 Taxonomic notes on Recent Foraminifera from the Continental Shelf-Slope Region of Southwestern Bay of Bengal, East Coast of India
Taxonomic notes on Recent Foraminifera from the Continental Shelf-Slope Region of Southwestern Bay of Bengal, East Coast of India
Article number: 22.3.55
https://doi.org/10.26879/811
Copyright Paleontological Society, September 2019
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Submission: 26 July 2017. Acceptance: 24 July 2019.
{flike id=2677}
ABSTRACT
A fully comprehensive work comprising the systematics of benthic and planktonic foraminifera is attempted for the first time from the continental shelf-slope region between Chennai and Cuddalore on the east coast of India. A total of 45 surface sediment samples and 75 subsamples from three gravity cores obtained from various depths (10-300m) were analysed to identify 286 foraminiferal species. Thus, this paper examines the taxonomic description of 262 benthic foraminifera in 117 genera and 24 planktonic foraminifera in 13 genera illustrated by SEM images.
Tabita K. Symphonia. Department of Earth Sciences, Pondicherry University, Puducherry- 605014, India. Present address: Department of Earth Sciences, Eritrea Institute of Technology, Mai Nefhi, Eritrea. tsymphonia@gmail.com
Nathan D. Senthil. Department of Earth Sciences, Pondicherry University, Puducherry- 605014, India. senthilom@gmail.com
Keywords: taxonomy; foraminifera; Bay of Bengal; East coast of India; Recent
Symphonia, Tabita K. and Senthil, Nathan D. 2019. Taxonomic notes on Recent Foraminifera from the Continental Shelf-Slope Region of Southwestern Bay of Bengal, East Coast of India. Palaeontologia Electronica 22.3.55A 1-89. https://doi.org/10.26879/811
palaeo-electronica.org/content/2019/2677-taxonomy-recent-foraminifera
Copyright: September 2019 Paleontological Society.
This is an open access article distributed under the terms of Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0), which permits users to copy and redistribute the material in any medium or format, provided it is not used for commercial purposes and the original author and source are credited, with indications if any changes are made.
creativecommons.org/licenses/by-nc-sa/4.0/
INTRODUCTION
Foraminifera are abundant unicellular microorganisms with pronounced diversity in the marine realm. There are around 6,800 recent species (Hayward et al., 2017) which are either planktonic or benthic in habit. They possess a mostly calcareous test although few species form agglutinated tests by cementing foreign material which are better preserved in the sediments as fossils. They are the most studied part in foraminifera as the variations in depth, salinity, temperature, nutrient flux prevalent during their short life span are recorded in their tests. These incorporated signatures make them the most reliable bioindicators to understand the present ecological conditions and reconstruct the past. A systematic morphological description is therefore essential for accurate classification of taxa and thereafter to identify the influence of environmental changes on their distribution pattern.
Previous Work
Foraminifera in the Bay of Bengal were first studied by Schwager (1866). Reports on foraminifera from the east coast were initially given by Carter (1880) followed by Cushman (1939a), Gnanamuthu (1943), Ganapati and Satyavati (1958), Sarojini (1958), and Rao and Vedantam (1968). Various aspects such as ecology and distribution of foraminifera (Rajasekhar, 1981), foraminiferal diversity (Naidu et al., 1990), changes in intertidal foraminifera due to oceanic disturbances (Gadi and Rajasekhar, 2007) etc. were part of further studies. The shelf off Chennai and Pondicherry (Puducherry) was studied by Setty (1976a and 1976b), Setty (1978), and Setty and Rao (1978). In the recent years the sediments from the offshore side of Chennai (Siva Kumar, 2002; Nandhakumar and Rao, 2008), Pondicherry, and Cuddalore areas (Ramesh, 2005) were analyzed for the distribution of foraminifera, but a continuous and detailed record from the south western Bay of Bengal is still lacking. Therefore, this paper aims at providing a comprehensive report on the systematics of foraminifera from the shelf-slope region off Chennai-Cuddalore.
STUDY AREA
The Bay of Bengal is the world’s largest semi-enclosed bay. This tropical bay is roughly triangular in shape and is situated in the northeastern part of the Indian Ocean. It covers a total area of about 2.2 million square kilometres (La Fond, 1966) and is bounded by the Ganges-Brahmaputra deltaic region on the north, the Burmese Peninsula on the east, the Andaman and Nicobar Islands on the southeast, the east coast of India on the west, and by the Indian Ocean on the south. The east coast of India enjoys a temperature range between 27 and 29°C, a salinity of 30-33‰ (Pannikar and Jayaraman, 1966), and organic carbon content of 0.88%, which is relatively less when compared to the west coast that exhibits 5-10% (Wiseman and Bennett, 1940; Subba Rao, 1960).
The bathymetry of the study area shows that the continental shelf has an average gradient of 1:115. The outer shelf is characterized by a steep gradient of 1:80 km while the inner shelf exhibits a gradient of 1:400 km. The shelf region between Chennai-Cuddalore is narrow with an average width of 35 km. Off Puducherry in the southern side the shelf is relatively narrower (only 25 km wide) than the broader off Chennai shelf (50 km wide). The Cuddalore shelf is concave shaped and narrow with an average width of 79 km and a gentle gradient up to 3,000 m of water depth (Murthy et al., 2006). This region experiences three seasons influenced by the surface currents which are the south-west monsoon (June-September), north-east monsoon (October-January) and summer (February-May). There are also four minor eastward flowing rivers—Palar, Gingee, Ponnaiyar, and Gadilam.
MATERIAL AND METHODS
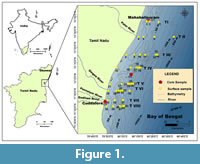 The present study is based on the analyses of a) surface samples and b) cores collected from the off-shore region between Chennai and Cuddalore (Figure 1). The co-ordinates of the sample locations and the depths are given in Table 1. This region lies within 11°40’0” N to 12°40’ 0” N longitude and 79°48’0” E to 80°28’0” E latitude in the southwestern Bay of Bengal.
The present study is based on the analyses of a) surface samples and b) cores collected from the off-shore region between Chennai and Cuddalore (Figure 1). The co-ordinates of the sample locations and the depths are given in Table 1. This region lies within 11°40’0” N to 12°40’ 0” N longitude and 79°48’0” E to 80°28’0” E latitude in the southwestern Bay of Bengal.
Sample Collection
Surface samples. A total of 45 surface sediment samples were collected by using a brass van Veen grab sampler during a cruise in May-June 2012 (cruise no. 05/2012) from ROV Sagar Paschimi, along eight transects across the shelf-slope region off Mahabalipuram to off Cuddalore Old Town. Samples were obtained from the sea floor at different water depths ranging from ~10 m to ~300 m except in transects I, II, and VII. Sampling was difficult from beyond 100 m depth (in Transects I and VII) and beyond 150 m (in Transect II) owing to the strong currents during the onset of southwest monsoon in the Bay. The CTD (Conductivity, Temperature, and Density) measurements for the vertical water column between ~10 m and ~300 m were recorded on board by using the CTD probe Sea bird-25. Fifty grams of the sediment from each sample was preserved immediately in Rose bengal-ethanol solution after collection in clean labelled vials and set aside for a fortnight. Samples were then oven dried at 60°C overnight and used for further analyses.
Core samples. In February 2013, core samples were procured from three different locations using a gravity corer on ORV Sagar Manjusha cruise no. 02/2013. Three undisturbed sediment cores collected from depths ranging from 12.5 to 60 m of water depth were used for the present study. On reaching the laboratory, all the three cores namely, Chennai core (C1: 35 cm long), Emerald Island core (C2: 60 cm long), and Ponnaiyar core (C3: 36 cm long) were sliced into 1 cm thick layers down to 5 cm and at 2 cm intervals in the remaining portion. Thus, a total of 74 subsamples—20 subsamples from C1 core, 33 subsamples from C2 core, and 21 subsamples from C3 core were obtained and investigated. Further, sediments till 10 cm depth were preserved in Rose bengal-ethanol solution and left to stand for a fortnight till further analyses.
Sample Preparation
The substrate sediments and the core samples were first washed through 40 μm, 63 μm, and 125 μm sieves in the laboratory. The residue was dried and for better identification of morphological features foraminifera were picked from >125 μm fraction. Around 300 foraminifera were picked from each subsample and sorted according to their genera and species level when possible. The list of foraminifer species with relative abundance of >5% as found in both surface and core samples are presented in Table 2, Table 3, Table 4, Table 5.
Classification
Digital photomicrographs of 286 species were obtained by Hitachi S-3400N Scanning Electron Microscope (SEM) at Pondicherry University. The images enabled the study of the morphological details and surface composition of the specimens.
The benthic foraminifera listed and described here are identified mainly based on the descriptions given by Loeblich and Tappan (1987) for supra generic level. Other references are Murray (1971), Jones (1994), Szarek (2001), Kaminski et al. (2002), Murray (2003), Javaux and Scott (2003), Riveiros and Patterson (2008), Margreth (2010), Milker and Schmiedl (2012), and Debenay (2012). A few taxa were referred from Hanagata and Nobuhara (2015).
The identification of planktonic species follows the taxonomic concepts given by Parker (1962), Saito et al. (1981), Kennett and Srinivasan (1983), and Ovechkina et al. (2010). The revised names for both benthic and planktic species are according to the WoRMS classification (World Register of Marine Species by Hayward et al., 2017). All identified specimens illustrated in this paper are deposited in the Palaeontology Laboratory, Department of Earth Sciences, Pondicherry University, Puducherry, India.
SYSTEMATIC CLASSIFICATION
Kingdom CHROMISTA Cavalier-Smith, 1981
Subkingdom HAROSA Cavalier-Smith, 2010
Infrakingdom RHIZARIA Cavalier-Smith, 2002
Phylum FORAMINIFERA d’Orbigny, 1826
Class FORAMINIFERA INCERTAE SEDIS Pawlowski, Holzmann, and Tyszka, 2013
Order LAGENIDA Delage and Hérouard, 1896
Superfamily NODOSARIOIDEA Ehrenberg, 1838
Family VAGINULINIDAE Reuss, 1860
Subfamily MARGINULININAE Wedekind, 1937
Genus AMPHICORYNA Schlumberger, 1881
Amphicoryna scalaris (Batsch, 1791)
Figure 2.1-2
1791 Nautilus (Ortoceras) scalaris Batsch, p. 1, 4, pl. 2, fig. 4a-b.
 2001 Amphicoryna scalaris (Batsch); Szarek, p. 201, pl. 14, fig. 18.
2001 Amphicoryna scalaris (Batsch); Szarek, p. 201, pl. 14, fig. 18.
2003 Amphicoryna scalaris (Batsch); Murray, p. 17, fig. 5.1.
2010 Amphicoryna scalaris (Batsch); Margreth, p. 153, pl. 13, figs. 2-3.
2012 Amphicoryna scalaris (Batsch); Milker and Schmiedl, p. 73, fig. 18.22-25.
2012 Amphicoryna scalaris (Batsch); Debenay, p. 162.
Description. Test elongate, wall calcareous, surface ornamented with many striae and basal apiculate spine; chambers globular, chamber arrangement uniserial; sutures depressed; aperture crescent-shaped or radiate at the end of a long neck, rimmed, enveloped by concentric ridges.
Amphicoryna scalaris (Batsch, 1791) macrospheric form
Figure 2.3
Description. In the macrospheric form, globular final chambers rapidly increase in size.
Subfamily LENTICULININAE Chapman, Parr, and Collins,1934
Genus LENTICULINA Lamarck, 1804
Lenticulina cultrata (de Montfort, 1808)
Figure 2.4
1808 Robulus cultratus de Montfort, p. 214, 540 genre.
2012 Lenticulina cultrata (de Monfort); Debenay, p. 223.
2015 Lenticulina cultrata (de Montfort); Hanagata and Nobuhara, p. 29, fig. 11.1-2.
Description. Test large, biconvex, wall finely perforate, surface smooth, no umbo; chambers many; periphery with a well-developed keel, sutures strongly curved; aperture terminal, radiate with many slits coalescing at the end of final chamber, a slit on the apertural side.
Lenticulina gibba (d’Orbigny, 1839a)
Figure 2.5
1839a Cristellaria gibba d’Orbigny, p. 292, no. 17.
2001 Lenticulina gibba (d’Orbigny); Szarek, p. 203, pl. 15, figs. 1-2.
2002 Lenticulina gibba (d’Orbigny); Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 172, pl. 2, fig. 6.
2010 Lenticulina gibba (d’Orbigny); Margreth, p. 150, pl. 12, fig. 7a-b.
2012 Lenticulina gibba (d’Orbigny); Debenay, p. 223.
Description. Test oblong, biconvex, wall finely perforate, surface smooth; chambers elongate, gradually increasing in size, chamber arrangement planispiral; periphery slightly keeled, sutures curved; aperture terminal, radiate, with a slightly longer slit on the apertural side.
Lenticulina orbicularis (d’Orbigny, 1826)
Figure 2.6-7
1826 Robulina orbicularis d’Orbigny, p. 288, pl. 15, figs. 8-9.
2010 Lenticulina orbicularis (d’Orbigny); Margreth, p. 150, pl. 12, fig. 8a-b.
2012 Lenticulina orbicularis (d’Orbigny); Debenay, p. 224.
2012 Lenticulina orbicularis (d’Orbigny); Milker and Schmiedl, p. 73, figs. 18.19-20.
Description. Test rounded, strongly biconvex, peripheral view lenticular, wall finely perforate, surface smooth, umbo prominent; chambers many, narrow; periphery with a thick keel, sutures strongly curved; aperture terminal, radiate, with a slit on the apertural side.
Lenticulina suborbicularis Parr, 1950
Figure 2.8-9
1950 Lenticulina (Robulus) suborbicularis Parr, p. 321, pl. 11, figs. 5-6.
2012 Lenticulina suborbicularis Parr; Debenay, p. 224.
Description. Test subcircular, biconvex, wall finely perforate, surface smooth, umbo broad and raised; chambers many; periphery with a thick keel, sutures strongly curved; aperture terminal, radiate, with a slit on the apertural face.
Genus MARGINULINOPSIS Silvestri, 1904
Marginulinopsis costata (Batsch, 1791)
Figure 2.10
1791 Nautilus (Orthoceras) costatus Batsch, p. 2, pl. 1, fig. 1.
2012 Marginulina costata (Batsch); Milker and Schmiedl, p. 73, fig. 18.26.
Description. Test elongate, cross section subcylindrical or oval, wall calcareous, surface ornamented with long striae; earlier chambers compact, final chambers inflated and subspherical, closely coiled initially, later uniserially arranged; sutures indistinct in the earlier chambers, straight and depressed in the final chambers; aperture terminal at the end of a short neck.
Family NODOSARIIDAE Ehrenberg, 1838
Subfamily NODOSARIINAE Ehrenberg, 1838
Genus DENTALINA d’Orbigny, 1826
Dentalina ittai Loeblich and Tappan, 1953
Figure 2.11
1953 Dentalina ittai Loeblich and Tappan, p. 56, pl. 10, figs. 10-12.
2008 Dentalina ittai Loeblich and Tappan; Riveiros and Patterson, p. 14, figs. 6.1a-b.
Description. Test free, elongate, slightly bent, wall calcareous, finely perforate, surface smooth; chambers elliptical, of uniform size, slightly overlapping, uniserially arranged; sutures distinct; aperture terminal, round, slightly raised.
Genus PYRAMIDULINA Fornasini, 1894
Pyramidulina catesbyi (d’Orbigny, 1839a)
Figure 2.12
1839a Nodosaria catesbyi d’Orbigny, p. 16, pl. 1, figs. 8-10.
2012 Pyramidulina catesbyi (d’Orbigny); Debenay, p. 168.
2012 Pyramidulina catesbyi (d’Orbigny); Milker and Schmiedl, p. 72, fig. 18.15-16.
Description. Test elongate, cross section subspherical or ovate, wall calcareous, finely perforate, surface ornamented with long striae and a prominent basal spine; chambers two, initial chamber globular, second chamber nearly pyriform, chamber arrangement uniserial; suture distinct, depressed; aperture terminal, radiate at the end of a well-developed neck.
Subfamily LINGULININAE Loeblich and Tappan, 1961
Genus PSEUDOLINGULINA McCulloch, 1977
Pseudolingulina bradii Silvestri, 1903
Figure 2.13-14
1903 Lingulonodosaria bradii Silvestri, p. 48.
2013 Pseudolingulina bradii Silvestri; Jones, p. 37, pl. 2, fig. 1.
Description. Test elongate, cross section circular, wall calcareous, surface smooth, no ornamentation; chambers subglobular, slightly overlapping, initial chamber tapering, gradually increasing in size as added, arranged linearly; sutures distinct and depressed; aperture terminal, radiate.
Family LAGENIDAE Reuss, 1862
Genus LAGENA Walker and Boys, 1784
Lagena aspera Reuss, 1861
Figure 2.15
1861 Lagena aspera Reuss, p. 305, pl. 1, fig. 5.
2006 Lagena aspera Reuss; Figueroa, Marchant, Gigglio and Ramírez, fig. 13.
Description. Test subglobular, wall calcareous, hyaline, surface ornamented with regular rows of spines; chamber single; aperture terminal, round at the end of a short neck.
Lagena perlucida (Montagu, 1803)
Figure 2.16
1803 Vermiculum perlucidum Montagu, p. 525, pl. 14, fig. 3.
2002 Lagena perlucida (Montagu); Gandhi, Rajamanickam and Nigam, p. 56, pl. 2, fig. 3.
Description. Test flask-shaped, unilocular, wall calcareous, finely perforate, surface smooth except for striations at the aboral end; aperture terminal, round at the end of a long neck.
Lagena semistriata Williamson, 1848
Figure 2.17
1848 Lagena striata (Montagu) var. semistriata Williamson, p. 14, pl. 1, figs. 9-10.
1994 Lagena semistriata Williamson; Jones, p. 64, pl. 57, figs. 14, 16.
Description. Test pear-shaped with a flat base, wall calcareous, finely perforate, surface smooth except for the striations in the basal half of the test; single chambered; aperture terminal at the end of a long, striated neck.
Lagena striata (d’Orbigny, 1839a)
Figure 2.18
1839a Oolina striata d’Orbigny, p. 21, pl. 5, fig. 12.
2002 Lagena striata (d’Orbigny); Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 172, pl. 2, fig. 4.
2012 Lagena striata (d’Orbigny); Milker and Schmiedl, p.75, fig. 18.33.
Description. Test globular, wall calcareous, hyaline, unilocular, surface ornamented with many long striae and an apical pseudospine; aperture crescent-like with polygonal corrugations at the base of a long neck.
Lagena strumosa Reuss, 1858
Figure 2.19
1858 Lagena strumosa Reuss, p. 434.
2012 Lagena strumosa Reuss; Debenay, p. 153.
2012 Lagena strumosa Reuss; Milker and Schmiedl, p. 70, fig. 18.34.
Description. Test globular, unilocular, flask-shaped with a long neck, wall perforate, surface ornamented with long striae, neck with irregular annuli and fine spines, distinct basal spine; aperture crescent-like, at the end of the neck, with a thick, expanded lip.
