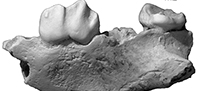
 A tiny new marsupial lion (Marsupialia, Thylacoleonidae) from the early Miocene of Australia
A tiny new marsupial lion (Marsupialia, Thylacoleonidae) from the early Miocene of Australia
Article number: 19.2.26A
https://doi.org/10.26879/632
Copyright Palaeontological Association, July 2016
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Submission: 24 January 2016. Acceptance: 20 May 2016
{flike id=1490}
ABSTRACT
Microleo attenboroughi, a new genus and species of diminutive marsupial lion (Marsupialia: Thylacoleonidae), is described from early Miocene freshwater limestones in the Riversleigh World Heritage Area, northwestern Queensland, Australia. A broken palate that retains incomplete cheektooth rows demonstrates that this new, very small marsupial lion possessed the elongate, trenchant P3 and predominantly subtriangular upper molars characteristic of thylacoleonids, while other features of the premolar support its placement in a new genus. Phylogenetic analysis suggests that Microleo attenboroughi is the sister taxon to all other thylacoleonids, and that Thylacoleonidae may lie outside Vombatomorphia as the sister taxon of all other wombat-like marsupials including koalas. However, given limited data about the cranial morphology of M. attenboroughi, Thylacoleonidae is concluded here, conservatively, to be part of the vombatomorphian clade. This new thylacoleonid brings to three the number of marsupial lion species that have been recovered from early Miocene deposits at Riversleigh and indicates a level of diversity previously not seen for this group. It is likely that the different size and morphology of the three sympatric taxa reflects niche partitioning and hence reduced competition. Thylacoleonids may have been the dominant arboreal predators of Cenozoic Australia.
Anna K. Gillespie*, Palaeontology, Geobiology and Earth Archives (PANGEA) Research Centre, School of Biological, Earth and Environmental Sciences, University of New South Wales, Sydney 2052, Australia; a.gillespie@unsw.edu.au, *corresponding author
Michael Archer, Palaeontology, Geobiology and Earth Archives (PANGEA) Research Centre, School of Biological, Earth and Environmental Sciences, University of New South Wales, Sydney 2052, Australia; m.archer@unsw.edu.au
Suzanne J. Hand, Palaeontology, Geobiology and Earth Archives (PANGEA) Research Centre, School of Biological, Earth and Environmental Sciences, University of New South Wales, Sydney 2052, Australia; s.hand@unsw.edu.au
Keywords: Thylacoleonidae; marsupial lion; new genus; new species; early Miocene; Riversleigh
Final citation: Gillespie, Anna K., Archer, Michael, and Hand, Suzanne J. 2016. A tiny new marsupial lion (Marsupialia, Thylacoleonidae) from the early Miocene of Australia. Palaeontologia Electronica 19.2.29A: 1-25. https://doi.org/10.26879/632
palaeo-electronica.org/content/2016/1490-new-marsupial-lion
http://zoobank.org/723FFDDC-F477-4BAB-9891-297A2959A2DB
INTRODUCTION
Marsupial lions (Thylacoleonidae) are extinct marsupials found in Australian late Oligocene to Pleistocene deposits. The family first gained renown through the description of the Pleistocene species Thylacoleocarnifex (Owen, 1858), a lion-sized animal (Wroe et al., 1999) that possessed uniquely hypertrophied third premolars resembling a pair of bolt-cutting blades. Thylacoleo carnifex has been shown (Wroe et al., 2005; Wroe, 2008) to have had a bite force quotient only slightly less than that of the African lion. To date, three genera and eight marsupial lion species have been described. Of these, T. carnifex is relatively common in the fossil record. Earlier members of the family that include domestic cat-sized species of the genus Priscileo (Rauscher, 1987; Gillespie, 1997) from the late Oligocene and early Miocene, the dog to leopard-sized species of Wakaleo (Clemens and Plane, 1974; Archer and Rich, 1982; Murray et al., 1987; Gillespie et al., 2014;Yates, 2015) from the early, middle and late Miocene, and the Pliocene species of Thylacoleo (Bartholomai, 1962; Pledge, 1977; Archer and Dawson, 1982) are poorly known. The diversity of the family throughout this time has been low with, until now, no more than two species known to co-exist at any point in time.
The scarce and fragmentary early fossil record of the group has resulted in a poor understanding of this family’s origins and early evolution. Although there is general agreement regarding the phylogenetic relationships of species within the genera, the evolutionary pathways leading to these lineages are still uncertain. Wakaleo and Thylacoleo have been proposed as sister taxa (Rauscher, 1987; Archer and Dawson, 1982) with Priscileo regarded as their sister taxon (Rauscher, 1987). Alternatively, others have proposed a sister-group relationship for Priscileo and Thylacoleo (Murray et al., 1987). The position of the family Thylacoleonidae among the largely wombat-like families that comprise Vombatiformes has also been debated (Ride, 1964; Aplin and Archer, 1987; Murray et al., 1987; Marshall et al., 1990).
The Riversleigh World Heritage Area (WHA) in northwestern Queensland has already been known to have hosted four thylacoleonid species (Gillespie, 1997; Gillespie et al., 2014). Now remains of a fifth, represented by paired left and right maxillary fragments and a lower molar, have been recovered from early Miocene sediments. The teeth of this species are the smallest known for any thylacoleonid. Furthermore, although the general morphology of the premolar and molars are characteristically those of a marsupial lion, some features of the dentition are unique within the family. This new taxon is the focus of the present paper.
MATERIALS AND METHODS
The material described here was recovered from one small block of limestone etched by acetic acid at the Vertebrate Palaeontology Laboratory of the University of New South Wales, and is registered in the palaeontology collection of the Queensland Museum. Serial homology of marsupial premolars and molars follows Flower (1867).
Institutional Abbreviations. AM F, palaeontological collection of the Australian Museum, Sydney; AR, palaeontological collection of the Vertebrate Palaeontology Laboratory, University of New South Wales, Sydney; CPC, Commonwealth palaeontological collection in the Australian Geological Survey Organisation, Canberra; NTM P, palaeontological collection of the Museums and Art Galleries of the Northern Territory, Alice Springs; QM F, palaeontological collection of the Queensland Museum, Brisbane; SAM P, palaeontological collection of the South Australian Museum, Adelaide.
SYSTEMATIC PALAEONTOLOGY
Class MAMMALIA Linnaeus, 1758
Superorder MARSUPIALIA Illiger, 1811
Order DIPROTODONTIA Owen, 1866
Suborder VOMBATIFORMES Woodburne, 1984
Family THYLACOLEONIDAE Gill, 1872
Genus MICROLEO gen. nov.
zoobank.org/24A55B6E-B7DE-4665-836A-907BD13819DB
Type Species.Microleo attenboroughi new genus and species
Etymology. From micro meaning small (Greek) and leo meaning lion (Latin). The species name honours Sir David Attenborough for his dedication and enthusiasm in promoting the natural history of the world and the palaeontological treasures of the Riversleigh World Heritage Area in particular.
Microleo attenboroughi new genus and species
Figure 1, Figure 2
zoobank.org/922FDD25-1D5D-40AD-9F49-94B57853CE51
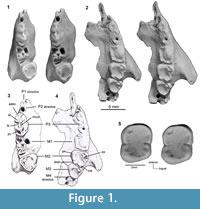 Holotype. QM F41143, an incomplete palate consisting of partial left and right maxillae (Figure 1.1-4, Figure 2). The left maxilla preserves M2-3, roots for P3-M1, alveoli for M4, and the maxillary root of the zygomatic arch. The right maxilla preserves P3-M2, alveoli for P1-2 and M1. QM F42676, paratype, is a left m3 (Figure 1.5).
Holotype. QM F41143, an incomplete palate consisting of partial left and right maxillae (Figure 1.1-4, Figure 2). The left maxilla preserves M2-3, roots for P3-M1, alveoli for M4, and the maxillary root of the zygomatic arch. The right maxilla preserves P3-M2, alveoli for P1-2 and M1. QM F42676, paratype, is a left m3 (Figure 1.5).
