 Xiphosurid from the Upper Permian of Tasmania confirms Palaeozoic origin of Austrolimulidae
Xiphosurid from the Upper Permian of Tasmania confirms Palaeozoic origin of Austrolimulidae
Article number: 22.3.62
https://doi.org/10.26879/1005
Copyright Paleontological Society, October 2019
Author biography
Plain-language and multi-lingual abstracts
PDF version
Submission: 6 June 2019. Acceptance: 26 August 2019.
ABSTRACT
Representatives of Austrolimulidae exhibit the most extreme morphological variation documented within true horseshoe crabs. Relative to the more standard morphologies of paleolimulids and limulids, austrolimulids have been described as oddball taxa. Despite their unique morphologies, austrolimulid diversity is somewhat understated. A horseshoe crab specimen from the Upper Permian Jackey Shale of Tasmania described in Ewington et al. (1989) is reconsidered here to develop the recent increased interest in the family. The specimen was originally assigned to Paleolimulus sp., but an array of unique features suggests it belongs within Austrolimulidae and Tasmaniolimulus patersoni gen. et sp. nov. is erected herein. Parsimony-based phylogenetic analyses support such a placement of T. patersoni in Xiphosurida and show that the new taxon is located at the base of Austrolimulidae. Furthermore, landmark-based geometric morphometric analyses corroborate the erection of the new taxon and illustrate a more extensive occupation of morphospace by austrolimulids than previously observed. Further studies of the paraphyletic genus Paleolimulus are needed to highlight the true diversity of austrolimulids and paleolimulids.
Russell D.C. Bicknell. Palaeoscience Research Centre, School of Environmental and Rural Science, University of New England, Armidale, New South Wales, 2351, Australia. rdcbicknell@gmail.com
Keywords: Xiphosurida; Permian; horseshoe crab; geometric morphometrics; new genus; new species
Bicknell, Russell D.C. 2019. Xiphosurid from the Upper Permian of Tasmania confirms Palaeozoic origin of Austrolimulidae. Palaeontologia Electronica 22.3.62 1-13. https://doi.org/10.26879/1005
palaeo-electronica.org/content/2019/2731-a-permian-austrolimulid
Copyright: October 2019 Paleontological Society.
This is an open access article distributed under the terms of Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0), which permits users to copy and redistribute the material in any medium or format, provided it is not used for commercial purposes and the original author and source are credited, with indications if any changes are made.
creativecommons.org/licenses/by-nc-sa/4.0/
http://zoobank.org/6DE9B99C-9554-47DB-B25B-9A962B2EC317
INTRODUCTION
Horseshoe crabs are one of the most well-studied marine chelicerate groups. The interest in xiphosurids reflects their unique array of biological and physiological features (Shuster Jr., 1982; Bicknell et al., 2018a-c), an exceptional fossil record that spans the Lower Ordovician to today (Rudkin and Young, 2009; Van Roy et al., 2010, 2015; Bicknell et al., 2019a), and the exhibition of evolutionary conservation since the Jurassic (Kin and Błażejowski, 2014; Bicknell et al., 2019b, c; d). Curiously, fossil horseshoe crab species numbers remained generally low across the Phanerozoic. However, there were two pulses of expansion, the first during the Pennsylvanian (Moore et al., 2007; Rudkin and Young, 2009) and a second during the Triassic (Bicknell et al., 2019a). During the Triassic, a family of xiphosurids with extreme morphologies--Austrolimulidae--diversified. Lamsdell (2016) suggested that the group had a mid to late Palaeozoic origin. As such, further examples of these oddball taxa may help fill in the gap between the Palaeozoic onset and the Triassic diversification event.
Australian fossil horseshoe crabs are currently represented by five taxa: Kasibelinurus amicorum Pickett, 1993, from the Upper Devonian Mandagery Sandstone, New South Wales (NSW); Paleolimulus sp. Ewington Clarke, and Banks, 1989, from the Upper Permian Jackey Shale, Tasmania; Austrolimulus fletcheri Riek, 1955, from the Middle Triassic Beacon Hill Shale, NSW; Dubbolimulus peetae Pickett, 1984, from the Middle Triassic Ballimore Formation, NSW and Victalimulus mcqueeni Riek and Gill, 1971, from the Lower Cretaceous Korumburra Group, NSW. Recent phylogenetic analyses highlighted that both A. fletcheri and D. peetae are austrolimulids (Lamsdell, 2016) and that Paleolimulus Dunbar, 1923, was a paraphyletic group (also see comments in Anderson and Selden, 1997, Babcock and Merriam, 2000, and Allen and Feldmann, 2005), such that select Paleolimulus species were austrolimulids. In light of these recent developments, I have reconsidered Paleolimulus sp. from the Jackey Shale to determine if this taxon is an austrolimulid, as suggested in Lerner et al. (2017). I remove the taxon from the family Paleolimulidae and the genus Paleolimulus, instead treating it as Tasmaniolimulus patersoni gen. et sp. nov.--a representative of Austrolimulidae.