Lagena substriata Williamson, 1848
Figure 2.20
1848 Lagena substriata Williamson, p. 15, pl. 2, fig. 12.
2003 Lagena substriata Williamson; Murray, p. 17, fig. 5.7.
Description. Test subglobular, wall calcareous, finely perforate, surface ornamented with long striae, base with short spines; single-chambered; aperture terminal, round at the end of a long neck twisted at the base.
Genus HYALINONETRION Patterson and Richardson, 1988
Hyalinonetrion gracillima (Seguenza, 1862)
Figure 2.21
1862 Amphorina gracillima Seguenza, p. 51, pl. 1, fig. 37.
2012 Hyalinonetrion gracillima (Seguenza); Debenay, p. 152.
2012 Hyalinonetrion gracillima (Seguenza); Milker and Schmiedl, p. 74, fig. 18.30.
Description. Test elongate, spindle-shaped with a long neck, aboral end acute, wall calcareous, hyaline with fine perforations, surface smooth; aperture terminal, rimmed by a phialine lip.
Genus PROCEROLAGENA Puri, 1954
Procerolagena gracilis (Williamson, 1848)
Figure 2.22
1848 Lagena gracilis Williamson, p. 13, pl. 1, fig. 5.
2008 Procerolagena gracilis (Williamson); Riveiros and Patterson, p. 16, fig. 6.7.
2015 Procerolagena gracilis (Williamson); Hanagata and Nobuhara, p. 37, fig. 13.12.
Description. Test slightly elongate, unilocular with a distinct basal spine, wall calcareous, finely perforate, surface ornamented with long striae extending only up to half of the neck; aperture terminal, at the end of the neck, bordered by a phialine lip.
Procerolagena sp.
Figure 2.23
Description. Test elongate, slightly compressed, wall calcareous, hyaline, surface ornamented with striae and a distinct, stout basal spine; single-chambered; aperture terminal, round at the end of a blunt neck, bordered by a thick rim.
Genus REUSSOOLINA Colom, 1956
Reussoolina laevis (Montagu, 1803)
Figure 2.24
1803 Vermiculum laeve Montagu, p. 524, pl. 1, fig. 9.
2008 Lagena laevis (Montagu); Riveiros and Patterson, p. 14, figs. 6.3a-b.
Description. Test flask-shaped with a rounded base and a subcylindrical neck; wall calcareous, finely perforate, surface smooth or with low costae developed on the basal half of the test; single-chambered; aperture terminal, round at the end of the neck, with a flared lip.
Superfamily POLYMORPHINOIDEA d’Orbigny, 1839
Family ELLIPSOLAGENIDAE Silvestri, 1923
Subfamily OOLININAE Loeblich and Tappan, 1961
Genus FAVULINA Patterson and Richardson, 1988
Favulina hexagona (Williamson, 1848)
Figure 2.25
1848 Entosolenia squamosa (Montagu) var. hexagona Williamson, p. 20, pl. 2, fig. 23.
2002 Favulina hexagona (Williamson); Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 172, pl. 2, fig. 8.
2010 Favulina hexagona (Williamson); Margreth, p. 107, pl. 14, fig. 7.
2012 Favulina hexagona (Williamson); Debenay, p. 144.
2012 Favulina hexagona (Williamson); Milker and Schmiedl, p.77, fig. 19.4.
Description. Test subglobular, wall calcareous, hyaline, surface ornamented with large, raised hexagonal reticulate ridges; chamber single; aperture terminal, rounded at the end of a short neck surrounded by a thick band-like lip.
Subfamily ELLIPSOLAGENINAE Silvestri, 1923
Genus FISSURINA Reuss, 1850
Fissurina laevigata Reuss, 1850
Figure 3.1
1850 Fissurina laevigata Reuss, p. 366, pl. 46, fig. 1.
2012 Fissurina laevigata Reuss; Debenay, p. 147.
Description. Test pyriform, unilocular, wall calcareous, finely perforate, surface smooth; aperture terminal, an oval opening.
Fissurina orbignyana Seguenza, 1862
Figure 3.2
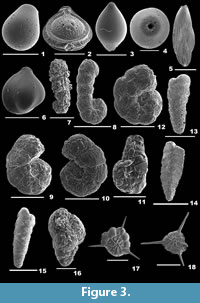 1862 Fissurina orbignyana Seguenza, p. 66, pl. 2, figs. 25-26.
1862 Fissurina orbignyana Seguenza, p. 66, pl. 2, figs. 25-26.
2003 Fissurina orbignyana Seguenza; Murray, p. 17, fig. 5.5-6.
2012 Fissurina orbignyana Seguenza; Milker and Schmiedl, p. 79, fig. 19.13.
Description. Test subrounded, wall calcareous, thickly perforated, surface ornamented with short spines; single-chambered; periphery with three prominent keels; aperture terminal, an irregular oval-shaped opening, bordered by a thick lip.
Family GLANDULINIDAE Reuss, 1860
Subfamily GLANDULININAE Reuss, 1860
Genus GLANDULINA d’Orbigny, 1839a
Glandulina ovula d’Orbigny, 1846
Figure 3.3-4
1846 Glandulina ovula d’Orbigny, p. 21, pl. 2, figs. 6-7.
2006 Glandulina ovula d’Orbigny; Figueroa, Marchant, Gigglio and Ramírez, fig. 11a-b.
2015 Glandulina ovula d’Orbigny; Hanagata and Nobuhara, p. 41, fig. 14.20-21.
Description. Test fusiform, wall calcareous, finely perforate, surface smooth, basal part with short spines; chamber more inflated; aperture terminal, several slits fuse to form one circular opening.
Family POLYMORPHINIDAE d’Orbigny, 1839
Subfamily POLYMORPHININAE d’Orbigny, 1839
Genus PSEUDOPOLYMORPHINA Cushman and Ozawa, 1928
Pseudopolymorphina sp.
Figure 3.5
1884 Polymorphina compressa (d’Orbigny); Brady, p. 564, pl. 72, figs. 9-11.
2012 Pseudopolymorphina sp. Debenay, p. 176.
Description. Test oval-shaped, compressed, wall calcareous, finely perforate, surface ornamented with many, continuous striations; initial chambers compressed, final chambers slightly inflated, rapidly increasing in size as added, chamber arrangement biserial; sutures depressed; aperture terminal, radiate.
Genus SIGMOIDELLA Cushman and Ozawa, 1928
Sigmoidella elegantissima (Parker and Jones, 1865)
Figure 3.6
1865 Polymorphina elegantissima Parker and Jones, p. 438.
2012 Sigmoidella elegantissima (Parker and Jones); Debenay, p. 248.
2015 Sigmoidella elegantissima (Parker and Jones); Hanagata and Nobuhara, p. 38, fig. 13.24-26.
Description. Test large, asymmetrical, cross section sigmoidal, wall calcareous, finely perforate, surface smooth; final chambers enveloping the initial ones on one side, coiling partially evolute on the other side; sutures slightly depressed; aperture terminal, radiate.
Class GLOBOTHALAMEA Pawlowski, Holzmann, Tyszka, 2013
Subclass TEXTULARIIA Mikhalevich, 1980
Order LITUOLIDA Lankester, 1885
Suborder LITUOLINA Lankester, 1885
Superfamily LITUOLOIDEA de Blainville, 1827
Family LITUOLIDAE de Blainville, 1827
Subfamily AMMOMARGINULININAE Podobina, 1978
Genus AMMOBACULITES Cushman, 1910
Ammobaculites agglutinans (d’Orbigny, 1846)
Figure 3.7
1846 Spirolina agglutinans d’Orbigny, p. 137, pl. 7, figs. 10-12.
1985 Ammobaculites agglutinans (d’Orbigny); Papp and Schmidt, p. 54, pl. 45, figs. 6-9.
2001 Ammobaculites agglutinans (d’Orbigny); Szarek, p. 85, pl. 5, fig. 2.
2010 Ammobaculites agglutinans (d’Orbigny); Margreth, p. 97, pl. 3, fig. 4a-b.
2012 Ammobaculites agglutinans (d’Orbigny); Debenay, p. 74.
2014 Ammobaculites agglutinans (d’Orbigny); Panchang and Nigam, pl. 1, figs. 21-24.
Description. Test elongate, umbilical area slightly concave, wall coarsely agglutinated, surface rough; early chambers small, compressed, and planispirally coiled, later chambers uncoiled, cylindrical, gradually increasing in size as added; periphery rounded, sutures obscured; aperture terminal, simple at the centre of the apertural face.
Ammobaculites exiguus Cushman and Brönnimann, 1948
Figure 3.8
1948 Ammobaculites exiguus Cushman and Brönnimann, p. 38, pl. 7, figs. 7-8.
2012 Ammobaculites exiguus Cushman and Brönnimann; Debenay, p. 74.
Description. Test elongate, wall agglutinated, surface smoothly finished; early chambers closely coiled, adult chambers rectilinearly arranged; periphery rounded, sutures distinct, depressed; aperture terminal, a circular opening.
Ammobaculites persicus Lutze, 1974
Figure 3.9-10
1974 Ammobaculites persicus Lutze, p. 9, pl. 2, figs. 27-35.
1999 Ammobaculites persicus Lutze; Nigam and Khare, p. 288, pl. 1, fig. 5.
Description. Test short, stout, umbilical region excavated, wall coarsely agglutinated, surface smooth; chambers initially closely coiled, final chambers arranged uniserially; periphery rounded, sutures distinct, depressed; aperture terminal and round.
Family DISCAMMINIDAE Mikhalevich, 1980
Genus AMMOSCALARIA Höglund, 1947
Ammoscalaria pseudospiralis (Williamson, 1858)
Figure 3.11
1858 Proteonina pseudospiralis Williamson, p. 2, pl. 1, figs. 2-3.
2003 Ammoscalaria pseudospiralis (Williamson); Murray, p. 11, fig. 2.3.
2014 Ammoscalaria pseudospiralis (Williamson); Panchang and Nigam, pl. 1, figs. 16a-b, 17.
Description. Test free, elongate, flattened laterally, wall coarsely agglutinated, surface rough; initial chambers compressed, planispirally arranged, later arranged linearly, aboral end broadly rounded, sutures indistinct; aperture terminal, rounded at the end of the final chamber.
Family HAPLOPHRAGMOIDIDAE Maync, 1952
Genus LABROSPIRA Höglund, 1947
Labrospira crassimargo (Norman, 1892)
Figure 3.12
1892 Haplophragmium crassimargo Norman, p. 17, pls. 7-8.
2008 Cribrostomoides crassimargo (Norman); Riveiros and Patterson, p. 5, fig. 2.2.
2012 Labrospira crassimargo (Norman); Hemleben, Spindler and Anderson, p. 332, pl. 2, figs. 16-17; pl. 7, figs. 6-8.
Description. Test robust, wall coarsely agglutinated, surface smooth, umbilical region depressed; chambers slightly inflated, coiling planispiral, partially evolute, gradually increasing in size as added; periphery broadly rounded, sutures distinct, depressed; aperture terminal, rounded.
Suborder SPIROPLECTAMMININA Mikhalevich, 1992
Superfamily SPIROPLECTAMMINOIDEA Cushman, 1927
Family SPIROPLECTAMMINIDAE Cushman, 1927
Subfamily SPIROPLECTAMMININAE Cushman, 1927
Genus SPIROPLECTAMMINA Cushman, 1927
Spiroplectammina sagittula (Defrance, 1824)
Figure 3.13
1824 Textularia sagittula Defrance, p. 177, pl. 13, fig. 5-5a.
2003 Textularia sagittula (Defrance); Murray, p. 15, fig. 3.12-14.
2006 Spiroplectinella sagittula (d’Orbigny); Oflaz, p. 129, pl. 1, fig. 2.
Description. Test elongate, slightly compressed laterally, outline subtriangular, wall agglutinated, surface smooth; chambers biserially arranged, gradually increasing in size as added; periphery subacute, sutures curved, slightly depressed; aperture terminal, low arched opening, bordered by a lip.
Genus SPIROPLECTINELLA Kisel'man, 1972
Spiroplectinella wrightii (Silvestri, 1903)
Figure 3.14
1903 Spiroplecta wrightii Silvestri, p. 59, text-figs. 1-6.
2001 Spiroplectinella wrightii (Silvestri); Szarek, p. 88, pl. 6, fig. 8.
2010 Spiroplectinella wrightii (Silvestri); Margreth, p. 133, pl. 3, fig. 6a-c.
2012 Spiroplectinella sagittula s.l. (Defrance); Milker and Schmiedl, p. 31, fig. 9.19-21.
2015 Spiroplectinella wrightii (Silvestri); Hanagata and Nobuhara, p. 14, fig. 6.7-8.
Description. Test triangular, wall finely agglutinated, surface smooth; chambers not inflated, compressed, gradually increasing in size as added, biserially arranged; periphery acutely angled, sutures distinct, depressed; aperture terminal, low arched opening, bordered by a lip.
Subfamily SPIROTEXTULARIINAE Saidova, 1975
Genus SPIROTEXTULARIA Saidova, 1975
Spirotextularia floridana (Cushman, 1922a)
Figure 3.15
1922a Textularia floridana Cushman, p. 24, pl. 1, fig. 7.
2001 Spirotextularia floridana (Cushman); Szarek, p. 88, pl. 6, figs. 11-13.
2012 Spirotextularia floridana (Cushman); Debenay, p. 95.
Description. Test elongate, narrow, much longer than wide, compressed laterally, apertural end broadly rounded, gently tapering towards the aboral end, wall finely agglutinated, surface smooth; chambers biserially arranged, gradually increasing in size as added, chamber ends compressed and projected along the sides; periphery subangular, peripheral margins nearly parallel, sutures very distinct and curved, slightly depressed; aperture terminal, small slit-like opening, bordered by a lip.
Suborder VERNEUILININA Mikhalevich and Kaminski, 2004
Superfamily VERNEUILINOIDEA Cushman, 1911
Family PROLIXOPLECTIDAE Loeblich and Tappan, 1985
Genus EGGERELLOIDES Haynes, 1973
Eggerelloides scaber (Williamson, 1858)
Figure 3.16
1858 Bulimina scabra Williamson, p. 65, pl. 3, figs. 136-137.
2003 Eggerelloides scaber (Williamson); Murray, p. 13, fig. 2.11.
2010 Eggerelloides scaber (Williamson); Margreth, p. 98, pl. 5, fig. 4.
2012 Eggerelloides scabrus (Williamson); Milker and Schmiedl, p. 37, fig. 10.9.
Description. Test subfusiform, wall coarsely agglutinated, surface smooth; chambers inflated, subglobular, trochospirally arranged initially, later triserially arranged with gradual increase in size; periphery rounded, sutures arcuate, depressed; aperture terminal, interiomarginal with a curved toothplate.
Family VERNEUILINIDAE Cushman, 1911
Subfamily VERNEUILININAE Cushman, 1911
Genus GAUDRYINA d’Orbigny, 1839
Gaudryina angulata Cushman, 1924
Figure 4.1
1924 Gaudryina triangularis (Cushman) var. angulata Cushman, p. 22.
2014 Gaudryina angulata Cushman; Panchang and Nigam, pl. 2, fig. 18a-b.
Description. Test triangular, wall arenaceous, surface smooth; chambers broad at the apertural end, pointed at the aboral end, angular on both sides; periphery acutely angled, sutures distinct, depressed; apertural side truncate, aperture terminal, low, slit-like depression.
Gaudryina convexa (Karrer, 1865)
Figure 4.2
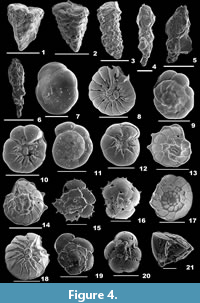 1865 Textilaria convexa Karrer, p. 78, pl. 16, fig. 8a-c.
1865 Textilaria convexa Karrer, p. 78, pl. 16, fig. 8a-c.
2012 Gaudryina convexa (Karrer); Debenay, p. 81.
2014 Gaudryina convexa (Karrer); Panchang and Nigam, pl. 2, fig. 19a-b.
Description. Test triangular, wall coarsely agglutinated, surface rough; final chambers broad, tapering towards the apex, flat on one side, strongly convex on the opposite side; periphery subacutely rounded, sutures depressed; apertural side obliquely truncate, aperture a low depression in the final chamber, bordered by a lip.
Suborder HORMOSININA Haeckel, 1894
Superfamily HORMOSINOIDEA Haeckel, 1894
Family REOPHACIDAE Cushman, 1927
Genus REOPHAX de Montfort, 1808
Reophax agglutinatus Cushman, 1913
Figure 4.3
1913 Reophax agglutinatus Cushman, p. 637, pl. 79 fig. 6.
2010 Reophax agglutinatus Cushman; Margreth, p. 96, pl. 2, fig. 1a-b.
2012 Reophax agglutinatus Cushman; Debenay, p. 89.
Description. Test subcylindrical, tapering towards the aboral end, wall coarsely agglutinated, composed of arenaceous material held together with grayish cement, surface rough; chambers many; aperture terminal on a slightly raised neck.
Reophax scorpiurus de Montfort, 1808
Figure 4.4-5
1808 Reophax scorpiurus de Montfort, p. 331.
2001 Reophax scorpiurus de Montfort; Szarek, p. 80, pl. 3, figs. 1-5.
2010 Reophax scorpiurus de Montfort; Margreth, p. 96, pl. 2, fig. 3a-b.
2011 Reophax scorpiurus de Montfort; Kaminski and Cetean, p. 65, pl. 2, figs. 23-25.
2012 Reophax scorpiurus de Montfort; Milker and Schmiedl, p. 32, fig 9.8.
2012 Reophax scorpiurus de Montfort; Debenay, p. 91.
2014 Reophax scorpiurus de Montfort; Panchang and Nigam, pl. 1, fig. 9.
Description. Test elongate, uniserial, wall coarsely agglutinated, surface irregular; chambers almost cylindrical or pyriform, increasing in size as added, arranged in an irregular series, early chambers slightly arcuate, final chamber fusiform or globular, tapering to the aperture; sutures horizontal, distinct and depressed; aperture simple, rounded, terminal on a short neck.
Genus NODULINA Rhumbler, 1895
Nodulina dentaliniformis (Brady, 1881)
Figure 4.6
1881 Reophax dentaliniformis Brady, p. 49.
1987 Nodulina dentaliniformis (Brady); Loeblich and Tappan, p. 58, pl. 44, figs. 10-11.
2001 Reophax dentaliniformis (Brady); Szarek, p. 79, pl. 2, figs. 14-15.
2011 Nodulina dentaliniformis (Brady); Kaminski and Cetean, p. 65, pl. 2, figs. 19-22.
2012 Reophax dentaliniformis Brady; Debenay, p. 90.
Description. Test elongate, slender, tapering, cross section circular, wall coarsely agglutinated, surface irregular, rough; chambers subcylindrical, arranged linearly; aperture terminal, rounded on a well-developed, distinct neck.
