. It has a deep and tear-shaped fossa on the anterior base of the transverse process.
Two lumbar vertebrae (Figure 16.6) have the anterior epiphyses ankylosed. Each lumbar vertebra has a very long wing-like transverse process; flat anterior and posterior surfaces of the body; and an anteroposteriorly long body. Vertebra F on Figure 16 is 139.3+ mm wide across the transverse processes and 54.4+ mm long, while another lumbar vertebra (not pictured) is 52.5 mm wide and 43.1 mm long.
 Ribs. In total, 12 ribs (Figure 17); two right and two left, and six others with only the mid-part of the shaft (their sides are uncertain) are preserved. Two right and left anterior double-headed ribs, damaged ventrally, are 180+ and 176+ mm chord length, respectively, anteroposteriorly flat and wide. There is a diffuse triangular and shallow cavity on the posterior surface. On the shaft, anterior and posterior surfaces are weakly swollen and flat, respectively. The rib portion converges distally.
Ribs. In total, 12 ribs (Figure 17); two right and two left, and six others with only the mid-part of the shaft (their sides are uncertain) are preserved. Two right and left anterior double-headed ribs, damaged ventrally, are 180+ and 176+ mm chord length, respectively, anteroposteriorly flat and wide. There is a diffuse triangular and shallow cavity on the posterior surface. On the shaft, anterior and posterior surfaces are weakly swollen and flat, respectively. The rib portion converges distally.
Four presumed mid-series of ribs show a conical shaft with slightly anteroposterior flattening, and with medial and lateral blunt margins. The longest preserved rib is 402+ mm chord length, around 13 mm length and 20 mm width around at the mid-shaft. The anterior and posterior surfaces are weakly swollen and flat, respectively. A left rib has the strong tubercle at around 70 mm distal to the capitulum on the lateral edge. Two right ribs show that both dorsal and ventral ends are widened dramatically, twice as wide as the mid-shaft. The dorsal end is short (or anteroposteriorly flat). The ventral end is thick and shows transversely long elliptical surface for the cartilaginous sternal ribs.
Sternum. The symmetrical flat manubrium (or presternum) (Figure 18) has incomplete outlines. Dimensions are 96.9+ mm long, 85.6+ mm wide, and 17.4 mm thick. The presumed ventral side is ventrally warped. The presumed anterior is wider, perhaps providing an area for the cartilaginous sternal ribs.
 Scapula. The right scapula is better preserved (Figure 19, Figure 20), but the left scapula (Figure 20) retains the coracoid process, which is broken on the right. For this description, the orientation of the scapula has the glenoid fossa ventral, and the suprascapular border dorsal as figured (Figure 19). The fan-shaped scapula is transversely thin and longer than high and has two projections (the acromion and coracoid) anteriorly.
Scapula. The right scapula is better preserved (Figure 19, Figure 20), but the left scapula (Figure 20) retains the coracoid process, which is broken on the right. For this description, the orientation of the scapula has the glenoid fossa ventral, and the suprascapular border dorsal as figured (Figure 19). The fan-shaped scapula is transversely thin and longer than high and has two projections (the acromion and coracoid) anteriorly.
The scapular blade is anteroposteriorly long (wide in lateral view), with the angle of the lines from the anterior and posterior borders is 106°. Anteriorly, the narrow prescapular (supraspinous) fossa and the large postscapular (infraspinous) fossa are separated by a rounded ridge. Both fossae have a smooth undulating surface; there is strong depression at the center of the scapula. The posterior convexity for the border between the infraspinatus, and the teres major lies slightly posterior to the center of the scapula. On medial surface, two ridges may be origins for tendons of the subscapularis muscle.
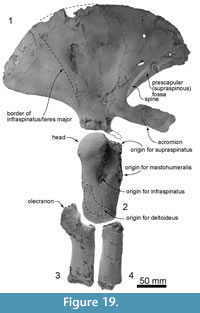 The acromion projects slightly ventrally when the glenoid cavity of the scapula is sitting on a flat plane, and curves gently laterally at the base and stops anteromedially. It provides a huge space for the supraspinatus muscle. The long (94 mm) acromion is parallel-sided anteriorly. A relatively small rod-like coracoid is long relative to diameter (25.3 mm long, versus diameters of 8.3 mm and 7.8 mm at mid-length), and robust at the base (13.7 mm width, 14.2 mm height). The neck of the scapula is strongly constricted. The scapula has a deep rounded glenoid cavity ventrally with a small foramen in the center.
The acromion projects slightly ventrally when the glenoid cavity of the scapula is sitting on a flat plane, and curves gently laterally at the base and stops anteromedially. It provides a huge space for the supraspinatus muscle. The long (94 mm) acromion is parallel-sided anteriorly. A relatively small rod-like coracoid is long relative to diameter (25.3 mm long, versus diameters of 8.3 mm and 7.8 mm at mid-length), and robust at the base (13.7 mm width, 14.2 mm height). The neck of the scapula is strongly constricted. The scapula has a deep rounded glenoid cavity ventrally with a small foramen in the center.
Humerus. The right and left humeri are well preserved (Figure 19.2 and Figure 20.5-8). The humerus is long, cylindrical (153 mm), and transversely slightly flattened (40 mm width and 65.4 mm length at the mid-shaft). For identifications of the muscle attachments, our direct examination of Globicephala melas and previous studies on the Odontoceti are used (Schulte and Smith, 1918; Howell, 1930; Smith et al., 1976; Strickler, 1978).
Proximally, a rounded, transversely long, elliptical, and smooth head is exposed laterally, occupying around 30% of the total length of the humerus. Medial to the head, there is a wide flat area for the subscapularis muscle. Below the head, there are weak anterior and posterior constrictions (proximal view). The small tuberosity lies posterior to the head and provides a small corner probably for the triceps muscle. Between the tuberosities, the bicipital groove appears to be absent. Anterior to the angle for the coracobrachialis, there is a large steep step for the supraspinatus muscle, formed by the proximal part of the humeral anterior margin. Anterior to the head, distal to the steep step for the supraspinatus, is a large proximodistally long depression for the infraspinatus muscle, beyond which is a large nutrient foramen at the level of the mid-shaft. The anterior margin of the humerus is thin and sigmoidal. Just slightly proximal to the fossa for the infraspinatus, there is a rough surface for the mastohumeralis muscle on the anterior edge. At the same level with the fossa for the infraspinatus, there is a small tubercle, probably for the serratus muscle on the posterior margin of the humerus. Just proximal to the distal end of the humerus is a weak crescent depression for the deltoid muscle. On the medial surface, muscle origins are uncertain (i.e., the teres major and minor). Distally, the joints for the ulna and radius are smooth with weak depressions on their centers. These joints are separated by a ridge. The posterior angle carries the joint for the ulna and has a small tubercle anteriorly and a strongly projected tubercle (an attachment for the triceps muscle) posteriorly. The anterior half of the distal end of the humerus is the joint for the radius.
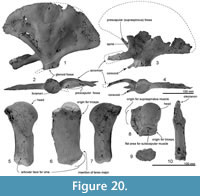 Ulna. The right ulna is robust proximally and transversely thin distally (Figure 19.3 and Figure 20.10). The olecranon projects posteriorly and is slightly curved laterally from proximal and distal view. The base of the olecranon and the proximal articular face for the humerus are thick (or wide). The trochlear notch is strongly curved (angle around 90°). The interosseous and the outer borders are straight, and gradually and weakly widened distally. On the distal end, the epiphysis is not fused. The distal end is an anteroposteriorly long ellipse (length 47.9+ mm and width 22.8+ mm).
Ulna. The right ulna is robust proximally and transversely thin distally (Figure 19.3 and Figure 20.10). The olecranon projects posteriorly and is slightly curved laterally from proximal and distal view. The base of the olecranon and the proximal articular face for the humerus are thick (or wide). The trochlear notch is strongly curved (angle around 90°). The interosseous and the outer borders are straight, and gradually and weakly widened distally. On the distal end, the epiphysis is not fused. The distal end is an anteroposteriorly long ellipse (length 47.9+ mm and width 22.8+ mm).