MATERIAL AND METHODS
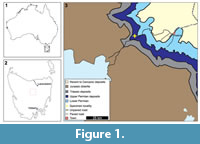 The single studied fossil specimen was originally described by Ewington et al. (1989) from an outcrop of the Jackey Shale. This is a formation within the Upper Parmeener Supergroup that preserves non-marine swamp, lake, and river deposits that may have experienced limited coastal influence (Banks, 1973; Ewington et al., 1989). The specimen was found close to the top of the Jackey Shale, 3 m from the base of the overlying Ross Sandstone at Poatina (Figure 1, 41°48'05"S 146°53'06"E; Ewington et al., 1989). The specimen (UTGD 123979) is housed at the Rock Library and Geological Museum of the University of
The single studied fossil specimen was originally described by Ewington et al. (1989) from an outcrop of the Jackey Shale. This is a formation within the Upper Parmeener Supergroup that preserves non-marine swamp, lake, and river deposits that may have experienced limited coastal influence (Banks, 1973; Ewington et al., 1989). The specimen was found close to the top of the Jackey Shale, 3 m from the base of the overlying Ross Sandstone at Poatina (Figure 1, 41°48'05"S 146°53'06"E; Ewington et al., 1989). The specimen (UTGD 123979) is housed at the Rock Library and Geological Museum of the University of 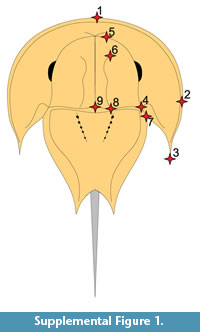 Tasmania (UTGD), Hobart, Tasmania, Australia. The fossil is preserved as a slightly domed internal mould without cuticle or a counterpart, on a slab of dark-yellow shale. UTGD 123979 was coated with ammonium chloride sublimate and photographed under LED lighting with a Canon EOS 5DS housed at the University of New England, Armidale, NSW, Australia. Images were stacked using Helicon Focus 7 (Helicon Soft Limited) stacking software. Description of UTGD 123979 hereinfollows the systematic taxonomy of Lamsdell (2013, 2016) and anatomical terms presented in Bicknell et al. (2018a, 2019a) and Bicknell and Pates (2019). The specimen was measured using the measuring tool in ImageJ. This approach was used to derive the most accurate length and width values of the specimen.
Tasmania (UTGD), Hobart, Tasmania, Australia. The fossil is preserved as a slightly domed internal mould without cuticle or a counterpart, on a slab of dark-yellow shale. UTGD 123979 was coated with ammonium chloride sublimate and photographed under LED lighting with a Canon EOS 5DS housed at the University of New England, Armidale, NSW, Australia. Images were stacked using Helicon Focus 7 (Helicon Soft Limited) stacking software. Description of UTGD 123979 hereinfollows the systematic taxonomy of Lamsdell (2013, 2016) and anatomical terms presented in Bicknell et al. (2018a, 2019a) and Bicknell and Pates (2019). The specimen was measured using the measuring tool in ImageJ. This approach was used to derive the most accurate length and width values of the specimen.
A geometric morphometric analysis using the method in Bicknell et al. (2019a) was conducted to quantitatively assess where UTGD 123979 is located in morphospace relative to other taxa. Contrasting Bicknell et al. (2019a) who used semilandmarks and landmarks, here only a landmark analysis of 21 specimens was conducted. The species considered are from the families Austrolimulidae and Paleolimulidae (sensu Lamsdell, 2016 and Lerner et al., 2017). Landmarking was conducted using the Thin-Plate Spline (TPS) suite. A TPS file was constructed using tpsUtil64 (v.1.7). The TPS file was imported into tspDig2 (v.2.26), which was used to place nine landmarks on the right cephalothorax (Supplementary Figure 1, Supplementary Table 1). Points are digitised as xy coordinates. Where the right side was poorly preserved, the left side was used instead, and the data mirrored. These points populated the TPS file with landmark data (Supplementary Information 1). The TPS file was imported into R. The ‘geomorph’ package (Adams and Otárola-Castillo, 2013) conducted the Procrustes Superimposition and Principal Components Analysis (PCA) of the data. Procrustes Superimposition standardises for size and orientation so that only shape variation was assessed. Only the first two Principal Components (PCs) were considered as they explain 69.9% of the variation in the data (Supplementary Information 2). It is important to note that results of geometric morphometric analyses can be impacted by taphonomy. However, Bicknell et al. (2019a) highlighted that the preservational mode of fossil horseshoe crabs does not impact the distribution of specimens in PC space. Finally, the generic and family assignment follows notes in Lamsdell (2016) and Lerner et al. (2017).
To evaluate the phylogenetic position of Tasmaniolimulus patersoni gen. et sp. nov., the taxon was coded into the matrix of Lamsdell (2016). This matrix contains a range of fossil and extant euchelicerates (Supplementary Information 3). The analysis was performed under equal-weights parsimony in TNT 1.5 (Goloboff and Catalano, 2016) and the “New Technology” tree search strategy using the default settings for searches, tree fusing, and drifting, together with 1000 iterations of the parsimony ratchet. All multistate characters were considered unordered, just as in the original analysis.