Order ROTALIIDA Delage and Hérouard, 1896
Superfamily ROTALIOIDEA Ehrenberg, 1839
Family ROTALIIDAE Ehrenberg, 1839
Subfamily AMMONIINAE Saidova, 1981
Genus AMMONIA Brünnich, 1772
Ammonia beccarii (Linnaeus, 1758)
Figure 4.7-8
1758 Nautilus beccarii Linnaeus, p. 710, pl. 1, fig. 1.
2001 Ammonia beccarii (Linnaeus); Szarek, p. 148, pl. 26, figs. 13-15.
2003 Ammonia beccarii (Linnaeus); Javaux and Scott, p. 10, fig. 2.2-3.
2005 Ammonia beccarii (Linnaeus); Debenay, Millet and Angelidis, p. 334, pl. 2, fig. 17.
2012 Ammonia beccarii (Linnaeus), Milker and Schmiedl, p. 117, fig. 27.1-2.
Description. Test calcareous, biconvex, wall perforate on both sides, surface smooth; chambers inflated, subglobular, trochospirally arranged, coiling evolute on the spiral side and involute on the umbilical side; periphery acute or slightly rounded, sutures radial and curved, thick and imperforate, depressed earlier and later incised on the spiral side, nearly radial and curved, deeply incised on the umbilical side; visible umbilical plug with pustules; aperture extraumbilical, interiomarginal, an arch shaped opening, sometimes covered by a calcitic boss.
Ammonia parkinsoniana (d’Orbigny, 1839b)
Figure 4.9-10
1839b Rosalina parkinsoniana d’Orbigny, p. 99, pl. 4, figs. 25-27.
2012 Ammonia beccarii (Linnaeus); Milker and Schmiedl, p. 117, figs. 27.3-4.
2015 Ammonia parkinsoniana (d’Orbigny); Hanagata and Nobuhara, p. 119, fig. 35.7-8.
Description. Test small, circular, spiral side convex, umbilical side flattened, wall calcareous, perforate, surface smooth; chambers subglobular, inflated on the spiral side, triangular on the umbilical side, arranged trochospirally; periphery broadly rounded, sutures nearly straight on the spiral side, radial on the umbilical side; umbilicus deeply sutured, with a calcitic knob; aperture an interiomarginal opening.
Ammonia tepida (Cushman, 1926)
Figure 4.11-12
1926 Rotalia beccarii (Linnaeus) var. tepida Cushman, p. 79, pl. 1.
2006 Ammonia tepida (Cushman); Oflaz, p. 231, pl. 9, figs. 13-15.
2012 Ammonia tepida (Cushman); Debenay, pp. 185-186.
2014 Ammonia tepida (Cushman); Panchang and Nigam, pl. 36, fig. 11a-c.
Description. Test small, biconvex, wall calcareous, densely perforate, surface smooth; chambers more inflated, subspherical on the spiral side, trochospirally arranged, gradually increasing in size as added; periphery slightly lobulated, broadly rounded, sutures almost straight, slightly depressed on the spiral side, radial, and more depressed on the umbilical side; umbilical plug missing; aperture terminal, an ovate slit-like opening.
Genus ASTEROROTALIA Hofker, 1950
Asterorotalia dentata (Parker and Jones, 1865)
Figure 4.13-14
1865 Rotalia beccarii (Linnaeus) var. dentata Parker and Jones, pp. 387-388, 422, pl. 19, fig. 18a-c.
2014 Asterorotalia dentata (Parker and Jones); Panchang and Nigam, pl. 36, fig. 12a-b.
Description. Test circular, wall calcareous, perforate, surface smooth; chambers many, arranged trochospirally, triangular and convex on the umbilical side; periphery keeled, with short, blunt spines, sutures distinct, thick, slightly curved on the spiral side, radial and deeply sutured on the umbilical side; aperture an arch-shaped opening at the terminal end of the apertural face.
Asterorotalia pulchella (d’Orbigny, 1839a)
Figure 3.17-18
1839a Rotalia (Calcarina) pulchella d’Orbigny, p. 80, pl. 5, figs. 16-18.
2001 Asterorotalia pulchella (d’Orbigny); Szarek, p. 147, pl. 27, figs. 11-12.
2015 Asterorotalia pulchella (d’Orbigny); Hanagata and Nobuhara, p. 119, figs. 35.9-10.
Description. Test small, outline nearly triangular, wall calcareous, perforate, surface strongly ornamented with knobs, pustules and ridges; chambers many, gradually increasing in size as added; periphery with imperforate keel and long triradiate spines, sutures nearly straight, raised and limbate on the dorsal side, radial and slightly curved on the ventral side; aperture an equatorial, terminal, ovate opening on the apertural side.
Asterorotalia inflata (Millett, 1904)
Figure 4.15-16
1904 Rotalia schroeteriana (Parker and Jones) var. inflata Millett, p. 504, pl. 10, fig. 5a-c.
2014 Asterorotalia inflata (Millett); Panchang and Nigam, pl. 36, fig. 13a-c.
Description. Test subcircular, flat dorsal and conical ventral side, wall calcareous, thin, perforate, surface smooth; chambers trochospirally arranged, compressed on the dorsal side, inflated, triangular and convex on the ventral side; periphery broadly rounded, with short spines, sutures distinct, slightly depressed on the spiral side, radial, curved and depressed on the ventral side; aperture two narrow, subovate openings on the ventral side.
Genus ROTALIDIUM Asano, 1936
Rotalidium annectens (Parker and Jones, 1865)
Figure 4.17-18
1865 Rotalia beccarii (Linnaeus) var. annectens Parker and Jones, pp. 387, 422, pl.19, fig. 11a-c.
2008 Rotalidium annectens (Parker and Jones); Panchang, p. 246, pl. 37, figs. 4a-c, 5a-c.
Description. Test large, round, wall calcareous, perforate, surface smooth, umbilicus with irregular calcitic knobs; chambers compressed on the dorsal side, ventral side convex, trochospirally arranged, peripheral margin acute, periphery with an imperforate keel, dorsal sutures nearly straight and limbate, radial and very much depressed on the ventral side; aperture interiomarginal, subcircular slit-like opening.
Genus ROTALINOIDES Saidova, 1975
Rotalinoides compressiuscula (Brady, 1884)
Figure 4.19-21
1884 Rotalia papillosa var. compressiuscula Brady, p. 708, pl. 107, fig. 1.
2015 Rotalinoides compressiuscula (Brady); Hanagata and Nobuhara, p. 119, fig. 35.11-12.
Description. Test subcircular, wall calcareous, perforate, surface smooth; initial chambers indistinct, gradually increasing in size as added, chamber arrangement trochospiral, flattened to slightly convex on the dorsal side, triangular and convex on the ventral side; periphery lobulated with a thin keel, sutures initially beaded, arcuate on the spiral side, radial on the umbilical side; aperture narrow, slit-like openings at the basal end of the apertural face.
Rotalinoides gaimardii (d’Orbigny, 1826)
Figure 5.1-2
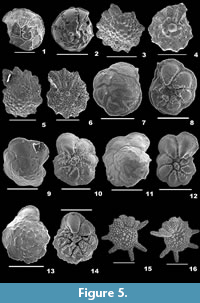 1826 Rotalia (Turbinulina) gaimardii d’Orbigny, p. 275, pl. 106.
1826 Rotalia (Turbinulina) gaimardii d’Orbigny, p. 275, pl. 106.
2001 Asterorotalia gaimardii (d’Orbigny); Szarek, p. 148, pl. 27, figs. 7-8.
2015 Rotalinoides gaimardii (Fornasini); Hanagata and Nobuhara, p. 119, figs. 36. 1-2.
Description. Test planoconvex, wall calcareous, perforate, surface smooth; chambers slightly convex on the dorsal side, highly convex on the ventral side, trochospirally arranged; periphery lobulated, strongly keeled, sutures raised, limbate on the spiral side, radial, deeply sutured, beaded on the umbilical side; aperture terminal, interiomarginal opening on the ventral side.
Subfamily PARAROTALIINAE Reiss, 1963
Genus NEOROTALIA Bermúdez, 1952
Neorotalia calcar (d’Orbigny, 1839a)
Figure 5.3-6
1839a Calcarina calcar d’Orbigny, p. 81, pl. 5, figs. 22-24.
2012 Neorotalia calcar (d’Orbigny); Debaney, pp. 204-205.
Description. Test biconvex, wall calcareous, perforate, surface ornamented with distinct, round and thick pustules on the dorsal side, ridges and many umbilical knobs on the ventral side; chambers many, slightly inflated and coiling evolute on the spiral side, involute on the umbilical side, trochospirally arranged; periphery angular, star-shaped with stout spines, sutures tilted and slightly depressed on the spiral side, radial and depressed on the umbilical side; primary aperture umbilical, low-arched opening bordered by a lip, secondary apertures peripheral, bordered by a lip.
Genus PARAROTALIA Le Calvez, 1949
Pararotalia boltovskoyi Jain and Bhatia, 1978
Figure 5.7-8
1978 Pararotalia boltovskoyi Jain and Bhatia, p. 165, pl. 2, figs. H-I.
2007 Pararotalia boltovskoyi Jain and Bhatia; Talib and Farroqui, p. 19, pl. 1, fig. 22a-c.
Description. Test subcircular, biconvex, umbilicus depressed, no plug, wall calcareous, distinctly perforate, dorsal surface smooth, umbilical surface rough, with pustules; chambers slightly convex, coiling evolute on the spiral side; convex, coiling involute on the ventral side, arranged trochospirally; periphery lobulated and angular with small spines at the chamber ends, sutures curved, limbate on the spiral side, deeply depressed on the umbilical side; aperture interiomarginal, umbilical, a narrow slit-like opening.
Pararotalia calcariformata McCulloch, 1977
Figure 5.9-10
1977 Pararotalia aff. P. calcariformata McCulloch, p. 428, pl. 177, figs. 10-11.
2013 Pararotalia calcariformata McCulloch; Meriç, Yokes, Avsar, Kirki-Elmas, Dinçer and Karhan, p. 3, figs. 2.1-2, 3.1-12.
Description. Test subrounded, biconvex, umbilicus with a plug, wall calcareous, perforate on both sides, surface smooth on the dorsal side, rough with many pustules on the ventral side; chambers slightly convex, coiling evolute on the spiral side, gradually increasing in size as added; convex and involute on the ventral side, arranged trochospirally; periphery lobulated and carinate with short spines at the chamber ends, prominent hyaline spine at the upper end of each chamber in the final whorl, sutures limbate and curved on the spiral side, very much depressed, broad and radiate on the umbilical side; aperture interiomarginal, umbilical, a low arched opening bordered by a thin rim.
Pararotalia nipponica (Asano), 1936
Figure 5.11-12
1936 Rotalia nipponica Asano, p. 614, pl. 31, fig. 2a-c.
2012 Pararotalia nipponica (Asano); Debenay, p. 206.
Description. Test subrounded, biconvex, umbilicus with a plug, wall calcareous, perforate on both sides, dorsal surface smooth, ventral surface rough, with pustules; chambers slightly convex, coiling evolute on the spiral side; involute and convex on the ventral side, arranged trochospirally; periphery lobulated and angular with small spines at the chamber ends, sutures curved, limbate on the spiral side, very much depressed on the umbilical side; aperture interiomarginal, umbilical, a slit-like opening.
Pararotalia venusta (Brady, 1884)
Figure 5.13-14
1884 Rotalia venusta Brady, p. 708, pl. 108, fig. 2c.
2012 Pararotalia venusta (Brady); Culver, Mallinson, Corbett, Leorri, Rouf, Shazili, Yaacob, Whittaker, Buzas and Parham, p. 114, fig. 3.4.
Description. Test sublenticular, wall calcareous, perforate, surface rough, granular on the dorsal side, pustular on the ventral side; chambers slightly convex on the spiral side, convex on the umbilical side, coiling evolute on the spiral side, involute on the umbilical side, arranged trochospirally, umbilicus with a knob; periphery lobulated, sutures curved on the dorsal side, radial and very much depressed on the ventral side; aperture interiomarginal, an elongate slit-like opening.
Family CALCARINIDAE d’Orbigny, 1826
Genus CALCARINA d’Orbigny, 1826
Calcarina hispida Brady, 1876
Figure 5.15-16
1876 Calcarina hispida Brady, p. 589.
2012 Calcarina hispida Brady; Debenay, p. 189.
2014 Calcarina hispida Brady; Panchang and Nigam, pl. 37, fig. 7a-b.
Description. Test hispid, subrounded, wall calcareous, perforate, surface rough, covered with distinct, granulated, calcitic knobs, umbilical side with radial ridges; chambers evolute on the spiral side, involute on the ventral side, final chambers narrow and inflated, arranged trochospirally; peripheral margin rounded, periphery with somewhat long, hispid spines, sutures depressed in the final chambers; apertures interiomarginal, terminal, mostly not seen.
Calcarina spengleri (Gmelin, 1791)
Figure 6.1-2
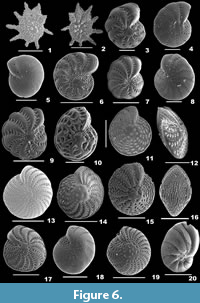 1788 Nautilus spengleri Gmelin, p. 3371.
1788 Nautilus spengleri Gmelin, p. 3371.
2005 Calcarina spengleri (Gmelin); Renema and Hohenegger, p. 16, pl. 1, figs. 1-10; text-fig. 1.
2014 Calcarina spengleri (Gmelin); Panchang and Nigam, pl. 37, figs. 9a-b, 10a-b.
Description. Test large, biconvex, side view lenticular, wall calcareous, coarsely perforate, surface ornamented with few, granulated calcitic knobs in the centre; chambers many, final chambers long, narrow and inflated, chamber arrangement trochospiral; periphery with long, tapering, blunt spines; sutures distinct only in the final chambers, slightly depressed on the spiral side, radial and depressed on the umbilical side; aperture not seen.
Family ELPHIDIIDAE Galloway, 1933
Subfamily ELPHIDIINAE Galloway, 1933
Genus CRIBROELPHIDIUM Cushman and Brönnimann, 1948
Cribroelphidium excavatum (Terquem, 1875)
Figure 6.3
1875 Polystomella excavatum, Terquem, p. 25, pl. 2, fig. 2a-f.
2008 Cribroelphidium excavatum (Terquem); Riveiros and Patterson, p. 32, fig. 14.1-5.
Description. Test free, slightly compressed, wall calcareous, surface smooth but for pustular sutures, umbilical region and apertural side, umbilicus slightly depressed, with a knob; chambers few, slightly inflated, gradually increasing in size as added, coiling involute, arranged planispirally; periphery broadly rounded, peripheral margin lobulate, sutures distinct, slightly depressed, curved backwards, each intercepted with large pores and short perpendicular sutural joints; aperture interiomarginal, multiple, a series of rounded openings on the apertural face.
Cribroelphidium poeyanum (d’Orbigny, 1839a)
Figure 6.4
1839a Polystomella poeyana d’Orbigny, p. 55, pl. 6, figs. 25-26.
2008 Elphidium poeyanum (d’Orbigny); Araújo and Machado, p. 34, pl. 2, fig. 5.
Description. Test free, slightly compressed, wall calcareous, finely perforate, surface smooth; chambers many, very slightly inflated, arranged planispirally, coiling involute; periphery rounded, margin slightly lobulate, sutures distinct, slightly depressed with very short and broad retral processes; aperture terminal, interiomarginal, series of circular openings on the apertural side.
Genus ELPHIDIELLA Cushman, 1936
Elphidiella hannai (Cushman and Grant, 1927)
Figure 6.5
1927 Elphidium hannai Cushman and Grant, p. 77, pl. 7, fig. 1.
2008 Elphidiella hannai (Cushman and Grant); Riveiros and Patterson, p. 34, figs. 15.5a-5c.
Description. Test round, wall calcareous, finely perforate, surface smooth, apertural side granular; chambers many, distinct, not inflated, arranged planispirally, coiling involute; umbilical region flattened; periphery rounded, sutures distinct, curved and limbate; aperture interiomarginal, multiple, small, round openings along the apertural face.
Genus ELPHIDIUM de Montfort, 1808
Elphidium advenum subsp. limbatum (Chapman, 1907)
Figure 6.6
1907 Polystomella macellum var. limbatum Chapman, p. 142, pl. 10, fig. 9.
2012 Elphidium limbatum (Chapman); Debenay, p. 220.
Description. Test large, wall calcareous, finely perforate, surface ornamented with granular pustules along the sutures, umbilical region and the apertural side, umbilicus with a definite boss; chambers many, increasing in size as added, final chambers more inflated, arranged planispirally; periphery rounded, margin slightly keeled, sutures distinct and curved, retral processes broad, closely spaced; aperture interiomarginal, multiple, small pores along the apertural face.
Elphidium advenum var. depressulum Cushman, 1933
Figure 6.7
1933 E lphidium advenum (Cushman) var. depressulum Cushman, p. 51, pl. 12, fig. 4a-b.
1939a Elphidium advenum (Cushman) var. depressulum Cushman, p. 61, pl. 17, fig.1.
Description. Test large, wall calcareous, finely perforate, surface smooth, pustular sutrues and umbilicus; chambers few, broad, inflated, planispirally arranged, coiling involute; umbilical region depressed, no definite boss, with a few large pits; periphery more lobulated, with a more prominent keel, sutures slightly curved and distinct, retral processes distinct, rod-like; aperture interiomarginal, multiple at the base of apertural face.
Elphidium asiaticum Polski, 1959
Figure 6.8
1959 Elphidium discoidale (d’Orbigny) var. asiaticum Polski, p. 585, pl. 78, fig. 2a-b.
2015 Cibrononion asiaticum Polski; Lei, Li, Bi, Cui, Song, Li, and Li, p. 250, pl. 1, fig. 4a-e.
Description. Test small, outline circular, wall finely perforate; surface smooth, extremely hyaline; chambers slightly inflated, gradually increasing in size, chamber arrangement planispiral, coiling involute; periphery rounded, sutures distinct and gently curved with rows of small pores; aperture interiomarginal, series of basal pores on the apertural side.
Elphidium botaniense Albani, 1981
Figure 6.9
1981 Elphidium botaniense Albani, p. 155, fig. 4j, n.
2012 Elphidium botaniense Albani; Debenay, p. 218.
Description. Test broad, large, biconvex, outline circular, wall calcareous, finely perforate, surface smooth, sutures, umbilicus and apertural area with pustules; chambers many, inflated, arranged planispirally, coiling involute; umbilicus with a roughly circular, definite boss; periphery rounded, margin more lobulated, with a very distinct keel, sutures curved, marked with distinct retral processes; aperture interiomarginal, multiple rounded openings along the apertural face.
Elphidium carteri Hayward in Hayward, Hollis, and Grenfell, 1997
Figure 6.10
1997 Elphidium carteri Hayward; Hayward, Hollis, and Grenfell, pp. 71-72, pl. 1, fig. 15; pl. 6, figs. 8-12.
1994 Elphidium jenseni (Cushman); Loeblich and Tappan, p. 169, pl. 381, figs. 4-5.
Description. Test small, slightly compressed, outline subcircular, wall calcareous, perforate, surface covered with pustules, more granulated on the apertural side; chambers many, arranged planispirally; periphery rounded, margin with a distinctly rounded keel, sutures distinct, curved, slightly raised and limbate, retral processes broad; aperture interiomarginal, a series of rounded pores at the basal end of the apertural face.
