 Uniformity in variety: Antler morphology and evolution in a predator-free environment
Uniformity in variety: Antler morphology and evolution in a predator-free environment
Article number: 21.1.9A
https://doi.org/10.26879/834
Copyright Paleontological Society, March 2018
Author biography
Plain-language and multi-lingual abstracts
PDF version
Submission: 9 November 2017 . Acceptance: 3 March 2018
{flike id=2159}
ABSTRACT
The Late Pleistocene mammal fauna of Crete was impoverished, as typical for oceanic islands, and consisted only of deer, dwarf elephants, an otter, a shrew and giant mice. Dwarf deer (Candiacervus spp.) were the dominant endemic herbivorous species. Here, I describe the adult antler morphology of this deer. Antler variety appears to be remarkably large, yet a few concise morphological groups without intermediate forms can be recognized, likely representing separate species. Antler variety is not a product of random variation induced by ecological release in a predator-free environment. Three new species are described here (Candiacervus spp. nov.), differing in antler and skull morphology, and the diagnosis of existing species (C. ropalophorus, C. rethymnensis) is emended based on new material. Antler variation can be explained by two evolutionary trends: showiness versus a classic fighting type. Divergence is driven and accelerated by intra-specific competition among males. The classic type is best explained as a result of allometric down-scaling during dwarfism. The display type is best explained as a result of restructuring of antler bauplan (simplification and extreme elongation of the main beam). Under predator-free scenarios, deer have the potential to evolve antler morphologies and behavioural changes unknown on the mainland.
Alexandra A.E. van der Geer. Naturalis Biodiversity Center, P.O. Box 9517, 2300 RA Leiden, the Netherlands. alexandra.vandergeer@naturalis.nl
Keywords: Candiacervus; endemic deer; new species; adaptive radiation
Final citation: van der Geer, Alexandra A.E. 2018. Uniformity in variety: Antler morphology and evolution in a predator-free environment. Palaeontologia Electronica 21.1.9A 1-31. https://doi.org/10.26879/834
palaeo-electronica.org/content/2018/2159-uniformity-in-variety
Copyright: March 2018 Paleontology Society
This is an open access article distributed under the terms of Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0), which permits users to copy and redistribute the material in any medium or format, provided it is not used for commercial purposes and the original author and source are credited, with indications if any changes are made.
creativecommons.org/licenses/by-nc-sa/4.0/
http://zoobank.org/336F2286-FE6B-48A0-A7DF-5A2389EB32B9
INTRODUCTION
Today, the native fauna of the island of Crete (Greece; Figure 1) includes the wild goat or Cretan ibex (Capra aegagrus cretica), 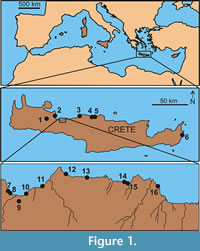 a wildcat (Felis sylvestris), the Cretan spiny mouse (Acomys minous), the Cretan hare (Lepus europaeus creticus), the Cretan lesser shrew (Crocidura suaveolens caneae), the Cretan stone marten (Martes foina bunites) and others. These were all introduced during the Holocene (Long, 2003; Gippoliti and Amori, 2006; Masseti, 2012). The Late Pleistocene mammal fauna, however, was very different from that of today: impoverished, as typical for oceanic islands, and consisting of nothing else but deer, dwarf elephants, an otter, a shrew and giant mice (van der Geer et al., 2010). Predators were limited to eagles and vultures. Dwarf deer (Candiacervus spp.) were the dominant endemic herbivorous species of that period. Fossil localities along most of its coastline as well as a few more inland localities (Iliopoulos et al., 2010), yielded literally hundreds of their antlers, bones and teeth, in contrast to sporadic finds of the larger species of the genus. One type of antler is truly bizarre because of its extreme length compared to the animal’s trunk and overall simplicity (Figure 2). This particular antler consists of a straight beam with a single basal tine and ends in a distal widening, described as club-like or bludgeon shaped (de Vos, 1984). What makes this antler most remarkable is that it is born by the smallest form of the endemic deer (Candiacervus ropalophorus) so that it is as long as its trunk or even slightly exceeding its length. It certainly does not represent an early ontogenetic stage with characteristic simplified bauplan, as revealed by characteristics of the burr and pedicle length of adult male skulls.
a wildcat (Felis sylvestris), the Cretan spiny mouse (Acomys minous), the Cretan hare (Lepus europaeus creticus), the Cretan lesser shrew (Crocidura suaveolens caneae), the Cretan stone marten (Martes foina bunites) and others. These were all introduced during the Holocene (Long, 2003; Gippoliti and Amori, 2006; Masseti, 2012). The Late Pleistocene mammal fauna, however, was very different from that of today: impoverished, as typical for oceanic islands, and consisting of nothing else but deer, dwarf elephants, an otter, a shrew and giant mice (van der Geer et al., 2010). Predators were limited to eagles and vultures. Dwarf deer (Candiacervus spp.) were the dominant endemic herbivorous species of that period. Fossil localities along most of its coastline as well as a few more inland localities (Iliopoulos et al., 2010), yielded literally hundreds of their antlers, bones and teeth, in contrast to sporadic finds of the larger species of the genus. One type of antler is truly bizarre because of its extreme length compared to the animal’s trunk and overall simplicity (Figure 2). This particular antler consists of a straight beam with a single basal tine and ends in a distal widening, described as club-like or bludgeon shaped (de Vos, 1984). What makes this antler most remarkable is that it is born by the smallest form of the endemic deer (Candiacervus ropalophorus) so that it is as long as its trunk or even slightly exceeding its length. It certainly does not represent an early ontogenetic stage with characteristic simplified bauplan, as revealed by characteristics of the burr and pedicle length of adult male skulls.
 The variation in antler size and morphology in the Cretan dwarf deer is unusually large relative to intra-specific variation in mainland deer (Kuss, 1965, 1975; de Vos, 1979, 1984, 2006). Kuss (1975) tentatively suggested an ontogenetic scheme and possible variations to explain the observed large variety. De Vos (1984) recognized five types of antlers, apart from an isolated very large burr, which he left unclassified. De Vos (1984) explains his findings of large antler and size variety within the Cretan deer Candiacervus by different taxonomic units, three of which were not formally named.
The variation in antler size and morphology in the Cretan dwarf deer is unusually large relative to intra-specific variation in mainland deer (Kuss, 1965, 1975; de Vos, 1979, 1984, 2006). Kuss (1975) tentatively suggested an ontogenetic scheme and possible variations to explain the observed large variety. De Vos (1984) recognized five types of antlers, apart from an isolated very large burr, which he left unclassified. De Vos (1984) explains his findings of large antler and size variety within the Cretan deer Candiacervus by different taxonomic units, three of which were not formally named.
Antlers show a high intraspecific variability in morphology as a result of a combination of ontogeny, environment and seasonal influence (Heintz, 1970). Nevertheless, a certain intra-specific variety is natural. Age, for example, has a profound influence on the shape of the antler, varying from a single, undivided spike in yearlings to maximal complexity and size in males in their prime age. Very old males have, again, simpler antler structures. The environment, e.g., reflected in nutrition, has influence on the potential expression of an antler type. The season dictates the developmental stage of the antler. The largest size and highest complexity is achieved around the breeding season. All these factors influence antler morphology, yet they are difficult or impossible to approach or quantify for an extinct deer, except for two obvious exceptions. One, the vast majority of antlers and antler fragments in the studied collections consist of shed antlers, and can thus be attributed to one and the same season, assuming that seasonality of reproduction had not changed between the age of the studied localities. Two, simple spikes and very slender antlers with small burrs without pearls are easily attributed to yearlings or two-year old males and can thus be separated from adult antler morphology. The effect of nutrition and other environmental factors like temperature and availability of fresh water cannot be reconstructed except in cases of obvious malformations. Apart from the variations due to the abovementioned factors, adult antlers are generally uniform in overall morphology (bauplan) within a species and are thus used for taxonomic and phylogenetic purposes.
Here, I amend and review earlier descriptions of antler morphology of Candiacervus, combining published specimens and new material, revise the taxonomic units of De Vos (1984) and establish three new species. I further tentatively explain patterns of antler morphology in terms of their potential ecological role. Life in isolation likely takes its toll on antler evolution, but hardly anything is known. For example, intra-specific variation might be amplified in island deer, as observed in Late Pleistocene Praemegaceros cazioti from Dragonara Cave, Sardinia (Caloi and Malatesta, 1974; see, however, Croitor et al., 2006). However, a large sample of Axis lydekkeri from Java (c. 60 complete antlers) on the other hand shows a very uniform morphology, without aberrant forms (de Vos, 1984, 34). Either way, uniformity or large variation, the antler variation of Candiacervus will give insight into evolutionary pathways in predator-free environments and the directions of ecological release on antler shape and size.
MATERIAL AND METHODS
Material
Antler material is curated at several institutions. Institutional abbreviations are: AMPG(V), Museum of Geology and Palaeontology of the National and Kapodistrian University of Athens (Greece); RGM, Naturalis Biodiversity Center, Leiden, the Netherlands, formerly Rijksmuseum voor Geologie en Mineralogie; NHMC, Natural History Museum of Crete, University of Crete, Heraklion; MPUR, Museum of Palaeontology, University of Rome La Sapienza, Italy. The material originates from 21 sites along the coasts of Crete and one inland site (Figure 1). Yearling antlers (spikes) and pathological antlers were excluded. Antler material of the two largest species (Candiacervus dorothensis, C. major) has not been retrieved; antler material of the two middle-sized species (C. cretensis, C. rethymnensis) is extremely rare, whereas the vast majority of antler material belongs to the dwarf sized forms (C. ropalophorus, Candiacervus sp. 2).
