 Lower Jurassic corals from Morocco with skeletal structures convergent with those of Paleozoic rugosan corals
Lower Jurassic corals from Morocco with skeletal structures convergent with those of Paleozoic rugosan corals
Article number: 22.2.48
https://doi.org/10.26879/874
Copyright Paleontological Society, August 2019
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Submission: 3 April 2018. Acceptance: 2 July 2019
{flike id=2654}
ABSTRACT
Neorylstonia nom. nov. pseudocolumellata, a replacement name for Mesophyllum pseudocolumellatum Beauvais, 1986, is only known from uppermost Sinemurian to uppermost Pliensbachian (Lower Jurassic) strata of Morocco. This solitary coral species went extinct during the Pliensbachian-Toarcian transition (~183.5 My), which is currently considered to be a second order biodiversity crisis linked to a period of rapid and global paleoenvironmental disturbances. Neorylstonia pseudocolumellata has a distinctive skeletal organization. The axial structure of the corallite, which has no equivalent in Mesozoic taxa, is reminiscent of the skeletal organization of some Paleozoic rugose corals such as Amygdalophyllum Dun and Benson, 1920, or Rylstonia Hudson and Platt, 1927. This similarity is based on the occurrence of a calicular boss also named pseudocolumella. As Rugosa and Scleractinia do not appear closely related, we consider that this feature cannot be due to evolutionary inheritance from a Rugosan ancestor. The morphological aspects of the skeleton have been assessed in order to understand the function and growth of this axial “calicular boss” as well as to more precisely describe the genus and species characters. The organization of the septal apparatus points to a typical Scleractinian pattern, which is also supported by the original aragonitic mineralogy of the skeleton. This implies a convergence phenomenon, leaving open the question of the functional significance of such a calicular organization. We posit that this feature was used for sexual reproduction and so did not always develop in a population.
Raphaël Vasseur. Université de Lorraine, CNRS, lab. GeoRessources, UMR 7359, BP 70239, 54506, Vandoeuvre-lès-Nancy Cedex, France. raphael.vasseur@univ-lorraine.fr
Simon Boivin. University of Geneva, Department of Earth Sciences, Rue des Maraîchers 13, 1205 Geneva, Switzerland. simon.boivin@unige.ch
Bernard Lathuilière. Université de Lorraine, CNRS, lab. GeoRessources, UMR 7359, BP 70239, 54506, Vandoeuvre-lès-Nancy Cedex, France. bernard.lathuiliere@univ-lorraine.fr
Iuliana Lazar. University of Bucharest, Faculty of Geology and Geophysics, Department of Geology and Paleontology, 1, N. Balcescu Ave., RO - 010041, Bucharest, Romania. iuliana.lazar@g.unibuc.ro
Christophe Durlet. UMR CNRS 6282 Biogéosciences, Univ. Bourgogne Franche-Comté, 21000 Dijon, France. christophe.durlet@u-bourgogne.fr
Rowan-Clare Martindale. Department of Geological Sciences, Jackson School of Geosciences, University of Texas at Austin, 2275 Speedway Stop C9000, Austin, Texas 78712, USA. martindale@jsg.utexas.edu
Stéphane Bodin. Aarhus University, Department of Geoscience, Høegh-Guldbergs Gade 2, 8000 Aarhus C, Denmark. stephane.bodin@geo.au.dk
Khalid Elhmidi. Département de l'Energie et des Mines Direction de la Géologie, Rue Abou Marouane Essaadi BP 6208, Haut Agdal, Rabat, Morocco. k.elhmidi@mem.gov.ma
Keywords: corals; Early Jurassic; CT scan; taxonomy; Rugosa; Scleractinia
Vasseur, Raphaël, Boivin, Simon, Lathuilière, Bernard, Lazar, Iuliana, Durlet, Christophe, Martindale, Rowan-Clare, Bodin, Stéphane, and Elhmidi, Khalid. 2019. Lower Jurassic corals from Morocco with skeletal structures convergent with those of Paleozoic rugosan corals. Palaeontologia Electronica 22.2.48A 1-32. https://doi.org/10.26879/874
palaeo-electronica.org/content/2019/2654-neorylstonia-paper
Copyright: August 2019 Paleontological Society.
This is an open access article distributed under the terms of Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0), which permits users to copy and redistribute the material in any medium or format, provided it is not used for commercial purposes and the original author and source are credited, with indications if any changes are made.
creativecommons.org/licenses/by-nc-sa/4.0/
http://zoobank.org/14F3D176-317B-4AFF-B165-5CD5CD1220F9
INTRODUCTION
The Pliensbachian/Toarcian boundary event (~183.5 Ma) and the Toarcian Oceanic Anoxic Event (Early Toarcian, ~183 Ma) are two intervals of worldwide environmental and ecological disruption during the Early Jurassic. This period is known for large-scale black shale deposits (e.g., Jenkyns, 1985, 1988; Jenkyns and Clayton, 1986; Schmid-Röhl et al., 2002) and multiple negative excursions of δ13C affecting exogenic carbon reservoirs and associated sediments (e.g., Pálfy et al., 2002; Littler et al., 2010; Bodin et al., 2016). These events have been associated with marine eutrophication, increased ocean anoxia (e.g., Röhl et al., 2001; Morard et al., 2003, Bodin et al., 2010), high global temperatures (e.g., Vakhrameyev, 1982; Jenkyns, 2003; Rosales et al., 2004, Suan et al., 2010), putative acidification of the oceans (Trecalli et al., 2012), and global disturbances of the carbon cycle attributed to the pulsed emplacement of the Karoo-Ferrar flood basalts (Duncan et al., 1997; Sell et al., 2014; Burgess et al., 2015). These paleoenvironmental disturbances appear to be contemporaneous with a second-order biodiversity crisis affecting various taxonomic groups such as ammonites, foraminifera, bivalves, and brachiopods (e.g., Raup and Sepkoski, 1984, 1986; Harries and Little, 1999; Caswell et al., 2009; Dera et al., 2010; Guex et al., 2012; Goričan et al., 2013; Caruthers et al., 2013; Danise et al., 2013, 2015; Caruthers et al., 2014; Martindale and Aberhan, 2017). Nevertheless, data about coral turnover are relatively sparse, and the only detailed study is based on a compilation of bibliographical data (Lathuilière and Marchal, 2009).
This publication focuses on one of the victims of the Pliensbachian/Toarcian boundary crisis; this unique coral has features that are reminiscent of Paleozoic coral structures, likely a case of convergent evolution rather than inheritance. As Rugosa and Scleractinia are not closely related, a case of inheritance is excluded (see for examples Oliver, 1980; Fedorowski, 1997). This taxon was initially described in Beauvais (1986) as Mesophyllum pseudocolumellatum but the genus name is currently unavailable because of homonymy; thus a nomen novum is required. The original description of this species was established on the basis of a narrow sampling of only seven poorly preserved specimens from Beni Tadjit in Morocco (Menchikoff collections), and the genus is based on these seven samples and six other specimens from the Jebel Bou Dahar found in the Du Dresnay collections. These other samples were considered by Beauvais as another potential species, but the preservation was too poor to validate this assumption. The present study is based on the analysis of 91 specimens of Mesophyllum collected and/or pictured by our team during field campaigns from the Dades Valley and the Amellagou region in the Central High Atlas as well as from the Guigou Plateau in the Middle Atlas of Morocco. This significant new sampling effort results in new insights about the morphology of this taxon, its taxonomy, its biostratigraphic attribution, and sedimentological context. This work also raises new questions about the phylogeny and function morphology of this coral taxon.
GEOLOGICAL SETTING
In Morocco, studied sites are located in the Amellagou area, the Dades Valley, and the Guigou Plateau; GPS locations are presented in Table 1. Liassic distal environments in Morocco contain abundant biostratigraphically useful ammonites (see synthesis in Dommergues and Meister, 2017) and so there is a relatively good biostratigraphical framework established for the coral occurrences reported here.
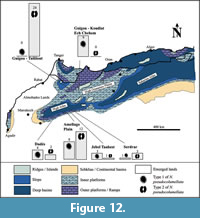
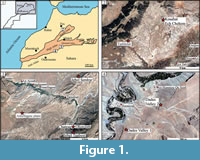 The Guigou Plateau is located in Middle Atlas (Figure 1.1, 1.2), where several huge carbonate buildups from Carixian (lower Pliensbachian sub-stage) to Domerian (upper Pliensbachian sub-stage) shape the landscape. This carbonate platform is generally interpreted as capping a tilted block limited in the southeast by the “Northern Middle-Atlasic Accident” (Colo, 1961; El Arabi et al., 1987), which marks the boundary between tabular Middle-Atlas and “folded” Middle-Atlas, and by the “Tizi n’Trettène accident” in the northwest, which separates two distinct plateaus of the Tabular Middle Atlas. One of the sampling areas has been concentrated especially on one of these bioconstructions: the Koudiat Ech Chehem. It is an approximately 100 m high buildup prograding towards the southwest and rising above limestone-marls deposits including early Domerian ammonites Protogrammoceras (Morabet, 1974 according to El Arabi et al., 1987). Marly limestone and marl alternations containing Hildaites gyralis, Hildaites subserpentinus, and Harpoceras cf. madagascariense (all known from the Serpentinum Zone, early Toarcian) onlap the flanks of the bioconstruction. The other outcrop for sampling is part of the “Taililout” section located in the western part of the Guigou Plateau, directly west from Koudiat Ech Chehem. This outcrop corresponds to lower or middle Carixian stage; it is topped by a level that stratigraphically corresponds to the Maftah 2 Formation (comforted by personal communications from André Charrière in the field), which contains the middle Carixian ammonite Tropidoceras (see Charrière et al., 2011, p. 62). The outcrop level sampled corresponds to the transition between the Maftah 1 and Maftah 2 formations. In fact, the core of Maftah 1 Formation shows mudstone to packstone limestone alternations containing bivalves, foraminifera, and algae, sometimes with cross-bedding. Then, the top of Maftah 1 Formation is made of cross-bedded oolitic and bioclastic calcarenites including corals in some beds. A major discontinuity showing a ferruginous hardground, including wood, marks the top of this Formation. The Maftah 2 Formation includes more silicified bioclastic and oolitic limestones or thin argillaceous limestones and is dated by the presence of middle Carixian ammonite Tropidoceras at the base and the upper Carixian ammonites Becheiceras gallicum and Fuciniceras aff. bastianii (Davoei zone) in the core of the member. The subsequent Maftah 3 Formation records deeper depositional environments with clay and limestones alternations including lower to middle Domerian ammonites.