Elphidium charlottense (Vella, 1957)
Figure 6.11-12
1957 Elphidiononion charlottensis Vella, p. 38, pl. 9, figs. 187-188.
2012 Elphidium charlottense (Vella); Debenay, p. 218.
Description. Test biconvex, slightly compressed, wall calcareous, finely perforate, surface with pustules along the sutures and the apertural face; umbilicus with a flat boss; chambers many, arranged planispirally, coiling involute; periphery rounded, margin very slightly lobulated, with a distinct keel, sutures slightly curved, distinct, retral processes short; aperture multiple openings at the basal end of the final chamber on the apertural side.
Elphidium craticulatum (Fichtel and Moll, 1798)
Figure 6.13
1798 Nautilus craticulatus Fichtel and Moll, p. 51, pl. 5, figs. h-k.
2007 Elphidium craticulatum (Fichtel and Moll); Talib and Farroqui, p. 21, pl. 1, fig. 24 a-b.
2012 Elphidium craticulatum (Fichtel and Moll); Debenay, p. 219.
2014 Elphidium craticulatum (Fichtel and Moll); Panchang and Nigam, pl. 38, fig. 5a-b.
Description. Test large, subglobose, strongly biconvex, wall calcareous, perforate, surface reticulate, ornamented with pustules; chambers many, slightly inflated, planispirally arranged; periphery smooth with a thin rounded keel, sutures radial; aperture interiomarginal, a row of small openings, weakly bordered by a rim.
Elphidium crispum (Linnaeus, 1758)
Figure 6.14
1758 Nautiluscrispus Linnaeus, p. 709, pl. 19, fig. 1d.
2001 Elphidium crispum (Linnaeus); Szarek, p. 150, pl. 28, fig. 3.
2006 Elphidium crispum (Linnaeus); Oflaz, p. 235, pl. 11, fig. 11.
2007 Elphidium crispum (Linnaeus); Talib and Farroqui, p. 21, pl.1, fig. 25a-b.
2012 Elphidium crispum (Linnaeus); Debenay, p. 216.
2012 Elphidium crispum (Linnaeus); Milker and Schmiedl, p. 120, fig. 27.13-14.
2014 Elphidium crispum (Linnaeus); Panchang and Nigam, pl. 38, figs. 6a-b, 7.
Description. Test large, lenticular, outline circular, wall calcareous, perforate, surface reticulate, ornamented with small pustules in the apertural region, umbilical boss with small, shallow pits; chambers many, narrow, final chambers inflated, chamber arrangement planispiral, coiling involute; periphery angular with thin carina, sutures strongly curved backwards; aperture interiomarginal, multiple, a series of openings bordered by a rim on the apertural face.
Elphidium hispidulum Cushman, 1936
Figure 6.15-16
1936 Elphidium hispidulum Cushman, p. 83, pl. 14, fig. 13a-b.
2007 Elphidium hispidulum Cushman; Horton, Culver, Hardbattle, Larcombe, Milne, Morigi, Whittaker, and Woodroffe, p. 58, pl. 1, fig. 9a-b.
2012 Parrellina hispidula Cushman; Debenay, p. 229.
Description. Test slightly compressed, circular, wall coarsely perforate, surface finely spinose, with fine rounded costae in the earlier portion; chambers indistinct, final chambers slightly inflated, planispirally arranged; umbilical area with thick spines, slightly raised; periphery rounded, sutures slightly curved and depressed; aperture multiple, small openings at the base of the apertural face, mostly obscured.
Elphidium macellum (Fichtel and Moll, 1798)
Figure 6.17
1798 Nautilus macellus Fichtel and Moll, p. 66, pl. 10, figs. h-k.
2002 Elphidium macellum (Fichtel and Moll); Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 178, pl. 5, fig. 11.
2012 Elphidium macellum (Fichtel and Moll); Debenay, p. 220.
2012 Elphidium macellum (Fichtel and Moll); Milker and Schmiedl, p. 122, fig. 27.21-22.
2014 Elphidium macellum (Fichtel and Moll); Panchang and Nigam, pl. 38, fig. 10a-b.
Description. Test large, compressed, outline circular, wall perforate, surface reticulate, ornamented with small pustules near apertural region and the periphery, umbilical region flat; chambers many, narrow, chamber arrangement planispiral, coiling involute; periphery acute or angular with thin keel, slightly rounded in the final chambers, sutures strongly curved backwards; aperture a series of interiomarginal openings.
Elphidium somaense Takayanagi, 1955
Figure 6.18
1955 Elphidium somaense Takayanagi, p. 52, fig. 28a-b.
1999 Elphidium somaense Takayanagi; Nigam and Khare, p. 300, pl. 7, fig. 15.
2000 Elphidium somaense Takayanagi; Scott, Takayanagi, Hasegawa and Saito, p. 21, fig. 7.125-126.
Description. Test nearly circular, small, wall calcareous, perforate, surface smooth, apertural face granulated; chambers distinct, planispirally arranged, coiling involute; periphery rounded, sutures distinct with small round pores and curved; aperture interiomarginal, multiple basal pores on the apertural side.
Elphidium striatopunctatum (Fichtel and Moll, 1798)
Figure 6.19
1798 Nautilus striato-punctatus Fichtel and Moll, p. 61, pl. 9, figs. a-c.
2008 Elphidium striatopunctatum (Fichtel and Moll); Meriç, Avşar and Yokeş, p. 323, pl. 9, figs. 13-15.
2011 Elphidium striatopunctatum (Fichtel and Moll); Pilarczyk, Reinhardt, Boyce, Schwarcz and Donato, p. 66, pl. 1, fig. 9.
Description. Test slightly large, lenticular, wall finely perforate, surface smooth, apertural side slightly pustular; chambers many, final chambers rapidly increase in size, inflated, coiling involute; periphery broadly rounded, sutures curved and slightly depressed, retral processes short, closely spaced, rod-like; aperture multiple, a row of small openings at the base of slightly convex apertural face.
Superfamily ASTERIGERINOIDEA d’Orbigny, 1839
Family AMPHISTEGINIDAE Cushman, 1927
Genus AMPHISTEGINA d’Orbigny, 1826
Amphistegina bicirculata Larsen, 1976
Figure 7.1-2
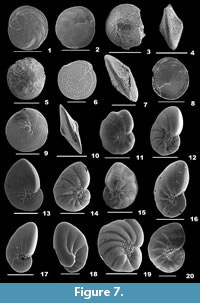 1976 Amphistegina bicirculata Larsen, p. 10, pl. 2, figs. 1-5; p. 16, text-figs. 9.2, 10.2.
1976 Amphistegina bicirculata Larsen, p. 10, pl. 2, figs. 1-5; p. 16, text-figs. 9.2, 10.2.
2002 Amphistegina bicirculata Larsen; Yordanova and Hohenegger, p. 178, pl. 31, figs. 9-12.
2007 Amphistegina bicirculata Larsen; Yordanova and Hohenegger, p. 1276, fig. 1.11.
2012 Amphistegina bicirculata Larsen; Debenay, p. 215.
2014 Amphistegina bicirculata Larsen; Hohenegger, p. 723, fig. 17.
2014 Amphistegina bicirculata Larsen; Panchang and Nigam, pl. 32, figs. 9a-b, 10a-b.
Description. Test flat, lenticular, unequally biconvex or slightly spiroconvex, wall calcareous, surface smooth; chambers initially radiate, later nicely curved at the periphery, chamber arrangement low trochospiral, coiling involute; peripheral margin acute, slightly keeled, sutures slightly depressed; aperture interiomarginal, a slit-like opening, surrounded by pustules, bordered by a lip.
Amphistegina gibbosa d’Orbigny, 1839b
Figure 7.3-4
1839b Amphistegina gibbosa d’Orbigny, p. 120, pl. 8, figs. 1-3.
2014 Amphistegina gibbosa d’Orbigny; Panchang and Nigam, pl. 32, fig. 11a-c.
Description. Test lenticular, strongly biconvex in the central portion, flattened at the sides, wall calcareous, surface smooth; chambers radial initially, later curved towards the periphery, chamber arrangement low trochospiral, coiling involute; peripheral margin subacute, sutures slightly depressed; aperture interiomarginal, a narrow slit, bordered by a lip.
Amphistegina lessonii d’Orbigny, 1826
Figure 7.5
1826 Amphistegina lessonii d’Orbigny, p. 304, pl. 17, figs. 1-4.
2002 Amphistegina lessonii d’Orbigny; Yordanova and Hohenegger, p. 178, pl. 31, figs. 4-8.
2003 Amphistegina lessonii d’Orbigny; Javaux and Scott, p. 11, fig. 2.4-6.
2007 Amphistegina lessonii d’Orbigny; Yordanova and Hohenegger, p. 1276, fig. 1.10.
2012 Amphistegina lessonii d’Orbigny; Debenay, p. 215.
2014 Amphistegina lessonii d’Orbigny; Hohenegger, p. 723, fig. 17.
2014 Amphistegina lessonii d’Orbigny; Panchang and Nigam, pl. 32, fig. 1a-c.
Description. Test lenticular, outline circular, more convex on the spiral side, wall calcareous, umbilicus transparent, imperforate, surface smooth, apertural face with pustules; chambers many, curved backwards at the periphery, chamber arrangement low trochospiral, coiling involute; periphery rounded, margin subacute, sutures slightly depressed; aperture interiomarginal, umbilical, a slit-like opening, surrounded by pustules, bordered by a narrow lip.
Amphistegina papillosa Said, 1949
Figure 7.6-7
1949 Amphistegina radiata (Fichtel and Moll) var. papillosa Said, p. 39, pl. 4, fig. 12.
2001 Amphistegina papillosa Said; Szarek, p. 142, pl. 23, figs. 1-2.
2002 Amphistegina papillosa Said; Yordanova and Hohenegger, p. 178, pl. 31, figs. 18-21.
2007 Amphistegina papillosa Said; Yordanova and Hohenegger, p. 1276, fig. 1.13.
2012 Amphistegina papillosa Said; Debenay, p. 216.
2014 Amphistegina papillosa Said; Hohenegger, p. 723, fig. 17.
2014 Amphistegina papillosa Said; Panchang and Nigam, pl. 32, figs. 3a-b, 4a-b.
Description. Test flat, biconvex, outline subcircular, wall calcareous, perforate, surface covered with raised papillae; chamber arrangement low trochospiral; periphery rounded, margin acute, sutures slightly depressed in the final chambers; aperture umbilical, small, round opening surrounded by pustules.
Amphistegina radiata (Fichtel and Moll, 1798)
Figure 7.8-10
1798 Nautilus radiatus Fichtel and Moll, p. 58, pl. 8, figs. a-d.
1999 Amphistegina radiata (Fichtel and Moll); Nigam and Khare, p. 298, pl. 6, fig. 2.
2002 Amphistegina radiata (Fichtel and Moll); Yordanova and Hohenegger, p. 178, pl. 31, figs. 13-17.
2007 Amphistegina radiata (Fichtel and Moll); Yordanova and Hohenegger, p. 1276, fig. 1.12.
2012 Amphistegina radiata (Fichtel and Moll); Debenay, p. 216.
2014 Amphistegina radiata (Fichtel and Moll); Hohenegger, p. 723, fig. 17.
2014 Amphistegina radiata (Fichtel and Moll); Panchang and Nigam, pl. 32, figs. 5a-c, 6a-b.
Description. Test flattened, outline circular, wall calcareous, finely perforate, surface smooth; chambers many, planispirally arranged; periphery rounded, margin slightly angular, sutures slightly raised, curved at the periphery; aperture a small slit, bordered by a thin lip, surrounded by pustules.
Superfamily NONIONOIDEA Schultze, 1854
Family NONIONIDAE Schultze, 1854
Subfamily ASTRONONIONINAE Saidova, 1981
Genus ASTRONONION Cushman and Edwards, 1937
Astrononion stelligerum (d'Orbigny, 1839b)
Figure 7.11
1839b Nonionina stelligera d’Orbigny, p. 128, pl. 3, figs. 1-2.
2006 Astrononion stelligerum (d’Orbigny); Oflaz, p. 301, pl. 9, figs. 9-10.
2012 Astrononion stelligerum (d’Orbigny); Milker and Schmiedl, p. 113, fig. 26.7-8.
Description. Test compressed, side view circular, umbilical area depressed, wall calcareous, finely perforate, surface smooth, umbilicus, apertural side and along the sutures covered with granular pustules; chambers distinct, elongate, narrow, slightly inflated, gradually increasing in size; periphery subrounded, margin slightly angular, sutures distinct, strongly curved, slightly depressed; aperture an interiomarginal, low arch, equatorial opening at the base of the apertural face, bordered by a rim.
Subfamily NONIONINAE Schultze, 1854
Genus NONION de Montfort, 1808
Nonion commune (d'Orbigny, 1846)
Figure 7.12
1846 Nonionina communis d’Orbigny, p. 106, pl. 5, figs. 7-8.
2010 Nonion commune (d’Orbigny); Vénec-Peyré and Poignant, p. 488, fig. 3I-J.
Description. Test elongate-ovate, compressed, umbilical area depressed, wall calcareous, finely perforate, surface smooth, umbilicus, apertural side and to a very little extent along the sutures granulated; chambers distinct, slightly inflated, increasing in size as added, final chamber elongate; periphery subacutely rounded, sutures distinct, very slightly depressed, limbate and strongly curved; aperture an interiomarginal opening at the base of the long apertural face.
Nonion costiferum Cushman, 1926
Figure 7.13
1926 Nonionina costifera Cushman, p. 90, pl. 13, fig. 2a-c.
2014 Nonion costiferum Cushman; Panchang and Nigam, pl. 33, fig. 10a-b.
Description. Test elongate, compressed, umbilical portion depressed, wall finely perforate, surface smooth, umbilicus and the apertural side covered with pustules; chambers many, distinct, not inflated, of uniform size; periphery acute, slightly keeled, sutures distinct, slightly raised and costate; aperture an interiomarginal, small, low arch opening at the basal end of the apertural face.
Nonion fabum (Fichtel and Moll, 1798)
Figure 6.20
1798 Nautilus faba Fichtel and Moll, p. 103, pl. 19, figs. a-c.
2010 Nonion fabum (Fichtel and Moll); Margreth, p. 123, pl. 36, fig. 2a-c.
2012 Nonion fabum (Fichtel and Moll); Milker and Schmiedl, p. 112, fig. 25.22-24.
2014 Nonion fabum (Fichtel and Moll); Panchang and Nigam, pl. 33, fig. 11a-b.
Description. Test ovate, compressed, wall calcareous, perforate, surface smooth, umbilici slightly depressed, ornamented with pustules; chambers many, inflated, planispirally arranged with rapid increase in size, coiling involute; periphery broadly rounded, sutures thick and curved backwards; aperture an interiomarginal slit-like opening.
Genus NONIONELLA Cushman, 1926
Nonionella basispinata (Cushman and Moyer, 1930)
Figure 7.14
1930 Nonion pizarrensis Berry var. basispinata Cushman and Moyer, p. 54, pl. 7, fig. 18.
2008 Pseudononion basispinata (Cushman and Moyer); Riveiros and Patterson, p. 30, fig. 12.6a-b.
2014 Nonion basispinata (Cushman and Moyer); Panchang and Nigam, pl. 33, fig. 8a-c.
Description. Test small, asymmetrical, wall calcareous, finely perforate, surface smooth, granular pustules filling the umbilicus, apertural side and to a little extent along the sutures; chambers slightly inflated, rapidly increasing in size, planispirally arranged, coiling involute; periphery subrounded, sutures distinct, very slightly depressed and curved; aperture an interiomarginal, narrow, equatorially placed slit at the base of the apertural face.
Genus NONIONELLINA Voloshinova, 1958
Nonionellina labradorica (Dawson, 1860)
Figure 7.15
1860 Nonionina labradorica Dawson, p. 191, text-fig. 4.
2008 Nonionellina labradorica (Dawson); Riveiros and Patterson, p. 29, fig. 12.7a-c.
2010 Nonionellina labradorica (Dawson); Margreth, p. 123, pl. 36, fig. 4.
2014 Nonionellina labradorica (Dawson); Panchang and Nigam, pl. 34, figs. 8a-c, 9a-c.
Description. Test small, symmetrical on both sides, wall calcareous, finely perforate, surface smooth, granular pustules along the sutures, apertural side and the umbilicus; chambers few, rapidly increasing in size, planispirally arranged, coiling involute; periphery subacutely rounded, sutures distinct, curved and very slightly depressed; aperture an interiomarginal, narrow slit at the base of a broadly triangular apertural face.
Genus NONIONOIDES Saidova, 1975
Nonionoides elongatum (d'Orbigny, 1852)
Figure 7.16
1852 Nonionina elongata d’Orbigny, p. 3, fig. 4.
1999 Nonion elongatum (d’Orbigny); Nigam and Khare, p. 298, pl. 6, fig. 6.
2014 Nonion elongatum (d’Orbigny); Panchang and Nigam, pl. 39, fig. 12a-c.
Description. Test elongate, biumbilicate, wall finely perforate, surface smooth, umbilical portion depressed, papillated, granular pustules to some extent along the sutures and the apertural face; chambers distinct, many, increasing rapidly in size as added, coiling involute; periphery broadly rounded, sutures distinct, slightly depressed and curved; aperture a interiomarginal, narrow, low arch opening on the apertural face.
Nonionoides grateloupii (d'Orbigny, 1839a)
Figure 7.17
1839a Nonionina grateloupi d’Orbigny, p. 46, pl. 6, figs. 6-7.
2006 Nonionoides grateloupi (d’Orbigny); Oflaz, p. 228, pl. 9, figs. 7-8.
2012 Nonionoides grateloupi (d’Orbigny); Debenay, p. 227.
2014 Nonionoides grateloupi (d’Orbigny); Panchang and Nigam, pl. 36, fig. 11a-c.
Description. Test elongate, symmetrical on both sides, compressed, wall calcareous, finely perforate, surface smooth, depressed umbilical area filled with granular pustules, apertural face pustular; chambers narrow, trochospirally arranged, final chambers rapidly increasing in length, coiling involute; periphery rounded, sutures distinct, slightly depressed; aperture interiomarginal, an equatorial slit.
Nonionoides turgida (Williamson, 1858)
Figure 7.18
1858 Rotalina turgida Williamson, p. 50, pl. 4, figs. 95-97.
2003 Nonionella turgida (Williamson); Murray, p. 25, fig. 9.4-5.
2012 Nonionella turgida (Williamson); Milker and Schmiedl, p. 113, fig. 26.1-6.
Description. Test elongate, compressed, wall calcareous, finely perforate, surface smooth, apertural side with distinct flap-like projection; chambers distinct, long and narrow, chamber arrangement trochospiral, rapidly increasing in size; periphery rounded, slightly lobulate, sutures distinct, not much depressed, aperture interiomarginal, an equatorial slit-like opening.