Type Locality and Horizon. The Type Locality is Neville’s Garden Site, D Site Plateau, Riversleigh World Heritage Area, Boodjamulla National Park, northwestern Queensland. Neville’s Garden Site is early Miocene in age (radiometrically dated at 18.24±0.29 Ma and 17.85±0.13 Ma: Archer et al., 1997; Arena, 2004; Travouillon et al., 2006; Woodhead et al., 2016).
Diagnosis.Microleo attenboroughi is attributed to Thylacoleonidae on the basis of its bicuspid, blade-like P3 and its weakly-crenulated, subtriangular bunodont molars. Microleo attenboroughi is smaller than all other thylacoleonids (see below). Generic distinction is based primarily on its unique P3 morphology.
Microleo attenboroughi differs from all other marsupial lions as follows in the morphology of its premolars. The P2 alveolus is large relative to that for P3 and occurs in an anterior rather than anterolingual position relative to P3. P3 is fusiform and has a buccally-convex, longitudinally-orientated blade, small medial cuspule between the anterior and posterior cusps, a posterior cusp that is markedly taller than the posterior end of the crown, anterior part of central v-shaped blade much shorter than the posterior part, a steep posterior longitudinal blade, a deep posterobuccal basin enclosed by a well-developed posterobuccal crest, lingual mid-crown basal swellings that are separated by a narrow groove, a rounded anterobuccal crest that extends anterodorsally from the anterior cusp, a short posterolingual crest that extends anterodorsally from the posterior cusp, a sinuous valley between the anterolingual and posterolingual crests, little buccal exposure of the roots, and wear facets on the longitudinal blade that are directed posterolingually rather than lingually. In terms of molar morphology, it differs from all other thylacoleonids in the following features. The molars are weakly-crenulated. The M2 differs from all other thylacoleonids in the following features; the protocone is taller than the paracone, metaconule is moderately-developed, lingual margin anterior to metaconule has a distinct notch, postmetacrista does not merge with postmetaconulecrista and the crown below the paracone lacks buccal inflation. M3 has a pyramidal paracone near the anterobuccal corner of the trigon basin. The m3 talonid basin has distinct grooves near the midpoint of its buccal and lingual margins.
Microleo attenboroughi is further distinguished from species of Priscileo and Wakaleo as follows: P3 has an accessory anterobuccal crest, lacks basal lingual development of the anterior crest, and has a narrow anterolingual crest (vs. lingual inflation or cuspule); m3 is rectangular with a trigonid basin and a very posteriorly-broad talonid basin. Microleo attenboroughi is also distinguished from species of Wakaleo and Thylacoleo in having the lingual and buccal margins of M2 similar in height. It is further distinguished from species of Thylacoleo in retaining M3-4, in that P3 has a prominent posterior cusp and the posterolingual crest does not reach the crown base.
Description
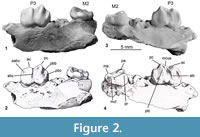 General. The holotype of Microleo attenboroughi is a partial palate consisting of portions of both maxillae (Figure 1.1-4, Figure 2). The right fragment measures 15.2 mm in length and preserves P3 and M2. Alveoli are present for M1. Two small premolar alveoli lie anterior to P3, which we interpret as housing a single-rooted P1 and P2. The left maxilla fragment measures 22.1 mm anteroposteriorly and preserves M2-3. The crowns of P3 and M1 have broken away at the level of the palate. Alveoli for M4 are present; only the anteromedial one is complete, the lateral and posterior sockets are broken. The cheektooth row is gently arcuate, with the molars having progressively more lingual positions relative to the preceding teeth. The region anterior to P3 is damaged, exposing the internal walls of the P2 alveolus and infraorbital canal. Only narrow portions (3-5 mm) of the palate medial to both tooth rows are preserved, and the presence of any palatal vacuities cannot be discerned. Fragments of bone from the posterior cranium are preserved and include a partial atlas. Dental dimensions are provided in Table 1.
General. The holotype of Microleo attenboroughi is a partial palate consisting of portions of both maxillae (Figure 1.1-4, Figure 2). The right fragment measures 15.2 mm in length and preserves P3 and M2. Alveoli are present for M1. Two small premolar alveoli lie anterior to P3, which we interpret as housing a single-rooted P1 and P2. The left maxilla fragment measures 22.1 mm anteroposteriorly and preserves M2-3. The crowns of P3 and M1 have broken away at the level of the palate. Alveoli for M4 are present; only the anteromedial one is complete, the lateral and posterior sockets are broken. The cheektooth row is gently arcuate, with the molars having progressively more lingual positions relative to the preceding teeth. The region anterior to P3 is damaged, exposing the internal walls of the P2 alveolus and infraorbital canal. Only narrow portions (3-5 mm) of the palate medial to both tooth rows are preserved, and the presence of any palatal vacuities cannot be discerned. Fragments of bone from the posterior cranium are preserved and include a partial atlas. Dental dimensions are provided in Table 1.
Dentition. P1 and P2 alveoli are small and round. P1 alveolus is broken but appears to be similar in size to P2. P2 alveolus is 1.0 mm long and 0.9 mm wide and sits at the anterior base of P3, directly behind P1.
P3 is sectorial and is fusiform in occlusal view. The crown is broadest centrally where there is a large lingual basal swelling at the level of the junction of the anterior and posterior roots. A second, smaller, lingual swelling lies directly anterior to the large swelling and is separated from the latter by a narrow groove. In occlusal view, the buccal side is gently undulating, the two main buccal crests forming slight swellings of the surface. A longitudinal blade extends the length of the crown, connecting two principal cusps: an anterior cusp that lies at the anterior third, and a posterior cusp that lies at the posterior third. In profile, crown forms an M-shape. Anterior cusp is shorter than posterior, although it is evident that the height of the former has been reduced by heavy wear; the broad, posterolingually-directed wear facet and exposed dentine suggest the anterior cusp was originally much taller. In occlusal view, the longitudinal blade that links the two cusps exhibits a weak buccal bow. In profile, it forms an uneven V- shape, having a very short anterior portion and a long posterior portion. About half way along the posterior portion, a small swelling is evident on both sides of the blade. The occlusal edge at this point forms a small circle indicating the presence of worn a medial cuspule. Slight wear facets are also discernible on the lingual edge of the longitudinal blade anterior to both the anterior and posterior cusps, and posterior to the posterior cusp.The anterior vertical crest descends from the anterior cusp at a gentle angle for a short distance, then curves medially and descends steeply to the base of the crown. On the anterobuccal surface of the crown, near the point where the anterior vertical crest descends, there is a short vertical accessory anterobuccal crest. This crest weakens as it nears the occlusal edge, where it again thickens and is cuspule-like. On the buccal surface, rounded, vertical crests descend in a slightly anterior direction from each of the principal cusps. These become weaker around the midcrown level and eventually fade near the base. The posterior longitudinal blade descends steeply from the posterior cusp to the posterior margin of the crown, where it then curves laterally forming a discrete posterobuccal crest that continues to descend for a short distance in an anterior direction, terminating at the lower third of the crown. This crest creates a small, deep, basin on the posterobuccal surface between the posterior cusp and the posterior margin of the tooth. On the lingual surface, a slightly anteriorly-directed crest (anterolingual crest) extends from the anterior cusp to a small basal swelling over the anterior root. Between this crest and the anterior margin the crown is gently concave.From the posterior cusp, a short, rounded, posterolingual crest extends in an anterior direction and merges with the large basal swelling that lies above the junction of the two roots. Posterior to the posterolingual crest, the lingual flank of the crown is broadly concave. A narrow, sinuous valley lies between the anterolingual and posterolingual crests. This valley is deepest directly anterior to the posterolingual crest, and becomes broader and shallower in its basal half. A weak, rounded, cingulum-like structure courses anteriorly and posteriorly from each basal swelling: the anterior part merges with the base of the anterior vertical crest, and the posterior part ascends the posterior edge of the crown to connect with the end of the longitudinal blade. P3 has two roots, the posterior being slightly larger than the anterior.