The Guigou Plateau is located in Middle Atlas (Figure 1.1, 1.2), where several huge carbonate buildups from Carixian (lower Pliensbachian sub-stage) to Domerian (upper Pliensbachian sub-stage) shape the landscape. This carbonate platform is generally interpreted as capping a tilted block limited in the southeast by the “Northern Middle-Atlasic Accident” (Colo, 1961; El Arabi et al., 1987), which marks the boundary between tabular Middle-Atlas and “folded” Middle-Atlas, and by the “Tizi n’Trettène accident” in the northwest, which separates two distinct plateaus of the Tabular Middle Atlas. One of the sampling areas has been concentrated especially on one of these bioconstructions: the Koudiat Ech Chehem. It is an approximately 100 m high buildup prograding towards the southwest and rising above limestone-marls deposits including early Domerian ammonites Protogrammoceras (Morabet, 1974 according to El Arabi et al., 1987). Marly limestone and marl alternations containing Hildaites gyralis, Hildaites subserpentinus, and Harpoceras cf. madagascariense (all known from the Serpentinum Zone, early Toarcian) onlap the flanks of the bioconstruction. The other outcrop for sampling is part of the “Taililout” section located in the western part of the Guigou Plateau, directly west from Koudiat Ech Chehem. This outcrop corresponds to lower or middle Carixian stage; it is topped by a level that stratigraphically corresponds to the Maftah 2 Formation (comforted by personal communications from André Charrière in the field), which contains the middle Carixian ammonite Tropidoceras (see Charrière et al., 2011, p. 62). The outcrop level sampled corresponds to the transition between the Maftah 1 and Maftah 2 formations. In fact, the core of Maftah 1 Formation shows mudstone to packstone limestone alternations containing bivalves, foraminifera, and algae, sometimes with cross-bedding. Then, the top of Maftah 1 Formation is made of cross-bedded oolitic and bioclastic calcarenites including corals in some beds. A major discontinuity showing a ferruginous hardground, including wood, marks the top of this Formation. The Maftah 2 Formation includes more silicified bioclastic and oolitic limestones or thin argillaceous limestones and is dated by the presence of middle Carixian ammonite Tropidoceras at the base and the upper Carixian ammonites Becheiceras gallicum and Fuciniceras aff. bastianii (Davoei zone) in the core of the member. The subsequent Maftah 3 Formation records deeper depositional environments with clay and limestones alternations including lower to middle Domerian ammonites.
The municipality of Amellagou is in the southern part of the Central High Atlas rift basin, in the Gueris Valley (Figure 1.1, 1.3). Some of the samples have been found in Pliensbachian-aged (from Ibex to Spinatum ammonite chronozones) coral and sponge bioherms, which grew on the upper slope that bordered the thick southern carbonate platform. Abundant lithiotid communities developed on subtidal lagoon paleoenvironments on this platform, outcropping at the top of Jebel Taabest, for example. Numerous other specimens have been collected in olistoliths that slipped eastwards and northwards, up to the lower slope, in a hemipelagic environment. These olistoliths are associated with slumps and calciturbidites, related to synsedimentary faults and tilting; they are attributed to the lateral transition between the Choucht and Ouchbis Formations. Olistoliths show a boundstone to floatstone texture, with solitary and colonial corals, sponges, gastropods, serpulid worms, crinoids, brachiopods, echinoids tests and spines, and occasional chaetetid sponges and microbialites. Some of the olistoliths also contain lithiotid bivalves (including Cochlearites and Opisoma genera). Numerous olistoliths originating from the carbonate platform slipped towards the basin during the latest Pliensbachian lowstand (Spinatum chronozone), just before the drowning of the Taabest platform, which occurred during the earliest Toarcian. Older specimens of presently studied corals have been collected from the Serdrar reef, a 200 m high basinal patch reef. The Serdrar reef is located on a local high, the foot wall of the Serdrar fault, which was tectonically active during the Early Jurassic (Sarih, 2008; Lachkar et al., 2009). Ammonites of the uppermost Sinemurian (Raricostatum chronozone) have been found on the distal slope deposits of this big pinnacle (see Table 1).
The Dades Valley also belongs to the southern margin of the central High Atlas rift basin (Figure 1.1, 1.4). Specimens come from the upper part of the Choucht Formation, which is a proximal equivalent of the Ouchbis Formation. This interval is dated by ammonite biostratigraphy and corresponds to the late Pliensbachian Emaciatum ammonite chronozone, more specifically the Elisa sub-chronozone (Ettaki et al., 2000). The age is confirmed by the presence of the nannofossil Lotharingius sigillatus (a marker for the nannofossil subzone NJT5b) found below the Choucht Formation in the Tizguin section (Mattioli et al., 2013; Bodin et al., 2016). The Choucht Formation is a succession of shallow-water deposits made of packstone to mudstone beds deposited in a carbonate platform environment, including phaceloid coral reefs with rare lithiotids bivalves. Nonetheless, ammonites, belemnites and Zoophycos ichnofossils are observed in this formation, in close association with the reefs, suggesting relatively deep (shoreface to offshore transition) depositional conditions for this formation (wave influence sedimentary structures have not been observed to date). Despite a thorough investigation of Toarcian outcrops from these regions, no N. pseudocolumellata was found in post-Pliensbachian strata, to date.
MATERIALS AND METHODS
Materials
The studied material is represented by 77 specimens collected by our team in the Lower Jurassic of Morocco although 14 additional specimens considered in this study come from the collection of the Muséum National d’Histoire Naturelle (MNHN) in Paris, including the holotype of the type species of the genus Mesophyllum Beauvais non Schlüter. Four specimens come from the Beni Tadjit (Menchikoff collections), and the others were in the collections of the Museum, associated with a note from Alloiteau but without any locality (they are suspected to come from Morocco, possibly specimens from Menchikoff collections and Du Dresnay collections cited by Beauvais, 1986).
Terminology: The Concept of Pseudocolumella
The samples correspond to scleractinian solitary corals with an axial structure corresponding to the definition of a pseudocolumella according to Milne-Edwards and Haime (1848) and later Berkowski and Weyer (2012). This term was introduced initially by Milne-Edwards and Haime (1848), as an alternative to the term columella, in view to describe different types of axial structures for skeleton-bearing corals (Rugosa, Scleractinia and Tabulata). The distinction was mainly based on the genesis of the axial edifice: septal origin in the case of pseudocolumella, non-septal origin in the case of a “columella propria” (Berkowski and Weyer, 2012). Even if the distinction was used by some authors (see for example Fedorowski, 1974) the term pseudocolumella has often been forgotten and the term columella applied to describe all skeletal axial structures in corals fossils, especially in the case of scleractinian description. Chevalier (1987) describes the columella of Scleractinia as a part of the skeleton occupying the axial part of the corallites. In this definition, the columella can have a septal origin or a non-septal origin. We choose to reintroduce here the terminology of Milne-Edwards and Haime (1848) for scleractinian corals because the columellae of septal origin described up until now was structurally distinct from the skeletal structure of the specimens described in this study. In fact, columellae of septal origin in scleractinians occur in the following ways: i) in a single lamellae in the main plan of the corallite (lamellar columella); ii) in the projection of septal trabeculae in the axis of the corallite (papillose and spongy columella); iii) in a thickening and welding of the inner edges of septa (“fausse columelle”—fake columella—in Chevalier, 1987). The axial structure described in this study is made of many septal lamellae separated from the main part of septa early in the coral’s ontogeny and organized in bilateral symmetry. These features correspond to the description of the axial structure of the example used by Milne-Edwards and Haime (1848): the genus Clisiophyllum Dana, 1846 (Berkowski and Weyer, 2012 proposed Clisiophyllum keyserlingi M’Coy, 1849 as the definitive morpho-terminological type of the term pseudocolumella). It also corresponds to the description of some pseudocolumella morphologies described by Fedorowski (1974) for the genus Lophophyllidium Grabau, 1928. Consequently, we propose that the term pseudocolumella be used for the singular cases of scleractinian corals showing a rugose-like axial structure made of several septal lamellae organized in a bilateral symmetry.