Nonionoides boueana (d'Orbigny, 1846)
Figure 7.19
1846 Nonionina boueana d’Orbigny, p. 108, pl. 5, figs. 11-12.
2014 Nonionoides boueanum (d’Orbigny); Panchang and Nigam, pl. 34, fig. 10a-c.
Description. Test broad, outline subcircular, wall calcareous, finely perforate, surface smooth, umbilical region depressed and filled with pustules; chambers many, inflated, gradually increasing in size; planispirally arranged, coiling involute; periphery slightly lobulate, sutures distinct and curved backwards, slightly depressed, limbate; aperture interiomarginal, a small, round opening at the basal end of the apertural side.
Genus HAYNESINA Banner and Culver, 1978
Haynesina depressula (Walker and Jacob, 1798)
Figure 7.20
1798 Nautilus depressulus Walker and Jacob, p. 641, pl. 14, fig. 33.
2006 Nonion depressulum (Walker and Jacob); Oflaz, p. 227, pl. 9, figs. 5-6 (not pl. 7, figs. 4-5).
2012 Haynesina depressula (Walker and Jacob); Debenay, p. 222.
2012 Nonion depressulum (Walker and Jacob); Milker and Schmiedl, p. 112, fig. 25.17-18.
Description. Test circular, biumbilicate, wall calcareous, finely perforate, surface smooth; chambers slightly inflated, planispirally arranged, gradually increasing in size, coiling involute; umbilici narrow and depressed with pustules; periphery subrounded, slightly lobulate, sutures radial, curved backwards, cut near umbilicus; aperture interiomarginal, a low arched opening.
Superfamily DISCORBOIDEA Ehrenberg, 1838
Family BAGGINIDAE Cushman, 1927
Subfamily BAGGININAE Cushman, 1927
Genus BAGGINA Cushman, 1926
Baggina indica (Cushman, 1921)
Figure 8.1-2
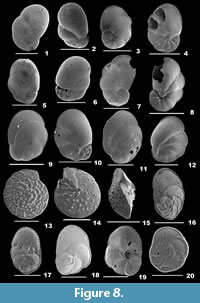 1921 Pulvinulina indica Cushman, p. 332, pl. 106, fig. 6.
1921 Pulvinulina indica Cushman, p. 332, pl. 106, fig. 6.
1999 Cancris indicus (Cushman); Nigam and Khare, p. 296, pl. 5, fig. 18.
2001 Baggina indica (Cushman); Szarek, p. 132, pl. 19, figs. 6-7.
2014 Baggina indica (Cushman); Panchang and Nigam, pl. 28, fig. 26a-c.
Description. Test oblong, wall calcareous, well perforated, umbilical region imperforate, surface smooth; chambers inflated, subglobular, rapidly increasing in size, final chamber much more inflated, chamber arrangement trochospiral; periphery broadly rounded, margin lobulated, sutures slightly depressed and curved on the spiral side, radial and much depressed on the umbilical side; aperture interiomarginal, a broad opening on the umbilical side at the base of the apertural face.
Family CANCRISIDAE Chapman, Parr and Collins, 1934
Genus CANCRIS de Montfort, 1808
Cancris auriculus (Fichtel and Moll, 1798)
Figure 8.3-4
1798 Nautilus auricula Fichtel and Moll, p. 108, pl. 20, figs. a-c.
2001 Cancris auriculus (Fichtel and Moll); Szarek, p. 132, pl. 19, figs. 1-3.
2003 Cancris auricula (Fichtel and Moll); Murray, p. 19, fig. 6.6-7.
2010 Cancris auriculus (Fichtel and Moll); Margreth, p. 28, pl. 29, fig. 10a-c.
2012 Cancris auriculus (Fichtel and Moll); Debenay, p. 189.
2012 Cancris auriculus (Fichtel and Moll); Milker and Schmiedl, p. 93, fig. 21.14-15.
2014 Cancris auriculus (Fichtel and Moll); Panchang and Nigam, pl. 28, fig. 27a-c.
Description. Test biconvex, outline auriculate, cross section lenticular, wall calcareous, perforate, umbilical area imperforate; surface smooth; chambers rapidly increasing in size, trochospirally arranged, coiling evolute on the spiral side, involute on the umbilical side; peripheral margin carinate and acute, sutures curved and slightly depressed on the spiral side, radial and much depressed on the umbilical side; aperture an interiomarginal, umbilical, slit-like opening with a broad apertural flap.
Cancris bubnanensis McCulloch, 1977
Figure 8.5-6
1977 Baggina bubnanensis McCulloch, p. 342, pl. 137, fig. 2a-c.
2012 Baggina bubnanensis McCulloch; Debenay, p. 187.
2014 Baggina bubnanensis McCulloch; Panchang and Nigam, pl. 28, fig. 25a-c.
Description. Test subovate, wall calcareous, well perforated, umbilical side above the aperture imperforate, surface glassy, smooth except for prominent ridges on the apertural side beneath the aperture; chambers inflated, final chamber very much inflated, rapidly increasing in size as added, chamber arrangement low trochospiral; periphery subacutely rounded, margin lobulated, sutures curved and depressed on the spiral side, radial and very depressed on the umbilical side; aperture interiomarginal, umbilical, a wide opening above the ridges, at the basal end of the apertural side.
Cancris carinata (Millet, 1904)
Figure 8.7-8
1904 Pulvinulina oblonga (Williamson) var. carinata Millett, p. 498, pl. 10, fig. 3.
2001 Cancris carinatus (Millet); Szarek, p. 133, pl. 19, fig. 5.
2014 Cancris carinata (Millet); Panchang and Nigam, pl. 28, figs. 28a-c, 29a-c.
Description. Test elongate, outline auriculate, cross section lenticular, wall calcareous, perforate, umbilical side imperforate, surface smooth; chambers many, rapidly increasing in size, arranged trochospirally, spiral side evolute, umbilical side involute; periphery acute and carinate, sutures curved and slightly depressed on the spiral side, radial and much depressed on the umbilical side; aperture an interiomarginal, umbilical, opening with a distinct, projecting apertural flap.
Cancris communis Cushman and Todd, 1942
Figure 8.9-10
1942 Cancris sagra (d’Orbigny) var. communis Cushman and Todd, p. 79, pl. 19, figs. 8-11; pl. 20, fig. 1.
2014 Cancris communis Cushman and Todd; Panchang and Nigam, pl. 28, fig. 30a-c.
Description. Test broad, outline auriculate, cross section lenticular, wall calcareous, perforate, umbilical side above the aperture imperforate, surface smooth; chambers flat on spiral side, inflated on the umbilical side, rapidly increasing in size, arranged trochospirally, spiral side evolute, umbilical side involute; periphery acute and carinate, sutures well arched, slightly depressed and limbate in the final chambers on the spiral side, radial and depressed on the umbilical side; aperture an interiomarginal, umbilical, broad opening.
Cancris sagra (d’Orbigny, 1839a)
Figure 8.11-12
1839a Rotalina sagra d’Orbigny, p. 77, pl. 5, figs. 13-15.
2012 Cancris sagrum (d’Orbigny); Debenay, p. 189.
2014 Cancris sagra (d’Orbigny); Panchang and Nigam, pl. 29, fig. 8a-c.
Description. Test broad, outline subelliptical, spiral side flat to gently convex, umbilical side convex, wall transparent to opaque, perforate, a selected portion above the aperture imperforate, surface smooth; chambers roughly triangular, inflated in the umbilical side, arched in the spiral side, final chambers rapidly increasing in size as added, chamber arrangement trochospiral; peripheral margin carinate, sutures curved and slightly depressed on the dorsal side, depressed and radial on the ventral side; aperture interiomarginal, umbilical, a narrow slit, with a flap-like lamina.
Family DISCORBIDAE Ehrenberg, 1838
Genus NEOEPONIDES Reiss, 1960
Neoeponides margaritifer (Brady, 1881)
Figure 8.13-15
1881 Truncatulina margaritifera Brady, p. 66.
2001 Heterolepa margaritifera (Brady); Szarek, p. 145, pl. 25, figs. 4-7.
2012 Heterolepa margaritifera (Brady); Debenay, p. 199.
Description. Test biconvex, wall calcareous, well perforated, surface richly ornamented with pustules; chambers many, trochospirally arranged, coiling evolute on the spiral side, involute on the umbilical side; periphery rounded, margin subcarinate and very slightly lobulate, sutures distinct on either side, marked with pustules; aperture interiomarginal, umbilical, small slit, bordered by a lip.
Family EPONIDIDAE Hofker, 1951
Subfamily EPONIDINAE Hofker, 1951
Genus EPONIDES de Montfort, 1808
Eponides cribrorepandus (Asano and Uchio, 1951)
Figure 8.16-17
1951 Poroeponides cribrorepandus Asano and Uchio in Asano, p. 18, text-figs. 134-135.
2001 Eponides cribrorepandus (Asano and Uchio); Szarek, p. 133, pl. 19, fig. 12.
2014 Eponides cribrorepandus (Asano and Uchio); Panchang and Nigam, pl. 29, fig. 11a-c.
Description. Test elongate, spiral side nearly flat, umbilical side more convex; wall calcareous, finely perforate, surface smooth; chambers rapidly increasing in size, final chambers more elongate, arranged trochospirally; periphery subangular, carinate with imperforate keel, sutures limbate and slightly arched on the spiral side, curved and slightly depressed on the umbilical side, primary aperture single, interiomarginal, low arch opening, secondary apertures few, areal.
Eponides repandus (Fichtel and Moll, 1798)
Figure 8.18-19
1798 Nautilus repandus Fichtel and Moll, p. 35, pl. 3, figs. a-d.
2001 Eponides repandus (Fichtel and Moll); Szarek, p. 133, pl. 19, figs. 9-11.
2003 Eponides repandus (Fichtel and Moll); Javaux and Scott, p. 14, fig. 3.1-2.
2012 Eponides repandus (Fichtel and Moll); Debenay, p. 196.
2014 Eponides repandus (Fichtel and Moll); Panchang and Nigam, pl. 29, fig. 12a-c.
Description. Test subrounded, spiral side slightly convex, umbilical side more convex; wall calcareous, finely perforate, surface smooth; chambers rapidly increasing in size, arranged trochospirally; periphery subangular, carinate with imperforate keel, sutures limbate and curved on the spiral side, curved and slightly depressed on the umbilical side, primary aperture interiomarginal, broad, low arch opening, secondary apertures areal on the ventral side.
Genus POROEPONIDES Cushman, 1944
Poroeponides lateralis (Terquem, 1878)
Figure 9.1-2
 1878 Rosalina lateralis Terquem, p. 25, pl. 2, fig. 11.
1878 Rosalina lateralis Terquem, p. 25, pl. 2, fig. 11.
2007 Poroeponides lateralis (Terquem); Talib and Farroqui, p. 25, pl. 1, fig. 18.
2012 Poroeponides lateralis (Terquem); Debenay, p. 210.
Description. Test biconvex, wall calcareous, finely perforate, surface smooth; chambers rapidly increasing in size, trochospirally arranged, coiling evolute on the dorsal side; periphery with a distinct keel, sutures curved, elevated, limbate on the spiral side, radial, slightly depressed on the umbilical side; aperture interiomarginal, slit-like opening, bordered by a rim, secondary apertures areal and round openings on the apertural face.
Family ROSALINIDAE Reiss, 1963
Genus ROSALINA d’Orbigny, 1826
Rosalina bradyi (Cushman, 1915)
Figure 9.3-4
1915 Discorbina globularis (d’Orbigny) var. bradyi Cushman, p. 12, pl. 8, fig. 1a-c.
2006 Rosalina bradyi (Cushman); Oflaz, p. 218, pl. 7, figs. 8-9.
2010 Rosalina bradyi (Cushman); Margreth, p. 120, pl. 31, fig. 5a-c.
2012 Rosalina bradyi (Cushman); Debenay, p. 211.
2012 Rosalina bradyi (Cushman); Milker and Schmiedl, p. 98, fig. 22.11-14.
2014 Rosalina bradyi (Cushman); Panchang and Nigam, pl. 30, fig. 5a-c.
Description. Test small, spiral side convex, umbilical side nearly flattened, wall calcareous, coarsely perforated on the spiral side, smooth and imperforate on the umbilical side; chambers on the spiral side inflated and convex, very slightly inflated chambers on the umbilical side, gradually increasing in size, arrangement trochospiral; periphery rounded, sutures curved backwards and limbate; aperture interiomarginal, extraumbilical arch on the umbilical side, bordered by a narrow, thick lip.
Rosalina floridana (Cushman, 1922a)
Figure 9.5-6
1922a Discorbis floridana Cushman, p. 39, pl. 5, figs. 11-12.
2012 Rosalina floridana (Cushman); Debenay, p. 211.
Description. Test subcircular, spiral side convex, umbilical side slightly convex, wall calcareous, coarsely perforate, umbilical region concave and imperforate; chambers on the spiral side inflated, subglobular and convex, very slightly inflated and triangular chambers on the umbilical side, gradually increasing in size, arrangement trochospiral; periphery rounded, margin subacute, slightly lobulate, sutures curved backwards and limbate on the spiral side, radial and depressed on the umbilical side; aperture interiomarginal, elongate arch on the umbilical side, bordered by a thin lip.
Rosalina globularis d'Orbigny, 1826
Figure 9.7-8
1826 Rosalina globularis d’Orbigny, p. 271, pl. 13, figs. 1-4.
2001 Rosalina globularis d’Orbigny; Szarek, p. 135, pl. 20, fig. 10.
2003 Rosalina globularis d’Orbigny; Javaux and Scott, p. 20, fig. 5.3-4.
2010 Rosalina globularis d’Orbigny; Margreth, p. 120, pl. 31, fig. 4a-c.
2012 Rosalina globularis d’Orbigny; Debenay, p. 211.
2012 Rosalina globularis d’Orbigny; Milker and Schmiedl, p. 98, fig. 22.15-16.
Description. Test subrounded, biconvex, wall calcareous, perforate, umbilical region concave and imperforate, surface smooth; chambers subglobular and convex on the spiral side, slightly inflated and triangular chambers on the umbilical side, gradually increasing in size, final chamber more inflated, arrangement trochospiral; periphery rounded and lobulate, sutures curved backwards, limbate and slightly depressed on the spiral side, radial and depressed on the umbilical side; aperture interiomarginal, a low arch opening on the umbilical side, almost covered by a thin, narrow lamina.
Rosalina macropora (Hofker, 1951)
Figure 8.20
1951 Discopulvinulina macropora Hofker, p. 460, figs. 312-313.
2012 Rosalina macropora (Hofker); Milker and Schmiedl, p. 99, fig. 22.17-18.
Description. Test subrounded, spiral side convex, umbilical side flattened, umbilical region depressed, wall calcareous, perforate, surface smooth; chambers convex on the spiral side, subtriangular on the umbilical side, gradually increasing in size, arrangement trochospiral; periphery rounded with a thick imperforate keel, sutures curved backwards, limbate and slightly depressed on the spiral side, radial on the umbilical side; aperture interiomarginal, extraumbilical elongate slit on the umbilical side.
Superfamily DISCORBINELLOIDEA Sigal, 1952
Family DISCORBINELLIDAE Sigal, 1952
Subfamily DISCORBINELLINAE Sigal, 1952
Genus DISCORBINELLA Cushman and Martin, 1935
Discorbinella bertheloti (d’Orbigny, 1839b)
Figure 9.9-10
1839b Rosalina bertheloti d’Orbigny, p. 135, pl. 1, figs. 28-30.
2001 Discorbinella bertheloti (d’Orbigny); Szarek, p. 137, pl. 21, figs. 1-3.
2006 Discorbinella bertheloti (d’Orbigny); Oflaz, p. 223, pl. 8, figs. 5-6.
2012 Discorbinella bertheloti (d’Orbigny); Debenay, p. 194.
2012 Discorbinella bertheloti (d’Orbigny); Milker and Schmiedl, p. 104, fig. 23.29-30.
2014 Discorbinella bertheloti (d’Orbigny); Panchang and Nigam, pl. 31, fig. 6a-c.
Description. Test compressed, outline circular, spiral side convex, umbilical side flat, wall, calcareous, perforate, surface smooth; chambers many, inflated, arranged trochospirally; periphery rounded, lobulated, sutures distinct and depressed, curved backwards on the spiral side; aperture interiomarginal, a low opening, bordered by a lip.
Superfamily BULIMINOIDEA Jones, 1875
Family SIPHOGENERINOIDIDAE Saidova, 1981
Subfamily SIPHOGENERINOIDINAE Saidova, 1981
Genus STILOSTOMELLOIDES Ujiie and Rifardi, 1993
Stilostomelloides virgula (Brady, 1879b)
Figure 9.11
1879b Sagrina virgula Brady, p. 275, pl. 8, figs. 19-21.
2001 Allassoida virgula (Brady); Szarek, p. 127, pl. 17, fig. 14.
2012 Allassoida virgula (Brady); Debenay, p. 197.
2014 Siphogenerina virgula (Brady); Panchang and Nigam, pl. 25, fig. 32.
Description. Test long, straight, aboral end bluntly tapering, wall calcareous, perforate, surface ornamented with short, sharp spines; chambers rapidly increasing in size, chamber arrangement trochospiral initially, later chambers uniserially arranged, globular, final chamber pyriform; aperture terminal, broadly round, slightly produced, bordered by a distinctly reversed lip.
Subfamily TUBULOGENERININAE Saidova, 1981
Genus RECTUVIGERINA Mathews, 1945
Rectuvigerina phlegeri Le Calvez, 1959
Figure 9.12
1959 Rectuvigerina phlegeri Le Calvez in Berthois and Le Calvez, p. 363, pl. 1, fig. 11.
2002 Rectuvigerina phlegeri Le Calvez; Kaminski, Aksu, Box, Hiscott, Filipescu and Al-Salameen, p. 174, pl. 3, fig. 9.
2006 Rectuvigerina phlegeri Le Calvez; Oflaz, p. 208, pl. 6, fig. 3.
2009 Rectuvigerina phlegeri Le Calvez; Milker, Schmiedl, Betzler, Römer, Jaramillo-Vogel and Siccha, p. 216, pl. 2, fig. 19.
2012 Rectuvigerina phlegeri Le Calvez; Milker and Schmiedl, p. 86.
Description. Test elongate, wall calcareous, perforate, surface of chambers ornamented with long costae; early chambers triserially arranged, later biserial and finally unserially arranged; periphery subrounded, sutures initially inclined, later horizontal and depressed; aperture terminal, rounded on a short neck, bordered by a rim.
Family UVIGERINIDAE Haeckel, 1894
Subfamily UVIGERININAE Haeckel, 1894
Genus UVIGERINA d'Orbigny, 1826
Uvigerina asperula Cžjžek, 1848
Figure 9.13
1848 Uvigerina asperula Cžjžek, p. 146, pl. 13, figs. 14-15.
2014 Euuvigerina asperula Cžjžek; Panchang and Nigam, pl. 26, fig. 30.
2015 Euuvigerina asperula Cžjžek; Hanagata and Nobuhara, p. 86, fig. 26.21-22.
Description. Test long, distal end bluntly pointed, wall calcareous, perforate, surface ornamented with fine spines; chambers subglobular, biserially arranged, rapidly increasing in size; periphery lobulated, sutures depressed and curved; aperture terminal, round on a cylindrical neck, bordered by a thin rim.