The crown of M1 is not preserved. In the left maxillary fragment, the M1 crown is broken away at the level of the palate, indicating it has a quadrilateral basal outline. M1 has three roots: the medial and posterior roots are subequal in size and larger than the anterior root.
M2 is trapezoidal and dominated by a large trigon basin. The paracone is located approximately 1mm internally from the anterobuccal corner, is pyramidal, similar in height to the protocone and taller than the metacone. From the paracone, a short preparacrista extends to the anterior edge of the tooth. A postparacrista curves posterobuccally to connect with the buccal margin midway along the crown. These connecting cristae form the lingual margin of a small basin that lies between the paracone and the anterobuccal edge of the tooth. A variably developed transverse crest connects the paracone and the protocone. This crest is unbroken on the right M2, but on the left tooth it is interrupted by a narrow notch about midway between the cusps. The lateral portion of this crest bows anteriorly. A narrow anterior cingulum lies at the anterior margin of the crown and extends between the preparacrista and the protocone. The protocone shows slightly more wear than the paracone. Below the protocone the crown bulges lingually. A gently rounded postprotocrista extends posterobuccally toward the metaconule. About two-thirds of the way to the metaconule, the postprotocrista terminates at a narrow notch, which separates it from the short premetaconulecrista. The metaconule is gently rounded and positioned on a distinct posterior projection of the crown. This projection gives the crown its trapezoidal shape. The metacone is shorter than the paracone and protocone, but taller than the metaconule. Two short, rounded, transverse crests extend towards each other from the metacone and metaconule. These two crests are separated by a narrow groove that connects with the narrow basin of the posterior cingulum. The posterior cingulum is short and extends around the posterior edge from the metacone to the metaconule.Very small grooves are found on the periphery of the trigon basin, suggesting that it may have been weakly crenulated, and it is possible that wear has obliterated crenulations that may have been present in the centre of the basin. M2 has three roots.
M3 is triangular and smaller than M2. M3 abuts and is slightly lingually rotated relative to M2. The occlusal surface is dominated by a large trigon basin. The paracone and protocone are subequal in height. The paracone is pyramidal in contrast to the other cusps which are gently rounded. From the paracone a short preparacrista extends anteriorly to the edge of the crown and a postparacrista curves posterobuccally connecting with the buccal margin half way between the paracone and metacone. These two cristae and the anterobuccal edge enclose a small, shallow, anterobuccal basin. A short crest also lies on the lingual face of the paracone and extends about one quarter of the distance to the protocone. The anterior edge is broadly rounded from this crest to the protocone.The base of the protocone is gently rounded, and the underlying crown is deep but not lingually-inflated. A postprotocrista courses posteriorly around the margin to connect with the metacone. The metacone is weakly developed.The trigon basin appears to lack crenulations; however, small grooves on its buccal margin and small, shallow, depressions near the protocone suggest that originally it may have been weakly crenulated and that these crenulations have been lost through wear. There is no posterior cingulum. M3 is anchored by three roots.
Facial Skeleton. The left maxillary fragment is the larger of the two fragments and preserves the region just dorsal and posterior to the infraorbital foramen and the wing of the zygomatic arch. The medial edge of the maxillary fragment is uneven and the presence of, or extent of a posterior palatal fenestra cannot be determined. The infraorbital foramen is directed anteriorly and forms the anterior margin of a triangular infraorbital canal. Dorsal and posterior to the foramen the maxilla is convex and at the base of the zygomatic wing it becomes slightly concave. The zygomatic arch projects posterolaterally dorsal to M1-M2. The lateral extremity of the maxillary root bears a narrow groove near its inferior border marking the articulation area of the jugal. A part of the jugal is preserved on the anterodorsal surface of the zygomatic arch.
The internal dorsal surface of the maxilla bears three fossae. The anterior fossa lies anterior to the level of the infraorbital foramen and is only partially preserved. The middle and posterior fossae are separated by a remnant of the lacrimal. Along the lateral wall of the anterior fossa, a shallow channel extends posteriorly and is confluent with a sulcus (nasolacrimal canal) that is present on the lateral wall of the middle fossa. Posteriorly, the sulcus terminates just anterior to the fragment of lacrimal that lies at the base of the anterior wall of the orbit.The posterior fossa is concave and broad and forms the anterior floor of the orbit. The maxillary foramen lies near the posteromedial margin of the fossa and is totally enclosed by the maxilla (as it is in Wakaleo vanderleueri). Indentations on the maxillary bone that forms the roof of the foramen indicate that the lacrimal extends medially to this region. Posteriorly, the maxilla extends to the level of the M4 alveoli and forms a flat medial shelf at the inferior margin of the orbit.
A narrow fragment of the palatine (3.5 mm long) is preserved on the medial edge of the maxillary fragment and extends from the level of M3 to the posterior alveolus of M4. The roughness of this bone makes it difficult to determine whether this bone marks the lateral margin of the posterior palatal fenestra.
A small, triangular remnant of the jugal is present on the superior margin of the zygomatic arch. At its anterior end the dorsal margin of the jugal forms a tiny projection, below which lies the opening for a shallow sulcus that traverses its anteromedial surface. This sulcus lies parallel to the maxillojugal suture and terminates at the level of the lacrimal and is probably a canal for the lacrimal gland. Medially, the jugal makes a narrow contact with the remains of the lacrimal. Posterior to this contact, the maxillojugal suture curves superiorly and laterally along the anteromedial surface of the zygomatic arch.
Description of Referred Specimen. A left m3, QM F42676 (Figure 1.5), was recovered from the concentrate associated with the holotype and is interpreted to be from the same individual. The tooth is rectangular and measures 3.0 mm long and 2.5 mm wide. A large, shallow talonid basin dominates the tooth. The paraconid, protoconid, metaconid and hypoconid are subequal in height. A weak crest extends across the anterobuccal corner of the talonid basin, creating a shallow anterobuccal basin which separates the paraconid from the rest of the talonid basin. The anterobuccal, anterolingual and posterobuccal corners of the tooth are rounded. The talonid basin rises more steeply to the hypoconid than to the other cusps. At approximately the midpoint of the buccal and lingual sides of the tooth, the margins of the talonid basin are indented. The talonid basin is very weakly crenulated, the surface of the basin bearing very thin, faint grooves, and the margins of the basin bearing small ridges that descend towards its centre. The m3 has two roots of equal size.
Comparisons
The blade-like P3 and predominantly (post-M1) subtriangular upper molars of Microleo attenboroughi are characteristic of thylacoleonids. However, it also exhibits a number of features that clearly differentiate it from other members of the family.
Microleo differs from all other thylacoleonids in its very small size. Its P3 is approximately half the length of P3 of Priscileo roskellyae, the smallest taxon previously known; one quarter the length of Wakaleo vanderleueri; and one tenth the length of Thylacoleo carnifex (Table 2). Its body mass is estimated to be one third that of Priscileo roskellyae, the next smallest marsupial lion (see below for body mass estimates).
Microleo shares with species of Priscileo a plesiomorphic dental formula (three premolars and four molars). It also shares the relatively quadrilateral M1 and more triangular succeeding molars, features that are also possibly plesiomorphic for thylacoleonids.