Methods
The samples have been prepared and analyzed using photographs, polished sections, thin sections, and computed tomography (CT) scans. The usual qualitative observations on polished surfaces and thin sections have been supplemented by morphometric measurements. Nine characters were measured on photographs by the lead author using the measurement software Canvas 9 (see Table 2); these characters are: great calicular diameter (Gd): the longest linear length passing across the center of the calice; small calicular diameter (Sd): the shortest linear length passing across the center of the calice; number of calicular septa (Ns): septa counted along the corallite periphery; homogeneous septal density (hsd): in order to compare all samples with a homogeneous parameter, the septal density of only S1 and S2 (first and second septal size orders) has been measured over a 5 mm distance at the periphery of the calice; length of S1 (lS1): measurement of the longest and the shortest septa of the first size order S1; length of S2 (lS2): measurement of the longest and the shortest S2; length of S3 (lS3): measurement of the longest and the shortest S3; greater pseudocolumella diameter (dp): the longest linear length passing across the center of the calice from the edges of the pseudocolumella in transverse section; number of pseudocolumellar septa (nps): septa counted along the pseudocolumella periphery.
Using these measurements as a basis, four types of transformed data have been produced for the statistical analysis—extrapolated number of calicular septa (exns): the total number of calicular septa has been estimated including hidden or broken septa. In fact, the preservation of samples is often poor, and it is frequently hard to determine if the third order of septa is present; in some cases these septa are partially or completely obscured by stereome thickening deposits or recrystallization. Middle of variation range value for length of S1 (mlS1): middle of variation range value of septa length is obtained by adding the highest and the lowest measured value of septal length for one order and then calculating the average of this sum. Middle of variation range value for length of S2 (mlS2). Middle of variation range value for length of S3 (mlS3).
Finally, two other pieces of information have been added to the database. Sampling outcrop (Os)—some numbers have been allocated to different sampling zones for each sample: 1, Amellagou plain; 2, MNHN; 3, Dades Valley; 4, Jebel Taabest; 5, Beni Tadjit (Menchikoff collection); 6, Jebel Serdrar; 7, Koudiat Ech Chehem; 8, Taililout. Pseudocolumella shape (ps): the two main shapes of pseudocolumella observed have been labelled with associated numbers and each sample has been associated with an apparent pseudocolumella shape. Examples of pseudocolumella observed in transverse section among the samples are presented in Appendix 1.
Measurements have been used to perform univariate and multivariate statistical analysis, in order to distinguish characters and identify potential connections between them. These analyses were conducted with the statistic software PAST (PAleontological STatistics; Hammer et al., 2001). In addition to the classical univariate and bivariate analysis, principal component, correspondence, and discriminant analyses have been performed on three distinct sets of data to maximize the number of samples considered in each analysis (the missing values written “n.c.” in Table 2 rule out the corresponding samples in the analysis). Set 1 includes all the characters (Gd, Sd, exns, hsd, mlS1, mlS2, mlS3, ps, dp) but with a number of only 23 samples. Set 2 includes set 1 but excludes the characters mlS1, mlS2, and mlS3 for a total of 44 samples. Set 3 includes set 1 but excludes the character mls3 for a total of 34 samples in the analysis
In the analysis, the points corresponding to the samples have been differentiated in accordance with the apparent morphology of pseudocolumella observed (point for type 1 and cross for type 2); and sometimes also in accordance with the sampling location.
 Three-dimensional CT-scan reconstructions have been created using the nanotom S phoenix 180 kV of Georessources laboratory in Nancy. The X-ray CT is a non-destructive technique used to inspect the internal structure of solid specimen based on recording abnormal attenuations levels of X-rays after passing them through a specimen. The X-ray measurements are dependent on density contrasts within studied specimen (Pryvalov et al., 2017). Two samples (A57331-1 and AM16161) have been chosen for their sufficient density contrasts between skeleton and matrix. Their internal skeletal structure has been observed in the three directions of a defined space as three sets of successive slices perpendicular to each axis of the define space (Figure 2). Successive transverse sections along the height of the corallites of each sample are presented in Appendix 2 and Appendix 3.
Three-dimensional CT-scan reconstructions have been created using the nanotom S phoenix 180 kV of Georessources laboratory in Nancy. The X-ray CT is a non-destructive technique used to inspect the internal structure of solid specimen based on recording abnormal attenuations levels of X-rays after passing them through a specimen. The X-ray measurements are dependent on density contrasts within studied specimen (Pryvalov et al., 2017). Two samples (A57331-1 and AM16161) have been chosen for their sufficient density contrasts between skeleton and matrix. Their internal skeletal structure has been observed in the three directions of a defined space as three sets of successive slices perpendicular to each axis of the define space (Figure 2). Successive transverse sections along the height of the corallites of each sample are presented in Appendix 2 and Appendix 3.
SYSTEMATIC PALEONTOLOGY
Phylum CNIDARIA Verrill, 1865
Class ANTHOZOA Ehrenberg, 1834
Sub-Class HEXACORALLIA Haeckel, 1866
Order SCLERACTINIA Bourne, 1900
Sub-Order CARYOPHYLLIINA Vaughan and Wells, 1943
Super-Family VOLZEIOIDEA Melnikova, 1974
Family incertae sedis
Neorylstonia, nomen novum pro Mesophyllum Beauvais, 1986 non Schlüter, 1889
Type species. Mesophyllum pseudocolumellatum Beauvais, 1986.
Derivatio nominis. Neorylstonia in reference to its doppelganger Paleozoic Rylstonia
Nomenclatural remarks. Mesophyllum Beauvais, 1986, is a junior homonym of Mesophyllum Schlüter, 1889 (p. 67). Unfortunately, as already indicated, several other junior homonyms were reported and among them another Liassic coral. The genus Mesophyllum Hahn, 1911 (p. 555), is a junior homonym of Mesophyllum Schlüter, 1889 (p. 67). Archaeosmilia Melnikova, 1975, could be an available synonym but a comparison with the type material is still necessary. If these two genera are justified in terms of taxonomy, a nomen novum should be created for Mesophyllum Hahn. Another junior homonym is also reported with the mention of “Mesophyllum Barrois, 1889 (see Barrois, 1889, p. 67)” by Neave (1940, p. 119). This quotation was present in other earlier indexes (e.g., Waterhouse, 1902, p. 219), but the citation could not be found in the 1888 nor the 1889 works of Barrois. Nevertheless, Barrois (1898, p. 241) gave a faunal list with "Mesophyllum lissingenense, Schlu." [Schlüter] and "Mesophyllum nov. sp."; the latter of which might be the origin of the erroneous entry. In conclusion, for these reasons, "Mesophyllum Barrois" is unavailable. Mesophyllum Hahn, 1911, is also a senior homonym of Mesophyllum Vieira, 1942, and Mesophyllum Beauvais, 1986, all of them depending on the zoological code. Incidentally, Mesophyllum is also the name of a red alga.
The family is given as incertae sedis because this taxon presents some characters of both Gardineriidae and Volzeiidae. The genus shares smooth lateral faces of septa with the Gardineriidae but differs to them by the presence of an endotheca; and the microstructure of septa is close to the one of Volzeiidae (according to the description of Volzeia badiotica in Cuif, 1974) but with no lateral spines or granulation.
Neorylstonia pseudocolumellata (Beauvais, 1986)
zoobank.org/52069B98-79EA-4F0E-AD01-C1EFF035D5F2
v. 1986 Mesophyllum pseudocolumellatum Beauvais, 1986, p. 20, Pl. 4, fig. 1a-1b
Originally included material. 7 specimens.
 Type designation. Mesophyllum pseudocolumellatum Beauvais, 1986 MNHN.F.R11604, Menchikoff collection. Holotype by original designation Beauvais, 1985, p. 20, figured plate 4 figure 1a, 1b; Paratypes figured plate 4, figure 1c, d and e.
Type designation. Mesophyllum pseudocolumellatum Beauvais, 1986 MNHN.F.R11604, Menchikoff collection. Holotype by original designation Beauvais, 1985, p. 20, figured plate 4 figure 1a, 1b; Paratypes figured plate 4, figure 1c, d and e.
Type locality. Beni Tadjit, Lower Jurassic.
Status. Available and valid.
Dimensions. Great diameter: 9-29 mm, Small diameter: 7.5-22.1 mm, Number of septa: 41-104 (mostly around 96), Septal density (S1 + S2): 4-10 for 5 mm.
Description. Solitary coral. Corallum ceratoid, straight, or curved. The calice is subcircular in outline. Costosepta straight or slightly curved (possibly post-mortem distortion), free, compact, occasionally rhopaloid. Some first-order (S1) septa rarely reach the axial structure but in most cases the inner edge abruptly dips in the fossa (see Figure 3.1-2). Lateral faces and distal edge are smooth. No palus. Radial elements are commonly arranged in three distinct size orders with different length and width in transverse sections but S1 and S2 are occasionally indistinct because of similar sizes. In contrast, some other samples show four clearly distinguishable size orders of septa. In the latter case, the first and second size orders geometrically correspond to a subdivision of the S1. Lastly, the S3 are sometimes missing.