Uvigerina akitaensis Asano, 1950
Figure 9.14
1950 Uvigerina akitaensis Asano, p. 14, pl. 14, figs. 60-62.
2014 Uvigerina akitaensis Asano; Panchang and Nigam, pl. 27, fig. 10.
Description. Test elongate, slender, tapering aboral end, wall calcareous, perforate, surface of chambers ornamented with long costae; chambers biserially and later uniserially arranged; periphery subrounded and carinate, sutures horizontal and depressed; aperture terminal, rounded on a short neck, bordered by an everted, phialine lip.
Uvigerina auberiana d'Orbigny, 1839b
Figure 9.15
1839b Uvigerina auberiana d’Orbigny, p. 106, pl. 2, figs. 23-24.
2001 Uvigerina ex gr. auberiana d’Orbigny; Szarek, p. 130, pl. 18, figs. 11-12.
2010 Uvigerina auberiana d’Orbigny; Margreth, p. 117, pl. 29, fig. 3a-b.
2014 Euuvigerina auberiana d’Orbigny; Panchang and Nigam, pl. 24, figs. 32-33.
Description. Test robust, aboral end bluntly pointed, wall calcareous, perforate, surface ornamented with minute spines; chambers subglobular, somewhat compressed, earlier chambers triserially arranged, later biserially and uniserially arranged, rapidly increasing in size; periphery slightly lobulated, sutures depressed and curved; aperture terminal, round on a slightly long neck, bordered by an everted lip.
Uvigerina ex gr. semiornata d’Orbigny, 1846
Figure 9.16
1846 Uvigerina semiornata d’Orbigny, p. 189, pl. 11, figs. 23-24.
2012 Uvigerina ex gr. semiornata d’Orbigny; Nisha and Singh, p. 788, pl. 2, fig. 41.
Description. Test long, wall calcareous, perforate, surface of chambers ornamented with longitudinal costae; earlier chambers triserially and later biserially arranged; periphery carinate, sutures nearly horizontal, curved in later chambers, depressed; aperture terminal, round on a short neck, bordered by an everted lip.
Uvigerina mediterranea Hofker, 1932
Figure 9.17
1932 Uvigerina mediterrenea Hofker, p.118, text-fig. 32a-g.
2002 Uvigerina mediterranea Hofker; Kaminski, Aksu, Box, Hiscott, Filipescu and Al-Salameen, p. 174, pl. 3, fig. 10.
2010 Uvigerina mediterranea Hofker; Margreth, p. 117, pl. 29, fig. 1a-b.
2012 Uvigerina mediterranea Hofker; Milker and Schmiedl, p. 90, fig. 20.28.
Description. Test ovate, broad than long, wall calcareous, perforate, surface ornamented with long costae; chambers inflated, gradually increase in size, triserially arranged; periphery carinate, sutures distinct, slightly inclined and depressed; aperture terminal, circular opening on a short neck, bordered by a lip.
Uvigerina peregrina Cushman, 1923
Figure 9.18
1923 Uvigerina peregrina Cushman, p. 166, pl. 42, figs. 7-10.
2001 Uvigerina peregrina Cushman; Szarek, p. 130, pl. 18, fig. 13.
2010 Uvigerina peregrina Cushman; Margreth, p. 117, pl. 29, fig. 2a-b.
2012 Uvigerina cf. U. peregrina Cushman; Debenay, p. 184.
2012 Uvigerina peregrina Cushman; Milker and Schmiedl, p. 90, fig. 20.29.
2014 Uvigerina peregrina Cushman; Panchang and Nigam, pl. 27, fig. 17.
Description. Test subovate, slightly long, wall calcareous, perforate, surface ornamented with long costae and pustules; chambers inflated, gradually increase in size, chamber arrangement triserial; periphery carinate, sutures distinct, slightly oblique and depressed; aperture terminal, round on a short neck, bordered by a lip.
Uvigerina pygmaea d'Orbigny, 1826
Figure 9.19
1826 Uvigerina pygmea d’Orbigny, p. 269, pl. 12, figs. 8-9.
2010 Uvigerina pygmea d’Orbigny; Margreth, p. 117, pl. 29, fig. 5a-b.
2014 Uvigerina pygmea d’Orbigny; Panchang and Nigam, pl. 27, fig. 18.
Description. Test ovate, wall calcareous, perforate, surface ornamented with long costae; chambers inflated, rapidly increasing in size, triserially arranged; periphery carinate, sutures distinct and depressed; aperture terminal, circular opening on a short neck, bordered by a lip.
Genus SIPHOUVIGERINA Parr, 1950
Siphouvigerina porrecta (Brady, 1879b)
Figure 9.20
1879b Uvigerina porrecta Brady, p. 274, pl. 8, figs. 15-16.
2012 Neouvigerina porrecta (Brady); Debenay, p. 181.
2014 Neouvigerina porrecta (Brady); Panchang and Nigam, pl. 27, fig. 5.
Description. Test long, fusiform, wall calcareous, perforate, surface ornamented with prominent long costae; chambers arranged linearly, sutures distinct; aperture terminal, round at the end of a long neck, bordered by an everted lip.
Siphouvigerina proboscidea (Schwager, 1866)
Figure 9.21-22
1866 Uvigerina proboscidea Schwager, p. 250, pl. 7, fig. 96.
2001 Neouvigerina proboscidea (Schwager); Szarek, p. 130, pl. 18, fig. 10.
2012 Neouvigerina proboscidea (Schwager); Debenay, p. 181.
2014 Euuvigerina proboscidea (Schwager); Panchang and Nigam, pl. 27, figs. 1-2.
Description. Test long, wall calcareous, perforate, surface ornamented with short spines; chambers subglobular, initially triserially arranged, later biserially and uniserially arranged, periphery lobulated, sutures distinct, curved and depressed; aperture terminal, round at the end of a long, cylindrical neck, bordered by an everted phialine lip.
Subfamily ANGULOGERININAE Galloway, 1933
Genus TRIFARINA Cushman, 1923
Trifarina angulosa (Williamson, 1858)
Figure 9.23
1858 Uvigerina angulosa Williamson, p. 67, pl. 5, fig. 140.
2003 Trifarina angulosa (Williamson); Murray, p. 26, fig. 10.5.
2010 Angulogerina angulosa (Williamson); Margreth, p. 118, pl. 29, fig. 6a-b.
2012 Trifarina angulosa (Williamson); Debenay, p. 182.
2012 Angulogerina angulosa (Williamson); Milker and Schmiedl, p. 90, fig. 21.2-4.
2014 Angulogerina angulosa (Williamson); Panchang and Nigam, pl. 27, fig. 22.
Description. Test elongate, cross section triangular, wall calcareous, perforate, surface ornamented with thick, long striae; initial chamber arrangement triserial, later biserially arranged; periphery carinate, sutures limbate and depressed; aperture terminal, ovate, at the end of a short neck, bordered by a rim.
Family BULIMINIDAE Jones, 1875
Genus BULIMINA d’Orbigny, 1826
Bulimina aculeata d'Orbigny, 1826
Figure 10.1
 1826 Bulimina aculeata d’Orbigny, p. 269, fig. 7.
1826 Bulimina aculeata d’Orbigny, p. 269, fig. 7.
2001 Bulimina aculeata d’Orbigny; Szarek, p. 128, pl. 17, fig. 19.
2002 Bulimina aculeata d’Orbigny; Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 174, pl. 3, fig. 3.
2010 Bulimina aculeata d’Orbigny; Margreth, p. 116, pl. 27, fig. 8.
2012 Bulimina aculeata d’Orbigny; Milker and Schmiedl, p. 87, fig. 20.19.
Description. Test small, lateral view ovate, tapering distal end, wall calcareous, finely perforate, surface smooth, chamber ends with pseudospines; chambers inflated, rapidly increasing in size, triserially arranged; periphery rounded, sutures distinct, oblique and depressed; aperture terminal, subcircular, loop-like opening, bordered by a thick rim.
Bulimina marginata d'Orbigny, 1826
Figure 10.2
1826 Bulimina marginata d’Orbigny, p. 269, pl. 12, figs. 10-12.
2001 Bulimina marginata d’Orbigny; Szarek, p. 129, pl. 18, figs. 2-5.
2003 Bulimina marginata d’Orbigny; Murray, p. 27, fig. 10.7-8.
2006 Bulimina marginata d’Orbigny; Oflaz, p. 210 pl. 6, figs. 5-7.
2010 Bulimina marginata d’Orbigny; Margreth, p. 116, pl. 27, fig. 9a-b.
2012 Bulimina marginata d’Orbigny; Debenay, p. 179.
2012 Bulimina marginata d’Orbigny; Milker and Schmiedl, p. 88, fig. 20.23.
Description. Test small, outline triangular, basal end tapering sharply, wall calcareous, finely perforate, surface smooth, basal and chamber ends with short, blunt pseudospines; chambers inflated, rapidly increasing in size, triserially arranged; aperture terminal, loop-shaped opening, bordered by thick rim, with a distinct toothplate.
Bulimina striata d’Orbigny in Guérin-Méneville, 1843
Figure 10.3-4
1843 Bulimina striata d’Orbigny in Guérin Méneville, p. 9, pl. 2, fig. 16.
2001 Bulimina striata d’Orbigny; Szarek, p. 129, pl. 18, fig. 6.
2010 Bulimina striata d’Orbigny; Margreth, p. 116, pl. 27, fig. 10a-c.
2012 Bulimina striata d’Orbigny; Debenay, p. 180.
Description. Test short, outline subovate, aboral end sharply pointed, wall calcareous, finely perforate, surface smooth in the upper half, ornamented with many, long costae hanging as projections in the lower portion of the test; chambers rapidly increasing in size, final chambers more inflated, triserially arranged; sutures distinct and depressed; aperture terminal, obliquely placed, slightly wide, slit-like opening, bordered by a weakly everted lip.
Genus GLOBOBULIMINA Cushman, 1927
Globobulimina turgida (Bailey, 1851)
Figure 10.5
1851 Bulimina turgida Bailey, p. 12, figs. 28-31.
2010 Globobulimina turgida (Bailey); Margreth, p. 117, pl. 28, figs. 1a-c.
Description. Test ovate, aboral end round, broad in the middle, apertural end narrow, wall smooth, finely perforate, surface smooth; chambers distinct, inflated; periphery rounded, sutures distinct and depressed; aperture terminal, a subovate opening with a slightly raised, broad tooth, bordered by a thin lip.
Family REUSSELLIDAE Cushman, 1933
Genus REUSSELLA Galloway, 1933
Reussella haizumensis Asano, 1950
Figure 10.6-8
1950 Reussella haizumensis Asano, p. 12, fig. 51.
2010 Reussella haizumensis Asano; Kathal and Singh, p. 70, pl. 1, fig. 2a-b.
Description. Test pyramidal, cross section triangular, aboral end pointed, wall calcareous, perforate, surface ornamented with regular rows of slightly produced, small, round pores; chambers many, triserially arranged; periphery acute and carinate, sutures slightly raised and curved; aperture terminal, triangular opening, bordered by a distinct, imperforate lip, secondary openings in a large perforated toothplate.
Reussella spinulosa (Reuss, 1850)
Figure 10.9
1850 Verneuilina spinulosa Reuss, p. 374, pl. 47, fig. 12.
2001 Reussella spinulosa (Reuss); Szarek, p. 131, pl. 18, figs. 18-19.
2006 Reussella spinulosa (Reuss); Oflaz, p. 212, pl. 6, fig. 10.
2012 Reussella spinulosa (Reuss); Debenay, p. 182.
2012 Reussella spinulosa (Reuss); Milker and Schmiedl, p. 92, fig. 21.6-7.
2014 Reussella spinulosa (Reuss); Panchang and Nigam, pl. 28, figs. 8-9.
Description. Test pyramid-shaped, triangular, wall calcareous, coarsely perforate, surface ornamented with short spines at chamber margins; chambers triserially arranged, gradually increase in size; periphery acute and strongly carinate, sutures distinct, curved and thick; aperture interiomarginal, slightly concave opening, bordered by a thick, imperforate lip, with a toothplate.
Genus VALVOBIFARINA Hofker, 1951
Valvobifarina mackinnonii (Millett, 1900a)
Figure 10.10
1900a Bifarina mackinnonii Millet, p. 281, pl. 2, fig. 15a-b.
1951 Valvobifarina mackinnoni (Millett); Hofker, p. 40, figs. 16-17.
1994 Valvobifarina mackinnoni (Millett); Loeblich and Tappan, pl. 254, figs. 9-12.
Description. Test long, wall calcareous, surface ornamented with slightly produced circular pores and acicular spines in the final chambers; chambers initially triserially arranged, later biserially and uniserially arranged; periphery angular, sutures distinct; oblique in later chambers; aperture a broad, horizontal slit-like opening, bordered by a thick lip.
Superfamily BOLIVINITOIDEA Cushman, 1927
Family BOLIVINITIDAE Cushman, 1927
Subfamily BOLIVINITINAE Cushman, 1927
Genus BOLIVINA d’Orbigny, 1839
Bolivina obscura He, Hu, and Wang 1965
Figure 10.11
1965 Bolivina obscura He, Hu, and Wang, pl. 7, figs. 1-3.
2014 Bolivina obscura He, Hu, and Wang; Panchang and Nigam, pl. 23, fig. 5.
Description. Test broad, compressed laterally, outline V-shaped, wall calcareous, finely perforate; chambers rapidly increasing in size, final chambers more inflated, biserially arranged; periphery acute and slightly lobulate, sutures distinct, depressed; aperture low, terminal opening.
Bolivina cistina Cushman, 1936
Figure 10.12
1936 Bolivina cistina Cushman, p. 55, pl. 8, fig. 4.
2012 Bolivina cistina Cushman; Milker and Schmiedl, p. 80, fig. 19.19-20.
Description. Test triangular, compressed, wall calcareous, perforate, surface ornamented with intermittent corrugations; chambers rapidly increasing in size, biserially arranged; periphery truncate; aperture terminal, subovate opening, bordered by a lip.
Bolivina dilatata Reuss, 1850
Figure 10.13
1850 Bolivina dilatata Reuss, p. 381, pl. 48, fig. 15a-c.
2002 Brizalina dilatata Reuss; Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 173, pl. 2, fig. 13.
2010 Brizalina dilatata Reuss; Margreth, p. 113, pl. 24, fig. 2a-c.
2012 Brizalina dilatata Reuss; Milker and Schmiedl, p. 81, fig. 19.30.
2014 Bolivina dilatata Reuss; Panchang and Nigam, pl. 23, figs. 27-28.
Description. Test subtriangular, compressed, wall calcareous, perforate, surface smooth; chambers gradually increasing in size, biserially arranged; periphery subacute, sutures inclined and slightly depressed; aperture terminal, an elongate slit-like opening, bordered by a lip, with a tooth.
Bolivina kuriani Seibold, 1975
Figure 10.14-15
1975 Bolivina kuriani Seibold, p.185, text-fig. 2a-b.
1999 Bolivina kuriani Seibold; Nigam and Khare, p. 294, pl. 4, fig. 16a-b.
Description. Test long than broad, aboral end tapering, wall calcareous, perforate, surface smooth; chambers many, gradually increasing in size, biserially arranged; periphery lobulated, sutures distinct, curved and depressed; aperture terminal, a subovate opening, bordered by a lip.
Bolivina persiensis Lutze, 1974
Figure 10.16-17
1974 Bolivina persiensis Lutze, p. 25, pl. 5, fig. 81.
2014 Latibolivina persiensis Lutze; Panchang and Nigam, pl. 24, figs. 3-4.
Description. Test robust, lateral view ovate, wall calcareous, surface ornamented with intermittent rigdes; chambers gradually increasing in size; periphery broadly rounded, sutures obscured; aperture terminal slit, bordered by a thin lip.
Bolivina pusilla Schwager, 1866
Figure 10.18-19
1866 Bolivina pusilla Schwager, p. 254, pl. 7, fig. 101.
2001 Brizalina pusilla Schwager; Szarek, p. 124, pl. 16, figs. 8-9.
Description. Test subtriangular, compressed, wall calcareous, perforate, surface smooth; chambers gradually increasing in size, biserially arranged; periphery carinate and subacute, sutures distinct and limbate; aperture terminal, slit-like opening, bordered by a lip.
Bolivina spathulata (Williamson, 1858)
Figure 10.20
1858 Textularia variabilis var. spathulata Williamson, p. 76, pl. 6, figs. 164-165.
2002 Brizalina spathulata (Williamson); Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 172, pl. 2, fig. 9.
2003 Brizalina spathulata (Williamson); Murray, p. 19, fig. 6.3.
2012 Brizalina spathulata (Williamson); Milker and Schmiedl, p. 82, fig. 20.1-2.
2014 Brizalina spathulata (Williamson); Panchang and Nigam, pl. 23, figs. 42-43.
Description. Test broad, elongate and compressed, wall calcareous, perforate, more at chamber margins, surface smooth; chambers gradually increase in size, biserially arranged; periphery subacutely rounded, sutures inclined, slightly depressed in the final chambers; aperture elongate, slit-like opening, bordered by a lip.
Bolivina striatula Cushman, 1922a
Figure 10.21
1922a Bolivina striatula Cushman, p. 27, pl. 3, fig. 10.
2002 Brizalina striatula (Cushman); Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 172, pl. 2, fig. 10.
2006 Brizalina striatula (Cushman); Oflaz, p. 203, pl. 5, fig. 14.
2010 Bolivinellina striatula (Cushman); Margreth, p. 114, pl. 24, fig. 5a-c.
2012 Brizalina striatula (Cushman); Milker and Schmiedl, p. 82, fig. 20.3.
2014 Brizalina striatula (Cushman); Panchang and Nigam, pl. 23, fig. 44.
Description. Test elongate, slightly compressed, wall calcareous, perforate, surface ornamented with long striae in the earlier chambers, final chambers smooth; chambers gradually increasing in size, slightly inflated, biserially arranged; periphery broadly rounded, sutures slightly depressed; aperture terminal, long, slit-like opening, bordered by a thick rim, with a tooth.
Genus PSEUDOBRIZALINA Zweig-Strykowski and Reiss, 1976
Pseudobrizalina lobata (Brady, 1881)
Figure 10.22
1884 Bolivina lobata Brady, p. 425, pl. 53, figs. 22-23.
2002 Bolivina lobata (Brady); Gandhi, Rajamanickam and Nigam, p. 56, pl. 2, fig. 5.
2012 Pseudobrizalina lobata (Brady); Debenay, p. 176.
2014 agrinella lobata (Brady); Panchang and Nigam, pl. 25, fig. 23.
Description. Test elongate, wall calcareous, surface smooth; chambers gradually increasing in size, biserially arranged; periphery lobulate, sutures prominent, elevated bars; aperture terminal, elongate opening bordered by a distinct lip.