Radius. The right and left radii (Figure 19.4) are preserved. The right radius shows an exact fit with the humerus. The shaft is weakly curved anteriorly and slightly thinner than the ulna. Both proximal and distal epiphyses are not fused. The proximal end is triangular with a small projection at the anterior end of the posteriorly-tilted articular face for the humerus. The anterior margin is thin, and the posterior margin shows a flattened area. The shaft widens gradually to the distal end.
Carpus. A single carpus (Figure 20.9) is 40+ mm long, 26 mm wide, and 18.3 mm thick. It may be a proximal carpus because it is very wide and is almost the same width as the ulna and radius.
PHYLOGENETIC ANALYSIS
The phylogenetic position of Otekaikea huata was analyzed using the matrix of Tanaka and Fordyce (2014) with 16 additional characters from previous studies (de Muizon, 1987; Fordyce, 1994; Bianucci et al., 2013; Gutstein et al., 2014) (see Appendices 1-6). Originally, the matrix was modified from that of Murakami et al. (2012b), which was designed to focus on Odontoceti phylogeny, particularly Delphinoidea. The matrix of Tanaka and Fordyce (2014) included 76 extant and extinct taxa, all named and described, and 278 characters (with 31 soft tissue characters) cited and/or modified from previous studies (Barnes, 1985a, 1985b; Heyning, 1989; Barnes, 1990; Fordyce, 1994; Arnold and Heinsohn, 1996; Heyning, 1997; Messenger and McGuire, 1998; Geisler and Sanders, 2003; Lambert, 2005; Lambert and Post, 2005; Lambert et al., 2008; Aguirre-Fernández et al., 2009; Geisler et al., 2011; Murakami et al., 2012a, 2012b; Murakami et al., 2014).
The matrix of Tanaka and Fordyce (2014) was modified in minor ways (see Appendix 5). Five taxa were added: Otekaikea huata of this study, the archaic Odontoceti Simocetus rayi (based on study of the holotype USNM 256517, on a cast at OU, and on Fordyce, 2002), Phocageneus venustus (based on study of referred specimen USNM 21039, and on Kellogg, 1957), Microcetus hectori (based on study of the holotype MoNZ Ma 653) , M. ambiguus (based on the type teeth photos), M. sharkovi (a cast of the type held at OU), and an undescribed species (based on study of OU 22125). An archaeocete, Georgiacetus vogtlensis, was the outgroup taxon. (The new genus Huaridelphis Bianucci et al. (2014) appeared in print when this article was in review, too late for formal inclusion in the cladistic analysis, although we offer some comments elsewhere in the text.) Percentages of missing data were: for Otekaikea huata 43% (includes soft tissue characters) and 36% (excludes soft tissue), Simocetus rayi 48% (includes soft tissue characters) and 41% (excludes soft tissue), Phocageneus venustus 89% (includes soft tissue characters) and 88% (excludes soft tissue), Microcetus hectori 86% (includes soft tissue characters) and 85% (excludes soft tissue), M. ambiguus 97.6% (includes soft tissue characters) and 97.3% (excludes soft tissue), M. sharkovi 90% (includes soft tissue characters) and 97.3% (excludes soft tissue) , OU 22125 73% (includes soft tissue characters), and 70% (excludes soft tissue). In summary, the present matrix has 83 taxa and 292 characters.
 Character data and tree data were managed using Mesquite 2.75 (Maddison and Maddison, 2011). The matrix files are in Appendices 1 (nexus format), 2 (tnt format), and 3 (text). Two sorts of cladistic analyses were performed with TNT, 1.1 (Goloboff et al., 2008). All characters were treated as unweighted and unordered (analysis 1), or implied weights with K=3 without ordering (analysis 2). Both analyses used New Technology Search with the setting: recover minimum length trees = 1000 times. To measure node stability, we used the decay index method (Bremer, 1994) for the strict consensus trees. After the analyses, as above, species in the more-diverse families crown-ward of Papahu (particularly Physeteroidea, Ziphiidae, Phocoenidae and Delphinidae) were merged for ease of illustrating. The full cladograms which show positions for all OTUs are provided as tree tif files in Appendix 7 and Appendix 8.
Character data and tree data were managed using Mesquite 2.75 (Maddison and Maddison, 2011). The matrix files are in Appendices 1 (nexus format), 2 (tnt format), and 3 (text). Two sorts of cladistic analyses were performed with TNT, 1.1 (Goloboff et al., 2008). All characters were treated as unweighted and unordered (analysis 1), or implied weights with K=3 without ordering (analysis 2). Both analyses used New Technology Search with the setting: recover minimum length trees = 1000 times. To measure node stability, we used the decay index method (Bremer, 1994) for the strict consensus trees. After the analyses, as above, species in the more-diverse families crown-ward of Papahu (particularly Physeteroidea, Ziphiidae, Phocoenidae and Delphinidae) were merged for ease of illustrating. The full cladograms which show positions for all OTUs are provided as tree tif files in Appendix 7 and Appendix 8.
Phylogenetic Relationships
Analysis 1; unweighted and unordered. The phylogenetic analysis shows 1266 of the shortest trees of 1858 steps each. The strict consensus tree (Figure 21) shows almost the same topology as the Tanaka and Fordyce (2014) unweighted and unordered strict consensus tree, except that Papahu taitapu now appears immediately crownward to the clade Platanistoidea, thus clustering with later-diverging Odontoceti.
Analysis 2; implied weighting. The phylogenetic analysis recovers 18 trees with a score of 138.02 (Figure 21). The strict consensus tree also shows almost the same topology as the Tanaka and Fordyce (2014) single tree, except that Papahu taitapu is again crownward to the Platanistoidea.
 In both analyses, Otekaikea belongs in a clade with the Platanistidae and Waipatiidae. In analysis 2, the clade with Otekaikea is broadly the same as the Platanistoidea proposed by de Muizon (1987). Otekaikea huata forms a clade with O. marplesi, separate from Waipatia. The genus Otekaikea is supported by three synapomorphies: the ventromedial edge of the internal opening of infraorbital foramen is formed by the both maxilla and palatine (Character 44); there is a fused accessory ossicle on the periotic (Character 199); and an intra-premaxillary foramen is present bilaterally, bounded by the premaxilla and maxilla.
In both analyses, Otekaikea belongs in a clade with the Platanistidae and Waipatiidae. In analysis 2, the clade with Otekaikea is broadly the same as the Platanistoidea proposed by de Muizon (1987). Otekaikea huata forms a clade with O. marplesi, separate from Waipatia. The genus Otekaikea is supported by three synapomorphies: the ventromedial edge of the internal opening of infraorbital foramen is formed by the both maxilla and palatine (Character 44); there is a fused accessory ossicle on the periotic (Character 199); and an intra-premaxillary foramen is present bilaterally, bounded by the premaxilla and maxilla.
DISCUSSION
Function - Face
The excavated and voluminous facial fossa would have held enlarged nasofacial muscles, inserting around the nasal diverticula, implying a well-developed echolocation apparatus, but there are no features that clearly show particular muscle origins or imply particular acoustic specializations. The premaxillary sac fossa has a transversely convex surface, rather than the concave surface seen in Waipatia ; implications for shape and function of the premaxillary sac are uncertain. The antorbital notch appears to have been shallow, judging from the remnant preserved on the right, but the size of the antorbital process, if any, is uncertain.