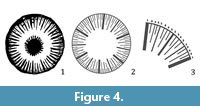 On the basis of the described arrangement of septal length and thicknesses a very classical model of septal insertion can be proposed, namely 96 septa distributed in five ontogenetical cycles according to 6+6+12+24+48 septa (see Figure 4). This model of septal insertion is statistically significant by univariate statistical analysis, in which the number 96 shows a well-marked mode. Ghosts of septal structures can sometimes be observed in thin sections (Figure 3.4). They consist in transverse section of a darker axial zone within the septum, a brighter zigzag line or some dark spots in a line considered as remains of “centers of calcification” or centers of rapid accretion (Stolarski, 2003). Growth lines in “ogive” or “gloves fingers” can be seen in longitudinal sections, resulting from the deposition of different successive layers of stereome thickening the septa (thickening deposits of Stolarski, 2003).
On the basis of the described arrangement of septal length and thicknesses a very classical model of septal insertion can be proposed, namely 96 septa distributed in five ontogenetical cycles according to 6+6+12+24+48 septa (see Figure 4). This model of septal insertion is statistically significant by univariate statistical analysis, in which the number 96 shows a well-marked mode. Ghosts of septal structures can sometimes be observed in thin sections (Figure 3.4). They consist in transverse section of a darker axial zone within the septum, a brighter zigzag line or some dark spots in a line considered as remains of “centers of calcification” or centers of rapid accretion (Stolarski, 2003). Growth lines in “ogive” or “gloves fingers” can be seen in longitudinal sections, resulting from the deposition of different successive layers of stereome thickening the septa (thickening deposits of Stolarski, 2003).
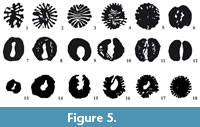 Axial structure is present, in the form of a Rugose-like calicular boss made of septa and endotheca. This pseudocolumella presents different morphologies throughout the polyp’s life (Figure 5, Appendix 1, Appendix 2, Appendix 3), giving the fossils a very characteristic aspect in transverse section. The pseudocolumella is made of 16 to 26 septal lamellae that seem to correspond to septa in some samples but, contrary to the septal system, are organized in a bilateral symmetry rather than the strict radial symmetry. Tabulae in the axial porosity (when present) and dissepiments between the lamellae sustain this structure. Most commonly the pseudocolumella consists, on average, of 21 to 22 lamellae organized in two main skeletal morphologies: Type 1 morphology—lamellae organized in the form of a star with opposite septa sometimes linked in the calicular axial zone (Figure 5.3). In some cases, the bilateral symmetry plan is materialized by a stereome deposit between lamellae, which should not be confused with a rugose-like axial lamellae (Figure 6.6). Type 2 morphology—lamellae organized in two soft crescents (the cross sections look like two kidney beans, see Figure 5.12) separated by an axial cavity regularly sealed by tabulae, which seem to serve as basis for the genesis of new lamellae (Figure 6.2). These tabulae probably correspond to stasis phases in the growth of the polyp alternating with stages of vertical increase associated with the secretion of lamellae by the soft ectodermal tissues of the animal.
Axial structure is present, in the form of a Rugose-like calicular boss made of septa and endotheca. This pseudocolumella presents different morphologies throughout the polyp’s life (Figure 5, Appendix 1, Appendix 2, Appendix 3), giving the fossils a very characteristic aspect in transverse section. The pseudocolumella is made of 16 to 26 septal lamellae that seem to correspond to septa in some samples but, contrary to the septal system, are organized in a bilateral symmetry rather than the strict radial symmetry. Tabulae in the axial porosity (when present) and dissepiments between the lamellae sustain this structure. Most commonly the pseudocolumella consists, on average, of 21 to 22 lamellae organized in two main skeletal morphologies: Type 1 morphology—lamellae organized in the form of a star with opposite septa sometimes linked in the calicular axial zone (Figure 5.3). In some cases, the bilateral symmetry plan is materialized by a stereome deposit between lamellae, which should not be confused with a rugose-like axial lamellae (Figure 6.6). Type 2 morphology—lamellae organized in two soft crescents (the cross sections look like two kidney beans, see Figure 5.12) separated by an axial cavity regularly sealed by tabulae, which seem to serve as basis for the genesis of new lamellae (Figure 6.2). These tabulae probably correspond to stasis phases in the growth of the polyp alternating with stages of vertical increase associated with the secretion of lamellae by the soft ectodermal tissues of the animal.
 Between these two extreme morphologies are some transitional states (Figure 5), and both morphologies sometimes coexist in one single sample. In such cases, the pseudocolumella always shows the same ontogenic pattern starting with a type 2 morphology in the early stages of development then vertically turning to a type 1 morphology in the oldest stages of development. Once the type 1 morphology is established, it never transforms (back) to type 2. Another point is that type 1 pseudocolumella are associated with a more conical shape of the corallite and, in this conformation, the axial structure continues to grow when the vertical increase of the wall stopped, sometimes highly surpassing the calix rim (Figure 6.9-11). Type 2 pseudocolumella never surpass the distal edges of septa and are associated with more cylindrical corallite shapes suggesting a continuous growth during the animal’s life.
Between these two extreme morphologies are some transitional states (Figure 5), and both morphologies sometimes coexist in one single sample. In such cases, the pseudocolumella always shows the same ontogenic pattern starting with a type 2 morphology in the early stages of development then vertically turning to a type 1 morphology in the oldest stages of development. Once the type 1 morphology is established, it never transforms (back) to type 2. Another point is that type 1 pseudocolumella are associated with a more conical shape of the corallite and, in this conformation, the axial structure continues to grow when the vertical increase of the wall stopped, sometimes highly surpassing the calix rim (Figure 6.9-11). Type 2 pseudocolumella never surpass the distal edges of septa and are associated with more cylindrical corallite shapes suggesting a continuous growth during the animal’s life.
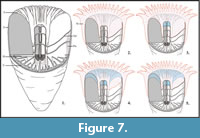
The pseudocolumella is generally wrapped into continuous deposits of stereome, which separate it from the lumen. It is observed independently of the morphology of the axial structure (Figure 6.3-5) and excludes the possibility of post-secretion modifications of the skeleton to explain the variations observed in morphologies (for example a case of symbiosis associated with specific bioerosion is excluded). These stereome deposits attenuate the spiky aspect of the pseudocolumella periphery in cross section. A model of step-by-step skeletal growth is proposed in Figure 7.
 In most cases, hexameral symmetry can be established on the basis of the different septa systems. The superimposed bilateral symmetry is evidenced by the spatial organization of the axial structure. Endotheca abundant and made of tabular dissepiments regularly arranged in the axial zone. Parathecal wall is present and the paratheca is continuous with the dissepiments. Synapticulae absent. A true epitheca with growth striae and wrinkles encloses the corallite (Figure 3.3).
In most cases, hexameral symmetry can be established on the basis of the different septa systems. The superimposed bilateral symmetry is evidenced by the spatial organization of the axial structure. Endotheca abundant and made of tabular dissepiments regularly arranged in the axial zone. Parathecal wall is present and the paratheca is continuous with the dissepiments. Synapticulae absent. A true epitheca with growth striae and wrinkles encloses the corallite (Figure 3.3).
As Neorylstonia is monospecific, the specific diagnosis is identical to the generic diagnosis.
Range. Uppermost Sinemurian (Raricostatum ammonite chronozone) to uppermost Pliensbachian (Spinatum ammonite Chronozone), with a probable acme during Late Pliensbachian (Domerian).
Occurrence. High Atlas and Middle Atlas of Morocco
STATISTICAL APPROACH
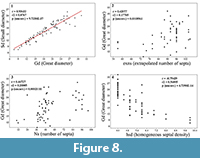 Univariate and Bivariate Analyses
Univariate and Bivariate Analyses
A number of trends can be deduced from the univariate and bivariate analysis (Figure 8). As it appears in Figure 8.2, the extrapolated number of septa (exns) seems to increase to some extent with the increase of the calicular diameter, but the link between these two characters is moderate (r = 0.42077), which is quite unexpected for scleractinian corals. A part of this weak correlation between diameters and number of septa is probably related either to a preservation bias or ontogeny. The process of extrapolation of the number of septa can result in an overestimation of the true number of septa in the sample; this may also erase the gradual establishment of the different orders of septa during the growth of the corallum. On the other hand, the apparent number of septa can lead to an underestimate of the true number of septa if some are hidden or broken. Nevertheless, a similar weak correlation is obtained with the non-extrapolated number of apparent septa in the samples (Figure 8.3). The points in Figure 8.3 are grouped in four packages (samples having approximately 12, 24, 48, and 96 septa) corresponding to different stages of development of the septal apparatus in the samples (see Figure 4), but all the samples from the package corresponding to approximately 48 septa in the septal apparatus could potentially be corals with a hidden third order of septa. Consequently, the possibility of making different species from these three packages, as old typological methods could have done, is not advised. Finally, the only conclusion that can be drawn from the values in Figure 8.2 and 8.4 is that N. pseudocolumellata develops its three orders even with small calicular diameters. This supports the idea that the growth size of the coral is more influenced by environmental parameters than the number of septa, which could be more linked to age.