SYSTEMATIC PALAEONTOLOGY
Subphylum CHELICERATA Heymons, 1901
Order XIPHOSURIDA Latreille, 1802
Suborder LIMULINA Richter and Richter, 1929
Family AUSTROLIMULIDAE Riek, 1955
Genus Tasmaniolimulus gen. nov.
zoobank.org/4E73EC90-EEBF-4CC1-9E3A-EDD13F0A10D2
Etymology. Generic name Tasmaniolimulus is suggested as the specimen is from the Australian state of Tasmania and Limulus is the genus of the iconic extant North American horseshoe crab.
Type species. Tasmaniolimulus patersoni, type and only species.
Diagnosis. Austrolimulid with genal spines extending posteriorly to the terminus of the thoracetron, without substantial splay; a prominent M-shaped terminus to the ophthalmic ridges, and thoracetron smaller than cephalothorax.
Tasmaniolimulus patersoni sp. nov.
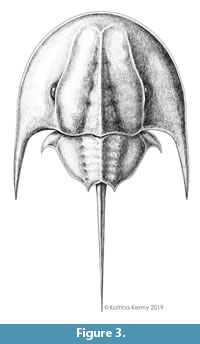
Figure 2 and Figure 3
zoobank.org/6DE9B99C-9554-47DB-B25B-9A962B2EC317
 1989 Paleolimulus sp. Ewington et al., p. 128, fig. 1.
1989 Paleolimulus sp. Ewington et al., p. 128, fig. 1.
1993 Paleolimulus sp. Selden, p. 12.
2003 Paleolimulidae Itow et al., fig. 7.
2005 Paleolimulus sp. Allen and Feldmann, p. 594.
2013 Paleolimulus sp. Lamsdell et al., p. 344.
2017 “Paleolimulus” sp. Lerner et al., p. 294.
Diagnosis. Same as for genus.
Etymology. The trivial name patersoni was chosen in recognition of John R. Paterson who has contributed extensively to Australian arthropod palaeontology and xiphosurid research across his career.
Holotype. UTGD 123979. The specimen is preserved on a slab of dark-yellow shale as an internal mould with a small degree of relief.
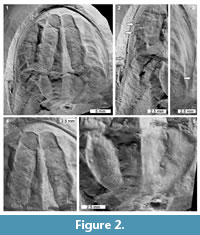
Description. UTGD 123979 is an articulated cephalothorax and partial thoracetron without a telson preserved as an internal mould (Figure 2, Figure 3). Specimen is 24.14 mm long, slightly domed, with a prominent axial ridge. Cephalothorax preserved well on both sides, parabolic in shape, and 16.9 mm long at midline. Cephalothoracic width across the posterior margin of lateral rims 23.8 mm. A cephalothoracic rim is preserved along margins and attains a greatest width of 0.83 mm. Left side of cephalothoracic doublure is partly preserved and visible. Ophthalmic ridges 14 mm long, pronounced, with slight concavity towards lateral borders, converging anteriorly to form an ‘M’-shaped terminus (Figure 2.1, 2.4). A putative lateral compound eye is discerned on the left ophthalmic ridge, 4.46 mm anteriorly from the posterior cephalothoracic boarder. A pronounced cardiac lobe is noted. Lobe is 5.55 mm wide posteriorly, tapering anteriorly into a triangular shape with a width of 1.37 mm. Cardiac lobe has a medial ridge that is 13.9 mm long. Interophthalmic ridges bordering the cardiac lobe are 11.43 and 11.31 mm long (left and right, respectively). Ocelli are not observed. Both genal spines are preserved, but the posterior edge of right genal spine is broken. Left genal spine is 11.24 mm long and terminates posteriorly of the thoracetron end. Genal spine tip is 12.4 mm from the sagittal line. Angle between the left genal spine and left side of the thoracetron is 59°. Inner margin of the left genal spine is curved slightly anteriorly, with a small indentation in the first fifth of the spine length (Figure 2.2, Figure 3). Right genal spine 4.7 mm long, terminating at rock edge, half way along thoracetron (Figure 2.3). Angle between right genal spine and right side of thoracetron 64°. Inner margin of the right genal spine is curved slightly anteriorly, with small indentation within the first fifth of the spine length (Figure 2.3). Hinge between cephalothorax and thoracetron is pronounced, 12.4 mm wide and has a thickness of 0.3 mm. Impressions of distal sections of walking legs, likely the tibiotarsus and dactylopodite, are preserved on the left side of the cephalothorax (Figure 2.2).