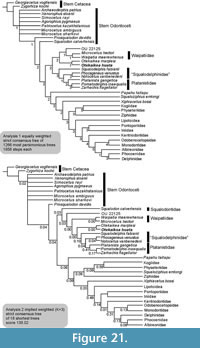 The newly named intra-premaxillary foramen is one of many dorsal foramina of the facial fossa that are part of the infraorbital canal-foramen complex (Figure 22). Previously, Tanaka and Fordyce (2014) reported the foramen in Otekaikea marplesi as lying between the posterior extremities of the maxilla and premaxilla, and used the name posterior accessory foramen, earlier proposed by Vélez-Juarbe and Pyenson (2012) for a comparable foramen in the Monodontidae. Closer study shows that the foramina in the Monodontidae and in Otekaikea have different bone relationships, below, suggesting independent origins. Thus, the foramen in Otekaikea is here named the intra-premaxillary foramen. Both species of Otekaikea have the foramen associated with the maxilla and the bifurcated premaxilla (the posterolateral plate and posteromedial splint). The posterolateral plate passes back toward the nearly-vertical medial part of the maxilla, which is elevated to form a vertex crest. The intra-premaxillary foramen opens in the premaxilla in the fold between the maxillary vertex crest and posterolateral plate, partly covered by the premaxilla. Conversely, in Monodon and Delphinapterus, the posterior accessory foramen is formed mainly by the maxilla, because of shortening of the premaxilla, more-lateral to the nares, and well below the vertex. The premaxilla may contribute to the anterior part of the posterior accessory foramen, but does not cover most of the foramen; further, the homologies in terms of posterolateral plate and posteromedial splint are uncertain. Thus, the foramen is exposed as a long groove on the maxilla, posterior to the ascending process of the premaxilla. The intra-premaxillary foramen should not be confused with the premaxillary foramen, which supplies vessels to the anterior part of the premaxillary sac fossa.
The newly named intra-premaxillary foramen is one of many dorsal foramina of the facial fossa that are part of the infraorbital canal-foramen complex (Figure 22). Previously, Tanaka and Fordyce (2014) reported the foramen in Otekaikea marplesi as lying between the posterior extremities of the maxilla and premaxilla, and used the name posterior accessory foramen, earlier proposed by Vélez-Juarbe and Pyenson (2012) for a comparable foramen in the Monodontidae. Closer study shows that the foramina in the Monodontidae and in Otekaikea have different bone relationships, below, suggesting independent origins. Thus, the foramen in Otekaikea is here named the intra-premaxillary foramen. Both species of Otekaikea have the foramen associated with the maxilla and the bifurcated premaxilla (the posterolateral plate and posteromedial splint). The posterolateral plate passes back toward the nearly-vertical medial part of the maxilla, which is elevated to form a vertex crest. The intra-premaxillary foramen opens in the premaxilla in the fold between the maxillary vertex crest and posterolateral plate, partly covered by the premaxilla. Conversely, in Monodon and Delphinapterus, the posterior accessory foramen is formed mainly by the maxilla, because of shortening of the premaxilla, more-lateral to the nares, and well below the vertex. The premaxilla may contribute to the anterior part of the posterior accessory foramen, but does not cover most of the foramen; further, the homologies in terms of posterolateral plate and posteromedial splint are uncertain. Thus, the foramen is exposed as a long groove on the maxilla, posterior to the ascending process of the premaxilla. The intra-premaxillary foramen should not be confused with the premaxillary foramen, which supplies vessels to the anterior part of the premaxillary sac fossa.
The phylogenetic patterns for the intra-premaxillary foramen are not well documented. The foramen is absent in the basal platanistoid Waipatia. In some Squalodontidae and putative Dalpiazinidae, the fold between the posteromedial splint and posterolateral plate is fissured, possible for a vessel. In squalodontid OU 21798, a large dorsal infraorbital foramen opens close to the fold, but slightly laterally. Similarly, some platanistoids, extant P. gangetica, Zarhachis flagellator USNM 10911, reported by Kellogg (1926) and Huaridelphis raimondii Lambert et al. (2014) show a foramen on the medial part of the maxilla, just posterolateral to the end of the premaxilla. Could such a foramen arise by the migration, medially, of an existing dorsal infraorbital foramen, or might it be a neomorphic structure? To consider the origin and significance of foramina on the face, all the taxa in the phylogenetic analysis have one dorsal infraorbital foramen (Agorophius pygmaeus, Otekaikea marplesi, and Papahu taitapu), or sometimes two as an adjacent pair (Simocetus rayi, Waipatia maerewhenua, Zarhachis flagellator, and Notocetus vanbenedeni). None has a set of three, which if one foramen were to migrate medially, might otherwise be a precursor situation for the structure in Otekaikea huata.
 Function - Basicranium
Function - Basicranium
Ventrally, Otekaikea lacks the enlarged orbital fossae for the basicranial pterygoid sinuses that, in Platanista (Fraser and Purves, 1960), Notocetus and Huaridelphis (Bianucci et al., 2014; Lambert et al., 2014), are linked to development of maxillary thickenings or crests on the face. The presence of a subtemporal crest in O. huata further suggests that lobes of the pterygoid sinus did not invade the orbit. Shallow fossae on the alisphenoid for parts of the pterygoid sinus are comparable in development to those of Waipatia. There is no evidence that the pterygoid sinus fossa had a bony lateral lamina running forward from the falciform process. The tympanosquamosal recess, which forms the fossa for the middle sinus, is prominent, with a distinct posteromedial sulcus marking the probable origin of the sinus from the middle ear via the epitympanic hiatus. The periotic sits snugly in the skull without evidence, however, of bony fusion with the squamosal. Cavities between the periotic and periotic fossa indicate a marked volume of soft tissues, but whether vascular or pneumatic sinus is uncertain. Whether these are truly parts of the foramen spinosum must be established by dissection of modern material. The significance is uncertain for the elongate, tongue-like paroccipital process; there are no obvious modern equivalents. Its transversely elongate structure could have been a laterally extensive base for a widened stylohyal, or alternatively may have allowed the base of the stylohyal to move in a transverse plane during feeding (for the role of the stylohyal in feeding, see Reidenberg and Laitman, 1994).
Systematics - Family Waipatiidae
The family Waipatiidae was established for the single species Waipatia maerewhenua Fordyce, 1994. Recently, Tanaka and Fordyce (2014) used cladistic analysis to identify Otekaikea marplesi as a second species in the family. The cladistic analysis here allows revision of the Waipatiidae to include only two species (W . maerewhenua and Microcetus hectori), with the clade characterized by having a squared postglenoid process in lateral view (character 115). Analyses 1 and 2 (Figure 21) now show two different relationships for W. maerewhenua. Analysis 1 (equally weighted characters) shows a clade with five species: Waipatia maerewhenua and Microcetus hectori as sister-species in Waipatiidae and, more basally, the unnamed OU 22125, and the two Otekaikea sister-species. Analysis 2 (implied weights) also recovers Waipatia maerewhenua and Microcetus hectori as sister-species in Waipatiidae, with the unnamed OU 22125 immediately basal, but the two Otekaikea species now lie immediately crown-ward. The clade Waipatiidae + Otekaikea and OU 22125 of equal-weights analysis 1 is supported by six characters, three of which have mosaic state distributions in different lineages. These homoplasous characters are down-weighted in the implied-weights analysis 2. Two of six synapomorphies in analysis 1 are reversals (or show the same states as the outgroup). Only one character is a synapomorphy for the clade Waipatiidae + Otekaikea and OU 22125 (poorly defined ventromedial keel of the tympanic bulla: character 212). One taxonomically important and enigmatic species included here, Microcetus hectori, is known from a single incomplete specimen for which 86% of skeletal characters could not be coded. It is axiomatic that relationships will probably change as new material becomes available to study. Such fossils include five more recently-identified small odontocetes, with skulls, from the upper Otekaike Limestone of Hakataramea Valley.
Systematics - Content of the Platanistoidea
Another difference between analyses 1 and 2 is the position of the genus Squalodon. In analysis 1, Squalodon appears as a stem Odontoceti, but in analysis 2, Squalodon appears within the Platanistoidea sensu lato. Prosqualodon does not form a clade with Squalodon in this study. The Platanistoidea sensu stricto (namely, with Squalodon excluded) is supported by six synapomorphies: presence of the posterior dorsal infraorbital foramina of the maxilla (character 59); C-shaped or weakly curved parabullary sulcus (character 169); presence of the articular rim (character 186); presence of the anterior spine of the tympanic bulla (character 195); presence of the anterolateral convexity of the tympanic bulla with anterolateral notch (character 196); and presence of the ventral groove (median furrow) of bulla anteriorly (character 212). Derived states for one of six characters (character 59) are seen in other linages, not only for the Platanistoidea sensu lato. Other synapomorphies of the Platanistoidea sensu lato were mentioned in previous studies (de Muizon, 1987, 1991; Fordyce, 1994; Lambert et al., 2014; Tanaka and Fordyce, 2014). Monophyly of the Platanistoidea is supported by relatively low decay indices, because this taxon contains only one extant species, plus related fossils. It is axiomatic that future analyses with new fossils will probably change topology and clade content.