In profile the P3 of Microleo is similar to that of Priscileo roskellyae and species of Wakaleo in that it is blade-like, with two main cusps connected by a V-shaped longitudinal crest. Its anterior margin is relatively steep, similar to that in species of Wakaleo, in contrast to the more rounded and reclined margins seen in P. roskellyae and species of Thylacoleo. Its occlusal outline is most similar to that of T. hilli, i.e., it is broadest at midcrown level and tapers at both ends. This similarity is primarily due to the swelling of the crown posterior to the anterobuccal crest that is found in both these species. The P3 of T. crassidentatus and T. carnifex is broadest at the level of the anterior cusp, tapers posteriorly and has a midcrown width that is similar to the width at the level of the posterolingual crest. In contrast, in P. roskellyae and species of Wakaleo the rear half of the crown of P3 is inflated, and thus it is broader posteriorly and narrower anteriorly. In occlusal view, the overall curvature of the buccal surface of P3 is convex in Microleo, whereas in species of Wakaleo, although the anterior and posterior ends of the buccal flank are convex, the overall curvature is concave as it is in species of Thylacoleo.
The lingual surface between the anterior and posterior cusps of P3 in Microleo also differs from other thylacoleonids. In Priscileoroskellyae and species of Wakaleo, a narrow depression occurs immediately posterior to the anterolingual crest, but beyond this the lingual surface expands to form a broadly convex flank. In Microleo, a tall, angled, sinuous valley occurs between the anterolingual and posterolingual crests. In species of Thylacoleo this region of the lingual flank is broad and vertical.
In profile, the P3 of Microleo appears to differ from all thylacoleonids in having an anterior cusp that is shorter than the posterior cusp. However, a large exposure of dentine on the anterior cusp indicates that the apex of this cusp has been broken off and/or has been subject to heavy wear. Thus, it is probable that its original height was at least similar to that of the posterior cusp, which is the condition seen in Priscileo roskellyae and species of Wakaleo.
The P3 of Microleo also differs from those of other thylacoleonids in having a longitudinal blade that has a very slight buccal bow between the major cusps (rather than bowing lingually) and in having the notch of the blade closer to the anterior rather than the posterior cusp. It also differs in having a markedly short posterior end of the crown relative to the height of the posterior cusp and in having a much steeper posterior portion of the longitudinal blade. In Priscileoroskellyae and species of Wakaleo, the posterior end of the crown is only slightly shorter than the posterior cusp and the posterior portion of the blade forms a relatively low angle (20o-30o) with respect to this cusp; in contrast, the angle for Microleo is much steeper (approx. 60o). The posterior cusp in species of Thylacoleo is barely discernible (T.hilli and T. crassidentatus) or absent (T. carnifex), presumably because of the posterior enlargement of the crown in these species. In these taxa the posterior end of the longitudinal blade forms a convex arc as it descends to the end of the tooth. Another significant feature of the Microleo P3 that is lacking in all other thylacoleonids is the small swelling of the longitudinal blade just anterior to the posterior cusp that suggests the original (in the unworn condition) presence of a medial cuspule on the blade at this point.
The anterior vertical crest of P3 of Microleo is more similar to the anterior crest seen in species of Thylacoleo in lacking the distinct lingually-enlarged basal crest present in Priscileo roskellyae and species of Wakaleo. However, this crest does differ from that in species of Thylacoleo, merging with the weak basal lingual swelling to form a very weak anterolingual cingulum, a feature that is absent in Thylacoleo.
The long anterolingual crest is also most similar to that seen in species of Thylacoleo, reaching the base of the crown and showing little basal inflation. In Priscileo roskellyae, the basal part of this crest is inflated and in species of Wakaleo a cuspule rather than a crest is present. The Microleo P3 also shares with species of Thylacoleo the presence of a posterolingual crest, although it is directed anterodorsally rather than posterodorsally which is the condition seen in Thylacoleo. Priscileo roskellyae and species of Wakaleo lack a posterolingual crest.
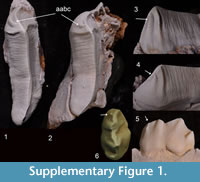 Another feature of the P3 shared withThylacoleocarnifex is the small accessory crest on the anterobuccal edge of the longitudinal blade, although it is variably present within that species. In a sample of 14 T. carnifex P3s examined in the Australian Museum collection, this feature was distinct in two specimens (AM F106836, AM F16609; see Supplementary Figure 1) but was absent in the remaining specimens. Within that sample, premolars that exhibited relatively little wear bore a short, broad, gently-rounded swelling in this region and in heavily worn specimens the entire anterobuccal surface was smooth, which suggests that this feature may be relatively common but obliterated early by wear.
Another feature of the P3 shared withThylacoleocarnifex is the small accessory crest on the anterobuccal edge of the longitudinal blade, although it is variably present within that species. In a sample of 14 T. carnifex P3s examined in the Australian Museum collection, this feature was distinct in two specimens (AM F106836, AM F16609; see Supplementary Figure 1) but was absent in the remaining specimens. Within that sample, premolars that exhibited relatively little wear bore a short, broad, gently-rounded swelling in this region and in heavily worn specimens the entire anterobuccal surface was smooth, which suggests that this feature may be relatively common but obliterated early by wear.
Compared to other thylacoleonids, the anterobuccal crest of P3 of Microleo is relatively broader, more obtuse and inclined in an anterodorsal direction, rather than posterodorsally as in Priscileo roskellyae and species of Wakaleo, or vertically as in species of Thylacoleo. The different inclination of this crest results in a relatively taller, more rectangular valley between the buccal crests, in contrast to the broader V-shape valley in P. roskellyae and species of Wakaleo. The posterobuccal crest of the Microleo P3 is more strongly developed and its posterobuccal basin relatively deeper than in P. roskellyae and species of Wakaleo. Both these features are absent in species of Thylacoleo.
The Microleo P3 also differs from all other thylacoleonids in its patterns of occlusal wear. In all other marsupial lions significant occlusal wear on the P3 is characteristically found evenly distributed along the lingual edge of the longitudinal blade with the wear facet extending uninterrupted from the anterior to the posterior cusps. This wear is created by occlusion with the longitudinal blade of p3. In Priscileo roskellyae and species of Wakaleo, less extensive wear is also present on the lingual edge of the blade that lies posterior to the posterior cusp, and is created by occlusion with the buccal face of the trigonid of m1. In Microleo, although wear on P3 is evident along the blade between the two major cusps, the distribution of this wear is not even. Wear on the anterior cusp is extremely heavy, but this heavy wear does not extend to the posterior cusp. The blade ascending to the posterior cusp exhibits only relatively light wear, and slightly heavier wear is evident on the posterior end of the blade as it descends from the posterior cusp to the rear of the crown. The orientation of these wear facets also differs slightly from those observed in other thylacoleonids. The wear facets on P3 in all other species of marsupial lion are primarily directed lingually. In Microleo, although all the facets lie on the lingual side of the tooth, both the facet on the anterior cusp and the facet posterior to the posterior cusp have a predominantly posterior direction. In addition, the facet immediately anterior to the posterior cusp has a more horizontal than lingual orientation. The wear facet on P3 of Thylacoleo carnifex becomes more horizontal with increased age and thus this pattern in the Microleo specimen may reflect an older individual. The wear pattern and distribution may also suggest that the morphology of the Microleo p3 and m1 is different to those of species of Priscileo and Wakaleo.
Although not preserved, the M1 of Microleo probably resembles that of Priscileo roskellyae because like the M1 of that species, it is quadrilateral (based on the preserved roots) and they also share similar M2 morphologies.
The M2 of Microleo is essentially similar to M2 of Priscileo roskellyae, but differs in having: a squarer posterior margin, a notch in the posterolingual margin just anterior to the metaconule, failure of the postmetacrista and postmetaconulecrista to meet, and the paracone more centrally located, resulting in a relatively longer preparacrista. It also differs in lacking anterobuccal expansion of the crown below the paracone, and in having a very weakly crenulated trigon basin. The M2 of species of Wakaleo are much larger, exhibit buccal expansion of the crown, enlargement of the protocone, reduction of the metaconule and are strongly crenulated. The M2 is absent in species of Thylacoleo.