For the three-dimensional (3D) reconstructions of the fossils, antlers and a skull were scanned using a NextEngine HD laser scanner. The surface geometries were imported in the open-source computer graphics program Blender 3D (BLENDER.ORG, Amsterdam [the Netherlands]; https://www.blender.org) and high-quality rendering of the surface was obtained based on three-dimensional polygon meshes (see Garwood and Dunlop, 2017, for an explanation of the advantages of this approach). This technique allows for the modelling of missing parts and the reconstruction of the mirrored antler. The resulting virtual reconstruction could then be rotated in any direction for correct comparison.
Technical Terms
Pedicle. The bony process of the skull supporting the antler.
Burr. Proximal end of the antler that forms the attachment zone of the antler to the pedicle. The ring of pearls (small bead-like nodules) at its circumference is also known as rosary or coronet.
Tine. Ramification (or point). Four types can be observed in Candiacervus. The sub-basal tine, generally known as the brow tine, is situated at or at a very short distance from the burr. It is extremely rare in Candiacervus. The basal tine or bez tine is situated at a short distance from the burr on the anterior side and is the first tine of the beam. This tine is present in all adult specimens, hence the original denomination as Anoglochis by Simonelli (1907). The second tine or middle tine (trez tine) is situated distally from the basal tine at the same side and generally has the same orientation. The back tine or posterior tine is situated at the posterior side of the beam, placed further distally than the basal tines.
Palmation. A flat and widened distal part of the antler that connects individual tines.
Beam. The main shaft of the antler, or the part from which the tines grow.
Base. The first segment of the beam, between the burr and the place of origin of the first tine.
Spike. Undivided short beam, typical of young animals.
Second segment. The shortest distance between the first and second tine.
Third segment. The shortest distance between the second and the third tine.
Morphological Description
De Vos (1984) describes five types of adult antlers, numbered 1 to 5. The antlers described here correspond largely to these groups, with a few noteworthy exceptions. First, a number of new antlers are here considered mere varieties of a main type. This is the case with antlers with curved main beams instead of straight beams. Second, de Vos (1984) follows a different description and does not recognize the back tine as such. This is largely due to a large amount of material that was unavailable to de Vos (1984). Third, there are a couple of antlers of varying morphology that share a sub-basal tine at the anterior side of the burr, in the form of a very small tine or protuberance (a separate class 5 in de Vos, 1984). The antlers with such a rudimentary brow tine from Liko Cave are AMPG(V) 1738 (left shed antler fragment; de Vos, 1984, plate 9) and AMPG(V)2153 (left shed antler fragment). They both originate from a layer deeper than 75 cm below the surface. However, new material, unavailable to de Vos (1984) showed that such sub-basal tines are present in other localities as well, on antlers with various morphologies, which I here interpret as mere variations, and not as a category in its own right. Apart from the adult specimens from deep and potentially geologically older layers of Liko Cave, they seem to mostly belong to young or juvenile individuals (AMPG[V]561 [Liko Cave OD], AMPG[V] 2108 [Liko D], AMPG[V] 2111 [Liko D], NHMH A.II.958.962 [Panajia 2]), which is suggestive for an atavistic ontogenetic feature that may disappear in adulthood. This may also apply to rudimentary back tines. Antlers with a sub-basal tine and rudimentary back tines are here thus regarded as varieties of other types.
 Antler Size
Antler Size
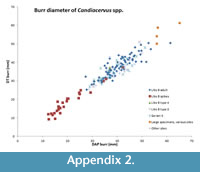
For minimal total length and for burr diameters, see Appendix 1, for burr diameters, see also Appendix 2. The burr is always oval in Candiacervus with the largest diameter mostly in antero-posterior direction. The burr is set obliquely on the pedicle (as in Dama). The smallest burr is from Liko B (AMPG[V] Li-2554; DAP = 27.8 mm; DT = 25.2 mm), the largest burrs are from Sifanos (NHMC F538/30; DAP = 5.6 cm; DT = 5 cm), Grida Avlaki (NHMC F538/53; DAP = 5.6 cm; DT = 5.4 cm), Mavro Muri 4 (NHMR MV4c-670; DAP = 56.4 mm; DT = 58.6 mm), and Melidoni (NHMC skull M; DAP = 65.2 mm; DT = 61.1 [average for both sides]).
SYSTEMATIC PALAEONTOLOGY
Class MAMMALIA Linnaeus, 1758
Family CERVIDAE Goldfuss, 1820
Subfamily CERVINAE Goldfuss, 1820
Genus CANDIACERVUS Kuss, 1975
Type species. Candiacervus cretensis (Simonelli, 1907) from the Late Pleistocene of Rethymnon Area (Crete, Greece), unknown site, possibly near Grida Avlaki (de Vos, 1984).
Included species. Candiacervus rethymnensis Kuss, 1975; Candiacervus ropalophorus de Vos, 1984; Candiacervus spp. nov.; Candiacervus dorothensis (Capasso Barbato, 1990), Candiacervus major (Capasso Barbato and Petronio, 1986).
Range. Late middle Pleistocene and late Pleistocene.
Occurrence. Crete (Greece).
Diagnosis. Pleistocene deer of the islands of Crete.
Emended diagnosis. Antlers with generally strongly compressed beams with longitudinal grooves and without pearling. Antlers with at most four tines (two anterior, one posterior, one sub-basal tine), but mostly a single anterior (basal) tine and often one posterior (back) tine. No crown is developed; rudimentary palmation may be present.
Candiacervus ropalophorus de Vos, 1984
Figure 3
Type locality. Gerani 4, 40 uppermost centimetres of the cave filling.
 Types. Holotype: male skull Ge4-46, figured in de Vos (1984: plate 10). Paratype: antler RGM 438460. Types stored at Naturalis Biodiversity Center, Leiden, the Netherlands.
Types. Holotype: male skull Ge4-46, figured in de Vos (1984: plate 10). Paratype: antler RGM 438460. Types stored at Naturalis Biodiversity Center, Leiden, the Netherlands.
Referred material. AMPG(V) 560 (Gerani, right shed antler), AMPG(V) 2107 (Rethymnon area, distal part), AMPG(V) 2133 (Rethymnon area, undivided beam), AMPG(V) 2134 (Rethymnon area, beam fragment), AMPG(V) 2135 (Rethymnon area, distal fragment), AMPG(V) 2139 (Kalo Chorafi, distal fragment), NHMC F538/103 (Kalo Chorafi, beam fragment), NHMC KC.111/63.227 (Kalo Chorafi, beam fragment), RGM 437170 (Gerani 4, beam fragment), RGM 437171 (Gerani 4, beam fragment), RGM 437176 (Gerani 4, beam fragment), RGM 438780 (Gerani 4, distal part), RGM 438788 (Gerani 4, distal part), RGM Ge4-3520 (Gerani 4, middle part).
Diagnosis. Very small deer (shoulder height about 40 cm) with post-cranial skeletal elements of size 1 of de Vos (1979, 1984). Metatarsal length varies between 110-131 mm (mean 121 mm). Metacarpal relatively short with length varying between 88.4-114.1 mm (mean 98.6 mm). Limb bones relatively more massive than in Cervus elaphus. Metatarsals (6%) may be fused with the navico-cuboid; navico-cuboids (68%) may be further fused with the central cuneiform (de Vos, 1979). Antlers very long (up to 77 cm), with single tine at c. 10 cm from the burr. At the level of the tine the beam curves backwards and immediately forwards again; the distal end of the beam is bludgeon-shaped.
Differential diagnosis. Smallest species of Candiacervus. Candiacervus ropalophorus further differs from all other Candiacervus species by its very long antler with single basal tine and a bludgeon-shaped end.
Emended diagnosis. The antler beam can be either straight or curved. Antlers may exceed total head-to-tail length.
Description. For the holotype skull, see de Vos (1984, 13-14, plates 10-11).
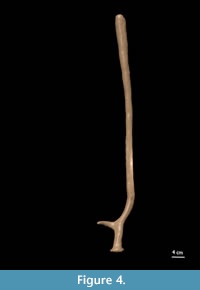
Antler RGM 438460 (Figure 3.1, Figure 4): Very long beam (minimal length 77 cm in a straight line) with a single (basal) tine. Burr oval with longest diameter anterior-posterior. Antler base oval with largest diameter medio-lateral. Basal tine arises c. 6.5 cm from the burr and forms an angle of c. 90° degrees with the base. Basal tine broken off about 1 cm from its base. Beam bends backwards and after c. 7 cm forwards again just above the tine so that the distal, straight part of the beam is again parallel to the base (lyre shaped). Overall impression is that of a straight beam. Beam after basal tine round, gradually more compressed towards distal end with the last 20 cm bludgeon-shaped. Maximum diameter of this distal widening is c. 3.6 cm.
 The new material includes a complete right shed antler (AMPG[V] 560) (Figure 3.2, Figure 5). The beam is undivided after the tine and very long (c. 60 cm). The beam gently curves upwards and sideward, ends in a blunt tip and is strongly compressed till halfway after which it widens and is fairly round. The burr is moderately pearled and almost round. The basal tine arises c. 7.7 cm from the burr and is broken off at 2 cm from its base.