Paleoecology
Otekaikea marplesi and O. huata are similar in geological age, and the uncertainties about exact ages make it possible that the species could have been coeval. In addition, their skull sizes are similar. To what extent, then, might these species have had similar ecologies? Otekaikea huata can be distinguished clearly from O. marplesi by characters given in the diagnosis. To consider some possibly functional differences, the dentition of O. huata is near-homodont, while O. marplesi is probably heterodont (based on the presence of at least one denticulate double-rooted tooth). Other platanistoids from New Zealand (here, Otekaikea, Waipatia maerewhenua, Microcetus hectori and OU 22125) show better-developed double rooted posterior teeth. In addition, uniquely O. huata has an extremely large tusk (197+ mm, admittedly not in place, and worn) and smaller long tusks (see description and Figure 10) with a long root. The largest tusk originally had a crown estimated at 280 mm long, based on the proportions of the procumbent incisors of Waipatia maerewhenua, and of OU 22397. Otekaikea marplesi and W. maerewhenua also have tusks (Fordyce, 1994; Tanaka and Fordyce, 2014), but smaller than in O. huata. Large tusks in odontocetes have been reported from other lineages (at least the Kentriodontidae, Monodontidae, Odobenocetopsidae, Dalpiazinidae, and Ziphiidae) (de Muizon and Domning, 2002; Nweeia et al., 2012). Tusk function is problematic (de Muizon et al., 2002; Fordyce et al., 2013). Erupted mandibular tusks are sexually dimorphic in living Ziphiidae, occurring in adult males, and perhaps in some fossil ziphiids (Lambert et al., 2010). Other structural differences imply differences in function and thus ecology: O. huata has a relatively deeper facial fossa, more asymmetrical nasals, and a tongue-like paroccipital process. The latter implies a stylohyal articulation, and presumably mode of action, without a comparable modern equivalent.
Taphonomy
At the locality, the Otekaike Limestone is subhorizontal and diffusely bedded, suggesting deposition below storm wave base and in conditions with limited traction currents. The matrix of Otekaikea huata contains well-preserved macroinvertebrates, consistent with quiet waters. The ostracod Bisulcocythere (Cytheroidea; Ostracoda) implies a paleodepth of roughly mid-shelf (50-100 m) (Ayress and Swanson, 1992; Ayress, 1993). Molluscan studies suggest that the Otekaike Limestone represents a sheltered, warm water, middle shelf setting (Fordyce, 2009; Fordyce and Maxwell, 2003).
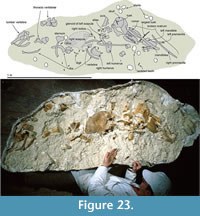 Otekaikea huata was preserved right side down. Figure 23 shows the specimen, which is exposed with the lower side visible and the lower part of the field plaster jacket partly removed. The upper figure is based on the original distribution of bones, as traced on plastic sheets by Andrew Grebneff during preparation in 1996. The skeleton (from the skull to lumbar vertebrae) is not articulated, but most of the elements (include both scapulae and mandible, and a digit close to the scapula) are near to original positions in the 2 m long limestone field block. Such semi-articulated specimens are not reported commonly (Kellogg, 1925; Lancaster, 1986; Fordyce and de Muizon, 2001). In contrast to the inferred preservation of O. huata, Schäfer and Craig (1972) noted that marine mammal carcasses may "drift for weeks on the surface of the sea," with parts such as the mandibles and skull lost (dropped) from the trunk early on. Such preservation has been noted in previous studies (Bianucci et al., 2010; Boessenecker, 2013).
Otekaikea huata was preserved right side down. Figure 23 shows the specimen, which is exposed with the lower side visible and the lower part of the field plaster jacket partly removed. The upper figure is based on the original distribution of bones, as traced on plastic sheets by Andrew Grebneff during preparation in 1996. The skeleton (from the skull to lumbar vertebrae) is not articulated, but most of the elements (include both scapulae and mandible, and a digit close to the scapula) are near to original positions in the 2 m long limestone field block. Such semi-articulated specimens are not reported commonly (Kellogg, 1925; Lancaster, 1986; Fordyce and de Muizon, 2001). In contrast to the inferred preservation of O. huata, Schäfer and Craig (1972) noted that marine mammal carcasses may "drift for weeks on the surface of the sea," with parts such as the mandibles and skull lost (dropped) from the trunk early on. Such preservation has been noted in previous studies (Bianucci et al., 2010; Boessenecker, 2013).
The carcass sank to the sea floor with little loss of bones, presumably shortly after death. The lack of bedding in the limestone suggests that traction currents were weak, but water movement may have sufficed to move a carcass made almost buoyant by decomposition gas. Immediately postmortem, the rostrum and mandible were broken off, filled by matrix, and scattered along with small bone fragments. A rib and left forelimb elements lay close to the skull. Bioturbators probably were invertebrates, such as burrowing crustaceans, with additional movement from scavengers such as bony fish and chondrichthyans. Most damage probably occurred before full burial, but some bioerosion could have been infaunal. The braincase was also filled by matrix with well-preserved shells. Molluscan scars include inferred homing marks from limpets such as Crepidula and Sigapatella. Figure 22 shows some of the large shells around the dolphin carcass. Traces of bioactivity on bone surface include some bite marks, albeit not abundant (Figure 14.4). There is no sign of death from predation. The skeleton was slightly scattered by bioturbation and scavenging before burial.
CONCLUSION
The archaic dolphin Otekaikea huata (Otekaike Limestone, Oligo/Miocene boundary), from Hakataramea Valley, South Canterbury, New Zealand, is described as a new species of Otekaikea. The single known specimen adds to odontocete structural and taxonomic diversity during the Waitakian Stage, latest Oligocene to earliest Miocene (22.28 to 24.61 Ma) in the Southwest Pacific Ocean. Otekaikea huata and other dolphins from the Oligocene of New Zealand (O. marplesi and Waipatia maerewhenua) were at least 2.6 m long. The type specimen of Otekaikea huata preserves at least one large tusk and an elevated face for the nasofacial muscles implicated in production of echolocation sounds. New terms for skull features are: the anterolateral process of the nasal and the intra-premaxillary foramen. The phylogenetic analyses based on morphological features place Otekaikea huata as a near-basal member of the clade Platanistoidea. The new species implies wider diversity of the superfamily Platanistoidea in latest Oligocene time.
ACKNOWLEDGEMENTS
We thank many who helped in diverse ways. The late A. Grebneff (University of Otago) prepared the type specimen, with later preparation by S.E. White, R.E. Fordyce, and Y. Tanaka. In the field, A. Grebneff, B.V.N. Black, and O. Sarll (all University of Otago) helped R.E. Fordyce recover the specimen. The management of Foveran Station and Waitaki Transport (the then-owners and operators, respectively, of Hakataramea Quarry) granted access and permission to collect. D.A. Teagle (University of Southampton) graciously provided the strontium date. M. Murakami (Waseda University), G. Aguirre-Fernandez (University of Otago) and F.G. Marx (National Museum of Nature and Science) discussed phylogenetics. S.E. White (University of Otago) discussed possible Maori names for the fossil. We particularly thank the two journal peer-reviewers, J. H. Geisler and O. Lambert, for their constructive comments that helped improve the manuscript. J. Galkin (American Museum of Natural History), M.A. Reguero (Museo de La Plata), T.K. Yamada (National Museum of Nature and Science), and D.J. Bohaska and N.D. Pyenson (National Museum of Natural History) provided access to comparative specimens. Field and lab work was supported with research funds from the National Geographic Society (grants 4024-88 and 4341-90 to R.E. Fordyce) and the Department of Geology, University of Otago.
REFERENCES
Abel, O. 1905. Les odontocètes du Boldérien (Miocène supérieur) d'Anvers. Mémoires du Musée royal d'Histoire Naturelle de Belgique, 3:1-155.
Abel, O. 1914. Die Vorfahren der Bartenwale. Denkschriften der Akademie der Wissenschaften, Wien, Mathematisch-naturwissenschaftliche Klasse, 90:155-224.