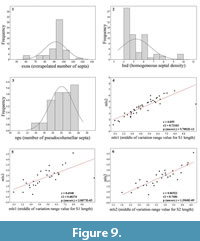
The homogeneous septal density (hsd) is very clearly negatively correlated with the calicular diameter (Figure 8.4). As demonstrated in Figure 9.5 the main number of extrapolated septa conforms well to the model with 96 septa exposed in the diagnosis. Figure 9.4, 9.5, and 9.6 emphasize the correlation between the length of septa from one cycle of septa to another cycle of septa.
Multivariate Analyses
 Principal components analysis (PCA) gave a sum of variance percentages greater than 93% for PC1 + PC2 in every set of data considered. The most influential character is the extrapolated number of septa (exns) in each case, followed by the great calicular diameter (Gd) and small calicular diameter (Sd) and after come the pseudocolumella diameter (dp) and the middle of variation range values for length of septa (mls1-3). The major role of dimensions and number of septa in PCA results is already well known for corals and corresponds to the traditional measurements used in taxonomy since the nineteenth century. Interestingly, the samples with a type 1 pseudocolumella are associated to high diameters and number of septa when the samples with a type 2 pseudocolumella correspond to high homogeneous septal density values and smaller calicular diameters (Figure 10). Similar results are observed with correspondence analysis and discriminant analysis, and the same trends are recorded for each set of data. Discriminant analysis gives one more important piece of information as it shows an “outcrop effect”; the corals from a same site are morphologically linked and the distribution of type 1 and type 2 pseudocolumella follows the same trends within a single sampling site. To avoid duplications, we choose to show only two graphic results; one from PCA on set 3 (Figure 10) and one from discriminant analysis on set 2 (Figure 11).
Principal components analysis (PCA) gave a sum of variance percentages greater than 93% for PC1 + PC2 in every set of data considered. The most influential character is the extrapolated number of septa (exns) in each case, followed by the great calicular diameter (Gd) and small calicular diameter (Sd) and after come the pseudocolumella diameter (dp) and the middle of variation range values for length of septa (mls1-3). The major role of dimensions and number of septa in PCA results is already well known for corals and corresponds to the traditional measurements used in taxonomy since the nineteenth century. Interestingly, the samples with a type 1 pseudocolumella are associated to high diameters and number of septa when the samples with a type 2 pseudocolumella correspond to high homogeneous septal density values and smaller calicular diameters (Figure 10). Similar results are observed with correspondence analysis and discriminant analysis, and the same trends are recorded for each set of data. Discriminant analysis gives one more important piece of information as it shows an “outcrop effect”; the corals from a same site are morphologically linked and the distribution of type 1 and type 2 pseudocolumella follows the same trends within a single sampling site. To avoid duplications, we choose to show only two graphic results; one from PCA on set 3 (Figure 10) and one from discriminant analysis on set 2 (Figure 11).
DISCUSSION
 Without considering the question of the pseudocolumella, univariate, and multivariate statistical analyses show significant overlap in coral dimensions and number of septa, thus we conclude that all the samples belong to a single species. Since there is a correlation between locality and morphology, it can also be concluded that this taxon is strongly influenced by its living environment and not only by the slightly different ages of the samples. Nevertheless, the variability of the pseudocolumella shape makes the case more complex. The two morphologies of pseudocolumella are associated with different trends in overall morphologies of the corallites. In fact, type 2 is preferentially associated with small calicular diameters, low numbers of septa, and high septal density, which could correspond to young development stages.
Without considering the question of the pseudocolumella, univariate, and multivariate statistical analyses show significant overlap in coral dimensions and number of septa, thus we conclude that all the samples belong to a single species. Since there is a correlation between locality and morphology, it can also be concluded that this taxon is strongly influenced by its living environment and not only by the slightly different ages of the samples. Nevertheless, the variability of the pseudocolumella shape makes the case more complex. The two morphologies of pseudocolumella are associated with different trends in overall morphologies of the corallites. In fact, type 2 is preferentially associated with small calicular diameters, low numbers of septa, and high septal density, which could correspond to young development stages. 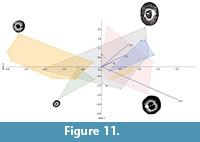 On the other hand, type 1 is more often associated with greater calicular diameters, a higher number of septa, and smaller septal density. This can be attributed to an older or more mature development stage. In addition, the distribution of pseudocolumella morphologies in one population of corals is obviously strongly linked with the sampling area of this population (Figure 11). Taililout and Koudiat Ech Chehem populations show two extreme situations with all samples exhibiting the same pseudocolumella type (see Figure 12); thus excluding the possibility of a random sampling effect in the distributions. If there is an equal chance of sampling either pseudocolumella morphology (i.e., a 50/50 chance of sampling type 1 or type 2), the probability of observing exclusively one pseudocolumella type in a Neorylstonia population (e.g., the Taililout population) would be extremely low; i.e., 24 examples of one type = (1/2)24 = 0.6 x 10-7. These unequal distributions from one outcrop to the other can be explained by three hypotheses: the type of pseudocolumella is the only difference between two distinct species within the genus Neorylstonia; the change in the type of pseudocolumella is the result of an evolutionary novelty among one single species and the different stratigraphic positions explain the different distributions or the type of pseudocolumella is constrained by the paleoecological conditions occurring during the life of the animals.
On the other hand, type 1 is more often associated with greater calicular diameters, a higher number of septa, and smaller septal density. This can be attributed to an older or more mature development stage. In addition, the distribution of pseudocolumella morphologies in one population of corals is obviously strongly linked with the sampling area of this population (Figure 11). Taililout and Koudiat Ech Chehem populations show two extreme situations with all samples exhibiting the same pseudocolumella type (see Figure 12); thus excluding the possibility of a random sampling effect in the distributions. If there is an equal chance of sampling either pseudocolumella morphology (i.e., a 50/50 chance of sampling type 1 or type 2), the probability of observing exclusively one pseudocolumella type in a Neorylstonia population (e.g., the Taililout population) would be extremely low; i.e., 24 examples of one type = (1/2)24 = 0.6 x 10-7. These unequal distributions from one outcrop to the other can be explained by three hypotheses: the type of pseudocolumella is the only difference between two distinct species within the genus Neorylstonia; the change in the type of pseudocolumella is the result of an evolutionary novelty among one single species and the different stratigraphic positions explain the different distributions or the type of pseudocolumella is constrained by the paleoecological conditions occurring during the life of the animals.
 As Koudiat Ech Chehem is stratigraphically above Taililout, it is possible that the appearance of a type 1 pseudocolumella could be interpreted as an evolutionary novelty appearing at the end of the ontogeny in more recent Neorylstonia. That said, other outcrops reveal that there is no consistency in the temporal distribution of the pseudocolumella types; for example, the Choucht Formation in the Dades Valley belongs to the uppermost Domerian and includes both type 1 and 2 pseudocolumellae. The first and second hypotheses are rejected due to the coexistence of both morphologies in the same localities and even within a single sample (see Appendix 2, sample A57331-1). The co-occurrences of both morphologies in one outcrop, together with the results from univariate analysis, suggest that the type 2 pseudocolumella corresponds to a younger growth stage than the type 1.
As Koudiat Ech Chehem is stratigraphically above Taililout, it is possible that the appearance of a type 1 pseudocolumella could be interpreted as an evolutionary novelty appearing at the end of the ontogeny in more recent Neorylstonia. That said, other outcrops reveal that there is no consistency in the temporal distribution of the pseudocolumella types; for example, the Choucht Formation in the Dades Valley belongs to the uppermost Domerian and includes both type 1 and 2 pseudocolumellae. The first and second hypotheses are rejected due to the coexistence of both morphologies in the same localities and even within a single sample (see Appendix 2, sample A57331-1). The co-occurrences of both morphologies in one outcrop, together with the results from univariate analysis, suggest that the type 2 pseudocolumella corresponds to a younger growth stage than the type 1.
The third hypothesis proposed is the preferred explanation for the data presented here, but the succession of the two morphologies in one single sample must be explained. As the type 1 morphology seems to be a facultative mature stage constrained by paleoecological conditions, we propose that, as a secondary character, the type 1 pseudocolumella is a mature morphology linked to sexual reproduction. This explanation would require that the type 1 shape could be associated with gonadic fixation/activity when the environment is favorable for sexual reproduction. This would imply that unfavorable conditions for sexual reproduction would lead to asexual reproduction or the total lack of breeding activity, associated with type 2 morphology. This last hypothesis is supported by the lack of budding scars on the corals, which implies that budding was restricted to the soft parts of the animals or was totally absent. Unfortunately, due to the lack of studies about the relationship between present-day coral skeletons and sexual maturity, we cannot confirm this idea with a modern coral as an analogous example. In addition, direct field observations provide no evidence of the precise ecological factors impacting Neorylstonia pseudocolumellata morphology.
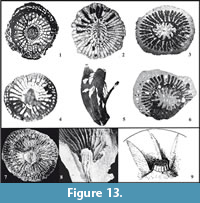 The mode of pseudocolumella growth in Neorylstonia is analogous to that observed in the Rugosa subfamily Amygdalophyllinae described in Fedorowski (1970) and Żołyński (2000) (Figure 13). Another rugose coral with analogous features for the axial structure corresponds to the genus Lophophyllidium sensu Fedorowski (1974), which can present a lamello-tabellar pseudocolumella made of septal lamellae sustained at their base by axial tabellae. These alternations are interpreted by the author as changes in the rhythm of the secretion of the structural elements. In Lophophyllidium, the pseudocolumella is wrapped into concentric endothecal layers in continuity with the tabulae of the lumen (see Fedorowski, 1974) and, like Neorylstonia, shows a sustained increase of the axial structure when the calix does not grow anymore.