The new material includes a complete right shed antler (AMPG[V] 560) (Figure 3.2, Figure 5). The beam is undivided after the tine and very long (c. 60 cm). The beam gently curves upwards and sideward, ends in a blunt tip and is strongly compressed till halfway after which it widens and is fairly round. The burr is moderately pearled and almost round. The basal tine arises c. 7.7 cm from the burr and is broken off at 2 cm from its base.
Several bludgeon-shaped fragments and undivided beams with bludgeon-shaped ends are present: AMPG(V) 2107 (straight beam), AMPG(V) 2133 (curved beam; Figure 3.3), AMPG(V) 2135, AMPG(V) 2139, NHMC F538/103 (straight beam), RGM 438780, RGM 438788, RGM Ge4-3520 (straight beam). These beams are strongly compressed distally but round just before the onset of the widening (where preserved). A number of beam fragments is similar in morphology to other referred material and is included here: AMPG(V) 2134 (straight beam), NHMC KC.111/63.227 (curved beam), RGM 437170 (curved beam), RGM 437171 (curved beam) and RGM 437176 (curved beam).
Occurrence. Gerani 2 (upper layer), 4, 5 and 6, Mavro Muri 4c, Sourida, Rethymnon fissure and Kalo Chorafi.
Remarks. This is the single Candiacervus species at the localities Gerani 4, 5, 6 and the upper deer layer of Gerani 2, with the exception of one out of 57 antlers at Gerani 4 that belongs to the second new species (see below). Curved beams are represented by AMPG(V) 560, AMPG(V) 2133, RGM 437170, RGM 437171, RGM 437176 and NHMC KC.111/63.227.
Candiacervus size 2 de Vos, 1984
 Description. A heterogeneous group of deer with post-cranial elements of size 2. Metatarsal length varies between 123.2-152.5 mm (mean 138.4 mm); metacarpal relatively short, length varies between 99.2-122.3 mm (mean 110.8 mm). Limb bones more massive than in Cervus elaphus. The length of P2-M3 ranges from 62.7-77.5 mm (mean 69.9 mm).
Description. A heterogeneous group of deer with post-cranial elements of size 2. Metatarsal length varies between 123.2-152.5 mm (mean 138.4 mm); metacarpal relatively short, length varies between 99.2-122.3 mm (mean 110.8 mm). Limb bones more massive than in Cervus elaphus. The length of P2-M3 ranges from 62.7-77.5 mm (mean 69.9 mm).
Remarks. Generally referred to as Candiacervus sp. II (de Vos, 1984, 53; de Vos, 1996, figure 9.1; van der Geer et al., 2010, 50, 333, 334, 364, 375, 395; van der Geer et al., 2014, 4, 9, 11, 15, figure 5), Candiacervus sp. 2 (van der Geer et al., 2010, 48), or Candiacervus ropalophorus (Capasso Barbato, 1989, 155 [partim], 165 [partim], figure 19 [partim], figure 25 [partim], figure 27). Based on morphological differences in skull, teeth and antlers, three different taxonomic units are distinguished, here described as three new species (see below).
Candiacervus devosi sp. nov.
Figure 6
zoobank.org/76A2EAB1-C2BC-4ED8-BD3E-5A98FC524EAE
v*1984 Candiacervus IIb; de Vos, p. 44, 48, plate 13.
v.1987 Candiacervus IIb; Dermitzakis and de Vos, p. 393, figure 10c.
p.1992 Megaloceros (Candiacervus) ropalophorus; Capasso Barbato, pp. 72-73.
p.1995 Megaceroides (Candiacervus) ropalophorus; Capasso Barbato, p. 243.
v.1996 Candiacervus IIb; de Vos, p. 113.
v.2000 Candiacervus IIb; de Vos, p. 127.
v.  2006 Candiacervus IIb; van der Geer, Dermitzakis and de Vos, p. 120.
2006 Candiacervus IIb; van der Geer, Dermitzakis and de Vos, p. 120.
v.2010 Candiacervus IIb; van der Geer, Lyras, de Vos and Dermitzakis, pp. 54, 58.
Type locality. Liko Cave, 75 uppermost centimetres of the cave filling (Likotinara, Crete, Greece).
Types. Holotype: male skull AMPG(V)1735, figured in de Vos (1984: plate 13). Paratype: right antler AMPG(V) 1733. Types stored at Museum of Palaeontology and Geology, National and Kapodistrian University of Athens (Greece).
Derivatio nominis. In honour of Dr. John de Vos, author of the first extensive monographic work on the systematics of Candiacervus.
Material. RGM 442704 (Liko B, right shed antler), RGM 442725 (Liko B, left antler fragment), RGM 442727 (Liko B, left shed antler fragment), RGM 442726 (Liko B, right antler fragment), RGM 442730 (Liko D, left shed antler), AMPG(V) Li-687 (Liko B, right shed proximal part), AMPG(V) 2102 (Liko C, antler fragment), AMPG(V) 2103 (Liko D; left shed antler fragment), AMPG(V) 2109 (Liko D, shed antler part), AMPG(V) 2113 (Liko O, antler fragment), NHMC MM.IV.69.48 (Mavro Muri 4, shed antler fragment).
Diagnosis. Candiacervus of size 2 of de Vos (1979, 1984). Orbit circular. Antler with a double basal tine, a back tine and an upwards pointing and long last segment (c. 1.5 the length of the previous segments combined), tapering into a sharp point. Double basal tines share the same origin and are connected for about 1-5 cm by a webbed, flat structure. Total straight line length is c. 40-50 cm. Additional bifurcation of the basal tines may occur.
Differential diagnosis. Larger than C. ropalophorus and smaller than C. cretensis, C. rethymnensis, C. dorothensis, C. major. Differs from all other Candiacervus species by its typical antler with a double basal tine, a very small back tine and a very long, up-going last segment.
Description. For a description of the holotype skull, see de Vos (1984, 14, skull c).
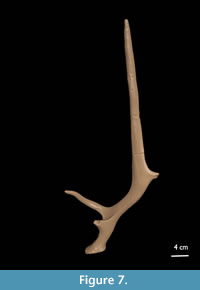 Paratype (Figure 7): The burr (DAP = 4.5 cm; DT = 4.2 cm) is damaged and minimally pearled. The two basal tines lie in a horizontal plane and are medio-laterally compressed. The base is c. 6 cm long. The outer tine (10.5 cm) bends upwards and then inwards. The two tines are connected with a webbing structure. The inner tine is broken off at about 2 cm from its base. The base is posteriorly convex and anteriorly concave. The second segment bends slightly backwards and is 10.3 cm long. The back tine is small, and its tip is broken off at 1.7 cm. The third, undivided segment makes an angle of 136° with the second segment, is medio-laterally compressed towards the distal end and tapering into a point. The third segment is 29.6 cm long. The minimal total length is ca. 44.5 cm. Deep longitudinal grooves are present over the entire beam.
Paratype (Figure 7): The burr (DAP = 4.5 cm; DT = 4.2 cm) is damaged and minimally pearled. The two basal tines lie in a horizontal plane and are medio-laterally compressed. The base is c. 6 cm long. The outer tine (10.5 cm) bends upwards and then inwards. The two tines are connected with a webbing structure. The inner tine is broken off at about 2 cm from its base. The base is posteriorly convex and anteriorly concave. The second segment bends slightly backwards and is 10.3 cm long. The back tine is small, and its tip is broken off at 1.7 cm. The third, undivided segment makes an angle of 136° with the second segment, is medio-laterally compressed towards the distal end and tapering into a point. The third segment is 29.6 cm long. The minimal total length is ca. 44.5 cm. Deep longitudinal grooves are present over the entire beam.
Other occurrences. Liko layer deeper than 75 cm below the surface; Mavro Muri 4.
Remarks. Referred to as skull type c with antler type 3 in de Vos (1984). Specimen AMPG(V) 2109 has an additional bifurcation of the double basal tine. This specimen originates from a deeper layer, and may thus represent a chronologically earlier form. A rudimentary sub-basal tine may be present (RGM 442730), again from a deeper layer; this specimen also has a rudimentary back tine. The connective webbing may be lacking with the tines arising separately from the beam (RGM 442726).
Candiacervus listeri sp. nov.
Figure 8
zoobank.org/F9A99042-A55C-4810-93B3-A2B5FF235E60
*1907 Anoglochis cretensis; Simonelli, p. 62, figure 16.
v.1965 Megaceros cretensis; Kuss, figure 3a-b.
v.1969 Cervus cretensis; Kuss, figure 2d-e.
v.1975 Candiacervus cretensis; Kuss, figure 3a.
 v*1984 Candiacervus sp. IIa; de Vos, pp. 44, 48, plate 12.
v*1984 Candiacervus sp. IIa; de Vos, pp. 44, 48, plate 12.
v.1987 Candiacervus sp. IIa; Dermitzakis and de Vos, p. 393, figure 10b.
p.1990 Megaceros (Candiacervus) ropalophorus; Capasso Barbato, p. 268, figure 2.
p.1992 Megaloceros (Candiacervus) ropalophorus; Capasso Barbato, pp. 72-73.
p.1995 Megaceroides (Candiacervus) ropalophorus; Capasso Barbato, pp. 243.
v.1996 Candiacervus sp. IIa; de Vos, p. 113.
v.2000 Candiacervus sp. IIa; de Vos, p.127.
v.2006 Candiacervus sp. IIa; van der Geer, Dermitzakis and de Vos, p. 120.
v.2010 Candiacervus sp. IIa; van der Geer, Lyras, de Vos and Dermitzakis, pp. 54, 58.