Aguirre-Fernández, G. and Fordyce, R.E. 2014. Papahu taitapu, gen. et sp. nov., an early Miocene stem odontocete (Cetacea) from New Zealand. Journal of Vertebrate Paleontology, 34(1):195-210.
Aguirre-Fernández, G., Barnes, L.G., Aranda-Manteca, F.J., and Fernández-Rivera, J.R. 2009. Protoglobicephala mexicana, a new genus and species of Pliocene fossil dolphin (Cetacea; Odontoceti; Delphinidae) from the Gulf of California, Mexico. Boletin de la Sociedad Geologica Mexicana, 61(2):245-265.
Allen, G.M. 1921. A new fossil cetacean. Bulletin of the Museum of Comparative Zoology, Harvard, 65(1):1-14.
Allen, G.M. 1923. The black finless porpoise, Meomeris. Bulletin of the Museum of Comparative Zoology, 65(7):233-256.
Anderson, J. 1878. Anatomical and zoological researches: Comprising an account of the zoological results of the two expeditions to Western Yunnan in 1868 and 1875; and a monograph on the two cetacean genera, Platanista and Orcella. 2 vols. B. Quaritch, London.
Arnold, P.W. and Heinsohn, G.E. 1996. Phylogenetic status of the Irrawaddy dolphin Orcaella brevirostris (Owen in Gray): a cladistic analysis. Memoirs of the Queensland Museum, 39(2):141-204.
Ayress, M.A. 1993. Ostracod biostratigraphy and paleoecology of the Kokoamu Greensand and Otekaike Limestone (late Oligocene to early Miocene), North Otago and South Canterbury, New Zealand. Alcheringa, 17(1-2):125-151.
Ayress, M. and Swanson, K. 1992. New genera and species of Ostracoda (Crustacea) from the Cenozoic of South Island, New Zealand. New Zealand Natural Sciences, 18:1-18.
Barnes, L.G. 1984a. Fossil odontocetes (Mammalia: Cetacea) from the Almejas Formation, Isla Cedros, Mexico. Museum of Paleontology, University of California.
Barnes, L.G. 1984b. Whales, dolphins and porpoises; origin and evolution of the Cetacea. In Gingerich, P.D. and Badgle, C.E. (eds.), Mammals. Notes for a short course, University of Tennessee, Department of Geological Science.
Barnes, L.G. 1985a. Evolution, taxonomy and antitropical distributions of the porpoises (Phocoenidae, Mammalia). Marine Mammal Science, 1(2):149-165.
Barnes, L.G. 1985b. Fossil pontoporiid dolphins (Mammalia: Cetacea) from the Pacific coast of North America. Contributions to Science, Natural History Museum of Los Angeles County, 363:1-34.
Barnes, L.G. 1990. The fossil record and evolutionary relationships of the genus Tursiops, p. 3-26. In Leatherwood, S. and Reeves, R.R. (eds.), The Bottlenose Dolphin. Academic Press Inc, San Diego, New York.
Barnes, L.G. 2008. Miocene and Pliocene Albireonidae (Cetacea, Odontoceti), rare and unusual fossil dolphins from the eastern North Pacific Ocean. Geology and Vertebrate Paleontology of Western and Southern North America, Contributions in Honor of David P. Whistler. Natural History Museum of Los Angeles County Science Series, 41:99-152.
Bianucci, G. 2005. Arimidelphis sorbinii a new small killer whale-like dolphin from the Pliocene of Marecchia River (Central eastern Italy) and a phylogenetic analysis of the Orcininae (Cetacea: Odontoceti). Rivista Italiana di Paleontologia e Stratigrafia, 111(2):329-344.
Bianucci, G., Lambert, O., and Post, K. 2010. High concentration of long‐snouted beaked whales (genus Messapicetus) from the Miocene of Peru. Palaeontology, 53(5):1077-1098.
Bianucci, G., Urbina, M., and Lambert, O. 2014. A new record of Notocetus vanbenedeni (Squalodelphinidae, Odontoceti, Cetacea) from the Early Miocene of Peru. Comptes Rendus Palevol, 14(1):5-13.
Bianucci, G., Lambert, O., Salas-Gismondi, R., Tejada, J., Pujos, F., Urbina, M., and Antoine, P.-O. 2013. A Miocene relative of the Ganges River dolphin (Odontoceti, Platanistidae) from the Amazonian Basin. Journal of Vertebrate Paleontology, 33(3):741-745.
Boessenecker, R.W. 2013. A new marine vertebrate assemblage from the Late Neogene Purisima Formation in Central California, part II: Pinnipeds and Cetaceans. Geodiversitas, 35(4):815-940.
Bremer, K. 1994. Branch support and tree stability. Cladistics, 10(3):295-304.
Brisson, A. 1762. Regnum Animale in Classes IX distributum sive synopsis methodica. Edito altero auctior, Theodorum Haak, Leiden.
Brownell, R.L., Findley, L.T., Vidal, O., Robles, A., and Silvia Manzanilla, N. 1987. External morphology and pigmentation of the vaquita, Phocoena sinus (Cetacea: Mammalia). Marine Mammal Science, 3(1):22-30.
Cotten, P.B., Piscitelli, M.A., McLellan, W.A., Rommel, S.A., Dearolf, J.L., and Pabst, D. 2008. The gross morphology and histochemistry of respiratory muscles in bottlenose dolphins, Tursiops truncatus. Journal of Morphology, 269(12):1520-1538.
Cranford, T.W., Amundin, M., and Norris, K.S. 1996. Functional morphology and homology in the odontocete nasal complex: implications for sound generation. Journal of Morphology, 228(3):223-285.
Curry, B.E. 1992. Facial anatomy and potential function of facial structures for sound production in the harbor porpoise (Phocoena phocoena) and Dall's porpoise (Phocoenoides dalli). Canadian Journal of Zoology, 70(11):2103-2114.
de Muizon, C. 1983. Pliopontos littoralis un nouveau Platanistidae (Cetacea) du Pliocène de la côte péruvienne. Comptes Rendus de l’Académie des Sciences Paris, 296:625-628.
de Muizon, C. 1984. Les vertébrés fossiles de la Formation Pisco (Pérou). deuxiéme partie: les Odontocétes (Cetacea, Mammalia) du Pliocéne inférieur de Sud-Sacaco. Travaux de l’Institut Français d’Études Andines, 27:1-188.
de Muizon, C. 1985. Nouvelles données sur le diphylétisme des Dauphins de rivière (Odontoceti, Cetacea, Mammalia). Comptes rendus l'Academie des Sciences series 2,, 301:359-362.
de Muizon, C. 1987. The affinities of Notocetus vanbenedeni, an Early Miocene platanistoid (Cetacea, Mammalia) from Patagonia, southern Argentina. American Museum novitates, 2904:1-27.
de Muizon, C. 1988. Les relations phylogenetiques des Delphinida (Cetacea, mammalia). Annales de paléontologie, 74(4):159-227.
de Muizon, C. 1991. A new Ziphiidae (Cetacea) from the Early Miocene of Washington State (USA) and phylogenetic analysis of the major groups of odontocetes. Bulletin du Muséum National d'Histoire Naturelle, 12(3-4):279-326.
de Muizon, C. 1994. Are the squalodonts related to the platanistoids? Proceedings of the San Diego Society of Natural History, 29:135-146.
de Muizon, C. and Domning, D.P. 2002. The anatomy of Odobenocetops (Delphinoidea, Mammalia), the walrus‐like dolphin from the Pliocene of Peru and its palaeobiological implications. Zoological Journal of the Linnean Society, 134(4):423-452.
de Muizon, C., Domning, D.P., and Ketten, D.R. 2002. Odobenocetops peruvianus, the walrus-convergent delphinoid (Mammalia: Cetacea) from the Early Pliocene of Peru. Smithsonian Contributions to Paleobiology, 93:223-261.
de Smet, W.M.A. 1977. The regions of the cetacean vertebral column volume 3, p. 59-80. In Harrison, R.J. (ed.), Functional Anatomy of Marine Mammals. Academic Press, London.