 Thoracetron trapezoidal, at least 6.74 mm long and 9.96 mm wide anteriorly, increasing to a width of 12.7 mm at 2 mm along the thoracetron (Figure 2.5). Increase in width is associated with a possible anterolateral spine and extension of a free lobe (Figure 2.2). Thoracetron then tapers to at least 3.65 mm posteriorly. Thoracetron is less wide and shorter than cephalothorax. Right side of the thoracetron preserved more completely than left. Axial lobe present, triangular in shape, tapering from 5.59 mm anteriorly to 3.14 mm posteriorly. At least five segmentary axial furrows are visible, ranging between 0.98 and 1.3 mm in length. At least five, possibly six, pairs of apodemal pits are visible (Figure 2.5). Medial ridge present, 6.61 mm in length. Left pleural lobe has limited relief and no segments, 3.16 mm wide anteriorly, tapering to 1.16 mm posteriorly. Marginal rim not preserved. Right pleural lobe wider than the left, 3.6 mm wide anteriorly, tapering to 1.16 mm posteriorly. No segmentation noted. Marginal rim present and is 0.55 mm wide along its length. No evidence of movable or fixed spines. Telson not preserved.
Thoracetron trapezoidal, at least 6.74 mm long and 9.96 mm wide anteriorly, increasing to a width of 12.7 mm at 2 mm along the thoracetron (Figure 2.5). Increase in width is associated with a possible anterolateral spine and extension of a free lobe (Figure 2.2). Thoracetron then tapers to at least 3.65 mm posteriorly. Thoracetron is less wide and shorter than cephalothorax. Right side of the thoracetron preserved more completely than left. Axial lobe present, triangular in shape, tapering from 5.59 mm anteriorly to 3.14 mm posteriorly. At least five segmentary axial furrows are visible, ranging between 0.98 and 1.3 mm in length. At least five, possibly six, pairs of apodemal pits are visible (Figure 2.5). Medial ridge present, 6.61 mm in length. Left pleural lobe has limited relief and no segments, 3.16 mm wide anteriorly, tapering to 1.16 mm posteriorly. Marginal rim not preserved. Right pleural lobe wider than the left, 3.6 mm wide anteriorly, tapering to 1.16 mm posteriorly. No segmentation noted. Marginal rim present and is 0.55 mm wide along its length. No evidence of movable or fixed spines. Telson not preserved.
Remarks. UTGD 123979 was originally considered a paleolimulid by Ewington et al. (1989). They compared UTGD 123979 to Paleolimulus and Limulitella Størmer, 1952, and noted that the parabolic cephalothorax was twice as long as the thoracetron, a condition unique to the taxon and potentially indicative of a new genus. Advances in horseshoe crab taxonomy, morphometrics, and phylogeny highlight that this specimen is indeed representative of a unique genus as explored in Lerner et al. (2017). The feature that excludes UTGD 123979 from Paleolimulidae, and relates the specimen to Austrolimulidae, is the overdevelopment of the genal spines that terminate near the level of the telson (Lerner et al., 2017). Such genal spine development is not known in limulids or paleolimulids, but is observed in select belinurids (Haug et al., 2012; Haug and Rötzer, 2018). However, the completely fused thoracetron and lack of ophthalmic spines precludes the assignment of UTGD 123979 to Belinuridae. Additional characteristics that UTGD 123979 exhibits supporting an austrolimulid assignment are pronounced apodemal pits and absence of fixed spines. Furthermore, the thoracetron in UTGD 123979 is half the size of the cephalothorax, a feature known in Dubbolimulus peetae and Austrolimulus fletcheri. This difference in cephalothorax and thoracetronic size is related to the austrolimulid tendency towards reduction or accentuation of the xiphosurid body plan. Finally, UTGD 123979 has an array of unique characteristics relative to other austrolimulids that warrant the erection of Tasmaniolimulus patersoni gen. et sp. nov. These features are the posteriorly projected genal spines with a slight indentation, comparable to Sloveniolimulus rudkini Bicknell, Žalohar, Miklavc, Celarc, Križnar, and Hitij, 2019, and the notable absence of the swallowtail feature, the condition where posteriormost fixed thoracetronic spines “are swept back and elongated” into a swallowtail-like morphology (Lamsdell, 2016, p. 185). Finally, the assignment of UTGD 123979 to the new genus and to Austrolimulidae is supported by geometric morphometric results (discussed below).
Occurrence. The species is known only from its type locality: Jackey Shale at Poatina, 41°48'05"S 146°53'06"E, 3 m below the Ross Sandstone; Upper Permian.