The mode of pseudocolumella growth in Neorylstonia is analogous to that observed in the Rugosa subfamily Amygdalophyllinae described in Fedorowski (1970) and Żołyński (2000) (Figure 13). Another rugose coral with analogous features for the axial structure corresponds to the genus Lophophyllidium sensu Fedorowski (1974), which can present a lamello-tabellar pseudocolumella made of septal lamellae sustained at their base by axial tabellae. These alternations are interpreted by the author as changes in the rhythm of the secretion of the structural elements. In Lophophyllidium, the pseudocolumella is wrapped into concentric endothecal layers in continuity with the tabulae of the lumen (see Fedorowski, 1974) and, like Neorylstonia, shows a sustained increase of the axial structure when the calix does not grow anymore.
The Neorylstonia pseudocollumella axial structure is totally unique among Mesozoic corals. Despite a throrough literature search, no similar features could be found in Cenozoic or present-day corals either. Axial structures in post-Paleozoic corals are equally from a septal and/or endothecal origin, but the organization in septal lamellae, the bilateral symmetry, the ontogenic variations, the axial porosity, and the overcalicular apical growth found in Neorylstonia are singular characters that are only known in rugose corals. Type 1 and type 2 pseudocollumella are both facultative states as each can be absent in samples studied.
Another point is the question of the link between N. pseudocolumellata axial structure and the pseudocolumella of some rugose corals from Paleozoic (Figure 13). As phylogenetic inheritance is excluded between Paleozoic Rugosa and post-Paleozoic Scleractinia, only a convergence phenomenon can be purposed to explain the similarity of the axial structure in N. pseudocolumellata and some Rugosa. The affiliation of N. pseudocolumellata to Scleractinia is evidenced by the typical scleractinian organization of its septal apparatus in hexameral symmetry and by the probable aragonitic nature of the original skeleton (i.e., before diagenesis). Scleractinians almost exclusively biomineralize aragonitic skeletons, although rare examples of calcitic Scleractinia have been discovered (Stolarski, 2007, 2008). An original aragonitic skeleton is interpreted for N. pseudocolumellata because all samples collected have been inverted to calcite (losing its initial microstructural architecture during neomorphism), whereas a calcitic coral would not be altered and thus would conserve its microstructural features. So, this similar axial organization appears to be a surprising convergence phenomenon. The key of the functional significance of this organization may be in the understanding of its ecological role in similar rugose corals. In Rugosans it is either interpreted as a structure associated with sexual reproduction (Poty, 1981) because of its development (seasonally or environmentally influenced), or as a reinforcement structure for the skeleton (Poty, personal communication, 2017). More generally, the increase of calcification associated with the formation of the pseudocolumella can be interpreted as a way to eliminate calcium carbonate (CaCO3) excess in the body tissues resulting from photosynthetic activity, or also as a way to manage hypoxia (see Wooldridge, 2013 concerning general processes of calcification). In this study, we also proposed the possibility that the pseudocolumella was used to help soft tissues lift up during the growth of the septal apparatus.
CONCLUSION
Neorylstonia pseudocolumellata is a small solitary Moroccan coral that lived nearshore in the far end of a western Tethyan Gulf during the late Sinemurian and Pliensbachian stages. This scleractinian coral disappeared at the Pliensbachian/Toarcian boundary. Its skeletal structure is reminiscent of the pseudocolumella structure found in some rugose corals. In Rugosa, the pseudocolumella occasionally exhibits a seasonal or environmentally-dependent development. This comparable organization results in a convergence phenomenon between some Paleozoic rugosa made of calcite and this originally aragonitic scleractinian coral. Concerning N. pseudocolumellata, the pseudocolumella axial structure shows two main different morphologies between the different samples and vertically within a single sample (transition stages observed). One of these morphologies (type 1) is associated with an advanced degree of maturity; it is always observed as a final stage when present, is sometimes present since the beginning of the pseudocolumella formation, but can also be absent. This environmentally-dependent type 1 morphology is proposed to be linked to sexual reproduction, possibly as a gonads-fixation structure. The type 2 morphology would thus be conditioned by the conformation of the soft tissues at the base of the polyp, in this case more relaxed and pleated. This conformation would correspond to unfavorable environmental conditions for sexual reproduction. Despite important variability in dimensions and number of septa and septal lamellae among the samples, statistical analyses do not distinguish two different species under the genus Neorylstonia. Due to the large variability in dimensions as well as the ontogenic modifications of the axial structure, N. pseudocolumellata appears to be a significant example of the possible intraspecific variability in scleractinian coral species and a unique case of true pseudocolumella in Scleractinia.
ACKNOWLEDGMENTS
We would like to thank S. Charbonnier and J. Pacaud for their help in the collections of MNHN. Our field campaign in Middle Atlas has been successfully leaded by A. Charrière and H. El Arabi, we express our profound appreciation for that. We are equally grateful to M. Ousri and his family for the hospitality and help in the field in Morocco. We express gratitude to our friend and colleague R. Martini for her collaboration in this work and also to P. Neige and C. Meister for their precious help with the ammonite identifications. J. Sterpenich and C. Morlot are thanked for carrying out CT-Scan analysis. R. Vasseur would like to acknowledge A. Flammang and J. Moine from the laboratory of lithopreparation of Georessources, as well as C. Delangle and the association Terrae Genesis for their help in the production of thin sections. R. Martindale thanks the University of Texas at Austin for a 2015 Jackson School of Geosciences seed grant, which funded the travel associated with this work. We also would like to thank our anonymous rewievers for the wise advices that significantly enhanced the quality of this article.
REFERENCES
Ait Addi, A., Benshili, K., Benzaggagh, M., Chafiki, D., El Hariri, K., Elmi, S., Fedan, B., Ibhou, H., and Sadki, D. 2005. Le Jurassique Marocain: Transversale du Rif à la Bordure Saharienne, Livret Guide d’excursion du Groupe Français d’Etude du Jurassique. Groupe Français d’Etude du Jurassique (publisher’s locality unknown).
Barrois, C. 1889. Faune du Calcaire d’Erbray (Loire Inférieure). Mémoires de la Société Géologique du Nord, 3:1-348.
Barrois, C. 1898. Des relations des mers dévoniennes de Bretagne avec celles des Ardennes. Annales de la Société Géologique du Nord, 27:231-259.
Beauvais, L. 1986. Monographie des madréporaires du Jurassique inférieur du Maroc. Palaeontographica, Abteilung A, 194(1/3):1-68.
Berkowski, B. and Weyer, D. 2012. Hamaraxonia, a new pseudocolumellate genus of Middle Devonian deep-water Rugosa (Anthozoa) from Morocco. Geologica Belgica, 15(4):245-253.
Bodin, S., Krencker, F.-N., Kothe, T., Hoffmann, R., Mattioli, E., Heimhofer, U., and Kabiri, L. 2016. Perturbation of the carbon cycle during the late Pliensbachian - Early Toarcian: New insight from high-resolution carbon isotope records in Morocco. Journal of African Earth Sciences, 116:89-104. https://doi.org/10.1016/j.jafrearsci.2015.12.018
Bodin, S., Mattioli, E., Fröhlich, S., Marshall, J.D., Boutib, L., Lahsini, S., and Redfern, J. 2010. Toarcian carbon isotope shifts and nutrient changes from the Northern margin of Gondwana (High Atlas, Morocco, Jurassic): Palaeoenvironmental implications. Palaeogeography, Palaeoclimatology, Palaeoecology, 297:377-390. https://doi.org/10.1016/j.palaeo.2010.08.018
Bourne, G.C. 1900. The Anthozoa, p. 1-84. In Ray Lankester, E. (ed.), A Treatise on Zoology. The Porifera and Coelentera. Adam & Charles Black, London. https://doi.org/10.5962/bhl.title.11637
Brahmi,C., Kopp, C., Domart-Coulon, I., Stolarski, J., and Meibom, A. 2012. Skeletal growth dynamics linked to trace-element composition in the scleractinian coral Pocillopora damicornis. Geochimica et Cosmochimica Acta, 99:146-158. https://doi.org/10.1016/j.gca.2012.09.031
Burgess, S.D., Bowring, S.A., Fleming, T.H., and Elliot, D.H. 2015. High-precision geochronology links the Ferrar large igneous province with early-Jurassic ocean anoxia and biotic crisis. Earth and Planetary Science Letters, 415:90-99. https://doi.org/10.1016/j.epsl.2015.01.037
Caruthers, A.H., Smith, P.L., and Gröcke, D.R. 2013. The Pliensbachian-Toarcian (Early Jurassic) extinction, a global multi-phased event. Palaeogeography, Palaeoclimatology, Palaeoecology, 386:104-118. https://doi.org/10.1016/j.palaeo.2013.05.010
Caruthers, A.H., Smith, P.L., and Gröcke, D.R. 2014. The Pliensbachian-Toarcian (Early Jurassic) extinction: A North American perspective. Geological Society of America Special Papers, 505:225-243. https://doi.org/10.1130/2014.2505(11)
Caswell, B.A., Coe, A.L., and Cohen, A.S. 2009. New range data for marine invertebrate species across the early Toarcian (Early Jurassic) mass extinction. Journal of the Geological Society, 166:859-872. https://doi.org/10.1144/0016-76492008-0831
Charrière, A., Ibouh, H., and Haddoumi, H. 2011. The Central High Atlas from Beni Mellal to Imilchil. Notes et Mémoires du Service Géologique du Maroc, 559(4):1-168.