Dickson, M.R. 1964. The skull and other remains of Prosqualodon marplesi, a new species of fossil whale. New Zealand Journal of Geology and Geophysics, 7(3):626-635.
Doran, A.H. 1878. Morphology of the mammalian ossicula auditus. Transactions of the Linnaean Society, Series 2, Zoology, 1(18):371-497.
Dubrovo, I.A. and Sanders, A.E. 2000. A new species of Patriocetus (Mammalia, Cetacea) from the late Oligocene of Kazakhstan. Journal of Vertebrate Paleontology, 20:577-590.
Dubrovo, I. and Sharkov, A. 1971. Kit iz verkhnego Oligotsena Mangyshlaka [A whale from the Upper Oligocene of Mangyshlak]. Doklady Akademia Nauk SSSR, 194(6):1403-1406.
Evans, H.E. and de Lahunta, A. 2013. Miller's anatomy of the dog. Elsevier, St. Louis, Missouri.
Fajardo-Mellor, L., Berta, A., Brownell, R.L., Boy, C.C., and Goodall, N.P. 2006. The phylogenetic relationships and biogeography of true porpoises (Mammalia: Phocoenidae) based on morphological data. Marine Mammal Science, 22(4):910-932.
Flower, W.H. 1867. Description of the skeleton of Inia geoffrensis and the skull of Pontoporia blainvillii, with remarks on the systematic position of these animals in the Order Cetacea. Transactions of the Zoological Society of London, 6(3):87-116.
Flower, W.H. 1868. On the osteology of the cachalot or sperm-whale (Physeter macrocephalus). Transactions of the Zoological Society of London, 6(6):309-372.
Flower, W.H. 1872. On the recent ziphioid whales, with a description of the skeleton of Berardius arnouxi. Transactions of the Zoological Society of London, 8(3):203-234.
Flower, W.H. 1884. On the characters and divisions of the Family Delphinidae. Proceedings of the Zoological Society of London, 1883:466-513.
Flower, W.H. 1885a. An introduction to the osteology of the Mammalia. Macmillan and Co., London.
Flower, W.H. 1885b. List of the specimens of Cetacea in the Zoological Department of the British Museum. British Museum, London.
Flynn, T.T. 1948. Description of Prosqualodon davidi Flynn, a fossil cetacean from Tasmania. Transactions of the Zoological Society of London, 26(2):153-197.
Fordyce, R.E. 1981. Systematics of the odontocete whale Agorophius pygmaeus and the Family Agorophiidae (Mammalia, Cetacea). Journal of Paleontology , 55(5):1028-1045.
Fordyce, R.E. 1994. Waipatia maerewhenua, new genus and new species (Waipatiidae, new family), an archaic Late Oligocene dolphin (Cetacea: Odontoceti: Platanistoidea) from New Zealand. Proceedings of the San Diego Society of Natural History, 29:147-176.
Fordyce, R.E. 2002. Simocetus rayi (Odontoceti: Simocetidae, new family): A bizarre new archaic Oligocene dolphin from the eastern North Pacific. Smithsonian Contributions to Paleobiology, 93:185-222.
Fordyce, R.E. 2003. New Oligocene dalpiazinids from the Waitaki region: increasing the diversity of platanistoid dolphins. Geological Society of New Zealand miscellaneous publication, 116a:53.
Fordyce, R.E. 2009. Fossil sites, p. 459-466. In Perrin, W.F., Thewissen, J.G.M., and Würsig, B. (eds.), Encyclopedia of Marine Mammals. Elsevier, San Diego.
Fordyce, R.E. 2014. A southern view of Oligocene marine tetrapods: Kokamu Greensand and Otekaike Limestone, Hakataramea Valley, New Zealand. Abstracts, Secondary Adaptations of Tetrapods to Life in the Water 7th triennial meeting, George Mason University, Virginia, June 2014. events2.gmu.edu/secad2014/category/abstracts/.
Fordyce, R.E., Aguirre-Fernandez, G., Loch, C., and Tanaka, Y. 2013. Tusks in extinct Oligocene dolphins: feeding, display, agonism, or sensory? Abstracts, 20th Biennial Conference on the Biology of Marine Mammals 2013 (Society for Marine Mammalogy):71.
Fordyce, R.E., and de Muizon, C. 2001. Evolutionary history of whales: a review, p. 169-234. In Mazin, J.-M. and de Buffrenil, V. (eds.), Secondary Adaptation of Tetrapods to Life in Water. Pfeil, München, Germany.
Fordyce, R.E. and Jones, C.M. 1990. The history of penguins, and new fossil penguin material from New Zealand, p. 419-446. In Davis, L.S. and Darby, J.D. (eds.), Penguin Biology. Academic Press, San Diego.
Fordyce, R.E. and Maxwell, P.A. 2003. Canterbury Basin paleontology and stratigraphy (Field trip 8, Annual Conference, Geological Society of New Zealand). Geological Society of New Zealand miscellaneous publication, 116b(4):1-18.
Fordyce, R.E., Quilty, P.G., and Daniels, J. 2002. Australodelphis mirus, a bizarre new toothless ziphild-like fossil dolphin (Cetacea : Delphinidae) from the Pliocene of Vestfold Hills, East Antarctica. Antarctic Science, 14(1):37-54.
Fraser, F.C. and Purves, P.E. 1960. Hearing in cetaceans: evolution of the accessory air sacs and the structure of the outer and middle ear in recent cetaceans. Bulletin of the British Museum of Natural History (Zoology) 7:1-140.
Gage, M. 1957. The geology of Waitaki subdivision. New Zealand Geological Survey Bulletin , 55:1-135.
Geisler, J.H., Colbert, M.W., and Carew, J.L. 2014. A new fossil species supports an early origin for toothed whale echolocation. Nature, 508(7496):383-386.
Geisler, J.H., Godfrey, S.J., and Lambert, O. 2012. A new genus and species of late Miocene inioid (Cetacea, Odontoceti) from the Meherrin River, North Carolina, USA. Journal of Vertebrate Paleontology, 32(1):198-211.
Geisler, J.H. and Luo, Z. 1996. The petrosal and inner ear of Herpetocetus sp.(Mammalia: Cetacea) and their implications for the phylogeny and hearing of archaic mysticetes. Journal of Paleontology:1045-1066.
Geisler, J.H., McGowen, M.R., Yang, G., and Gatesy, J. 2011. A supermatrix analysis of genomic, morphological, and paleontological data from crown Cetacea. BMC Evolutionary Biology, 11:112.
Geisler, J.H. and Sanders, A.E. 2003. Morphological evidence for the phylogeny of Cetacea. Journal of Mammalian Evolution, 10(1/2):23-129.
Geisler, J.H., Sanders, A.E., and Luo, Z.-X. 2005. A new protocetid whale (Cetacea: Archaeoceti) from the late middle Eocene of South Carolina. American Museum Novitates, 3480:1-68.
Godfrey, S.J. 2013. On the olfactory apparatus in the Miocene odontocete Squalodon sp. (Squalodontidae). Comptes Rendus Palevol, 12(7):519-530.
Goloboff, P.A., Farris, J.S., and Nixon, K.C. 2008. TNT, a free program for phylogenetic analysis. Cladistics, 24(5):774-786.
Gottfried, M.D., Fordyce, R.E., and Rust, S. 2012. A new billfish (Perciformes, Xiphioidei) from the late Oligocene of New Zealand. Journal of Vertebrate Paleontology, 32(1):27-34.
Graham, I.J., Morgans, H.E.G., Waghorn, D.B., Trotter, J.A., and Whitford, D.J. 2000. Strontium isotope stratigraphy of the Oligocene-Miocene Otekaike Limestone (Trig Z section) in southern New Zealand: age of the Duntroonian/Waitakian Stage boundary. New Zealand Journal of Geology and Geophysics, 43(3):335-347.
Gray, J.E. 1863. On the arrangement of the cetaceans. Proceedings of the Scientific Meetings of the Zoological Society of London, 3:197-202.
Gutstein, C.S., Figueroa-Bravo, C.P., Pyenson, N.D., Yury-Yañez, R.E., Cozzuol, M.A., and Canals, M. 2014. High frequency echolocation, ear morphology, and the marine-freshwater transition: A comparative study of extant and extinct toothed whales. Palaeogeography, Palaeoclimatology, Palaeoecology, 400:62-74.