GEOMETRIC MORPHOMETRICS
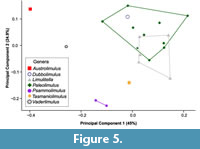
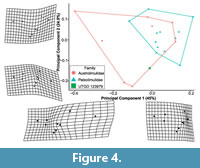 The PCA plot shows how PC1 (45% shape variation) describes the posterior extension of the most distal genal spine point (Landmark 3, see Supplementary Figure 1) relative to the cephalothorax (Figure 4). Austrolimulus fletcheri is therefore located in distinctly negative PC1 space. PC2 (24.9% shape variation) describes the lateral extent of the outer-most cephalothoracic edge relative to the sagittal line (Landmark 2, see Supplementary Figure 1) and the lateral extent of the posterior edge of the ophthalmic ridge relative to the sagittal line (Landmark 8, see Supplementary Figure 1). UTGD 123979 is located in a PC1 space of ~0 (PC1 = -0.01) and a slightly negative PC2 space (PC2 = -0.14) (Figure 4, Figure 5). The genus falls within the outer limits of the shape space occupied by austrolimulids (as defined by the convex hull around austrolimulid specimens in PC space) (Figure 4) and outside the limits of the other genera when considering the generic distribution of specimens (Figure 5).There is an overlap between Paleolimulus taxa and Limulitella. Furthermore, Dubbolimulus peetae is located within the Paleolimulus convex hull.
The PCA plot shows how PC1 (45% shape variation) describes the posterior extension of the most distal genal spine point (Landmark 3, see Supplementary Figure 1) relative to the cephalothorax (Figure 4). Austrolimulus fletcheri is therefore located in distinctly negative PC1 space. PC2 (24.9% shape variation) describes the lateral extent of the outer-most cephalothoracic edge relative to the sagittal line (Landmark 2, see Supplementary Figure 1) and the lateral extent of the posterior edge of the ophthalmic ridge relative to the sagittal line (Landmark 8, see Supplementary Figure 1). UTGD 123979 is located in a PC1 space of ~0 (PC1 = -0.01) and a slightly negative PC2 space (PC2 = -0.14) (Figure 4, Figure 5). The genus falls within the outer limits of the shape space occupied by austrolimulids (as defined by the convex hull around austrolimulid specimens in PC space) (Figure 4) and outside the limits of the other genera when considering the generic distribution of specimens (Figure 5).There is an overlap between Paleolimulus taxa and Limulitella. Furthermore, Dubbolimulus peetae is located within the Paleolimulus convex hull.
PHYLOGENETIC ANALYSIS
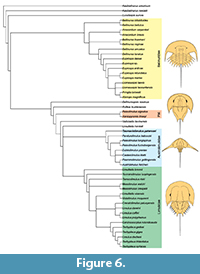
 The phylogenetic analysis resulted in five most parsimonious trees (CI: 0.473, RI: 0.880). The topology of the strict consensus tree is similar to those in Selden et al. (2015), Lamsdell (2016) and Bicknell et al. (2019d) (Figure 6). Tasmaniolimulus patersoni gen. et sp. nov. is resolved as the earliest diverging representative of a monophyletic Austrolimulidae (sensu Lamsdell, 2016), superseding Panduralimulus babcocki Allen and Feldmann, 2005, at the base of the clade. No other changes are noted.
The phylogenetic analysis resulted in five most parsimonious trees (CI: 0.473, RI: 0.880). The topology of the strict consensus tree is similar to those in Selden et al. (2015), Lamsdell (2016) and Bicknell et al. (2019d) (Figure 6). Tasmaniolimulus patersoni gen. et sp. nov. is resolved as the earliest diverging representative of a monophyletic Austrolimulidae (sensu Lamsdell, 2016), superseding Panduralimulus babcocki Allen and Feldmann, 2005, at the base of the clade. No other changes are noted.
DISCUSSION
 The morphometric, phylogenetic, and systematic assessments presented here confirm that Tasmaniolimulus patersoni gen. et sp. nov. is a valid taxon and as such represents the oldest described Gondwanan austrolimulid (Lerner et al., 2017), increasing the diversity of Australian austrolimulid genera, and indeed species, to three. These results have also built on Lamsdell (2016) and Lerner et al. (2017) to confirm that Paleolimulus has been somewhat treated as a waste-basket genus. Taxa within this paraphyletic genus that exhibit accentuated or diminished features (see discussion in Lerner et al., 2017) are therefore in need of revision. This research effort will undoubtedly impact the phylogenetic construction of Austrolimulidae and, more broadly, Xiphosurida.
The morphometric, phylogenetic, and systematic assessments presented here confirm that Tasmaniolimulus patersoni gen. et sp. nov. is a valid taxon and as such represents the oldest described Gondwanan austrolimulid (Lerner et al., 2017), increasing the diversity of Australian austrolimulid genera, and indeed species, to three. These results have also built on Lamsdell (2016) and Lerner et al. (2017) to confirm that Paleolimulus has been somewhat treated as a waste-basket genus. Taxa within this paraphyletic genus that exhibit accentuated or diminished features (see discussion in Lerner et al., 2017) are therefore in need of revision. This research effort will undoubtedly impact the phylogenetic construction of Austrolimulidae and, more broadly, Xiphosurida.