Type locality. Liko Cave, 75 uppermost centimetres of the cave filling (Likotinara, Crete, Greece).
Types. Holotype: male skull AMPG(V) 1734, figured in de Vos (1984, plate 12). Paratype: partial skull with left and right antler AMPG(V) 1726. Types stored at the Museum of Palaeontology and Geology, National and Kapodistrian University of Athens (Greece).
Derivatio nominis. In honour of Dr. Adrian Lister, specialist on fossil megacerine deer.
Referred material. AMPG(V) 1725 (Liko B; right shed antler), AMPG(V) 1727 (Liko B, left antler), AMPG(V) 1729 (Liko B, antler fragment), AMPG(V) 2106 (Kalo Chorafi?, right shed antler), AMPG(V) 2114 (Gerani, left shed antler), AMPG(V) 2115 (Rethymnon area, left shed antler), AMPG(V) 2121 (Rethymnon area, left shed antler), AMPG(V) 2137 (Peristeri 2, right shed antler), AMPG(V) 2143 (Gerani 2, right antler), AMPG(V) 2147 (Gumbes B, right shed antler), AMPG(V) 2218 (Rethymnon area, right shed antler), AMPG(V) GE764 (Gerani, partial skull with right antler), AMPG(V) Li-1721 (Liko B, partial beam), AMPG(V) Li-2593 (Liko B, right shed antler), AMPG(V) Li-2596 (Liko B, partial beam), NHMC F538/30 (Sifanos, left antler fragment), NHMC F538/53 (Grida Avlaki, left antler fragment), NHMC Ge.I.69.94 (Gerani 1, left shed antler), NHMC Ge.I.69.95 (Gerani 1, left antler), NHMC K.C.109.63 (Kalo Chorafi, right shed antler; Kuss, 1965, figure 3b), NHMC K.C.110/63 (Kalo Chorafi, right shed antler; Kuss, 1965, figure 3a), NHMC M.M.III.67 (Mavro Muri 3, left shed antler; Kuss, 1969, figure 2e), NHMC M.M.IV.67 (Mavro Muri 4, left shed antler; Kuss, 1969, figure 2d), NHMC n.n. (Gerani 1, left shed antler; Kuss 1975, figure 3a), RGM 438498 (Gerani 4, right shed antler fragment), RGM 442695 (Liko B, right shed antler), RGM 442696 (Liko B, right antler), RGM 442698 (Liko B, left shed antler), RGM 442720 (Liko B, left antler fragment), RGM 442723 (Liko B, right antler fragment), RGM 442731 (Liko OD, right shed antler).
 Diagnosis. Candiacervus of size 2 as defined by de Vos (1979). Dorsal rim of orbital wall very thick; orbit circular. P2 same morphology as in C. ropalophorus. Antlers long (up to 60-70 cm), consisting of a beam with single (basal) tine. Antler varieties with additional back tine tend to be shorter (c. 30-40 cm). Beam either straight or gently curved after the tine and not distally widening. Angle made by the two antlers is c. 100°. The tine arises at c. 9 cm from the burr.
Diagnosis. Candiacervus of size 2 as defined by de Vos (1979). Dorsal rim of orbital wall very thick; orbit circular. P2 same morphology as in C. ropalophorus. Antlers long (up to 60-70 cm), consisting of a beam with single (basal) tine. Antler varieties with additional back tine tend to be shorter (c. 30-40 cm). Beam either straight or gently curved after the tine and not distally widening. Angle made by the two antlers is c. 100°. The tine arises at c. 9 cm from the burr.
Differential diagnosis. Larger than C. ropalophorus and smaller than C. cretensis, C. rethymnensis, C. dorothensis, C. major. Differs further from C. ropalophorus by lacking a bludgeon-shaped distal end of the antlers. Dorsal rim of orbital wall thicker than in C. devosi.
Description. For a description of the holotype, see de Vos (1984, 14, skull b).
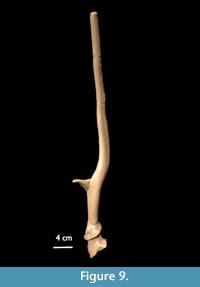
Paratype: Two antlers from the same individual and still attached to the pedicles with parts of the frontals preserved. The antlers are among the larger specimens from Liko B. The left antler consists of a rather straight beam with an upwards pointing tine at 8.7 cm above the burr. The burr (DAP = 4.5 cm; DT = 4.4 cm) is slightly damaged and lightly pearled. The base is 6.4 cm long and round. Shallow longitudinal grooves are present on the posterior side of the base and the beam. Just after the tine, the beam curves slightly backwards and immediately forwards again (lyre-shaped), and is strongly anterior-posteriorly compressed. The tip is chewed off. The right antler (Figure 9) is similar in morphology. The burr (DAP = 4.6 cm; DT = 4.5 cm) is lightly pearled. The tine starts at 8.8 cm from the burr. Shallow longitudinal grooves are present at the posterior side of the base and the dorsal side of the rest of the beam. The distal end is broken off. The angle made by both beams is 100°. The minimal total preserved length is 46 cm and 45.5 cm, respectively. An estimated 10-15 cm is missing.
 AMPG(V) Li-2593 (Figure 10): Right antler (= type specimen for antler type 2 of de Vos, 1984). The burr (DAP = 4.4 cm; DT = 4.0 cm) is moderately pearled. The base is 6 cm long and dorso-ventrally compressed. Moderately deep longitudinal grooves are present at the anterior side. The tine is 11.2 cm long, points upwards and inwards and arises from the outside of the beam. Its tip is chewed off. Just after the tine, the beam bends backwards and then immediately forwards, yielding a lyre-shaped appearance. The beam is anterior-posteriorly compressed after the tine. The total preserved minimal length is 47 cm. An estimated 15 cm is missing.
AMPG(V) Li-2593 (Figure 10): Right antler (= type specimen for antler type 2 of de Vos, 1984). The burr (DAP = 4.4 cm; DT = 4.0 cm) is moderately pearled. The base is 6 cm long and dorso-ventrally compressed. Moderately deep longitudinal grooves are present at the anterior side. The tine is 11.2 cm long, points upwards and inwards and arises from the outside of the beam. Its tip is chewed off. Just after the tine, the beam bends backwards and then immediately forwards, yielding a lyre-shaped appearance. The beam is anterior-posteriorly compressed after the tine. The total preserved minimal length is 47 cm. An estimated 15 cm is missing.
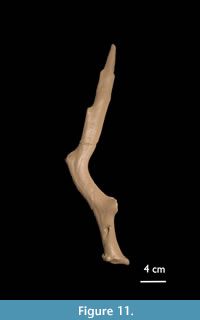 AMPG(V) 2106 (Figure 8.3 and Figure 11): The burr is roundish (DAP = 4.1 cm; DT = 3.7 cm) and moderately pearled. The base is 5.4 cm long and round (diameter c. 2.1 cm). The first tine is broken off at 1 cm from its base. The small (vestigial?) back tine is broken off at its base. The second segment is 7 cm long. The third segment is 21.3 cm long and has a serrated posterior edge. The serrations are less than 0.5 cm high and here interpreted as a minimal palmation. The beam is strongly anterio-posteriorly compressed and shows longitudinal grooves over its entire length.
AMPG(V) 2106 (Figure 8.3 and Figure 11): The burr is roundish (DAP = 4.1 cm; DT = 3.7 cm) and moderately pearled. The base is 5.4 cm long and round (diameter c. 2.1 cm). The first tine is broken off at 1 cm from its base. The small (vestigial?) back tine is broken off at its base. The second segment is 7 cm long. The third segment is 21.3 cm long and has a serrated posterior edge. The serrations are less than 0.5 cm high and here interpreted as a minimal palmation. The beam is strongly anterio-posteriorly compressed and shows longitudinal grooves over its entire length.
AMPG(V) 2115 (Figure 8.4): The beam is lyre-shaped. The burr is damaged and lightly pearled (DAP = c. 5.0 cm; DT = 4.5 cm). The base is 5.2 cm long. The first tine is broken off at 5.7 cm. The second segment is 10.8 cm long and bends backwards and sideward. The small back tine is broken off at c. 1 cm from its base. The third, undivided segment is long and directed up and backwards; the distal end is chewed or broken off at 11 cm. The beam is strongly compressed over its entire preserved length. No longitudinal grooves are present.
Other occurrences. Gerani 1, Gerani 2, Gerani 4, Grida Avlaki, Gumbes B, Kalo Chorafi, Mavro Muri 3, Mavro Muri 4, Peristeri 2, Sifanos.
Remarks. The skull has been referred to as type b with antler type 2 in de Vos (1984). Rudimentary palmation in the form of a serrated posterior border is represented by AMPG(V) 2106 (Figure 11) and NHMC Ge.I.69.94, and, inferred from rarity, probably is an atavistic feature. Curved beams are represented by AMPG(V) 2143, AMPG(V) 1729 and NHMC K.C. 110/63. The burrs of NHMC F538/53 (DAP = 5.6 cm; DT = 5.4 cm) and NHMC F538/30 (DAP = 5.6 cm; DT = 5 cm) are very large and may fall outside the range for size 2 (de Vos, 1984, figure 7) and may either represent C. cretensis, or a geologically earlier form of C. listeri. Antlers with a back tine are AMPG(V) 2114, AMPG(V) 2115, AMPG(V) 2121, AMPG(V) 2137, NHMC Ge.I.69.95, NHMC M.M.IV.67, RGM 438498, RGM 442720 and RGM 442696 (rudimentary back tine). Only one out of 57 antlers from Gerani 4 belongs to this type, probably indicating that the back tine was lost in extreme dwarf forms but before the acquisition of a bludgeon-shaped distal end.