The M3 of Microleo has the same triangular shape as M3 of Wakaleo oldfieldi and W. vanderleueri but differs in having a well-developed pyramidal paracone in the anterobuccal corner of the trigon basin. The latter feature has been lost or incorporated into the edge of the trigon basin in these other species. M3 is not yet known for species of Priscileo and is absent in species of Thylacoleo.
The m3 of Microleoattenboroughi is similar to that tooth in Priscileo roskellyae being rectangular, having a small trigonid basin that is similar in height to the talonid basin, and that is located at the anterobuccal corner of the crown, and in having a broad talonid basin. It differs from all other thylacoleonids in having much weaker crenulations and in having distinct mid-crown indentations on the buccal and lingual edges of the talonid basin. In species of Wakaleo, the m3 lacks a trigonid basin, the talonid basin is relatively narrow compared to crown width, and the anterior end of the crown is relatively taller. The m3 paratype, QM F42676, has a low-crowned, bunoid, quadritubercular morphology similar to that seen in the posterior lower molars of pilkipildrid, petaurid and burramyid possums. However, it differs from these families in lacking a metastylid (which is present in pilkipildrids and burramyids), being more weakly-crenulated (in contrast to pilkipildrids), in lacking development of hemilophs (which are present in petaurids), and in having a distinct trigonid basin (in contrast to burramyids). The reasons for concluding that QM F42676 is conspecific with the holotype are as follows: 1) both specimens represent a marsupial lion of equivalent size and have low-crowned molars that display a similar degree of molar crenulation; 2) the paratype occludes well with M3 of the holotype; 3) this is a rare taxon and is currently only known from Neville’s Garden Site; 4) for palaeoecological reasons, it is improbable that the two specimens would represent two different rare but equivalently-sized and morphologically similar species; and 5) it was recovered from the same small piece of limestone as the holotype.
PHYLOGENETIC ANALYSIS
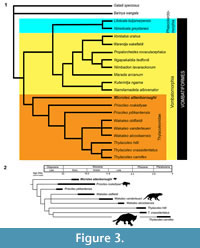 To investigate the evolutionary relationships of Microleo attenboroughi within the Suborder Vombatiformes, a phylogenetic analysis employing PAUP * 4.0b10 (Swofford, 2002) was carried out using a modified version of the data matrix of Black et al. (2012) (see Appendix 1 and Appendix 2). The analysis resulted in nine most-parsimonious trees of 272 steps in which M. attenboroughi emerged as the sister-group to all other thylacoleonids in a strongly-supported thylacoleonid clade (Figure 3; see Appendix 1 and Supplementary Figure 2 for bootstrap values).
To investigate the evolutionary relationships of Microleo attenboroughi within the Suborder Vombatiformes, a phylogenetic analysis employing PAUP * 4.0b10 (Swofford, 2002) was carried out using a modified version of the data matrix of Black et al. (2012) (see Appendix 1 and Appendix 2). The analysis resulted in nine most-parsimonious trees of 272 steps in which M. attenboroughi emerged as the sister-group to all other thylacoleonids in a strongly-supported thylacoleonid clade (Figure 3; see Appendix 1 and Supplementary Figure 2 for bootstrap values).
Previous hypotheses (Rauscher, 1987; Gillespie, 1997) which placed Priscileo outside of a Wakaleo - Thylacoleo clade (Archer and Dawson, 1982; Archer and Rich, 1982) were supported. Unequivocal synapomorphies for this latter clade are an increase in P3 length relative to the cheektooth row length (> 0.4) and loss of M4/m4 (Appendix 2). Apparent paraphyly of Priscileo may be the result of the large amount of missing data for P. pitikantensis. Synapomorphies uniting a Priscileo - Wakaleo - Thylacoleo clade include P3 having a posterior longitudinal blade that is gently angled and a confluent postprotocrista and premetaconulecrista on M2 (Appendix 2). The similar molar morphologies and dental formulae shared by Microleo attenboroughi and Priscileoroskellyae suggest the possibility that species of Priscileo evolved from a Microleo ancestor. With respect to M. attenboroughi, P. roskellyae has retained the plesiomorphic quadrilateral M1 and trapezoidal/semi-triangular M2 but has an enlarged and posteriorly-inflated P3.
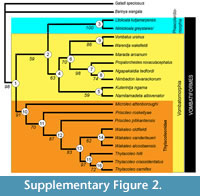 According to our analysis, many similarities shared by the diminutive Microleo attenboroughi and species of Thylacoleo, in particular T. hilli, appear to be homoplasies or retained symplesiomorphies. These features include, in P3, a posterolingual crest, a small accessory anterobuccal crest (variably present in T. carnifex; see Supplementary Figure 1), occlusal shape (T. hilli), lack of posterior inflation, simple, long and uninflated anterior and anterolingual crests, anterobuccal crests that do not extend posterodorsally, and a quadrilateral M1 (T. crassidentatusand T. carnifex).
According to our analysis, many similarities shared by the diminutive Microleo attenboroughi and species of Thylacoleo, in particular T. hilli, appear to be homoplasies or retained symplesiomorphies. These features include, in P3, a posterolingual crest, a small accessory anterobuccal crest (variably present in T. carnifex; see Supplementary Figure 1), occlusal shape (T. hilli), lack of posterior inflation, simple, long and uninflated anterior and anterolingual crests, anterobuccal crests that do not extend posterodorsally, and a quadrilateral M1 (T. crassidentatusand T. carnifex).
The results of the phylogenetic analysis support the starting assumption that Thylacoleonidae, including Microleo attenboroughi, is a clade (Figure 3.1). However, it does not provide support for the concept (Aplin and Archer, 1987) that this family is the sister-group of a clade containing all of the other vombatomorphian families. This is because Phascolarctomorphia (containing only the koala family Phascolarctidae) is not supported as the sister group of Vombatomorphia although most previous analyses have suggested it is (e.g., Aplin and Archer, 1987; Marshall et al., 1990; Black et al., 2012). Instead, Phascolarctomorphia appears to be the sister-group of a clade containing Vombatidae (wombats) and the extinct Diprotodontoidea, Maradidae, Wynyardiidae and Ilariidae but not Thylacoleonidae.
DISCUSSION
As well as Microleo attenboroughi, Neville’s Garden Site has yielded another much larger marsupial lion, a new, yet-to-be described plesiomorphic species of Wakaleo. Other Faunal Zone B sites at Riversleigh (Upper, Dirk’s Towers and Camel Sputum Sites) have yielded Priscileo roskellyae, a species intermediate in size between M. attenboroughi and this new species of Wakaleo. Hence the early Miocene rainforests of Riversleigh appear to have supported at least three marsupial lion species (Figure 3.2). This level of diversity is unmatched for the family at any other time in their evolutionary history.
Estimates of body mass indicate that Priscileo roskellyae (1813 g) is likely to have been three times heavier than Microleo attenboroughi (590 g). These body mass estimates were made using the regression equation of Myers (2001) from correlations of marsupial body mass with cranio-dental measurements (see Appendix 3). The equation used for the analysis was the highest possible ranked regression from the ‘Diprotodontians data set’ of Myers (2001, table 5, p.106) and employed the measurement for upper molar row length (UMRL, i.e., M1-M4). This equation (log y = -0.418 + 3.011 [log x], where x equals UMRL) was chosen because it was based on alveolar rather than molar measurements, the posterior teeth being absent in both taxa. The UMRL measurements for M. attenboroughi (11.3 mm), P. roskellyae (16.4 mm) and the new undescribed species of Wakaleo (23.3 mm) resulted in body weight estimates of 590 g, 1813 g and 5221 g, respectively. As recommended by Myers (2001), smearing estimates were applied to the prediction equation, and the calculated body mass estimates are likely to be underestimates. These weights are similar to those for females of living Eastern Quolls, Tiger Quolls and Tasmanian Devils, respectively (Jones and Barmuta, 2000).