Genus SAGRINELLA Saidova, 1975
Sagrinella durrandii (Millett, 1900b)
Figure 10.23
1900b Bolivina durrandii Millett, p. 544, pl. 4, fig. 7a-b.
2002 Bolivina durrandii (Millett); Gandhi, Rajamanickam and Nigam, p. 56, pl. 2, fig. 4.
2012 Sagrinella durrandii (Millett); Debenay, p. 178.
Description. Test elongate, compressed, wall calcareous, perforate, surface of chamber ends ornamented with intermittent spines; chambers very slightly inflated, rapidly increasing in length; periphery acute, carinate, sutures depressed; aperture terminal, an elongate slit-like opening, bordered by a reversed lip.
Genus SAIDOVINA Haman, 1984
Saidovina karreriana (Brady, 1881)
Figure 10.24
1881 Bolivina karreriana Brady, p. 28.
2015 Saidovina karreriana (Brady); Hanagata and Nobuhara, p. 81, fig. 25.22-23.
Description. Test elongate-ovate, wall calcareous, finely perforate, surface ornamented with long costae; chambers rapidly increasing in size; periphery strongly carinate, sutures obscured; aperture a terminal opening, bordered by a lip.
Subfamily FURSENKOININAE Loeblich and Tappan, 1961
Genus FURSENKOINA Loeblich and Tappan, 1961
Fursenkoina hadai Uchio, 1962
Figure 11.1
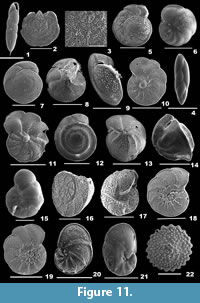 1962 Bolivina hadai Uchio, pp. 368-369, pl. 18, figs. 3-4.
1962 Bolivina hadai Uchio, pp. 368-369, pl. 18, figs. 3-4.
2014 Cassidellina hadai Uchio; Panchang and Nigam, pl. 25, fig. 7.
Description. Test elongate, wall calcareous, coarsely perforate, surface smooth, aboral end with a distinct basal spine; chambers of uniform size; periphery slightly lobulate, sutures limbate and oblique, slightly depressed; aperture terminal, subovate opening, bordered by a thick rim.
Fursenkoina pannikeri (Seibold, 1972)
Figure 11.2-3
1972 Cassidella pannikeri Seibold, p. 453.
1999 Cassidella pannikeri (Seibold); Nigam and Khare, p. 296, pl. 5, fig. 15.
Description. Test broad, very much compressed, outline subcircular, wall calcareous, surface ornamented with regular rows of slightly produced pores, apertural face with small pustules; chambers many, crescent-shaped; periphery rounded, sutures distinct, marked with pores.
Fursenkoina schreibersiana (Cžjžek, 1848)
Figure 11.4
1848 Virgulina schreibersiana Cžjžek, p. 147, pl. 13, figs. 18-21.
2006 Fursenkoina schreibersiana (Cžjžek); Oflaz, p. 212, pl. 6, figs. 11-12.
2012 Fursenkoina schreibersiana (Cžjžek); Debenay, p. 174.
Description. Test long than broad, very slightly compressed, wall calcareous, finely perforate, surface smooth; chambers slightly inflated, inclined, early chambers twisted; periphery broadly rounded, sutures distinct and depressed; aperture terminal, an elongate opening, bordered weakly by a rim.
Subfamily PARABRIZALININAE Zweig-Strykowski and Reiss, 1976
Genus LOXOSTOMINA Sellier de Civrieux, 1969
Loxostomina amygdalaeformis (Brady, 1881)
Figure 10.25
1881 Bolivina amygdalaeformis Brady, p. 59.
2001 Saidovina amygdalaeformis (Brady); Szarek, p. 127, pl. 17, figs. 11-13.
2014 Saidovina amygdalaeformis (Brady); Panchang and Nigam, pl. 25, figs. 24-27.
Description. Test elongate-ovate, wall calcareous, perforate, surface ornamented with thick, longitudinal costae, final chambers with depressions; chambers few; periphery strongly carinate, sutures obscured; aperture terminal, subovate opening, bordered by an everted lip.
Loxostomina mayori (Cushman, 1922a)
Figure 10.26
1922a Bolivina mayori Cushman, p. 27, pl. 3, figs. 5-6.
2001 Bolivina macella (Belford); Szarek, p. 124, pl. 16, fig. 12.
2014 Loxostomina mayori (Cushman); Panchang and Nigam, pl. 25, fig. 16.
Description. Test elongate, slender, wall calcareous, perforate, surface ornamented with long costae; chambers slightly inflated, biserially arranged; periphery slightly lobulate, sutures fairly distinct and slightly depressed at the periphery; aperture terminal, an elongate slit-like opening, bordered by a lip.
Superfamily PLANORBULINOIDEA Schwager, 1877
Family CIBICIDIDAE Cushman, 1927
Subfamily CIBICIDINAE Cushman, 1927
Genus CIBICIDES de Montfort, 1808
Cibicides kullenbergi Parker, 1953
Figure 11.5-6
1953 Cibicides kullenbergi Parker in Phleger et al., p. 49, pl. 11, figs. 7-8.
2012 Cibicides pachyderma (Rzehak); Debenay, p. 190.
Description. Test biconvex, slightly convex on the spiral side, strongly convex on the umbilical side, wall calcareous, dorsal side coarsely perforate, ventral side moderately perforate, surface smooth; periphery subacutely rounded, sutures curved backwards, slightly elevated on spiral side, radial and depressed in the final chambers on the umbilical side; aperture an interiomarginal, equatorial arch, under a thin lamina.
Cibicides pachyderma (Rzehak, 1886)
Figure 11.7-9
1886 Truncatulina pachyderma Rzehak, p. 87, pl. 1, fig. 5a-c.
2010 Cibicidoides pachyderma Rzehak; Margreth, p. 121, pl. 33, fig. 2a-c.
Description. Test lenticular, spiral side slightly convex, umbilical side strongly convex, wall calcareous, coarsely perforate, surface smooth; periphery rounded, sutures limbate, curved backwards, slightly depressed on spiral side, radial and depressed in the final chambers on the umbilical side; aperture an interiomarginal arch, bordered by a thick, everted lip.
Cibicides patalaensis Haque, 1956
Figure 11.10-11
1956 Cibicides patalaensis Haque, pl. 16, fig. 6.
2014 Cibicides patalaensis Haque; Panchang and Nigam, pl. 31, fig. 11a-c.
Description. Test circular in outline, much compressed, wall calcareous, coarsely perforate, surface smooth; chambers slightly inflated, gradually increasing in size; periphery carinate, lobulated in the final chambers, sutures limbate, curved on the dorsal side, radial and depressed on the umbilical side; umbilical plug visible on both sides; aperture an interiomarginal opening.
Cibicides praecinctus (Karrer, 1868)
Figure 11.12-14
1868 Rotalina praecincta Karrer, p. 189, pl. 5, fig. 7.
2001 Heterolepa praecincta (Karrer); Szarek, p. 145, pl. 25, figs. 1-3.
2012 Heterolepa praecincta (Karrer); Debenay, p. 199.
2014 Heterolepa praecincta (Karrer); Panchang and Nigam, pl. 35, fig. 12a-c.
Description. Test biconvex, spiral side slightly convex, umbilical side strongly convex, wall calcareous, coarsely perforate, surface smooth; chambers arranged trochospirally, coiling evolute on the dorsal side, involute on the ventral side; periphery slightly lobulate, margin carinate, sutures limbate on both sides, elevated circular bands on the spiral side, radial and slightly depressed on the umbilical side; aperture interiomarginal, low slit-like opening, bordered by a lip.
Cibicides pseudolobatulus Perelis and Reiss, 1975
Figure 11.15
1975 Cibicides pseudolobatulus Perelis and Reiss, pp. 77-78, pl. 4, figs. 1-7; pl. 5, figs. 1-2.
2012 Cibicides pseudolobatulus Perelis and Reiss; Debenay, p. 190.
2012 Cibicides pseudolobatulus Perelis and Reiss; Milker and Schmiedl, p. 107, fig. 24.12-13.
Description. Test subrounded, spiral side flat, umbilical side convex, wall calcareous, perforate, surface smooth; chambers trochospirally arranged, coiling evolute on the dorsal side, involute on the ventral side; periphery rounded and slightly lobulate, sutures limbate and curved on the spiral side, radial, curved and depressed on the umbilical side; aperture interiomarginal, extraumbilical opening, bordered by a rim.
Cibicides refulgens de Montfort, 1808
Figure 11.16-17
1808 Cibicides refulgens de Montfort, p. 123, text-fig. 122.
2003 Cibicides refulgens de Montfort; Murray, p. 21, pl. 7, figs. 1-2.
2006 Cibicides refulgens de Montfort; Oflaz, p. 224, pl. 8, fig. 7.
2010 Cibicides refulgens de Montfort; Margreth, p. 122, pl. 34, fig. 4a-c.
2012 Cibicides refulgens de Montfort; Debenay, p. 191.
2012 Cibicides refulgens de Montfort; Milker and Schmiedl, p. 107, fig. 24.14-16.
Description. Test planoconvex, flat spiral side, convex umbilical side, wall calcareous, perforate on both sides, surface smooth; chambers arranged trochospirally; periphery weakly carinate, sutures limbate and slightly elevated on the spiral side, curved and depressed on the umbilical side; aperture interiomarginal, extraumbilical opening.
Cibicides wuellerstorfi (Schwager, 1866)
Figure 11.18-19
1866 Anomalina wuellerstorfi Schwager, p. 258, pl. 7, figs. 105, 107.
2010 Fontbotia wuellerstorfi (Schwager); Margreth, p. 122, pl. 35, fig. 2a-c.
2012 Fontbotia wuellerstorfi (Schwager); Debenay, p. 196.
2014 Fontbotia wuellerstorfi (Schwager); Panchang and Nigam, pl. 31, fig. 13a-c.
Description. Test compressed, flat spiral side, convex umbilical side, wall calcareous, coarsely perforate, surface smooth; chambers many, curved and narrow; periphery acutely rounded, with a distinct keel, slightly lobulate, sutures more limbate, slightly raised and curved on the spiral side; aperture interiomarginal, an arch, bordered by a lip.
Genus HANZAWAIA Asano, 1944
Hanzawaia nitidula (Bandy, 1953)
Figure 11.20-21
1953 Cibicidina basiloba (Cushman) var. nitidula Bandy, p. 178, pl. 22, fig. 3.
1961 Hanzawaia nitidula (Bandy); Lehmann, p. 16, pl. 2, fig. 2a-c.
1973 Cibicidina basiloba var. nitidula (Bandy); Lankford and Phleger, p. 122, pl. 6, fig. 19.
Description. Test subovate, side view planoconvex, wall calcareous, finely perforate, surface smooth; chambers many, rapidly increasing in size, coiling involute; periphery subacute, slightly lobate, sutures arcuate, radial, strongly limbate on the spiral side, curved, limbate and slightly raised, slightly depressed in the later chambers on the umbilical side; aperture interiomarginal, a low arched opening, under a folium.
Genus LOBATULA Fleming, 1828
Lobatula lobatula (Walker and Jacob, 1798)
Figure 12.1-2
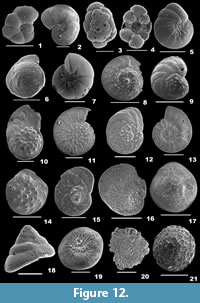 1798 Nautilus lobatulus Walker and Jacob, p. 642, pl. 14, fig. 36.
1798 Nautilus lobatulus Walker and Jacob, p. 642, pl. 14, fig. 36.
2003 Cibicides lobatulus (Walker and Jacob); Murray, p. 21, fig. 6.13-15.
2006 Lobatula lobatula (Walker and Jacob); Oflaz, p. 299, pl. 8, figs. 12-13.
2007 Cibicides lobatulus (Walker and Jacob); Talib and Farroqui, p. 19, pl.1, fig. 20a-b.
2010 Lobatula lobatula (Walker and Jacob); Margreth, p. 122, pl. 35, fig. 1a-c.
2012 Lobatula lobatula (Walker and Jacob); Debenay, p. 201.
2012 Lobatula lobatula (Walker and Jacob); Milker and Schmiedl, p. 107, fig. 24.17-20.
Description. Test with different morphology, planoconvex, spiral side flat, umbilical side convex, wall calcareous, perforate, surface smooth; chambers gradually increasing in size, trochospirally arranged; periphery carinate and lobulate, sutures curved backwards and slightly raised on the spiral side, radial and slightly depressed on the umbilical side; aperture interiomarginal, an equatorial arch, covered by a narrow lamina on the apertural side.
Family CYMBALOPORIDAE Cushman, 1927
Subfamily CYMBALOPORINAE Cushman, 1927
Genus CYMBALOPORETTA Cushman, 1928
Cymbaloporetta bradyi (Cushman, 1915)
Figure 12.3-4
1915 Cymbalopora poeyi (d’Orbigny) var. bradyi Cushman, p. 25, pl. 10, fig. 2; pl. 14, fig. 2.
2001 Cymbaloporetta bradyi (Cushman); p. 120, pl. 22, figs. 11-12.
2012 Cymbaloporetta bradyi (Cushman); Debenay, p. 236.
Description. Test subovate, slightly convex on the spiral side, flat umbilical side, umbilicus depressed and concave, spiral side coarsely perforate, umbilical side smooth and perforate; chambers subglobular on the dorsal side, subtriangular on the ventral side, increasing in size as added, chamber arrangement trochospiral initially, later annularly arranged; periphery round and lobulate, sutures distinct, slightly depressed; apertures three, two sutural and an umbilical opening in each chamber, bordered by a thick, everted lip.
Family PLANORBULINIDAE Schwager, 1877
Subfamily PLANORBULININAE Schwager, 1877
Genus PLANORBULINA d’Orbigny, 1826
Planorbulina mediterranensis d’Orbigny, 1826
Figure 11.22
1826 Planorbulina mediterranensis d’Orbigny, p. 280, pl. 14, figs. 4-6.
2002 Planorbulina mediterranensis d’Orbigny; Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 176, pl. 4, fig. 3a-b.
2006 Planorbulina mediterranensis d’Orbigny; Oflaz, p. 299, pl. 8, figs. 14-15.
2012 Planorbulina mediterranensis d’Orbigny; Milker and Schmiedl, p. 108, fig. 24.21-14.
Description. Test attached, outline circular, slightly convex on the spiral side, flat on the umbilical side, wall calcareous, coarsely perforate, surface with pustules; chamber arrangement trochospiral initially, later arranged in a circular plan; periphery lobulated, sutures depressed on the dorsal side, thick, imperforate and limbate on the ventral side; primary aperture interiomarginal, two openings diametrically placed on chamber ends, bordered by a lip, secondary extraumbilical openings.
Superfamily CHILOSTOMELLOIDEA Brady, 1881
Family ANOMALINIDAE Cushman, 1927
Genus RIMINOPSIS Revets, 1996
Riminopsis asterizans (Fichtel and Moll, 1798)
Figure 12.5
1798 Nautilus asterizans Fichtel and Moll, p. 37, pl. 3, figs. c-h.
1999 Nonion asterizans (Fichtel and Moll); Nigam and Khare, p. 298, pl. 6, fig. 3.
2014 Nonion asterizans (Fichtel and Moll); Panchang and Nigam, pl. 33, fig. 7a-b.
Description. Test symmetrical, outline circular, side view rounded, wall calcareous, finely perforate, surface smooth, umbilical region depressed, ornamented with granular pustules, pustules also along the sutures and the apertural face; chambers many, broad and low, chamber arrangement planispiral, coiling involute or slightly evolute; periphery angular, sutures distinct, slightly depressed and curved; aperture interiomarginal, short, narrow or rounded opening.
Family GAVELINELLIDAE Hofker, 1956
Subfamily GAVELINELLINAE Hofker, 1956
Genus HANSENISCA Loeblich and Tappan, 1987
Hansenisca soldanii (d’Orbigny, 1826)
Figure 12.6-7
1826 Gyroidina soldanii d’Orbigny, p. 278, no. 5.
2010 Gyroidina soldanii (d’Orbigny); Margreth, p. 125, pl. 40, fig. 1a-c.
2012 Hansenisca soldanii (d’Orbigny); Debenay, p. 197.
2012 Gyroidinoides soldanii (d’Orbigny); Milker and Schmiedl, p. 116, fig. 26.20-21.
Description. Test round in outline, flat on the spiral side, strongly convex on the umbilical side, wall calcareous, finely perforate, surface smooth; chambers arranged trochospirally, coiling evolute on the dorsal side, involute on the ventral side; periphery truncate, sutures radial, slightly curved and depressed on the umbilical side; aperture interiomarginal, an equatorial low arch opening, bordered by a lip, secondary apertures partially covered by an external flap.
Superfamily NUMMULITOIDEA de Blainville, 1827
Family NUMMULITIDAE de Blainville, 1827
Genus OPERCULINA d’Orbigny, 1826
Operculina ammonoides (Gronovius, 1781)
Figure 12.8
1781 Nautilus ammonoides Gronovius, p. 282, pl. 19, figs. 5-6.
2004 Operculina ammonoides (Gronovius); Hohenegger, p. 151, text-fig. 2.1.
2011 Operculina ammonoides (Gronovius); Hohenegger, p. 659, fig. 2b.
2012 Operculina ammonoides (Gronovius); Debenay, p. 228.
Description. Test large, planispiral, compressed, surface well-ornamented, tubercles slightly raised in the umbilical area; chambers flat, not divided, coiling evolute; periphery with a thin keel, sutures marked with pustules, slightly depressed at the periphery; aperture at the end of the apertural face.
Operculina bartschi Cushman, 1921
Figure 12.9
1921 Operculina bartschi Cushman, p. 376, text-fig. 13.
2012 Operculina bartschi Cushman; Debenay, p. 228.
Description. Test lenticular, surface ornamented with nodules in between the chambers, umbilical region with slightly raised granular knobs; chambers narrow, rapidly increasing in width, slightly inflated, not flaring, coiling evolute; periphery rounded with a thin keel, sutures distinct and depressed; aperture at the base of the apertural face.
Operculina complanata (Defrance in de Blainville, 1822)
Figure 12.10
1822 Lenticulites complanata Defrance, p. 453.
2004 Operculina complanata (Defrance); Hohenegger, p. 151, text-fig. 2.3.
2011 Operculina complanata (Defrance); Hohenegger, p. 659, fig. 2e.
2014 Operculina complanata (Defrance); Panchang and Nigam, pl. 39, fig. 3.
Description. Test thin, planispiral, surface ornamented with granular pustules in and around the umbilical region; chambers narrow, not separate, rapidly increasing in size; coiling evolute; septal flap flaring, slightly folded; periphery with a distinct keel, sutures distinct, marked with pustules initially, later faintly distinct.
Operculina discoidalis d’Orbigny, 1826
Figure 12.11
1826 Nummulina (Assilina) discoidalis d’Orbigny, p. 296, no. 88.
2011 Operculina discoidalis (d’Orbigny); Hohenegger, p. 659, fig. 2a.
2012 Operculina discoidalis (d’Orbigny); Debenay, p. 228.
Description. Test very flat, discoidal, wall calcareous, finely perforate, surface distinctly ornamented with granular pustules; chambers not separate; coiling involute initially, later nearly evolute; periphery rounded with a distinct keel, sutures slightly raised, limbate, marked with rows of pustules, slightly depressed at the periphery.