Candiacervus reumeri sp. nov.
Figure 12
zoobank.org/1DD7AD28-2F75-4C27-9B68-73BCF91F6520
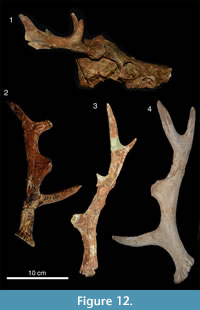 v*1984 Candiacervus sp. IIc; de Vos, pp. 44, 48, plate 14.
v*1984 Candiacervus sp. IIc; de Vos, pp. 44, 48, plate 14.
v.1987 Candiacervus sp. IIc; Dermitzakis and de Vos, p. 393, figure 10d.
p.1990 Megaceros (Candiacervus) ropalophorus; Capasso Barbato, p. 268, figure 2.
p.1992 Megaloceros (Candiacervus) ropalophorus; Capasso Barbato, pp. 72-73.
p.1995 Megaceroides (Candiacervus) ropalophorus; Capasso Barbato, p. 243.
v.1996 Candiacervus sp. IIc; de Vos, p. 113.
v.2000 Candiacervus sp. IIc; de Vos, p. 127.
v.2006 Candiacervus sp. IIc; van der Geer, Dermitzakis and de Vos, p. 120.
v.2010 Candiacervus sp. IIc; van der Geer, Lyras, de Vos and Dermitzakis, pp. 54, 58.
Type locality. Liko Cave, 75 uppermost cm of the cave filling (Likotinara, Crete, Greece).
Types. Holotype: male skull AMPG(V) 1736, figured in de Vos (1984, plate 14). Paratypes: left antler RGM 442702 (figured in de Vos (1984, plate 8). Types stored at Museum of Palaeontology and Geology, National and Kapodistrian University of Athens (Greece) (holotype) and Naturalis Biodiversity Center, Leiden, the Netherlands (paratype).
Derivatio nominis. In honour of Prof. dr. Jelle Reumer for his contribution to island palaeontology.
Referred material. AMPG(V) 562 (Liko OD, right shed antler), AMPG(V) 1738 (Liko OD, left shed antler fragment), AMPG(V) 2104 (Liko Cave OD, right shed antler), AMPG(V) 2105 (Peristeri 2, right shed antler), AMPG(V) 2136 (Peristeri 2, right shed antler), AMPG(V)2153 (Liko D, left shed fragment), NHMC G.A.68 (Grida Avlaki, right antler fragment), NHMC K.C.71.63 (Kalo Chorafi, right shed antler fragment; Kuss, 1965, figure 3c), RGM 442719 (Liko B, left antler fragment) and RGM 442717 (Liko B, left antler).
Diagnosis. Candiacervus of size 2. Dorsal rim of orbital wall very thin. Orbit oval, with the longer diameter anterior-posteriorly. Antler with two equi-distant tines and a back tine. Beam segments with straight or minimally concave posterior border. First three segments in a line. Anterior tines point anteriorly, lie in the same plane and are set at an angle of 90° with the beam. The fourth segments bend upwards.
Differential diagnosis. Larger than C. ropalophorus and smaller than C. cretensis, C. rethymnensis, C. dorothensis, C. major. Differs further from all other Candiacervus species by its typical antler with two anterior tines, a back tine and a non-lyre-shaped beam. Dorsal rim of orbital wall thinner than in C. devosi and C. listeri. Differs from C. devosi and C. listeri in oval orbit.
Description. For the holotype skull, see de Vos (1984, 14, skull d). P2 morphology unknown.
Antlers of the holotype: The right antler fragment consists of a moderately damaged burr (DAP = c. 3.9 cm; DT = 4.0 cm) with pearls, a base of 8.1 cm, a first tine, a second segment of 8 cm, a second tine and a third segment that is broken off at its origin and lies in the direction of the previous segments. Both tines point anteriorly and lie in the same plane. The first tine makes an angle of 90° with the beam, has a slightly oval base with the largest diameter in antero-posterior direction and bends slightly medially. Its tip is missing. The second tine is broken off at about 2 cm. The beam is strongly compressed except for the base, which is round, and has a straight posterior border. Longitudinal grooves are present. The left antler fragment is similar in morphology. The burr is damaged (DAP = c. 4.0 cm; DT = c. 4.0 cm). The base is 8.8 cm long, the second segment 7.4 cm and the second tine and third segment are both broken off at about 2 cm from their origin. The first tine is chewed at the end, and only 5 cm is preserved.
Paratype: Rather small antler with a minimal total length of c. 20 cm. Burr is round (DAP = 3.7 cm; DT = 3.7 cm) and lightly pearled, suggestive of a young individual. The beam curves slightly caudally. The first tine is placed laterally on the beam, points upwards and medially. The relatively broad second tine is broken off at its base. The third segment lies on a line with the previous two segments. The fourth segment is broken off a few cm after its origin, and makes an angle of 115° with the third segment. The back tine bends slightly caudally as a continuation of the second and third segments. The back tine is long (c. 6 cm) with its tip broken off at c. 5 cm. Longitudinal grooves are present over the entire beam.
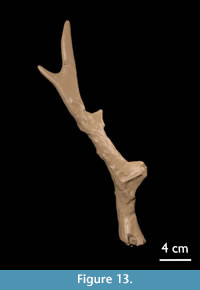 Antler AMPG(V) 562 (Figure 13): The burr is practically missing. The base is c. 7.3 cm long. The first tine is broken off at 1 cm from its base. The second segment bears a rudimentary second tine. The back tine lies in the continuation of the second and third segment and is short and pointed. The fourth segment makes an angle of 145° with the third segment and has a pointed end.
Antler AMPG(V) 562 (Figure 13): The burr is practically missing. The base is c. 7.3 cm long. The first tine is broken off at 1 cm from its base. The second segment bears a rudimentary second tine. The back tine lies in the continuation of the second and third segment and is short and pointed. The fourth segment makes an angle of 145° with the third segment and has a pointed end.
Antler AMPG(V) 2105 (Figure 12.4): The burr (DAP = 4.7 cm; DT = 4.2 cm) is moderately pearled. The base is c. 4.5 cm long. The first tine is very broad and compressed; only 1 cm is preserved. The second segment is c. 3.9 cm long. The third segment is c. 4.8 cm long. The first three segments and the back tine lie in one line. The posterior border is straight. The back tine is 3.8 cm long. The fourth segment bends up- and backwards and is 6.8 cm long. The beam is anterio-posteriorly compressed over its entire length. Shallow longitudinal grooves are present over the entire beam.
Other occurrences. Liko, layer deeper than 75 cm below the surface, Peristeri 2.
Remarks. Referred to as skull type d with antler type 4 in de Vos (1984; erroneously type c in the caption of plate 14). Specimens with a very small second basal tine (AMPG[V] 562, AMPG[V] 2104) are restricted to a layer of Liko Cave deeper than 75 cm below the surface, suggesting a geologically earlier stage (chronomorph). A rudimentary sub-basal tine may be present (AMPG[V] 1738, AMPG[V]2153, NHMC G.A.68, NHMC K.C.71.63), likely an ancestral feature. On the basis of overall morphology with a straight posterior border, the latter are included in C. reumeri. The first two are restricted to the same deep layer of Liko Cave, again suggestive of a geologically older stage.
Candiacervus cf. reumeri
Figure 14
.1980 Praemegaceros cretensis; Malatesta, pp. 18-68, 72, 80-82, 84-86, 91, plates 2-23.
 Material. AMPG(V) 490 (Simonelli 2, antler fragment), AMPG(V) 491 (Simonelli 2, left shed antler), AMPG(V) 492 (Simonelli 2, left shed antler), AMPG(V) 561 (Liko OD, left shed antler), AMPG(V) 2119 (Rethymnon area, left shed antler), AMPG(V) 2123 (Rethymnon area, right antler), AMPG(V) 2124 (Rethymnon area, left antler fragment), AMPG(V) 2125 (Rethymnon area, right shed antler), AMPG(V) 2138 (Kalo Chorafi, right shed antler), NHMC MM.III.67.252 (Mavro Muri 3, right shed antler; Kuss, 1969, figure 2a), RGM 442700 (Liko B, left antler), RGM 442718 (Liko B, left antler fragment), RGM 442722 (Liko B, right antler), MPUR (Simonelli Cave, all deer material).
Material. AMPG(V) 490 (Simonelli 2, antler fragment), AMPG(V) 491 (Simonelli 2, left shed antler), AMPG(V) 492 (Simonelli 2, left shed antler), AMPG(V) 561 (Liko OD, left shed antler), AMPG(V) 2119 (Rethymnon area, left shed antler), AMPG(V) 2123 (Rethymnon area, right antler), AMPG(V) 2124 (Rethymnon area, left antler fragment), AMPG(V) 2125 (Rethymnon area, right shed antler), AMPG(V) 2138 (Kalo Chorafi, right shed antler), NHMC MM.III.67.252 (Mavro Muri 3, right shed antler; Kuss, 1969, figure 2a), RGM 442700 (Liko B, left antler), RGM 442718 (Liko B, left antler fragment), RGM 442722 (Liko B, right antler), MPUR (Simonelli Cave, all deer material).