The significant size differences between these three early Miocene Riversleigh marsupial lion taxa probably reflects niche partitioning with reduction in competition for resources (food and shelter), as has been argued (Wroe, 2001) for other Australian Miocene marsupial carnivores. Five thylacinid species are known from middle Miocene Australian deposits, with at least two being sympatric in three assemblages. Wroe (2001) found marked differences in estimated body weights among these (from ~1 to 18 kg) suggesting considerable trophic diversity in that family at that time. There is speculation that in the Miocene, marsupial lions, thylacinids and bandicoots filled some ecological niches occupied today by dasyurids, which appear to have explosively radiated in the later Cenozoic, perhaps in response to the development of more open palaeoenvironments (Krajewski et al., 2000). There is evidence of an increase in size in the thylacoleonid and thylacinid lineages over time, with the late Miocene Powerful Thylacine (Thylacinus potens), Tasmanian Thylacine (Thylacinus cynocephalus) and Pleistocene Marsupial Lion (Thylacoleo carnifex) being by far the largest carnivorous marsupials known from Australia (Wroe et al., 1999; Wroe, 2001). Studies of living sympatric marsupial carnivores have found partitioning in body size and prey size and a strong relationship between body size and arboreal activity (Jones and Barmuta, 2000). Hence, considering the three early Miocene marsupial lions at Riversleigh, it is possible that the smaller Microleo attenboroughi and Priscileo roskellyae were arboreal in contrast to possibly more scansorial species of Wakaleo. The modern Thylacine was a terrestrial predator (Guiler, 1985). If species of Oligo-Miocene thylacinids were also cursorial it is possible that members of this group were the dominant ground-dwelling mammalian predators while thylacoleonids were the dominant arboreal/scansorial predators. Calcanea of Oligo-Miocene marsupial lions, currently under study, are similar in functional morphology to that of a modern brushtail possum, a predominantly arboreal species.
The palaeohabitat of Riversleigh Faunal Zone B assemblages including the Neville’s Garden Local Fauna, has been inferred to be a closed forest environment based on the high diversity of mammals and on the presence of groups that today are restricted to rainforests (e.g., musky rat-kangaroos, Hypsiprymnodon; lyrebirds, Menura ; logrunners, Orthonyx; and striped possums, Djaludjangi) (Archer et al., 1989; Boles, 1995; Brammall, 1998; Bates et al., 2014; Nguyen et al., 2014). Pseudocheirids have a greater abundance of sympatric species in rainforests (Bassarova and Archer, 1999) and some of Riversleigh’s early Miocene assemblages contain up to seven sympatric pseudocheirids (Travouillon et al., 2009). Cenogram analyses of Faunal Zone B (early Miocene) mammal assemblages suggested a high proportion of arboreal taxa which also suggests a rainforest habitat for these assemblages (Travouillon et al., 2009). The early Miocene age of the Neville’s Garden sediments correlates with a time of warm and wet ‘greenhouse’'climatic conditions (McGowran and Li, 1994; Woodhead et al., 2016) and further supports a closed forest palaeohabitat hypothesis.
The bunodont molar dentition of Microleo is similar to that seen in burramyids and petaurids, possums that feed predominantly on invertebrates as well as plant substances (Van Dyke and Strahan, 2008). Although it seems likely that M. attenboroughi also ate invertebrates, with an estimated body weight nearly one third greater than that of the largest petaurid (Dactylopsila trivirgata at 400 g, average; Handasyde and Martin, 1996) and highly carnassial, longitudinally-bladed P3s, it is likely to have taken larger prey items as well, such as the many small vertebrates known to have been present and abundant in the early Miocene communities of northwestern Queensland. Functional analysis of the opposing v-shaped sectorial P3s of Thylacoleo carnifex suggests that these entrapped hard foods, perhaps small bones, enabling them to be more readily cut by these carnassial teeth (Wells et al., 1982). Although the morphology of the lower P3 of Microleo is currently unknown, its v-notched P3 suggests it would have functioned in a manner similar to the carnassials of other thylacoleonids. The rest of the dentition of M. attenboroughi lacks any features that characterise herbivorous marsupials adding further support to the interpretation that this marsupial lion, like all others in the family, was faunivorous.
In terms of phylogenetic relationships, the scarce and fragmentary nature of relatively plesiomorphic, pre-Pleistocene marsupial lions, has frustrated development of understanding. Intrafamilial relationships have been, and continue to be, primarily based on assessment of dental characters because at present only three taxa ( Priscileo roskellyae,Wakaleo vanderleueri and Thylacoleo carnifex) are known from well-preserved cranial material. Further, known dental material for P. pitikantensis, W. alcootaensis and T. hilli is also relatively incomplete. Interfamilial relationships within the diprotodontian Suborder Vombatiformes have been difficult to resolve because of the highly autapomorphic features of the dentitions of many of the families.
Marsupial lions are the only carnivorous vombatiform marsupials as well as the only vombatiforms that have bunodont molars. While our phylogenetic analysis does not unambiguously resolve the position of thylacoleonids within the vombatiform radiation, it is clear that it represents a highly distinctive and phylogenetically deep-rooted group within the suborder. We have previously speculated (Archer, 1976, 1984a) that the ancestral molar morphology for diprotodontian marsupials may have been selenodonty similar to that seen in koalas, ringtail possums and the extinct ilariids with remnant selenodont patterns discernible in wombats and wynyardiids. In general, selenodonty is an indicator in living mammals of a folivorous diet. However, given that carnivorous adaptations are evident in the earliest known marsupial lions (the undoubted carnassial nature of P3/p3) and that none appear to have molars that exhibit undoubted aspects of selenodonty, it seems at least as likely that this group originated from carnivorous or at least omnivorous marsupials that did not have selenodont molars. Because thylacoleonids appear to be the sister-group of all other vombatiform marsupials, it is possible that families within this suborder that exhibit selenodonty, such as phascolarctids and ilariids, may have evolved this molar morphology more than once from bunodont ancestors. For the same reason, it is at least as likely that bunodonty rather than selenodonty was the ancestral pattern for diprotodontians as a whole.
ACKNOWLEDGEMENTS
We thank the following people for providing access to comparative specimens: R. Jones and Y. Zhen from the Palaeontology Section of the Australian Museum, Sydney; N. Pledge and M. Binnie of the South Australian Museum; J. Wilkinson, S. Hocknull and P. Wilson of the Queensland Museum; and G. Dally, from the Museum and Art Gallery of the Northern Territory. The Riversleigh Fossil Project is supported by the Australian Research Council grants to M. Archer, S.J. Hand and K.H. Black (DP1094569, LP100200486, DE130100467, DP130100197), Environment Australia, Queensland Parks and Wildlife Service, Queensland Museum, University of New South Wales, Outback at Isa, Mount Isa City Council, the Waanyi people of northwestern Queensland, and the Riversleigh Society. Invaluable assistance has been provided in the field and laboratory by Alan Rackham and family, many volunteers, staff and postgraduate students of the University of New South Wales. Our thanks go to R.M.D. Beck who provided helpful comments on an earlier draft and to two anonymous reviewers for their constructive comments that improved this manuscript.
REFERENCES
Aplin K.P. and Archer, M. 1987. Recent advances in marsupial systematics with a new syncretic classification, p. xv-lxxii. In Archer, M. (ed.), Possums and Opossums: Studies in Evolution. Surrey Beatty and Sons and the Royal Zoological Society of New South Wales, Sydney.
Archer, M. 1976. Phascolarctid origins and the potential of the selenodont molar in the evolution of diprotodont marsupials. Memoirs of the Queensland Museum, 17:367-371.
Archer, M. 1984a. The Australian marsupial radiation, p.633-808. In Archer, M. and Clayton, G. (eds.), Vertebrate Zoogeography and Evolution in Australasia. Hesperian Press, Perth.
Archer M. 1984b. On the importance of being a koala, p. 809-815. In Archer, M. and Clayton, G. (eds.), Vertebrate Zoogeography and Evolution in Australasia. Hesperion Press, Perth.