The erection of Tasmaniolimulus patersoni gen. et sp. nov. confirms the thesis that austrolimulids had a Palaeozoic origin and diversified during the Triassic―an event that occurred concurrently with the limulid diversification (Lamsdell, 2016; Lerner et al., 2017; Bicknell et al., 2019a). The austrolimulid radiation likely reflects the exploration and exploitation of freshwater environments (Lamsdell, 2016). It seems plausible that the larger genal spines characteristic of the family might have reduced the impact of uni-directional currents in freshwater flow regimes (Anderson, 1996), but may also have aided in sub-aerial activity or self-defence (Fisher, 1979). In the case of T. patersoni gen. et sp. nov., the austrolimulid would have experienced an extreme limit of freshwater conditions by inhabiting the glacial climatic regime of Tasmania in the Late Palaeozoic (Ewington et al., 1989).
Geometric morphometric analyses have previously been applied effectively to explore horseshoe crab evolution (Bicknell et al., 2019a). The preliminary geometric morphometric results of Bicknell et al. (2019a) illustrated mathematically how extreme the austrolimulid morphologies are when compared to other xiphosurid families. This same study also noted that if the definition of austrolimulids was changed, for example to follow definitions in Lamsdell (2016) and Lerner et al. (2017), the morphospace occupied by the family would require reconsideration. Here, I have shown that with a broader definition of the family, austrolimulids are far more diverse than previously considered. This broader definition has highlighted that geometric morphometric analyses of horseshoe crabs produce far more complex results than Bicknell et al. (2019a) had originally suggested and a degree of caution is needed when interpreting these outcomes. Alternatively, the exclusion of semi-landmarks in my analysis resulted in more overlap between groups. Future research should exploit both landmarks and semi-landmarks. Nonetheless, the overlap with Paleolimulidae and Austrolimulidae confirms that a careful reconsideration of paleolimulids is needed, as discussed above. In this context, one consideration is given to Dubbolimulus peetae. Originally placed into its own genus, Dubbolimulus, the species has also been placed within Paleolimulus (sensu Hauschke and Wilde, 1987, and Dunlop et al., 2019) but has been resolved within Austrolimulidae in phylogenetic analyses (Lamsdell, 2016). The geometric morphometric results presented here show that the taxon falls within the Paleolimulus morphospace, suggesting that the taxon could indeed be a paleolimulid. However, D. peetae has a much smaller thoracetron compared to the cephalothorax (see comments in Lerner et al., 2017). This reduction of body elements is typical of Austrolimulidae and, as such, D. peetae is most likely an austrolimulid (sensu Lamsdell, 2016, Figure 6). An extension of the geometric morphometric analyses that might highlight and explore this distinction is the inclusion of thoracetron data, an area of future research in the taxonomy of Xiphosurida. This new direction may also produce more distinct differentiations in morphospace and potentially uncover even more interesting evolutionary patterns exhibited by this iconic group of marine chelicerates.
CONCLUSIONS
Revision of a horseshoe crab specimen from the Late Permian of Tasmania has highlighted the need for a new taxon. The combination of systematic palaeontology, phylogenetic analyses, and geometric morphometrics was therefore used to erect Tasmaniolimulus patersoni. This taxon confirms the Palaeozoic origin of Austrolimulidae and illustrates new examples of curious morphologies for which the family is known. Further revision of Paleolimulus will undoubtedly result in the description of more austrolimulids. This research direction is key for uncovering the true diversity of these oddball xiphosurids.
ACKNOWLEDGMENTS
This research was supported by funding from an Australian Postgraduate Award, a Charles Schuchert and Carl O. Dunbar Grants-in-Aid award, a James R. Welch Scholarship, and a Betty Mayne Scientific Research Fund. I thank the UTGD and the curator thereof, I. von Lichtan, for kindly sending the specimen to UNE. I thank S. Pates for his insightful ideas regarding the text and for the permission to use his reconstructions of select taxa. I thank K. Kenny for her stunning reconstruction of Tasmaniolimulus patersoni. Finally, I thank the three anonymous reviewers and the handling editor, M. Hyžný, for their comments that thoroughly improved the text.
REFERENCES
Adams, D.C. and Otárola-Castillo, E. 2013. geomorph: an R package for the collection and analysis of geometric morphometric shape data. Methods in Ecology and Evolution, 4:393-399. https://doi.org/10.1111/2041-210x.12035
Allen, J.G. and Feldmann, R.M. 2005. Panduralimulus babcocki n. gen. and sp., a new Limulacean horseshoe crab from the Permian of Texas. Journal of Paleontology, 79:594-600. https://doi.org/10.1666/0022-3360(2005)079<0594:pbngas>2.0.co;2
Anderson, L.I. 1996. Taphonomy and Taxonomy of Palaeozoic Xiphosura, Unpublished PhD Thesis, University of Manchester, Manchester, UK.
Anderson, L.I. and Selden, P.A. 1997. Opisthosomal fusion and phylogeny of Palaeozoic Xiphosura. Lethaia, 30:19-31. https://doi.org/10.1111/j.1502-3931.1997.tb00440.x
Babcock, L.E. and Merriam, D.F. 2000. Horseshoe crabs (Arthropoda: Xiphosurida) from the Pennsylvanian of Kansas and elsewhere. Transactions of the Kansas Academy of Science, 103:76-94. https://doi.org/10.2307/3627941
Banks, M.R. 1973. General geology, p. 25-33. In Banks, M.R. (ed.), The Lake Country of Tasmania. Royal Society of Tasmania, Hobart.