Chevalier, J.-P. 1987. Ordre des scléractiniaires, p. 403-764. In Grassé, P.P. (ed.), Traité de Zoologie. Masson, Paris.
Colo, G. 1961. Contribution à l’étude du Jurassique du Moyen Atlas septentrional. Notes et Mémoires du Service Géologique du Maroc, 139:1-226.
Cuif, J.P. 1974. Recherches sur les madréporaires du Trias. II. Astraeoidea. Révision des genres Montlivaltia et Thecosmilia. Etude de quelques types structuraux du Trias de Turquie. Bulletin du Muséum National d’Histoire Naturelle de Paris, 275(40):293-400.
Dana, J.D. 1846. Genera of fossil corals of the family Cyathophyllidae. American Journal of Science, 1:181-184.
Danise, S., Twitchett, R.J., and Little, C.T.S. 2015. Environmental controls on Jurassic marine ecosystems during global warming. Geology, 43:263-266. https://doi.org/10.1130/G36390.1
Danise, S., Twitchett, R.J., Little, C.T.S., and Clémence, M.-E. 2013. The impact of global warming and anoxia on marine benthic community dynamics: An example from the Toarcian (Early Jurassic). PLoS ONE, 8(2):1-14. https://doi.org/10.1371/journal.pone.0056255
Dera, G., Neige, P., Dommergues, J.-L., Fara, E., Laffont, R., and Pellenard, P. 2010. High-resolution dynamics of Early Jurassic marine extinctions: The case of Pliensbachian-Toarcian ammonites (Cephalopoda). Journal of Geological Society of London, 167:21-33. https://doi.org/10.1144/0016-76492009-068
Dommergues, J.-L. and Meister, C. 2017. Ammonites du Jurassique inférieur (Hettangien, Sinémurien, Pliensbachien) d’Afrique du Nord (Algérie, Maroc et Tunisie). Atlas d’identification des espèces. Revue de Paléobiologie du Muséum de Genève, 36(2):189-367.
Duncan, R., Hooper, P., Rehacek, J., Marsh, J., and Duncan, A. 1997. The timing and duration of the Karoo igneous event, southern Gondwana. Journal of Geophysical Research, 102:18127-18138. https://doi.org/10.1029/97JB00972
Ehrenberg, C.G. 1834. Die corallenthiere des rothen Meeres physiologisch untersucht und systematisch verzeichnet. Beiträge zur physiologischen Kenntniss der Korallenthiere im allgemeinen, und besonders des rothen Meeres. Physicalische abhanblungen der königlischen Akademie der Wissenschaften zu Berlin, Berlin.
El Arabi, H., Canerot, J., and Charrière, A. 1987. Dynamique récifale et tectonique distensive en blocs basculés: L’exemple des récifs liasiques du bloc du Guigou (Causse moyen atlasique-Maroc). Archives des Sciences de la Société de Physique et d’Histoire Naturelle de Genève, 40(3):259-271.
Ettaki, M., Chellai, E.H., Milhi, A., Sadki, D., and Boudchiche, L. 2000. Le passage Lias moyen-Lias supérieur dans la région de Todrha-Dades: événements bio-sédimentaires et géodynamiques (Haut Atlas central, Maroc). Comptes Rendus de l'Academie des Sciences, Series IIA, Earth and Planetary Science, 331(10):667-674. https://doi.org/10.1016/S1251-8050(00)01458-0
Fedorowski, J. 1970. Some Upper Viséan columnate tetracorals from the Holy Cross Mountains (Poland). Acta Palaeontologica Polonica, 15(4):549-640.
Fedorowski, J. 1974. The Upper Palaeozoic tetracoral genera Lophophyllidium and Timorphyllum. Palaeontology, 17(3):441-473.
Fedorowski, J. 1997. Rugosa and Scleractinia - A commentary on some methods of phylogenetic reconstructions. Acta Palaeontologica Polonica, 42(3):446-456.
Garwood, E.J. 1913. The Lower Carboniferous succession in the north-west of England. Quarterly Journal of the Geological Society of London, 68:449-586.
Goričan, Š., Carter, E.S., Guex, J., O'Dogherty, L., De Wever, P., Dumitrica, P., Hori, R.S., Matsuoka, A., and Whalen, P.A. 2013. Evolutionary patterns and palaeobiogeography of Pliensbachian and Toarcian (Early Jurassic) Radiolaria. Palaeogeography, Palaeoclimatology, Palaeoecology, 386:620-636. https://doi.org/10.1016/j.palaeo.2013.06.028
Guex, J., Bartolini, A., Spangenberg, J., Vicente, J.-C., and Schaltegger, U. 2012. Ammonoid multi-extinction crises during the Late Pliensbachian-Toarcian and carbon cycle instabilities. Solid Earth Discussions, 4:1205-1228. https://doi.org/10.5194/sed-4-1205-2012
Haeckel, E.H.P.A. 1866. Generelle Morphologie der Organismen. Allgemeine Grundzüge der Organischen Formen-wissenschaft, Mechanisch Begründet Durch die von Charles Darwin Reformirte Descendenztheorie. Zweiter Band: Allgemeine Entwicklungsgeschichte der Organismen. Verlag von Georg Reimer, Berlin. https://doi.org/10.5962/bhl.title.3953
Hahn, F. 1911. Neue Funde in nordalpinen Lias der Achenseagegend und bei Ehrwald. Neues Jahrbuch für Mineralogie Geologie und Paläontologie, Beilage Band, 32:537-557.
Hammer, Ø., Harper, D.A.T., and Ryan, P.D. 2001. PAST: Paleontological statistics software package for education and data analysis. Palaeontologia Electronica, 4.1.4:1-9. https://palaeo-electronica.org/2001_1/past/issue1_01.htm
Harries, P.J. and Little, C.T.S. 1999. The early Toarcian (early Jurassic) and the Cenomanian-Turonian (Late Cretaceous) mass extinction: Similarities and contrasts. Palaeogeography Palaeoclimatology Palaeoecology, 154:39-66. https://doi.org/10.1016/S0031-0182(99)00086-3
Hill, D. 1981. Treatise on Invertebrate Paleontology, Part F, Coelenterata Suppl. I. Rugosa and Tabulata. Geological Society of America and University of Kansas Press, Boulder, Colorado, and Lawrence, Kansas. https://doi.org/10.17161/dt.v0i0.5501
Hudson, R.G.S and Platt, M.I. 1927. On the Lower Carboniferous corals: The development of Rylstonia benecompacta, gen. et. sp. n. Annals and Magazine of Natural History, 19(109):39-48. https://doi.org/10.1080/00222932708633571
Jenkyns, H.C. 1985. The Early Toarcian and Cenomanian-Turonian anoxic events in Europe: Comparisons and contrasts. International Journal of Earth Sciences (Geologische Rundschau), 74(3):505-518. https://doi.org/10.1007/BF01821208
Jenkyns, H.C. 1988. The early Toarcian (Jurassic) anoxic event: Stratigraphic, sedimentary and geochemical evidence. American Journal of Science, 288:101-151. https://doi.org/10.2475/ajs.288.2.101
Jenkyns, H.C. 2003. Evidence for rapid climate change in the Mesozoic-Palaeogene greenhouse world. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences, 361:1885-1916. https://doi.org/10.1098/rsta.2003.1240
Jenkyns, H.C. and Clayton, C.J. 1986. Black shales and carbon isotopes in pelagic sediments from the Tethyan Lower Jurassic. Sedimentology, 33:87-106. https://doi.org/10.1111/j.1365-3091.1986.tb00746.x
Kossovaya, O.L., Nowak, M., and Weyer, D. 2012. Sloveniaxon, a new genus of ahermatypic Rugosa (Anthozoa) from the basal Permian (Asselian) of Slovenia. Geologica Belgica, 15(4):361-369.
Lachkar, N., Guiraud, M., Harfi, A.E., Dommergues, J.-L., Dera, G., and Durlet, C. 2009. Early Jurassic normal faulting in a carbonate extensional basin: Characterization of tectonically driven platform drowning (High Atlas rift, Morocco). Journal of the Geological Society, 166:413-430. https://doi.org/10.1144/0016-76492008-084
Lathuilière, B. and Marchal, D. 2009. Extinction, survival and recovery of corals from the Triassic to Middle Jurassic Time. Terra Nova, 21:57-66. https://doi.org/10.1111/j.1365-3121.2008.00856.x
Littler, K., Hesselbo, S.P., and Jenkyns, H.C. 2010. A carbon-isotope perturbation at the Pliensbachian-Toarcian boundary: Evidence from the Lias Group, NE England. Geological Magazine, 147(2):181-192. https://doi.org/10.1017/S0016756809990458
Martindale, R.C. and Aberhan, M. 2017, Response of macrobenthic communities to the Toarcian Oceanic Anoxic Event in northeastern Panthalassa (Ya Ha Tinda, Alberta, Canada): Palaeogeography, Palaeoclimatology, Palaeoecology, 478:103-120. https://doi.org/10.1016/j.palaeo.2017.01.009
Mattioli, E., Plancq, J., Boussaha, M., Duarte, L.V., and Pittet, B. 2013. Calcareous nannofossil biostratigraphy: New data from the Lower Jurassic of the Lusitanian Basin. Comunicaçõe Geológicas, 100(I):69-76.