Archer, M. and Dawson, L. 1982. Revision of marsupial lions of the genus Thylacoleo Gervais (Thylacoleonidae: Marsupialia) and thylacoleonid evolution in the late Cainozoic, p. 477-494. In Archer, M. (ed.), Carnivorous Marsupials. Royal Zoological Society of New South Wales, Sydney.
Archer, M., Godthelp, H., Hand, S.J., and Megirian, D. 1989. Fossil mammals of Riversleigh, northwestern Queensland: preliminary overview of biostratigraphy, correlation and environmental change. Australian Zoologist, 25:29-65.
Archer, M., Hand, S.J., Godthelp, H., and Creaser, P. 1997. Correlation of the Cainozoic sediments of the Riversleigh World Heritage fossil property, Queensland, Australia, p. 131-152. In Aguilar, J-P., Legendre, S., and Michaux, J. (eds.), Actes du Congrès BiochroM '97. École Pratique des Hautes Études, Institut de Montpellier, Montpellier, France.
Archer, M. and Rich, T.H. 1982. Results of the Ray E. Lemley Expeditions. Wakaleo alcootaensis n. sp. (Thylacoleonidae: Marsupialia), a new marsupial lion from the Miocene of the Northern Territory, with a consideration of the early radiation of the family, p. 495-502. In Archer, M. (ed.), Carnivorous Marsupials. Royal Zoological Society of New South Wales, Sydney.
Arena, D. 2004. The geological history and development of the terrain at the Riversleigh World Heritage Area during the middle tertiary. Unpublished PhD thesis, University of New South Wales, Sydney, Australia.
Bartholomai, A. 1962. A new species of Thylacoleo and notes on some caudal vertebrae of Palorchestes azael. Memoirs of the Queensland Museum, 14:33-40.
Bassarova, M. and Archer, M. 1999. Living and extinct pseudocheirids (Marsupialia, Pseudocheiridae): phylogenetic relationships and changes in diversity through time. Australian Mammalogy, 21:25-27.
Bates, H., Travouillon, K.J., Cooke, B., Beck, R.M.D., Hand, S.J., and Archer, M. 2014. Three new species of musky rat-kangaroo (Hypsiprymnodontidae, Macropodoidea): description, phylogenetics, and paleoecology. Journal of Vertebrate Paleontology, 34:383-396.
Black, K. 2007. Maradidae: a new family of vombatomorphian marsupial from the late Oligocene of Riversleigh, northwestern Queensland. Alcheringa, 31:17-32.
Black, K. and Archer, M. 1997a. Silvabestius, a new genus and two new species of primitive zygomaturines (Marsupialia, Diprotodontidae) from Riversleigh, northwestern Queensland. Memoirs of the Queensland Museum, 41:181-208.
Black, K. and Archer, M. 1997b. Nimiokoala gen. nov. (Marsupialia, Phascolarctidae) from Riversleigh, northwestern Queensland, with a revision of Litokoala. Memoirs of the Queensland Museum, 41:209-228.
Black, K.H., Archer. M., and Hand, S.J. 2012. New Tertiary Koala (Marsupialia, Phascolarctidae) from Riversleigh, Australia, with a revision of phascolarctid phylogenetics, paleoecology, and paleobiodiversity. Journal of Vertebrate Paleontology, 32:125-138.
Boles, W.E. 1995. A preliminary analysis of the Passeriformes from Riversleigh, Northwestern Queensland, Australia, with the description of new species of Lyrebird. Courier Forschungsinstitut Senckenberg, 181:163-170.
Brammall, J.R. 1998. A new petaurid possum from the Oligo-Miocene of Riversleigh, northwestern Queensland. Alcheringa, 23:31-50.
Clemens, W.A. and Plane, M. 1974. Mid-Tertiary Thylacoleonidae (Marsupialia, Mammalia). Journal of Paleontology, 48:652-660.
Flower, W.H. 1867. On the development and succession of teeth in the Marsupialia. Philosophical Transactions of the Royal Society, 157:631-641.
Gill, T. 1872. Arrangement of the families of mammals with analytical tables. Smithsonian Miscellaneous Collections, 11:1-98.
Gillespie, A. 1997. Priscileo roskellyae sp. nov. (Thylacoleonidae, Marsupialia) from the Oligocene-Miocene of Riversleigh, northwestern Queensland. Memoirs of the Queensland Museum, 41:321-327.
Gillespie A.K. 2007. Diversity and systematics of marsupial lions from the Riversleigh World Heritage Area and the evolution of the Thylacoleonidae. Unpublished PhD thesis. University of New South Wales, Sydney, Australia.
Gillespie, A.K., Archer, M., Hand, S.J., and Black, K.H. 2014. New material referable to Wakaleo (Marsupialia: Thylacoleonidae) from the Riversleigh World Heritage Area, northwestern Queensland: revising species boundaries and distributions in Oligo/Miocene marsupial lions. Alcheringa, 38:513-527.
Guiler, E.R. 1985. Thylacine: the Tragedy of the Tasmanian Tiger. Oxford University Press, Oxford.
Hand, S., Archer, M., Rich, T., and Pledge, N. 1993. Nimbadon, a new genus and three species of Tertiary zygomaturines (Marsupialia, Diprotodontidae) from northern Australia, with a reassessment of Neohelos. Memoirs of the Queensland Museum,33:193-210.
Handasyde, K.A. and Martin, R.W. 1996. Field observations on the Common Striped Possum (Dactylopsila trivirgata) in north Queensland. Wildlife Research, 23:755-766.
Horovitz, I. and Sánchez-Villagra, M.R. 2003. A morphological analysis of marsupial mammal higher-level phylogenetic relationships. Cladistics, 19:181-212.
Hope, J. and Wilkinson, H. 1982. Warendja wakefieldi, a new genus of wombat (Marsupialia, Vombatidae) from Pleistocene sediments in Mc Eacherns Cave, western Victoria. Memoirs of the National Museum of Victoria, 43:109-212.
Illiger, C. 1811. Prodromus Systematis Mammalian et Avian Additus Terminus Zoographicis utriudque Classis, p. xviii + 301. C. Salfield, Berlin.
Jones, M.E. and Barmuta, L.A. 2000. Niche differentiation amongst sympatric Australian dasyurid carnivores. Journal of Mammalogy, 81:434-447.
Krajewski, C., Wroe, S., and Westerman, M. 2000. Molecular evidence for the pattern and timing of cladogenesis in dasyurid marsupials. Zoological Journal of the Linnean Society, 130:375-404.
Linnaeus, C. 1758.Systema Naturae per Regna tria Naturae, secundum Classes, Ordines, Genera, Species, cum Characteribus, Differentiis, Synonymis, Locis. Tenth edition. Laurentii Salvii, Stockholm, Sweden.
Louys, J. 2004. Cranial descriptions of archaic koalas and an examination of vombatiform phylogeny based on cranial characters. Unpublished Honours thesis, University of New South Wales, Sydney, Australia.
Luo, Z-X., Kielan-Jaworowska, Z., and Cifell,i R.L. 2002. In quest for a phylogeny of Mesozoic mammals. Acta Palaeontologica Polonica, 47:1-78.
Marshall, L.G., Case, J.A., and Woodburne, M.O. 1990. Phylogenetic relationships of the families of the marsupials, p. 433-505. In Genoways, H.H. (ed.), Current Mammalogy, Vol. 2. Plenum Press, New York.
McGowran, B.and Li, Q. 1994. The Miocene oscillation in southern Australia. Records of the South Australian Museum, 27:197-212.
Murray, P. 1986. Propalorchestes novaculacephalus gen. et sp. nov., a new palorchestid (Diprotodontoidea: Marsupialia) from the middle Miocene Camfield Beds, Northern Territory, Australia. The Beagle, 3:195-211.
Murray, P., Wells, R., and Plane, M. 1987. The cranium of the Miocene thylacoleonid, Wakaleo vanderleueri : click go the shears-a fresh bite at thylacoleonid systematics, p. 433-466. In Archer, M. (ed.), Possums and Opossums: Studies in Evolution. Surrey Beatty and Sons and the Royal Zoological Society of New South Wales, Sydney.