Heyning, J.E. 1989. Comparative facial anatomy of beaked whales (Ziphiidae) and a systematic revision among the families of extant Odontoceti. Contributions in Science, Natural History Museum of Los Angeles County, 405:1-64.
Heyning, J.E. 1997. Sperm whale phylogeny revisited: analysis of the morphological evidence. Marine Mammal Science, 13(4):596-613.
Howell, A.B. 1927. Contribution to the anatomy of the Chinese finless porpoise Neomeris phocaenoides. Proceedings of The United States National Museum, 70(13):1-43.
Howell, A.B. 1930. Myology of the narwhal (Monodon monoceros). American Journal of Anatomy , 46(2):187-215.
Hulbert Jr., R.C., Petkewich, R.M., Bishop, G.A., Bukry, D., and Aleshire, D.P. 1998. A new middle Eocene protocetid whale (Mammalia: Cetacea: Archaeoceti) and associated biota from Georgia. Journal of Paleontology, 72(5):907-927.
Ichishima, H. 2011. Do cetaceans have the mesethmoid? Memoir of the Fukui Prefectural Dinosaur Museum, 10:63-75.
Ichishima, H. and Kimura, M. 2005. Haborophocoena toyoshimai, a new Early Pliocene porpoise (Cetacea; Phocoenidae) from Hokkaido, Japan. Journal of Vertebrate Paleontology, 25(3):655-664.
International Code of Zoological Nomenclature 1999. International code of zoological nomenclature. International Trust for Zoological Nomenclature London, UK.
Jefferson, T.A. and Newcomer, M.W. 1993. Lissodelphis borealis. Mammalian Species, 425:1-6.
Jefferson, T.A., Webber, M.A., and Pitman, R.L. 2008. Marine Mammals of the World: A Comprehensive Guide to Their Identification. Academic Press, Oxford, UK.
Jenkins, D.G. 1965. Planktonic foraminiferal zones and new taxa from the Danian to Lower Miocene of New Zealand. New Zealand Journal of Geology and Geophysics, 8(6):1088-1126.
Kasuya, T. 1973. Systematic consideration of recent toothed whales based on the morphology of tympano-periotic bone. Scientific Reports of the Whales Research Institute , Tokyo, 25:1-103.
Kellogg, R. 1923. Description of an apparently new toothed cetacean from South Carolina. Smithsonian Miscellaneous Collections, 76:1-7.
Kellogg, R. 1925. Additions to the Tertiary history of the pelagic mammals on the Pacific coast of North America. Proceedings of the U.S. National Museum, 66(26):1-40.
Kellogg, A.R. 1926. Supplementary observations on the skull of the fossil porpoise Zarhachis flagellator. Proceedings of the U.S. National Museum, 67(28):1-18.
Kellogg, R. 1927. Kentriodon pernix, a Miocene porpoise from Maryland. Proceedings of the United States National Museum, 69(19):1-14.
Kellogg, R. 1936. A review of the Archaeoceti. Carnegie Institution of Washington publication , 482:1-366.
Kellogg, R. 1957. Two additional Miocene porpoises from the Calvert Cliffs, Maryland. Proceedings of the U.S. National Museum, 107(3387):279-337.
Klima, M., Oelschlaeger, H.A., and Wünsch, D. 1980. Morphology of the pectoral girdle in the Amazon dolphin Inia geoffrensis with special reference to the shoulder joint and the movements of the flippers. Zeitschrift Saugertierkunde, 45:288-309.
Kuzmin, A.A. 1976. Embryogenesis of the osseous skull of the sperm whale (Physeter macrocephalus Linnaeus, 1758). Invest Cetacea, 7:187-202.
Lambert, O. 2005. Phylogenetic affinities of the long-snouted dolphin Eurhinodelphis (Cetacea, Odontoceti) from the Miocene of Antwerp, Belgium. Palaeontology, 48(3):653-679.
Lambert, O. 2008. A new porpoise (Cetacea, Odontoceti, Phocoenidae) from the Pliocene of the North Sea. Journal of Vertebrate Paleontology, 28(3):863-872.
Lambert, O., Bianucci, G., and de Muizon, C. 2008. A new stem-sperm whale (Cetacea, Odontoceti, Physeteroidea) from the latest Miocene of Peru. Comptes Rendus Palevol, 7(6):361-369.
Lambert, O., Bianucci, G., and Post, K. 2010. Tusk-bearing beaked whales from the Miocene of Peru: sexual dimorphism in fossil ziphiids? Journal of Mammalogy, 91(1):19-26.
Lambert, O., Bianucci, G., and Urbina, M. 2014. Huaridelphis raimondii, a new early Miocene Squalodelphinidae (Cetacea, Odontoceti) from the Chilcatay Formation, Peru. Journal of Vertebrate Paleontology, 34(5):987-1004.
Lambert, O. and Post, K. 2005. First European pontoporiid dolphins (Mammalia: Cetacea, Odontoceti) from the Miocene of Belgium and The Netherlands. Deinsea, 11:7-20.
Lancaster, W. 1986. The taphonomy of an archaeocete skeleton and its associated fauna. Gulf Coast Association of Geological Societies publication, 119:119-131.
Leatherwood, S., Reeves, R.R., and Foster, L. 1983. Sierra Club handbook of whales and dolphins. Sierra Club Books, San Francisco.
Loch, C., Kieser, J.A., and Fordyce, R.E. 2015. Enamel Ultrastructure in Fossil Cetaceans (Cetacea: Archaeoceti and Odontoceti). PLoS ONE, 10(1):e0116557.
Luo, Z. and Marsh, K. 1996. Petrosal (periotic) and inner ear of a Pliocene kogiine whale (Kogiinae, Odontoceti): implications on relationships and hearing evolution of toothed whales. Journal of Vertebrate Paleontology, 16(2):328-348.
Maddison, W.P. and Maddison, D.R. 2011. Mesquite: a modular system for evolutionary analysis. Available at http://mesquiteproject.org.
Marsh, H., Lloze, R., Heinsohn, G.E., and Kasuya, T. 1989. Irrawaddy dolphin - Orcaella brevirostris, (Gray, 1866), p. 101-118. In Ridgway, S.H., and Harrison, S. (eds.), Handbook of marine mammals. Volume 4: river dolphins and the larger toothed whales. Academic Press, London.
McArthur, J.M., Howarth, R.J., and Shields, G.A. 2012. Chapter 7 - Strontium isotope stratigraphy, p. 127-144. In Gradstein, F.M., Ogg, J.G., Schmitz, M., and Ogg, G. (eds.), The Geologic Time Scale. Elsevier, Boston.
Mead, J.G. 1975. Anatomy of the external nasal passages and facial complex in the Delphinidae (Mammalia, Cetacea). Smithsonian Contributions to Zoology, 207:1-72.
Mead, J. 1989. Shepherd’s beaked whale Tasmacetus shepherdi Oliver, 1937. Handbook of marine mammals, 4:309-320.
Mead, J.G., and Fordyce, R.E. 2009. The therian skull: a lexicon with emphasis on the odontocetes. Smithsonian Contributions to Zoology, 627:1-248.
Mellor, L., Cooper, L.N., Torre, J., and Brownell, R.L., Jr. 2009. Paedomorphic ossification in porpoises with an emphasis on the Vaquita ( Phocoena sinus). Aquatic Mammals, 35(2):193-202.
Messenger, S. 1994. Phylogenetic relationships of platanistoid river dolphins (Odontoceti, Cetacea): assessing the significance of fossil taxa. Contributions in Marine Mammal Paleontology Honoring Frank Whitmore Jr. Proceedings of the San Diego Society of Natural History:125-133.
Messenger, S.L. and McGuire, J.A. 1998. Morphology, molecules, and the phylogenetics of cetaceans. Systematic Biology, 47(1):90-124.
Miller, G.S. 1918. A new river-dolphin from China. Smithsonian Miscellaneous Collections, 68(9):1-12.
Miller, G.S. 1923. The telescoping of the cetacean skull. Smithsonian Miscellaneous Collections , 76(5):1-70.