Description. Antlers with a long and robust basal tine, a generally very robust back tine, a broad upward pointing third segment and segments with straight posterior borders. The back tine is on a line with the preceding segments or sharply curves down and backwards. Orbit oval and dorsal rim of orbital wall very thin (Malatesta, 1980, plates 2-5).
AMPG(V) 561 (Figure 14.1): The burr (DAP = c. 3 cm) is damaged. Antler with a small sub-basal tine, a basal tine, and a second segment that ends in a bifurcation of more or less equal size. The sub-basal tine arises at c. 0.8 cm from the burr. The base is c. 6 cm long. The second segment is c. 5.5 cm long. The basal tine is 5.7 cm long and slender. The back tine lies in the continuation of the second segment and is c. 6 cm long. The third segment bends upwards and is c. 5.3 cm long. The distal tip is chewed off. The beam is antero-posteriorly flattened over its entire length. Shallow longitudinal grooves are present on the beam and tines. The antler may belong to a subadult individual with a poorly developed third segment.
AMPG(V) 2138 (Figure 14.2): The burr is relatively large (DAP = 4.4 cm; DT = 3.9 cm), but pearling is minimal. The base is 5.5 cm long and almost round, the second and third segment are compressed. The tine is 6.7 cm long and minimally curved. The second segment is 4.5 cm long and in the direction of the base. The third segment bends up-, back- and sideward. The back tine lies in the direction of the second segment, bends slightly backwards and is 4.2 cm long. The third, undivided segment is 13 cm long. The minimal length of the antler is 26.9 cm. Moderately developed longitudinal grooves are present on the beam and tines. A similar antler is AMPG(V) 2123, but with shorter back tine.
AMPG(V) 492: The basal tine is slender and spike-like instead of long and robust. The back tine lies in the direction of the second segment. The third segment bends upwards and is parallel to first tine. Longitudinal grooves are present over the entire beam, and the burr is moderately pearled excluding an early ontogenetic stage.
The back tine curves downward in the otherwise similar antlers AMPG(V) 2124, AMPG(V) 2125, RGM 442718. The third segment is broken off in several otherwise complete and morphologically similar antlers (AMGP[V] 491, AMPG[V] 492, RGM 442700 and RGM 442722). The vestigial back tine of AMPG(V) 491 is incorporated in a minimal posterior palmation.
Two fragments conform in morphology and are referred here: AMPG(V) 490 has a vestigial back tine in a posterior palmation onset and is very similar to the more complete AMPG(V) 491 from the same locality, and NHMC MM.III.67.252 preserves only the basal tine and the back tine and has the typical straight posterior border.
Occurrence. Simonelli Cave, Kalo Chorafi, Mavro Muri 3, Liko B.
Remarks. Antlers of this type are likely a variety of the typical C. reumeri antler, but lacking the second basal tine. The overall shape is most similar, notably in having straight segments, contrary to the curved or lyre-shaped second segment as seen in the other species. The morphology is also close to that seen at Kasos (Kuss, 1969, figure 1), but the latter specimens are conspicuously pearled and have deep longitudinal grooves on the entire surface, very unlike any Candiacervus antler. The vast majority of this antler morphology is found at Simonelli cave (see Accordi, 1972; Malatesta, 1980 for descriptions), which would be suggestive for a taxonomic unit in its own right, were it not for a few occurrences elsewhere (Kalo Chorafi, Mavro Muri 3, Liko layer B). In case the antlers here assigned to C. cf. reumeri are a simplification of the latter’s typical antlers, these forms might be a geologically younger form of the latter (chronomorph) or represent a different ecological niche (ecomorph). Skulls from Simonelli cave conform to the holotype skull of C. reumeri with oval orbit and very thin dorsal orbital rim. A sub-basal tine may be present, as shown by six shed antlers (four adults, two juveniles) with a sub-basal tine just above the burr (Malatesta, 1980). The back tine may be rudimentary and included in a rudimentary posterior palmation (AMPG[V] 490, AMPG[V] 491).
Candiacervus rethymnensis Kuss, 1975
Type locality. Mavro Muri 4, Crete.
Holotype. Right metacarpal figured by Kuss (1975, plate 6, figure o), stored at the Natural History Museum, University of Crete, Heraklion.
Material. NHMH 38.70 (skull marked M, probably from Melidoni). Figured: http://www.nhmc.uoc.gr/sites/default/files/private/images/83443_1.jpg.
Diagnosis. Candiacervus of size group 4 (de Vos, 1984). Metacarpal length varies between 185.0 and 206.2 mm (mean 194.8 mm; Cervus elaphus-like). Metatarsal length varies between 217.5 and 241.8 mm (mean 229.6 mm; Dama dama-like). The limb bones are about twice as long as those of size group 2.
Emended diagnosis. Basal tine, set at c. 3 cm from the burr, may be trifurcated. Adult burr diameter c. 6.5 cm. Interorbital distance c. 126 mm. Distance between supra-orbital foramina c. 67 mm, between pedicles c. 42 mm.
Description. Partial skull with preserved proximal antler fragments. Smallest distances between the pedicles is 42.4 mm, between supraorbital foramina 66.8 mm and between orbits 125.7 mm. Pedicles are 18.6 mm (left) and 21.0 mm (right) long. The basal tine of the antler is trifold, much like that of C. devosi, but with an additional tine, placed more distally.
Left side: Burr is round (DAP= 65.6 mm; DT= 63.6 mm) and lightly pearled. Longitudinal grooves only at the base. Second segment is round and broken off at c. 6.4 cm. First tine's origin at 27.2 mm. Proximal subtines broken off at c. 39.5 mm (outer) and 20.2 mm (inner), distal subtine chewed off at 24.6 mm. Right side has similar morphology. Measurements: burr (DAP = 64.8 mm; DT = 58.6 mm), first tine at 35.4 mm, proximal inner subtine broken off at 31.6 mm, proximal outer subtine broken off at 17.4 mm, distal subtine broken off at its origin, second segment broken off at its origin.
Other occurrences. Gerani 2 (lower deer layer), Sourida, Rethymnon fissure, Liko and possibly Simonelli Cave.
Remarks. This is the single Candiacervus species in the lower deer layer of Gerani 2. The layer above it contains only C. ropalophorus, which is suggestive for a temporal difference between these two species. A higher complexity of the antler confirms this view.
Candiacervus cf. devosi
Material. AMPG(V) 2101 (Liko C, right shed antler fragment), AMPG(V) 2122 (Rethymnon area, right shed partial antler), AMPG(V) 2148 (Gumbus B, left shed antler fragment), NHMC G.A.66 (Grida Avlaki, right shed antler), RGM 442699 (Liko B, right proximal fragment), RGM 442716 (Liko B, right proximal fragment).
Description. These antlers are all characterized by a bifurcated basal tine, a small back tine and an upwards pointing last segment. The bifurcation is situated a few centimetres after the origin of the tine.
AMPG(V) 2101: Bifurcation at c. 1.5 cm from its origin. The bifurcated end is chewed off. The second segment is broken off at 1 cm. The burr is oval (DAP = 34 mm; DT = 31 mm).
AMPG(V) 2122: Bifurcation at 6.5 cm; entire length of the basal tine is 8.3 cm. The burr is slightly damaged at its most anterior point and moderately pearled (DAP = c. 4.5 cm; DT = 3.5 cm). The base is round and 4.2 cm long. The second segment is 10.5 cm long. The back tine is broken off at its origin. The third segment bends upwards and is broad and strongly antero-posteriorly compressed, it is broken off at 3.5 cm. Shallow longitudinal ridges over the entire beam and tine are present.
AMPG(V) 2148: Bifurcation at 5.5 cm; total length of the basal tine is 8.6 cm. The tine is curved up- and frontward. The burr (DAP = 3.6 cm; DT = 3.3 cm) is moderately pearled. The base is 5.3 cm long, lightly longitudinally grooved and slightly compressed. The second segment is chewed off at 5.5 cm.
NHMC G.A.66 (Kuss, 1969, figure 2b): Only the tip of the basal tine is bifurcated. The back tine lies in the continuation of the second segment and the third segment makes an angle of 130° with the second segment.
RGM 442699 and 442716: The end of the basal tine bifurcates into two small tips, which are situated in a vertical plane relative to the beam in the first and in a horizontal plane in the second (length c. 8 cm in RGM 442716). The second segment bends backwards and medially (RGM 442716) or backwards (RGM 442699). The burrs are of medium size (RGM 442699: DT = 4 cm; RGM 442716: DAP = 3.7 cm; DT = 3.6 cm).
Occurrence. Liko Cave, Gumbus B, Grida Avlaki.
Remarks. These antlers likely represent a mere variety of typical C. devosi antlers. However, no complete antlers were found of this variety, hampering taxonomical classification.
DISCUSSION
Phylogenetic Considerations
The overall antler morphology suggest a phylogenetic placement closer to that of the genus Dama than to Megaloceros, especially when early middle Pleistocene forms without palmation are taken into consideration. Dama roberti has a long, straight and unforked beam and a single (basal) tine, much like C. ropalophorus and C. listeri. The palmation of D. roberti is nothing more than a distally flattened, gradually expanded blade-like shovel (Breda and Lister, 2013). The bludgeon-shaped distal end of C. ropalophorus may then be a modification of a simple palmation like that of D. roberti. Furthermore, the main palmation tines arise from the posterior border of the palm in Dama and from the anterior border in Megaloceros (see Gould, 1974). There are a few Candiacervus antlers with a rudimentary palmation in the form of a serrated posterior border, while there is only one antler with rudimentary anterior palmation tines. In addition, the configuration of the double basal tine as represented by C. devosi is seen in Dama mesopotamica (see Geist, 1998). At first sight, an important difference with Dama antlers is that in the latter genus the first (basal) tine arises directly at or very close to the burr, whereas this is at a few centimetres in Candiacervus. However, the rudimentary sub-basal tine present in a few specimens is here considered to represent the ancestral Dama-like condition. In that case, the first (basal) tine in Candiacervus is in fact the second tine. Candiacervus might be distantly related to D. mesopotamica, based on the bifurcated first tine and reduced palmation. The palmation of D. mesopotamica is similar to that of D. clactoniana or even less developed (van der Made et al., 2016).