Myers, T.J. 1994. Kuterintja ngama (Marsupialia, Ilariidae): a revised and extended systematic and phylogenetic analysis based on fossil material from Oligo-Miocene deposits at Riversleigh, northwestern Queensland, Australia. Unpublished Honours thesis, University of New South Wales, Sydney, Australia.
Myers, T.J. 2001. Prediction of marsupial body mass. Australian Journal of Zoology, 49:99-118.
Myers, T. and Archer, M. 1997. Kuterintja ngama (Marsupialia, Ilariidae): a revised systematic analysis based on material from the late Oligocene of Riversleigh, northwestern Queensland. Memoirs of the Queensland Museum, 41:379-392.
Nguyen, J.M.T., Boles, W.E., Worthy, T.H., Hand, S.J., and Archer, M. 2014. New specimens of the logrunner Orthonyx kaldowinyeri (Passeriformes; Orthonychidae) from the Olig-Miocene of Australia. Alcheringa, 38:245-254.
Owen, R. 1858. Odontology, p. 407-484. In Encyclopaedia Britannica, or dictionary of arts, sciences, and general literature. Eighth edition. 16, London.
Owen, R. 1866. On the fossil mammals of Australia. 2. Description of an almost entire skull of the Thylacoleo carnifex, Owen, from a freshwater deposit, Darling Downs, Queensland. Philosophical Transactions of the Royal Society, London, 156:73-82.
Pledge, N. 1977. A new species of Thylacoleo (Marsupialia, Thylacoleonidae) with notes on the occurrence and the distribution of Thylacoleonidae in South Australia. Records of the South Australian Museum, 17:277-283.
Rauscher, B. 1987. Priscileo pitikantensis, a new genus and species of thylacoleonid marsupial (Marsupialia: Thylacoleonidae) from the Miocene Etadunna Formation, South Australia, p. 423-432. In Archer, M. (ed.), Possums and Opossums: Studies in Evolution. Surrey Beatty and Sons and the Royal Zoological Society of New South Wales, Sydney.
Reig, O.A., Kirsch, J.A.W., and Marshall, L.G. 1987. Systematic relationships of the living and the neocenozoic American “opossum-like” marsupials (Suborder Didelphimorphia), with comments on the classification of these and of the Cretaceous and Paleogene New World and European metatherians, p. 1-89. In Archer, M. (ed.), Possums and Opossums: Studies in Evolution. Surrey Beatty and Sons and the Royal Zoological Society of New South Wales, Sydney.
Ride, W.D.L. 1964. A review of Australian fossil marsupials. Journal Proceedings of the Royal Society of Western Australia, 47:97-131.
Rougier, G.W., Wible, J.R., and Novacek, M.J. 1998. Implications of Deltatheridium specimens for early marsupial history. Nature, 396: 4459-4463.
Sanchez-Villagra, M.R. and Wible, J.R. 2002. Patterns of evolutionary transformation in the petrosal bone and some basicranial features in marsupial mammals, with special reference to didelphids. Journal of Zoological Systematics and Evolutionary Research 40: 26-45.
Springer, M.S., Kirsch, J.A.W., and Case, J.A. 1997. The chronicle of marsupial evolution, p. 129-161. In Givnish, T.J. and Sytsma, K.J. (eds.), Molecular Evolution and Adaptive Radiation. Cambridge University Press, Cambridge.
Swofford, D. L. 2002.PAUP*. Phylogenetic Analysis Using Parsimony (*And Other Methods), Version 4 (updated to 10 beta). Sinauer Associates, Sunderland, Massachusetts.
Travouillon, K.J., Archer, M., Hand, S.J., and Godthelp, H. 2006. Multivariate analyses of Cainozoic mammalian faunas from Riversleigh, northwestern Queensland. Alcheringa, Special Issue, 1:323-349.
Travouillon, K.J., Legendre, S., Archer, M., and Hand, S.J. 2009. Palaeoecological analyses of Riversleigh’s Oligo-Miocene sites: Implications for Oligo-Miocene climate change in Australia. Palaeogeology, Palaeoclimatology, Palaeoecology, 276:24-37.
Travouillon, K.J., Gurovich, Y., Beck, R.M.D, and Muirhead, J. 2010. An exceptionally well-preserved short-snouted bandicoot (Marsupialia; Peramelemorphia) from Riversleigh's Oligo-Miocene deposits, northwestern Queensland, Australia. Journal of Vertebrate Paleontology, 30:1528-1546.
Van Dyke, S. and Strahan, R. 2008. The Mammals of Australia. New Holland Publishing, Sydney.
Wells, R.T., Horton, D.R., and Rogers, P. 1982. Thylacoleo carnifex Owen (Thylacoleonidae): marsupial carnivore? p. 573-585. In Archer, M. (ed.), Carnivorous Marsupials. Royal Zoological Society of New South Wales, Sydney.
Woodburne, M.O. 1984. Families of marsupials: relationships, evolution and biogeography, p. 48-71. In Broadhead, T.W. (ed.), Mammals: Notes for a Short Course. University of Tennessee Department of Geological Science, Knoxville.
Woodburne, M.O., Tedford, R.H., Archer, M., and Pledge, N.S. 1987. Madakoala, a new genus and two species of Miocene koalas (Marsupialia: Phascolarctidae) from South Australia and a new species of Perikoala, p. 293-317. In Archer, M. (ed.), Possums and Opossums: Studies in Evolution. Surrey Beatty and Sons and the Royal Zoological Society of New South Wales, Sydney.
Woodhead, J., Hand, S.J., Archer, M., Graham, I., Sniderman, K., Arena, D.A., Black, K.H., Godthelp, H., Creaser, P., and Price, E. 2016. Developing a radiometrically-dated sequence for Neogene biotic change in Australia, from the Riversleigh World Heritage Area of Queensland. Gondwana Research, 29:153-167.
Wroe, S. 2001. Maximucinus muirheadae, gen. et sp. nov. (Thylacinidae: Marsupialia), from the Miocene of Riversleigh, northwestern Queensland, with estimates of bodyweights for fossil thylacinids. Australian Journal of Zoology, 49:604-613.
Wroe, S. 2008. Cranial mechanics compared in extinct marsupial and extant African lions using a finite-element approach. Journal of Zoology, 274:332-339.
Wroe, S., Brammall, J.R., and Cooke, B.N. 1998. The skull of Ekaltadeta ima (Marsupialia, Hypsiprymnodontidae?): an analysis of some marsupial cranial features and a re-investigation of propleopine phylogeny, with notes on the inference of carnivory in mammals. Journal of Paleontology, 72:738-751.
Wroe, S., Ebach. M., Ahyong, S., Muizon, C., and Muirhead, J. 2000. Cladistic analysis of dasyuromorphian (Marsupialia) phylogeny using cranial and dental characters. Journal of Mammalogy, 81:1008-1024.
Wroe, S., McHenry, C., and Thomason, J. 2005. Bite club: comparative bite force in big biting mammals and the prediction of predatory behaviour in fossil taxa. Proceedings of the Royal Society London B, 272:619-625.
Wroe, S., Myers, T.J., Wells, R.T., and Gillespie, A. 1999. Estimating the weight of the Pleistocene Marsupial Lion (Thylacoleo carnifex : Thylacoleonidae): implications for the ecomorphology of a marsupial super-predator and hypotheses of impoverishment of Australian marsupial carnivore faunas. Australian Journal of Zoology, 47:489-498.
Yates, A.M. 2015. New craniodental remains of Wakaleo alcootaensis (Diprotodontia: Thylacoleonidae) a carnivorous marsupial from the late Miocene Alcoota Local Fauna of the Northern Territory, Australia. Peer J, 3:e1408.
doi:10.7717/peerj.1408.