Moore, J.C. 1968. Relationships among the living genera of beaked whales with classifications, diagnoses and keys. Fieldiana: Zoology, 53(4):509-598.
Murakami, M., Shimada, C., Hikida, Y., and Hirano, H. 2012a. A new basal porpoise, Pterophocaena nishinoi (Cetacea, Odontoceti, Delphinoidea), from the upper Miocene of Japan and its phylogenetic relationships. Journal of Vertebrate Paleontology, 32(5):1157-1171.
Murakami, M., Shimada, C., Hikida, Y., and Hirano, H. 2012b. Two new extinct basal phocoenids (Cetacea, Odontoceti, Delphinoidea), from the upper Miocene Koetoi Formation of Japan and their phylogenetic significance. Journal of Vertebrate Paleontology , 32(5):1172-1185.
Murakami, M., Shimada, C., Hikida, Y., Soeda, Y., and Hirano, H. 2014. Eodelphis kabatensis , a new name for the oldest true dolphin Stenella kabatensis Horikawa, 1977 (Cetacea, Odontoceti, Delphinidae), from the upper Miocene of Japan, and the phylogeny and paleobiogeography of Delphinoidea. Journal of Vertebrate Paleontology, 34(3):491-511.
Noble, B. and Fraser, F. 1971. Description of a skeleton and supplementary notes on the skull of a rare porpoise Phocoena sinus Norris & McFarland 1958. Journal of Natural History, 5(4):447-464.
Norris, K.S. 1964. Some problems of echolocation in cetaceans, p. 317-336. In Tavolga, W.N. (ed.), Marine bio-acoustics. MacMillan, New York.
Norris, K.S. and Harvey, G.W. 1972. A theory for the function of the spermaceti organ of the sperm whale (Physeter catodon L). NASA Special Publication, 262:397.
Nweeia, M.T., Eichmiller, F.C., Hauschka, P.V., Tyler, E., Mead, J.G., Potter, C.W., Angnatsiak, D.P., Richard, P.R., Orr, J.R., and Black, S.R. 2012. Vestigial tooth anatomy and tusk nomenclature for Monodon monoceros. The Anatomical Record, 295(6):1006-1016.
Perrin, W.F. 1975. Variation of spotted and spinner porpoise (Genus, Stenella) in the eastern Pacific and Hawaii. Bulletin of the Scripps Institution of Oceanography, 21:1-206.
Pyenson, N.D. and Sponberg, S.N. 2011. Reconstructing body size in extinct crown Cetacea (Neoceti) using allometry, phylogenetic methods and tests from the fossil record. Journal of Mammalian Evolution, 18(4):269-288.
Reeves, R., Stewart, B., Clapham, P., and Powell, J. 2002. Marine mammals of the world. Chanticleer Press, Inc, New York.
Reidenberg, J.S. and Laitman, J.T. 1994. Anatomy of the hyoid apparatus in Odontoceli (toothed whales): Specializations of their skeleton and musculature compared with those of terrestrial mammals. The Anatomical Record, 240(4):598-624.
Rice, D.W. and Wolman, A.A. 1990. The stomach of Kogia breviceps. Journal of Mammalogy:237-242.
Rommel, S. 1990. Osteology of the bottlenose dolphin, p. 29-49. In Leatherwood, S. and Reeves, R.R. (eds.), The Bottlenose Dolphin. Academic Press Inc, San Diego, New York.
Sanders, A.E. and Barnes, L.G. 2002. Paleontology of the late Oligocene Ashley and Chandler Bridge formations of South Carolina, 2: Micromysticetus rothauseni, a primitive cetotheriid mysticete (Mammalia: Cetacea). Smithsonian Contributions to Paleobiology, 93:271-293.
Schäfer, W. and Craig, G.Y. 1972. Ecology and palaeoecology of marine environments. Oliver and Boyd, Edinburgh.
Schenkkan, E. 1971. The occurrence and position of the “connecting sac” in the nasal tract complex of small odontocetes (Mammalia, Cetacea). Beaufortia, 19(246):37-43.
Schulte, W. 1916. Anatomy of a foetus of Balaenopterus Borealis. Memoirs of the American Museum of Natural History, 1(6):389-502.
Schulte, H.v.W. 1917. The skull of Kogia breviceps Blainv. Bulletin of the American Museum of Natural History, 37:361-404.
Schulte, H.v.W. and Smith, M.D.F. 1918. The external characters, skeletal muscles, and peripheral nerves of Kogia breviceps (Blainville). Bulletin American Museum of Natural History, 38:7-72.
Simpson, G.G. 1945. The principles of classification and a classification of mammals. Bulletin of the American Museum of Natural History, 85:1-350.
Smith, G., Browne, K., and Gaskin, D. 1976. Functional myology of the harbour porpoise, Phocoena phocoena (L.). Canadian Journal of Zoology, 54(5):716-729.
Steeman, M.E., Hebsgaard, M.B., Fordyce, R.E., Ho, S.Y.W., Rabosky, D.L., Nielsen, R., Rahbek, C., Glenner, H., Sørensen, M.V., and Willerslev, E. 2009. Radiation of extant cetaceans driven by restructuring of the oceans. Systematic Biology, 58(6):573-585.
Strickler, T.L. 1978. Myology of the shoulder of Pontoporia blainvillei, including a review of the literature on shoulder morphology in the Cetacea. American Journal of Anatomy, 152(3):419-431.
Struthers, J. 1872. On the cervical vertebrae and their articulations in Fin-whales. Journal of Anatomy and Physiology, 7:1-55.
Tanaka, Y. and Fordyce, R.E. 2014. Fossil dolphin Otekaikea marplesi (latest Oligocene, New Zealand) expands the morphological and taxonomic diversity of Oligocene cetaceans. PLoS ONE, 9(9):e107972.
True, F.W. 1904. The whalebone whales of the western North Atlantic compared with those occurring in European waters with some observations on the species of the North Pacific. Smithson Contribution to Knowledge, 33:1-332.
True, F.W. 1912. Description of a New Fossil Porpoise of the Genus Delphinodon from the Miocene Formation of Maryland. Journal of the Academy of Natural Sciences of Philadelphia, 2:165-194.
Uhen, M.D. 2008. A new Xenorophus ‐ like odontocete cetacean from the Oligocene of North Carolina and a discussion of the basal odontocete radiation. Journal of Systematic Palaeontology, 6(4):433-452.
Van Valen, L. 1968. Monophyly or diphyly in the origin of whales. Evolution, 22:37-41.
Vandenberghe, N., Hilgen, F.J., Speijer, R.P., Ogg, J.G., Gradstein, F.M., Hammer, O., Hollis, C.J., and Hooker, J.J. 2012. Chapter 28 - The Paleogene Period, p. 855-921. In Gradstein, F.M., Ogg, J.G., Schmitz, M., and Ogg, G. (eds.), The Geologic Time Scale. Elsevier, Boston.
Vélez-Juarbe, J. and Pyenson, N.D. 2012. Bohaskaia monodontoides, a new monodontid (Cetacea, Odontoceti, Delphinoidea) from the Pliocene of the western North Atlantic Ocean. Journal of Vertebrate Paleontology, 32(2):476-484.
Whitmore, F.C. and Sanders, A.E. 1977. Review of the Oligocene Cetacea. Systematic Zoology , 25(4):304-320.
Wilson, L.E. 1973. A delphinid (Mammalia, Cetacea) from the Miocene of Palos Verdes Hills, California. University of California Press.
Yablokov, A. 1964. Convergence or parallelism in the evolution of cetaceans. International Geology Review, 7(8):1461-1468.
Yamada, M. 1953. Contribution to the anatomy of the organ of hearing of whales. Scientific Reports of the Whales Research Institute Tokyo, 8:1-79.
Zhou, K. 1982. Classification and phylogeny of the superfamily Platanistoidea, with notes on evidence of the monophyly of the Cetacea. Scientific Reports of the Whales Research Institute Tokyo, 34: 93-108.
Zhou, K., Qian, W., and Li, Y. 1979. Recent advances in the study of the baiji, Lipotes vexillifer. J. Nanjing Normal College (Natural Science), 1:8-13.