The composite mount of Candiacervus size 2 (van der Geer et al., 2005, 2006b) is actually incorrect, regarding its antler. The selected antler belongs to type A (= type 1 in de Vos, 1984). However, no bludgeon-shaped distal ends were so far retrieved at Liko Cave (de Vos, 1984; contrary to van der Geer et al., 2005), and the antler therefore should have been a C. listeri type of antler (= type 2 in de Vos, 1984). The choice was guided by the extremely robust type of skull, which is indeed identical in morphology, though somewhat larger in size, than those found at Gerani 4, where the only antlers are of the C. ropalophorus type. This implies that the antler would have been proportionally even longer than is presented in the composite mount on display, as the skeleton should have been of the smallest size with wither’s height of c. 40 cm instead of the c. 50 cm as it has now.
Health and Mortality
Diet is a crucial factor in antler growth, and it is well known that it is habitat rather than genetic background that has a major influence on antler size. This is nicely demonstrated by red deer on the island of Rum, where the average antler size declines with increasing population density as a direct outcome of higher competition for food (Clutton-Brock et al., 1985). Weather and time of birth were also noted to be of major influence, again related to food, with calves born late in the year and growing up in unfavourable weather (mainly low temperatures) developed smaller antlers than earlier-born calves. Antlers are among the fastest growing mammalian tissue and are complete within 3-4 months on average. Since antlers are composed of about 50% minerals, of which 45% is calcium and 19% is phosphorous (Chapman, 1975), this means that this rapid growth requires a considerable amount of minerals, mainly calcium and phosphorous. That said, the very long antlers of Candiacervus ropalophorus, C. listeri and the relatively large but more complicated antlers of C. devosi do not give the impression of poor conditions but rather the opposite. Although part of the mineral supply is sequestered internally from the skeleton, with the ribs and long bones decreasing almost 60% in density caused by bone resorption (osteoporosis), mainly in the trabecular bone (Hillman et al., 1973; Baxter et al., 1999), this is repleted later and cannot be annually repeated. The suggestion by van der Geer et al. (2014) on nutrient-poor diets of dwarf Candiacervus to explain a high selective pressure seems in this light untenable. The diet of the average dwarf Candiacervus stag certainly was not poor in minerals (except for a singular population or site, mentioned in Dermitzakis et al., 2006). This is confirmed by the overall very low percentage of nutrient-related pathological antlers (three out of hundreds of antlers from Liko and Gerani cave; de Vos, 1984) and the very low prevalence rate (below 5%) of possibly nutrition-based degenerative lesions in skeletal elements from Liko Cave (Lyras et al., 2016).
Evolution Towards Show-off
 Although several types of antlers can be distinguished within the genus Candiacervus, these types are easily explained as evolutionary derivations (stages) of two basic bauplans (Figure 15). These derivations are not gradualistic, in the sense that one morphotype, representing a species or ecomorph, evolved into the 'next' morphotype. Likely, these stages may very well represent contemporaneous, geographically separated species or ecomorphs. The connecting lines in this figure thus only show the order of changes needed to evolve another antler morphology. Intermediate forms are extremely rare, confirming that the different forms represent distinct species and that evolution was step-wise, not gradual.
Although several types of antlers can be distinguished within the genus Candiacervus, these types are easily explained as evolutionary derivations (stages) of two basic bauplans (Figure 15). These derivations are not gradualistic, in the sense that one morphotype, representing a species or ecomorph, evolved into the 'next' morphotype. Likely, these stages may very well represent contemporaneous, geographically separated species or ecomorphs. The connecting lines in this figure thus only show the order of changes needed to evolve another antler morphology. Intermediate forms are extremely rare, confirming that the different forms represent distinct species and that evolution was step-wise, not gradual.
Two basic lines of derivations can be discerned in the Cretan antler material. One consists of a lyre-shaped antler in which the number and length of tines is step-wise reduced whereas the length of the main beam is increased between the two extremes. This evolution is, tentatively, represented by the following stages from complex to simplified: presence of a double basal tine, a back tine and an upwards directed last segment of moderate length (as in C. devosi), reduction of the basal tine to a single tine (as in varieties of C. listeri with a back tine), loss of the back tine and marked elongation of the last segment (as in typical C. listeri), to an extreme elongation of the last segment (as in C. ropalophorus). The other bauplan consists of an antler with straight segments in which the robusticity and length of the tines seems to increase. The evolution constitutes of a few tentative stages (not represented here), the order of which is less straightforward than that of the first lineage, but could be as follows. First, the presence of a basal tine, a small second tine, a back tine and a forked end (as in varieties of C. reumeri), first and second tine equally robust (as in typical C. reumeri), loss of the second tine, larger back tine, marked length increase of the last segment (as in C. cf. reumeri). As said above, the evolution of the two lineages is definitely not an evolution in time, because several forms (stages) are contemporaneous. The evolution as presented here is merely an illustration of how the various antler shapes of Candiacervus may have evolved, simply by adding and reduction of tines and lengthening of the last segment. When considered in this way, it is clear that their bauplan is derived from a single ancestral bauplan following deductible patterns, and not a playful experimentation of Mother Nature in a predator-free environment where ‘everything’ is possible.
The obvious question now is what could have been the driving force behind the evolution of these antlers? The end product of the first lineage, as represented by the iconic C. ropalophorus, is clearly a product of showiness. It is difficult to imagine that an antler that is longer than the entire body of its bearer and lacks conspicuous tines is fit for fighting. Investment in showy structures requires allocation of resources to antler growth at the cost of behaviours like fighting. The end product of the second lineage, as represented either by C. reumeri or the deer from Simonelli Cave, dependent on the exact order of the stages, corresponds more to the classic outline of an antler fit for fighting and wrestling, likely consisting of an initial clashing of the antlers, followed by a pushing match as seen in many deer species (Clutton-Brock et al., 1979). In fact, these short, multi-tined antlers look most like size-reduced, miniature deer antlers, likely as result of allometric changes that took place during dwarfism. In both cases, the display type and the fighting type, the amount of resources needed is likely similar, and their evolution cannot be explained by resource limitation.
CONCLUSION
The large antler variety exhibited by the endemic Cretan deer (Candiacervus) from the Late Pleistocene is not a product of random variation induced by ecological release in a predator-free environment. On the contrary, antlers are distributed in a few concise morphological groups. Likely, these different morphologies represent actual species or ecomorphs because intermediate forms are extremely rare and some localities have at most two forms. Three new species are described here (C. devosi, C. listeri, C. reumeri), differing in antler and skull morphology, and the diagnosis of existing species (C. ropalophorus, C. rethymnensis) is emended based on new material. The variation in antlers can be reconstructed as variations of two basic evolutionary trends: showiness versus a classic fighting type, entirely driven by intra-specific competition among males. The classic type is best explained as a result of allometric scaling during dwarfism. The display type on the other hand is best explained as a result of restructuring of antler bauplan (simplification and extreme elongation of the main beam). Under predator-free scenarios, deer have the potential to evolve antler morphologies and behavioural changes unknown on the mainland.
ACKNOWLEDGEMENTS
First of all, I want to take the opportunity here to thank John de Vos (Naturalis, Leiden) for being a great teacher and introducing me not only to Candiacervus but to island studies in general. I thank V. Karakitsios and G. Lyras (Museum of Paleontology and Geology, University of Athens, Greece), B. Fassoulas (Natural History Museum of Crete, Heraklion), N. den Ouden (Naturalis, Leiden) for allowing me to study the collections in their care. I am indebted to G. Lyras, A. Lister, G. Iliopoulos, T. Strasser, S. Murray, L. Ruprecht, J.W.F. Reumer and M.R. Palombo for discussions we had the last years on this intriguing deer with its bizarre antlers. I finally thank four anonymous reviewers for their constructive comments.
REFERENCES
Accordi, B. 1972. Lo scavo della “Grotta Simonelli”, con cervi nani del Quaternario, effettuato a Creta nel 1971 dall’Istituto di Geologia e Paleontologia dell’Università di Roma, con il finanziamento dell’Accademia Nazionale dei Lincei. Atti della Accademia Nazionale dei Lincei, 167:3-17.
Baxter, B.J., Andrews, R.N., and Barrell, G.K. 1999. Bone turnover associated with antler growth in red deer (Cervus elaphus). Anatomical Record, 256:14-19. https://doi.org/10.1002/(SICI)1097-0185(19990901)256:1<14::AID-AR3>3.0.CO;2-A
Breda, M. and Lister, A. 2013. Dama roberti, a new species of deer from the early Middle Pleistocene of Europe, and the origins of modern fallow deer. Quaternary Science Reviews, 69:155-167. https://doi.org/10.1016/j.quascirev.2013.01.029
Caloi, L. and Malatesta, A. 1974. Il cervo Pleistocenico di Sardegna. Memorie dell'Istituto Italiano di Paleontologia Umana, 2:163-247.