Bicknell, R.D.C., Amati, L., and Ortega-Hernández, J. 2019b. New insights into the evolution of lateral compound eyes in Palaeozoic horseshoe crabs. Zoological Journal of the Linnean Society: zlz065. https://doi.org/10.1093/zoolinnean/zlz065
Bicknell, R.D.C., Brougham, T., Charbonnier, S., Sautereau, F., Hitij, T., and Campione, N.E. 2019c. On the appendicular anatomy of the xiphosurid Tachypleus syriacus and the evolution of fossil horseshoe crab appendages. The Science of Nature, 106:38. https://doi.org/10.1007/s00114-019-1629-6
Bicknell, R.D.C., Klinkhamer, A.J., Flavel, R.J., Wroe, S., and Paterson, J.R. 2018a. A 3D anatomical atlas of appendage musculature in the chelicerate arthropod Limulus polyphemus. PLoS ONE, 13:e0191400. https://doi.org/10.1371/journal.pone.0191400
Bicknell, R.D.C., Ledogar, J.A., Wroe, S., Gutzler, B.C., Watson III, W.H., and Paterson, J.R. 2018b. Computational biomechanical analyses demonstrate similar shell-crushing abilities in modern and ancient arthropods. Proceedings of the Royal Society B: Biological Sciences, 285:20181935. https://doi.org/10.1098/rspb.2018.1935
Bicknell, R.D.C., Lustri, L., and Brougham, T. 2019d. Revision of "Bellinurus" carteri (Chelicerata: Xiphosura) from the Late Devonian of Pennsylvania, USA. Comptes Rendus Palevol. https://doi.org/10.1016/j.crpv.2019.08.002
Bicknell, R.D.C., Paterson, J.R., Caron, J.-B., and Skovsted, C.B. 2018c. The gnathobasic spine microstructure of recent and Silurian chelicerates and the Cambrian artiopodan Sidneyia: Functional and evolutionary implications. Arthropod Structure & Development, 47:12-24. https://doi.org/10.1016/j.asd.2017.12.001
Bicknell, R.D.C. and Pates, S. 2019. Abnormal extant xiphosurids in the Yale Peabody Museum Invertebrate Zoology collection. Bulletin of the Peabody Museum of Natural History, 60:41-53. https://doi.org/10.3374/014.060.0102
Bicknell, R.D.C., Žalohar, J., Miklavc, P., Celarc, B., Križnar, M., and Hitij, T. 2019a. A new limulid genus from the Strelovec Formation (Middle Triassic, Anisian) of northern Slovenia. Geological Magazine, 1-14. https://doi.org/10.1017/S0016756819000323
Blake, F., Everard, G.B., Voisey, A.H., and McKellar, J.B.A. 1956. Greak Lake. Geological Atlas 1 mile Series. Zone 7, Sheet No. 53. Geological Survey of Tasmania Department of Mines.
Dunbar, C.O. 1923. Kansas Permian insects, Part 2, Paleolimulus, a new genus of Paleozoic Xiphosura, with notes on other genera. American Journal of Science, 5:443-454. https://doi.org/10.2475/ajs.s5-5.30.443
Dunlop, J.A., Penney, D., and Jekel, D. 2019. A summary list of fossil spiders and their relatives, accessed 01/05/2019. In World Spider Catalog, version 19.5, Natural History Museum Bern. https://wsc.nmbe.ch
Ewington, D.L., Clarke, M.J., and Banks, M.R. 1989. A Late Permian fossil horseshoe crab (Paleolimulus: Xiphosura) from Poatina, Great Western Tiers, Tasmania. Papers and Proceedings of the Royal Society of Tasmania, 123:127-131.
Fisher, D.C. 1979. Evidence for subaerial activity of Euproops danae (Merostomata, Xiphosurida), p. 379-447. In Nitecki, M.H. (ed.), Mazon Creek Fossils. Elsevier, New York. https://doi.org/10.1016/b978-0-12-519650-5.50022-9
Goloboff, P.A. and Catalano, S.A. 2016. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics, 32:221-238. https://doi.org/10.1111/cla.12160
Haug, C. and Rötzer, M.A.I.N. 2018. The ontogeny of the 300 million year old xiphosuran Euproops danae (Euchelicerata) and implications for resolving the Euproops species complex. Development Genes and Evolution, 228:63-74. https://doi.org/10.1007/s00427-018-0604-0
Haug, C., Van Roy, P., Leipner, A., Funch, P., Rudkin, D.M., Schöllmann, L., and Haug, J.T. 2012. A holomorph approach to xiphosuran evolution--a case study on the ontogeny of Euproops. Development Genes and Evolution, 222:253-268. https://doi.org/10.1007/s00427-012-0407-7
Hauschke, N., and Wilde, V. 1987. Paleolimulus fuchsbergensis n. sp. (Xiphosura, Merostomata) aus der oberen Trias von Nordwestdeutschland, mit einer Übersicht zur Systematik und Verbreitung rezenter Limuliden. Paläontologische Zeitschrift, 61:87-108. https://doi.org/10.1007/bf02985944
Heymons, R. 1901. Die Entwicklungsgeschichte der Scolopender. Zoologica, 13:1-244. https://doi.org/10.5962/bhl.title.1587
Itow, T., Kato, H., and Kato, K. 2003. Horseshoe crabs in Australia. Bulletin of the Education Faculty, Shizuoka University. Natural Science series, 53:11-22.