Melnikova, G.K. 1974. Osobennosti gistologicheskikh struktur i mikrostruktur septalnogo aparata u pozdnetriasovykh skleraktinii. Trudy (Institut geologii i geofiziki. Sibirskoe Otdelenie), 201:224-228.
Melnikova, G.K. 1975. Novye rannejurskie predstaviteli Amphiastraeina (Skleraktinii) jugo-vostocnogo Pamira, p. 108-120. In Dzalilov, V. (ed.), Voprocy Paleontologii Tadjikistana. Akademija Nauk Tadzikskoj SSR, Dushanbe.
Milne Edwards, H. and Haime, J. 1848. Recherches sur les polypiers. Mémoire 1. Observations sur la structure et le development des polypiers en general. Annales des Sciences Naturelles, Zoologie, 9(4):37-89.
Milne Edwards, H. and Haime, J. 1854. A Monograph of the British Fossil Corals. Paleontographical Society, London. https://doi.org/10.5962/bhl.title.11691
Moore, R.C. 1956. Treatise on Invertebrate Paleontology. Geological Society of America and University of Kansas Press, Boulder, Colorado, and Lawrence, Kansas.
Morabet, A. 1974. The Lias Reefs of the Middle Atlas Platform, Morocco. Unpublished Master of Science Thesis, University of South Carolina, Columbia, South Carolina, USA.
Morard, A., Guex, J., Bartolini, A., Morettini, E., and De Wever, P. 2003. A new scenario for the Domerian-Toarcian transition. Bulletin de la Société Géologique de France, 174(4):351-356. https://doi.org/10.2113/174.4.351
Neave, S.A. 1940. Nomenclator Zoologicus. A List of the Names of Genera and Subgenera in Zoology from the Tenth Edition of Linnaeus 1758 to the End of 1935, Vol. III. Zoological Society of London, London.
Oliver, W.A. 1980. The relationship of the scleractinian corals to the rugose corals. Paleobiology, 6(2):146-160. https://doi.org/10.1017/S0094837300006709
Pálfy, J., Smith, P.L., and Mortensen, J.K. 2002. Dating the end-Triassic and Early Jurassic mass extinctions, correlative large igneous provinces, and isotopic events. Catastrophic Events and Mass Extinctions: Impacts and Beyond. Geological Society of America Special Paper, 356:523-532. https://doi.org/10.1130/0-8137-2356-6.523
Poty, E. 1981. Recherches sur les Tétracoralliaires et les Hétérocoralliaires du Viséen de la Belgique. Mededelingen Rijks Geologische Dienst, 35:1-161.
Pryvalov, V.A., Pironon, J., Izart, A., and Morlot, C. 2017. Exploration of architecture and connectivity of cleat arrays in coals of the Lorraine CBM play by the means of X- ray computer tomography. 79th EAGE Conference and Exhibition 2017, Paris, p. 1388-1392. https://doi.org/10.3997/2214-4609.201700758
Raup, D.M. and Sepkoski, J.J. 1984. Periodicity of extinctions in the geologic past. Proceedings of the National Academy of Sciences, 81:801-805. https://doi.org/10.1073/pnas.81.3.801
Raup, D.M. and Sepkoski, J.J. 1986. Periodic extinction of families and genera. Science, 231:833-836. https://doi.org/10.1126/science.11542060
Röhl, H.-J., Schmid-Röhl, A., and Oschmann, W. 2001. The Posidonia Shale (Lower Toarcian) of SW-Germany: An oxygen-depleted ecosystem controlled by sea level and palaeoclimate. Palaeogeography, Palaeoclimatology, Palaeoecology, 165:27-52. https://doi.org/10.1016/s0031-0182(00)00152-8
Rosales, I., Quesada, S., and Robles, S. 2004. Paleotemperature variations of Early Jurassic seawater recorded in geochemical trends of belemnites from the Basque-Cantabrian basin, northern Spain. Palaeogeography, Palaeoclimatology, Palaeoecology, 203:253-275. https://doi.org/10.1016/S0031-0182(03)00686-2
Sarih, S. 2008. Geodynamique et Transferts Sédimentaires Gravitaires des Bassins Liasiques du Haut-Atlas Central (Maroc). Unpublished PhD Thesis, Université de Bourgogne, Dijon, France.
Schlüter, C. 1889. Anthozoen des reinischen Mittel-Devon. Abhandlungen zur geologischen Spezialkarte von Preussen und den Thüringischen Staaten, 8(4):259-465.
Schmid-Röhl, A., Röhl, H.J., and Oschmann, W. 2002. Paleoenvironmental reconstruction of Lower Toarcian epicontinental black shales (Posidonia Shale, SW Germany): Global versus regional control. Geobios, 35:13-20. https://doi.org/10.1016/S0016-6995(02)00005-0
Sell, B., Ovtcharova, M., Guex, J., Bartolini, A., Jourdan, F., Spangenberg, J.E., Vicente, J.-C., and Schaltegger, U. 2014. Evaluating the temporal link between the Karoo LIP and climatic-biologic events of the Toarcian Stage with high-precision U-Pb geochronology. Earth and Planetary Science Letters, 408:48-56. https://doi.org/10.1016/j.epsl.2014.10.008
Stolarski, J. 2003. Three-dimensional micro- and nanostructural characteristics of the scleractinian coral skeleton: A biocalcification proxy. Acta Palaeontologica Polonica, 48(4):497-530.
Stolarski, J., Meibom, A., Przenioslo, R., and Mazur, M. 2007. A Cretaceous Scleractinian coral with a calcitic skeleton. Science, 318:92-94. https://doi.org/10.1126/science.1149237
Stolarski, J., Meibom, A., Mazur, M., and Phillips, G.E. 2008. Calcitic Scleractinian corals: When, where and why? 2008 Joint Meeting of The Geological Society of America, Soil Science Society of America, American Society of Agronomy, Crop Science Society of America, Gulf Coast Association of Geological Societies with the Gulf Coast Section of SEPM, the Geological Society of America, Houston, p. 201.
Suan, G., Mattioli, E., Pittet, B., Lécuyer, C., Suchéras-Marx, B., Duarte, L.V., Philippe, M., Reggiani, L., and Martineau, F. 2010. Secular environmental precursors to Early Toarcian (Jurassic) extreme climate changes. Earth and Planetary Science Letters, 290(3-4):448-458. https://doi.org/10.1016/j.epsl.2009.12.047
Trecalli, A., Spangenberg, J., Adatte, T., Föllmi, K.B., and Parente, M. 2012. Carbonate platform evidence of ocean acidification at the onset of the early Toarcian oceanic anoxic event. Earth and Planetary Science Letters, 357-358:214-225. https://doi.org/10.1016/j.epsl.2012.09.043
Vakhrameyev, V.A. 1982. Classopollis pollen as an indicator of Jurassic and Cretaceous climate. International Geology Review, 24(10):1190-1196. https://doi.org/10.1080/00206818209451058
Vaughan, T.W. and Wells, J.W. 1943. Revision of the Suborders, Families, and Genera of the Scleractinia. Geological Society of America, New York. https://doi.org/10.1130/SPE44
Verrill, A.E. 1865. Classification of polyps. (Extract condensed from a synopsis of the Polypi of the North Pacific exploring expedition under Captains Ringgold and Rodgers, U.S.N.). Annals and Magazine of Natural History, 16:191-197. https://doi.org/10.1080/00222936508679407
Vieira, C.O.C. 1942. Ensaio monográfico sobre os quirópteros do Brasil. Arquivos de Zoologia do Estado de São Paulo, 3:219-417.
Waterhouse, C.O. 1902. Index Zoologicus. An Alphabetical List of Names of Genera and Subgenera Proposed for Use in Zoology as Recorded in the "Zoological Record" 1880-1900, Together with Other Names not Included in the "Nomenclator Zoologicus" of S.H. Scudder. Zoological Society of London and Gurney and Johnson, London. https://doi.org/10.5962/bhl.title.34443
Wells, J.W. 1969. The formation of dissepiments in Zoantharian corals, p. 17-26. In Campbell, K.S.W. (ed.), Stratigraphy and Palaeontology Essays in Honour of Dorothy Hill. Australian University Press, Canberra.
Wooldridge, S. 2013. A new conceptual model of coral biomineralisation: Hypoxia as the physiological driver of skeletal extension. Biogeosciences 10:2867-2884. https://doi.org/10.5194/bg-10-2867-2013
Żołyński, L. 2000. Amygdalophyllum sudeticum sp. nov. (Rugosa) from a Lower Visean gneissic conglomerate, Bardzkie Mts., Sudetes (Poland). Acta Geologica Polonica, 50(3):335-342.