Capasso Barbato, L. 1989. Cervidi Endemici del Pleistocene di Creta. Unpublished PhD Thesis, Università Consorziate, Modena, Italy.
Capasso Barbato, L. 1990. Les Cervidés endémiques de Crètes. Quaternaire, 3-4:265-270.
Capasso Barbato, L. 1992. Nuova specie di cervide del Pleistocene di Creta. Atti della Accademia Nazionale dei Lincei, Memorie Lincee, Scienze Fisische e Naturali (Serie 9), 1:183-220.
Capasso Barbato, L. 1995. Un tentativo di analisi cladistica applicata ad un cervide endemico del Pleistocene di Creta. Geologica Romana, 31:243-248.
Capasso Barbato, L. and Petronio, C. 1986. Cervus major n. sp. of Bate Cave (Rethymnon, Crete). Atti della Accademia Nazionale dei Lincei, Memorie, classe di Scienze fisischi, matematische e naturali (Serie 8, 2a), 18:59-100.
Chapman, D.I. 1975. Antlers-bones of contention. Mammal Review, 5:122-172. https://doi.org/10.1111/j.1365-2907.1975.tb00194.x
Clutton-Brock, T., Major, M., and Guinness, F.E. 1985. Population regulation in male and female red deer. Journal of Animal Ecology, 54:831-846. https://doi.org/10.2307/4381
Clutton-Brock, T.H., Albon, S.D., Gibson, R.M., and Guinness, F.E. 1979. The logical stag: adaptive aspects of fighting in red deer (Cervus elaphus L.). Animal Behaviour, 27:211-215. https://doi.org/10.1016/0003-3472(79)90141-6
Croitor, R., Bonifay, M.-F., and Bonifay, E. 2006. Origin and evolution of the late Pleistocene island deer Praemegaceros (Nesoleipoceros) caztioti (Depéret) from Corsica and Sardinia. Bulletin du Musée D'anthropologie Préhistorique de Monaco, 46:35-68.
Dermitzakis, M.D. and de Vos, J. 1987. Faunal succession and the evolution of mammals in Crete during the Pleistocene. Neues Jahrbuch Geologische und Paläontologische Abhandlungen, 173:377-408.
Dermitzakis, M., van der Geer, A.A.E., and Lyras, G. 2006. Palaeopathological observations on a population of fossil deer from the Late Pleistocene of Crete, pp. 43-51. In Kalofourtis, A., Papadopoulos, N., Spiliopoulou, C., Marabellas, K., and Chatzioannou, A. (eds.), Volume in Honor of Prof. A.S. Koutselinis. Parisianos, Athens. [In Greek with English abstract]
de Vos, J. 1979. The endemic Pleistocene deer of Crete. Proceedings of the Koninklijke Nederlandse Akademie van Wetenschappen B, 82:59-90.
de Vos, J. 1984. The endemic Pleistocene deer of Crete. Verhandelingen der Koninklijke Akademie van Wetenschappen, afd. Natuurkunde, Eerste Reeks, 31:1-100.
de Vos, J. 1996. Taxonomy, ancestry and speciation of the endemic Pleistocene deer of crete compared with the taxonomy, ancestry and speciation of Darwin’s finches, p. 111-124. In Reese, D. (ed.), Pleistocene and Holocene Fauna of Crete and Its First Settlers. Monographs in World Archaeology, 28. Prehistory Press, Madison.
de Vos, J. 2000. Pleistocene deer fauna in Crete: its adaptation, radiation and extinction. Tropics, 10:125-134.
de Vos, J. 2006. Notes about parallels in the evolution of the Pleistocene cervids from Greece (Crete, Kassos and Karpathos), Japan (the Ryukyu-islands) and Philippines (Masbate). Hellenic Journal of Geosciences, 41:127-140.
Garwood, R. and Dunlop, J. 2017. The walking dead: Blender as a tool for paleontologists with a case study of extinct arachnids. Journal of Paleontology, 88:735-746. https://doi.org/10.1666/13-088
Geist, V. 1998. Deer of the World: Their Evolution, Behaviour and Ecology. Stackpole Books, Mechanicsburg, PA.
Gippoliti, S. and Amori, G. 2006. Ancient introductions of mammals in the Mediterranean Basin and their implications for conservation. Mammal Review, 36:37-48. https://doi.org/10.1111/j.1365-2907.2006.00081.x
Gould, S.J. 1974. The origin and function of "bizarre" structures: antler size and skull size in the "Irish Elk," Megaloceros giganteus. Evolution, 28:191-220. https://doi.org/10.1111/j.1558-5646.1974.tb00740.x
Goldfuss, A. 1820. Handbuch der Zoologie. Johann Leonard Schrag, Nurnberg.
Heintz, E. 1970. Les Cervidés Villafranchiens de France et d'Espagne. Mémoirs Museum National d'Histoire Naturelle, N.S.C., 22. Museum National d'Histoire Naturelle, Paris.
Hillman, J.R., Davis, R.W., and Abdelbaki, Y.Z. 1973. Cyclic bone remodeling in deer. Calcified Tissue Research, 12:323-330. https://doi.org/10.1007/BF02013745s://doi.org/10.1007/BF02013745
Iliopoulos, G., Eikamp, H., and Fassoulas, C. 2010. A new Late Pleistocene mammal locality from western Crete. Bulletin of the Geological Society of Greece, 43:918-925.
Kuss, S.E. 1965. Eine pleistozäne Säugetierfauna der Insel Kreta. Berichte der Naturforschenden Gesellschaft zu Freiburg im Breisgau, 55:271-348.
Kuss, S.E. 1969. Die paläolithische osteokeratische "Kultur" der Insel Kreta (Griechenland). Berichte der Naturforschenden Gesellschaft zu Freiburg im Breisgau, 59:137-168.
Kuss, S.E. 1975. Die pleistozänen Hirsche der ostmediterranen Inseln Kreta, Kasos, Karpatos und Rhodos (Griechenland). Berichte der Naturforschenden Gesellschaft zu Freiburg im Breisgau, 65:25-79.
Linnaeus, C. 1758. Systema Naturae. Volume 1 (tenth edition). Engelman, Leipzig.
Long, J.L. 2003. Introduced Mammals of the World: Their History, Distribution and Influence. Csiro Publishing, Collingwood, Australia.
Lyras, G.A., Giannakopoulou, A., Lillis, T., Veis, A., and Papadopoulos, G. 2016. Bone lesions in a Late Pleistocene assemblage of the insular deer Candiacervus sp. II from Liko cave (Crete, Greece). International Journal of Paleopathology, 14:36-45. https://doi.org/10.1016/j.ijpp.2016.04.004
Malatesta, A. 1980. Dwarf deer and other Late Pleistocene fauna of the Simonelli Cave in Crete. Accademia Nazionale dei Lincei, 249:3-97.
Masseti, M. 2012. Atlas of Terrestrial Mammals of the Ionian and Aegean Islands. De Gruyter, Berlin.
Simonelli, V. 1907. Mammiferi quaternari dell’isola di Candia I. Memorie, Accademia delle Scienze. Istituto di Bologna, Classe di Scienze Fisiche, Serie 6, 4:455-471.
van der Geer, A., Dermitzakis, M., and de Vos, J. 2006a. Relative growth of the metapodals in a juvenile island deer: Candiacervus (Mammalia, Cervidae) from the Pleistocene of Crete. Hellenic Journal of Geosciences, 41:119-125.
van der Geer, A., de Vos, J., Dermitzakis, M., and Lyras, G. 2010 Evolution of Island Mammals: Adaptation and Extinction of Placental Mammals on Islands. Wiley-Blackwell, Oxford, UK. https://doi.org/10.1002/9781444323986
van der Geer, A.A.E., de Vos, J., Lyras, G., and Dermitzakis, M. 2005. The mounting of a skeleton of the fossil species Candiacervus sp. II from Liko Cave, Crete, Greece, p. 337-346. In Alcover, J.A. and Bover, P. (eds.), Proceedings of the International Symposium “Insular Vertebrate Evolution: the Palaeontological Approach”. Monografies de la Societat d’Història Natural de les Balears, 12. Societat d'Història Natural de les Balears, Palma de Mallorca.
van der Geer, A.A.E., de Vos, J., Lyras, G., and Dermitzakis, M. 2006b. New data on the Pleistocene Cretan deer Candiacervus sp. II (Mammalia, Cervinae), p. 131-137. In Kahlke, R.-D., Maul, L.C., and Mazza, P. (eds.), Late Neogene and Quaternary Biodiversity and Evolution: Regional Developments and Interregional Correlations. Proceedings of the 18th International Senckenberg Conference vol. II. Courier Forschungsinstitut Senckenberg, 256. Schweizerbart, Stuttgart.
van der Geer, A.A.E., Lyras, G.A., MacPhee, R.D.E., Lomolino, M., and Drinia, H. 2014. Mortality in a predator-free insular environment: the extinct dwarf deer from Crete. American Museum Novitates, 3807:1-29. https://doi.org/10.1206/3807.1
van der Made, J., Torres, T., Ortíz, J.E., Moreno-Pérez, L., and Fernández-Jalvo, Y. 2016. The new material of large mammals from Azokh and comments on older collections, p. 117-162. In Fernández-Jalvo, Y., King, T., Yepiskoposyan, L., and Andrews, P. (eds.), Azokh Cave and the Transcaucasian Corridor. Springer, Dordrecht. https://doi.org/10.1007/978-3-319-24924-7