Kin, A. and Błażejowski, B. 2014. The horseshoe crab of the genus Limulus: living fossil or stabilomorph? PLoS ONE, 9:e108036. https://doi.org/10.1371/journal.pone.0108036
Lamsdell, J.C. 2013. Revised systematics of Palaeozoic ‘horseshoe crabs’ and the myth of monophyletic Xiphosura. Zoological Journal of the Linnean Society, 167:1-27. https://doi.org/10.1111/j.1096-3642.2012.00874.x
Lamsdell, J.C. 2016. Horseshoe crab phylogeny and independent colonizations of fresh water: ecological invasion as a driver for morphological innovation. Palaeontology, 59:181-194. https://doi.org/10.1111/pala.12220
Lamsdell, J.C., Percival, I.G., and Poschmann, M. 2013. The problematic ‘chelicerate’ Melbournopterus crossotus Caster & Kjellesvig-Waering: A case of mistaken identity. Alcheringa, 37:344-348. https://doi.org/10.1080/03115518.2013.764681
Latreille, P.A. 1802. Histoire Naturelle, Générale et Particulière, des Crustacés et des Insectes, 3. Dufart, Paris.
Lerner, A.J., Lucas, S.G., and Lockley, M. 2017. First fossil horseshoe crab (Xiphosurida) from the Triassic of North America. Neues Jahrbuch für Geologie und Paläontologie-Abhandlungen, 286:289-302. https://doi.org/10.1127/njgpa/2017/0702
Moore, R.A., McKenzie, S.C., and Lieberman, B.S. 2007. A Carboniferous synziphosurine (Xiphosura) from the Bear Gulch Limestone, Montana, USA. Palaeontology, 50:1013-1019. https://doi.org/10.1111/j.1475-4983.2007.00685.x
Pickett, J.W. 1984. A new freshwater limuloid from the middle Triassic of New South Wales. Palaeontology, 27:609-621.
Pickett, J.W. 1993. A Late Devonian xiphosuran from near Parkes, New South Wales. Memoirs of the Association of Australian Palaeontologists, 15:279-287.
Richter, R., and Richter, E. 1929. Weinbergina opitzi n. g. n. sp., ein Schwertträger (Merost., Xiphos.) aus dem Devon (Rheinland). Senckenbergiana, 11:193-209.
Riek, E.F. 1955. A new xiphosuran from the Triassic sediments at Brookvale, New South Wales. Records of the Australian Museum, 23:281-282. https://doi.org/10.3853/j.0067-1975.23.1955.637
Riek, E.F., and Gill, E.D. 1971. A new xiphosuran genus from Lower Cretaceous freshwater sediments at Koonwarra, Victoria, Australia. Palaeontology, 14:206-210.
Rudkin, D.M. and Young, G.A. 2009. Horseshoe crabs-an ancient ancestry revealed, p. 25-44. In Tanacredi, J.T., Botton, M.L., and Smith, D.R. (eds.), Biology and Conservation of Horseshoe Crabs. Springer, New York. https://doi.org/10.1007/978-0-387-89959-6_2
Selden, P.A. 1993. Fossil chelicerates of Australia. The Fossil Collector, 41:11-20.
Selden, P.A., Lamsdell, J.C., and Qi, L. 2015. An unusual euchelicerate linking horseshoe crabs and eurypterids, from the Lower Devonian (Lochkovian) of Yunnan, China. Zoologica Scripta, 44:645-652. https://doi.org/10.1111/zsc.12124
Shuster Jr., C.N. 1982. A pictorial review of the natural history and ecology of the horseshoe crab Limulus polyphemus, with reference to other Limulidae. Progress in Clinical and Biological Research, 81:1-52.
Størmer, L. 1952. Phylogeny and taxonomy of fossil horseshoe crabs. Journal of Paleontology, 26:630-640.
Van Roy, P., Briggs, D.E.G., and Gaines, R.R. 2015. The Fezouata fossils of Morocco; an extraordinary record of marine life in the Early Ordovician. Journal of the Geological Society, 172:541-549. https://doi.org/10.1144/jgs2015-017
Van Roy, P., Orr, P.J., Botting, J.P., Muir, L.A., Vinther, J., Lefebvre, B., El Hariri, K., and Briggs, D.E.G. 2010. Ordovician faunas of Burgess Shale type. Nature, 465:215-218. https://doi.org/10.1038/nature09038