Operculina elegans Cushman, 1921
Figure 12.12
1921 Operculina elegans Cushman, p. 381, pl. 97, fig. 3.
2004 Operculina elegans Cushman; Hohenegger, p. 151, text-fig. 2.2.
2011 Operculina elegans Cushman; Hohenegger, p. 659, fig. 2d.
Description. Test compressed, wall calcareous, surface ornamented with granular pustules in and around the umbilical region, tubercles in between the chambers; coiling nearly evolute, chambers gradually increasing in width, slightly flaring, septal flap smooth; periphery rounded with a thin keel, sutures distinctly arched and depressed, marked with fine pustules in the final chambers; aperture at the basal end of the apertural face.
Operculina granulosa Leymerie, 1846
Figure 12.13
1846 Operculina granulosa Leymerie, p. 359, pl. 13, fig. 12a-c.
2014 Operculina granulosa Leymerie; Panchang and Nigam, pl. 39, fig. 4.
Description. Test large, compressed, wall calcareous, surface ornamented with more tubercles in the centre, final whorls sparsely covered by pustules; chambers narrow, coiling evolute; periphery with a distinct keel, sutures marked with pustules; aperture terminal, at the base of the apertural face.
Operculina hardiei d’Archiac and Haime, 1853
Figure 12.14
1853 Operculina hardiei d’Archiac and Haime, p. 346, pl. 35, fig. 6a-c.
2014 Operculina hardiei d’Archiac and Haime; Panchang and Nigam, pl. 39, fig. 5.
Description. Test small, wall calcareous, surface with nodules in the centre, irregularly covering the other portion, chambers high, coiling involute; periphery with distinct keel, sutures marked with pustules, aperture at the end of the apertural face.
Genus HETEROSTEGINA d’Orbigny, 1826
Heterostegina depressa d’Orbigny, 1826
Figure 12.15
1826 Heterostegina depressa d’Orbigny, p. 305, pl. 17, figs. 5-7.
2001 Heterostegina depressa d’Orbigny; Szarek, p. 151, pl. 28, fig. 5 (not fig. 6).
2008 Heterostegina depressa d’Orbigny; Meriç, Avşar, and Yokeş, p. 323, pl. 10, figs. 1-12.
2011 Heterostegina depressa d’Orbigny; Hohenegger, p. 659, fig. 2k.
Description. Test complanate, central portion thickened, wall calcareous, surface slightly rough; chambers many, elongate and curved, initial chambers undivided, thick and involute, final chambers subdivided by septa, thin and flaring; periphery thin and rounded, sutures distinct, very slightly depressed and limbate; aperture terminal, multiple openings on the apertural face.
Genus PLANOSTEGINA Banner and Hodgkinson, 1991
Planostegina operculinoides (Hofker, 1927)
Figure 12.16
1927 Heterostegina operculinoides Hofker, p. 67, pl. 34, figs. 2, 4, 5.
2004 Planostegina operculinoides (Hofker); Hohenegger, p. 151, text-fig. 2.5.
2011 Planostegina operculinoides (Hofker); Hohenegger, p. 659, fig. 2h.
2012 Heterostegina operculinoides (Hofker); Debenay, p. 222.
Description. Test very much flattened, thin, planispiral, wall calcareous, surface ornamented with granular pustules in the central portion; chambers arch-shaped, subdivided into chamberlets, rapidly increasing in size; sutures of chambers often marked with pustules, of chamberlets, distinct and depressed; aperture terminal, at the end of the apertural face.
Superfamily GLABRATELLOIDEA Loeblich and Tappan, 1964
Family GLABRATELLIDAE Loeblich and Tappan, 1964
Genus PILEOLINA Bermúdez, 1952
Pileolina patelliformis (Brady, 1884)
Figure 12.17-19
1884 Discorbis patelliformis Brady, p. 647, pl. 88, fig. 3a-c.
2010 Glabratella patelliformis (Brady); Margreth, p. 120, pl. 32, fig. 3a-c.
2012 Pileolina patelliformis (Brady); Debenay, p. 208.
2012 Pileolina patelliformis (Brady); Milker and Schmiedl, p. 102, fig. 23.16-17.
Description. Test subtriangular, planoconvex, wall calcareous, densely perforate on the spiral side and finely perforate on the umbilical side, surface rough, umbilical side depressed, ornamented with radiating pustules; chambers long, narrow and semicircular, spiral side strongly convex, conical with a bluntly pointing apex, gradually increasing in size as added, umbilical side flattened, arranged trochospirally, coiling evolute; periphery rounded, sutures curved on the spiral side; aperture an interiomarginal, low slit-like opening.
Superfamily ACERVULINOIDEA Schultze, 1854
Family ACERVULINIDAE Schultze, 1854
Genus PLANOGYPSINA Bermúdez, 1952
Planogypsina acervalis (Brady, 1884)
Figure 12.20
1884 Planorbulina acervalis Brady, p. 657, pl. 92, fig. 4.
2009 Planogypsina acervalis (Brady); Debenay, p. 246.
Description. Test attached, large, outline nearly discoid, dorsal side slightly convex, ventral side flattened, wall coarsely perforate; chambers inflated, subglobular, evenly added, and almost equidimensional, secondary chambers added irregularly and dispersed on the entire test surface; peripheral margin cuspate, periphery lobulated; primary apertures two arched openings at the juncture of earlier chambers, bordered by a rim, secondary apertures multiple, tiny sutural openings at either side of the test, peripheral and tubular openings.
Genus SPHAEROGYPSINA Galloway, 1933
Sphaerogypsina globulus (Reuss, 1848)
Figure 12.21
1848 Ceriopora globulus Reuss, p. 33, pl. 5, fig. 7.
2012 Sphaerogypsina globula (Reuss); Debenay, p. 249.
2012 Sphaerogypsina globula (Reuss); Milker and Schmiedl, p. 110, fig. 25.4.
Description. Test free, outline globular, side view spherical, wall calcareous, coarsely perforate, surface with thick, raised, imperforate septa; chambers many, small and roughly polygonal, closely packed; aperture secondary, multiple, small, circular openings at the basal end of chamber walls.
Superfamily TURRILINOIDEA Cushman, 1927
Family STAINFORTHIIDAE Reiss, 1963
Genus STAINFORTHIA Hofker, 1956
Stainforthia fusiformis (Williamson, 1858)
Figure 13.1
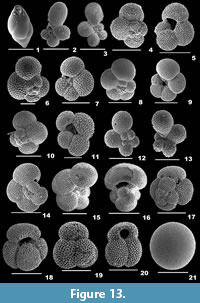 1858 Bulimina pupoides var. fusiformis Williamson, p. 63, pl. 5, figs. 129-130.
1858 Bulimina pupoides var. fusiformis Williamson, p. 63, pl. 5, figs. 129-130.
2003 Stainforthia fusiformis (Williamson); Murray, p. 26, figs. 10.1-10.
2010 Stainforthia fusiformis (Williamson); Margreth, p. 115, pl. 27, fig. 2a-c.
Description. Test small, elongate-ovate, fusiform, tapering, wall calcareous, finely perforate, surface smooth; chambers subglobose, sutures distinct and slightly depressed; aperture terminal, semi-circular opening bordered by a thick rim.
Suborder GLOBIGERININA Delage and Hérouard, 1896
Superfamily GLOBIGERINOIDEA Carpenter, 1862
Family GLOBIGERINIDAE Carpenter, 1862
Subfamily GLOBIGERININAE Carpenter, 1862
Genus BEELLA Banner and Blow, 1960
Beella digitata (Brady, 1879a)
Figure 13.2-3
1879a Globigerina digitata Brady, p. 59, pl. 80, figs. 6-10.
1983 Beella digitata (Brady); Kennett and Srinivasan, p. 232, pl. 58, figs. 2, 6-8.
Description. Test strongly convex, wall calcareous, perforate, surface with irregular ridge-like spine bases, chambers globular initially, rapidly increasing in size as added, radially elongate and subconical final whorl, chamber arrangement trochospiral; periphery rounded, peripheral outline stellate; sutures radial, depressed; aperture umbilical, broad open arch bordered by a reverted lip, without teeth.
Genus GLOBIGERINA d’Orbigny, 1826
Globigerina bulloides d’Orbigny, 1826
Figure 13.4-5
1826 Globigerina bulloides d’Orbigny, p. 277, no. 1.
1983 Globigerina bulloides d’Orbigny; Kennet and Srinivasan, p. 36, pl. 6, figs. 4-6.
2010 Globigerina bulloides d’Orbigny; Ovechkina, Bylinskaya and Uken, p. 239, fig. 5G-5I.
Description. Test subglobular, wall perforate, surface smooth, with spines; chambers spherical, gradually increasing in size as added, chamber arrangement trochospiral, periphery rounded, sutures distinct and depressed; aperture high arch, umbilical, interiomarginal.
Globigerina falconensis Blow, 1959
Figure 13.6-7
1959 Globigerina falconensis Blow, p. 177, pl. 9, figs. 40a-c, 41.
2010 Globigerina falconensis Blow; Ovechkina, Bylinskaya and Uken, p. 239, fig. 6A-6C.
Description. Test small, wall coarsely perforate, surface ornamented with spines; chambers globular, increasing in size as added, chamber arrangement medium to low trochospiral; periphery rounded, sutures distinct, depressed; aperture umbilical, interiomarginal with a thick lip.
Turborotalia quinqueloba Natland, 1938
Figure 13.8-9
1938 Globigerina quinqueloba Natland, p. 149, pl. 6, fig. 7.
2017 T urborotalita quinqueloba (Natland); Schiebel and Hemleben, p. 50, pl. 2.16, figs. 1-17.
Description. Test small, slightly flattened, wall coarsely perforate, surface ornamentes with spines; chambers subglobular, chamber arrangement low trochospiral, periphery rounded, sutures distinct, depressed; aperture umbilical, interiomarginal, bordered by a lip.
Genus GLOBIGERINELLA Cushman, 1927
Globigerinella calida (Parker, 1962)
Figure 13.10-11
1962 Globigerina calida Parker, p. 221, pl. 1, figs. 9-13, 15.
1983 Globigerinella calida (Parker); Kennet and Srinivasan, p. 240, pl. 60, figs. 7-9.
2010 Globigerinella calida (Parker); Ovechkina, Bylinskaya and Uken, p. 242, fig. 6G-6I.
Description. Test trochoid, wall perforate, surface ornamented with spines; chambers spherical initially, slightly radially elongate later, increase in size as added, chamber arrangement slightly trochospiral; periphery lobulated, sutures depressed; aperture high umbilical arch, with a narrow lip.
Globigerinella radians (Egger, 1893)
Figure 13.12-13
1893 Globigerina radians Egger, p. 170, pl. 13, figs. 22-24.
Description. Test very lobate, flattened, wall perforate, surface ornamented with circular spines; chambers radially elongate with rounded tips, rapid increase in size as added, chamber arrangement planispiral, coiling highly evolute; aperture an equatorial, high arch.
Globigerinella siphonifera (d’Orbigny, 1839a)
Figure 13.14-16
1839a Globigerina siphonifera d’Orbigny, p. 83, pl. 4, figs. 15-18.
1983 Globigerinella aequilateralis (Brady); Kennett and Srinivasan, p. 238, pl. 59, fig. 1; pl. 60, figs. 4-6.
2010 Globigerinella siphonifera (d’Orbigny); Ovechkina, Bylinskaya, and Uken, p. 244, fig. 7A, 7B, 7D, 7E.
Description. Test large, wall perforate, surface ornamented with round spines; chambers spherical initially, globular or ovoid in the final stage, chamber arrangement nearly planispiral, coiling involute; periphery rounded, sutures distinct and depressed; aperture high, symmetrical, equatorial arch.
Genus GLOBIGERINOIDES Cushman, 1927
Globigerinoides conglobatus (Brady, 1879a)
Figure 13.17-18
1879a Globigerina conglobata Brady, p. 28B.
1983 Globigerinoides conglobatus (Brady); Kennett and Srinivasan, p. 58, pl. 12, figs. 4-6.
2010 Globigerinoides conglobatus (Brady); Ovechkina, Bylinskaya and Uken, p. 245, fig. 8A-8D.
Description. Test subquadrate, outline subglobular, wall calcareous, perforate, surface heavily cancellate, ornamented with spines; chambers subspherical, rapidly increasing in size as added, chamber arrangement medium to high trochospiral; sutures distinct; primary aperture interiomarginal, umbilical, elongate, low arch-shaped, secondary apertures small arched openings.
Globigerinoides ruber (d’Orbigny, 1839a)
Figure 13.19-20
1839a Globigerina rubra d’Orbigny, p. 82, pl. 4, figs. 12-14.
1983 Globigerinoides ruber (d’Orbigny); Kennett and Srinivasan, p. 78, pl. 10, figs. 6; pl. 17, figs. 1-3.
2010 Globigerinoides ruber (d’Orbigny); Ovechkina, Bylinskaya and Uken, p. 245, fig. 9A-9E.
Description. Test small, wall calcareous, coarsely perforate, surface ornamented with spines; chambers subglobular, of uniform size, chamber arrangement trochospiral; periphery rounded, sutures distinct, depressed; primary aperture semicircular, above the suture of two chambers.
Globigerinoides sacculifer (Brady, 1877)
Figure 14.1-4
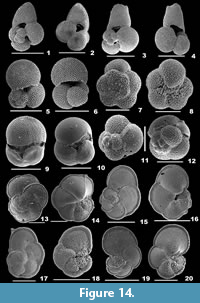 1877 Globigerina sacculifera Brady, p. 535.
1877 Globigerina sacculifera Brady, p. 535.
1983 Globigerinoides sacculifer (Brady); Kennet and Srinivasan, p. 66, pl. 14, figs. 4-6.
2010 Globigerinoides sacculifer (Brady); Ovechkina, Bylinskaya and Uken, p. 246, fig. 8G-8I.
Description. Test large, wall coarsely perforate, surface ornamented with spines; chambers globular, increasing in size as added, final chamber elongate, slightly flattened, chamber arrangement low trochospiral; periphery rounded, sutures well depressed; primary aperture umbilical, interiomarginal, low, wide symmetrical arch, bordered by an imperforate lip in few, secondary apertures subelliptical.
Globigerinoides trilobus (Reuss, 1850)
Figure 14.5-6
1850 Globigerina triloba Reuss, p. 374, pl. 47, fig. 11a-c.
1983 Globigerinoides triloba (Reuss); Kennet and Srinivasan, p. 62, pl. 10, fig. 4; pl. 13, figs. 1-3.
2010 Globigerinoides trilobus (Reuss); Ovechkina, Bylinskaya and Uken, p. 246, fig. 8E, 8F.
Description. Test trilobate, wall coarsely perforate, surface ornamented with spines; chambers globular, increasing in size as added, chamber arrangement trochospiral; periphery rounded, sutures very distinct and depressed; primary aperture umbilical, slit-like or low, slightly wide symmetrical arch, secondary apertures subrounded.
Genus GLOBOTURBOROTALITA Hofker, 1976
Globoturborotalita sp.
Figure 14.7-8
Description. Test small, evolute, wall moderately coarse agglutinate, surface rough; chambers trochospirally arranged, gradually increase in size as added; periphery rounded, sutures radial and depressed; aperture interiomarginal.
Genus ORBULINA d’Orbigny, 1839
Orbulina universa d’Orbigny, 1839a
Figure 13.21
1839a Orbulina universa d’Orbigny, p. 3, pl. 1, fig. 1.
1983 Orbulina universa d’Orbigny; Kennet and Srinivasan, p. 86, pl. 20, figs. 4-6.
2010 Orbulina universa d’Orbigny; Ovechkina, Bylinskaya and Uken, p. 248, fig. 9H.
Description. Test spherical, wall calcareous, perforate, surface ornamented with many small pores interspersed amidst few larger pores; chambers many, globular, chamber arrangement trochospiral; sutures distinct and depressed; aperture interiomarginal, umbilical, bordered by an imperforate lip in juveniles, areal in adults.
Genus SPHAEROIDINELLA Cushman, 1927
Sphaeroidinella dehiscens (Parker and Jones, 1865)
Figure 14.9-10
1865 Sphaeroidinella bulloides d’Orbigny var. dehiscens Parker and Jones, p. 369, pl. 19, fig. 5a-b.
2010 Sphaeroidinella dehiscens (Parker and Jones); Ovechkina, Bylinskaya, and Uken, p. 248, fig. 9F, 9G.
Description. Test large, ovoid, outline weakly lobulate, wall calcareous, coarsely perforate, surface smooth and glossy in adults; chambers globular, rapidly increasing in size as added, chamber arrangement trochospiral, coiling tight, periphery broadly rounded, sutures depressed initially, later obscured by surface cortex in adults; aperture interiomarginal, umbilical in juveniles, slit-like in adults, with a continuous crenulated lip along the cortical margins.
Superfamily GLOBOROTALIOIDEA Cushman, 1927
Family CANDEINIDAE Cushman, 1927
Subfamily GLOBIGERINITINAE Bermúdez, 1961
Genus CANDEINA d’Orbigny, 1839c
Candeina nitida d’Orbigny, 1839c
Figure 14.11
1839c Candeina nitida d’Orbigny, p. 107, pl. 2, figs. 27-28.
1983 Candeina nitida d’Orbigny; Kennett and Srinivasan, p. 228, pl. 57, figs. 6-8.
Description. Test subglobular, shiny, wall calcareous, very finely perforate, surface smooth, without spines; chambers radial, coiling tight; periphery rounded, sutures distinct; aperture multiple, small circular to elongate, rimmed openings along the sutures of the final and also the penultimate chamber, in rare cases.
Genus GLOBIGERINITA Brönnimann, 1951
Globigerinita glutinata (Egger, 1893)
Figure 14.12
1893 Globigerina glutinata Egger, p. 371, pl. 13, figs. 19-21.
1983 Globigerinita glutinata (Egger); Kennet and Srinivasan, p. 224, pl. 56, figs. 1, 3-5.
2010 Globigerinita glutinata (Egger); Ovechkina, Bylinskaya and Uken, p. 239, figs. 5B, 5E.
Description. Test subrounded, wall calcareous; chambers spherical, of nearly uniform size, chamber arrangement trochospiral, periphery rounded, sututres distinct; primary aperture umbilical, low arch opening, bordered by a lip, umbilical side sutures covered by bulla.
Family GLOBOROTALIIDAE Cushman, 1927
Genus GLOBOROTALIA Cushman, 1927
Globorotalia cultrata (d’Orbigny, 1839a)
Figure 14.13-14
1839a Rotalina cultrata d’Orbigny, p. 76, pl. 5, figs. 7-9.
2010 Globorotalia cultrata (d’Orbigny); Ovechkina, Bylinskaya and Uken, p. 235, fig. 3A, 3D.
Description. Test broad, biconvex, wall calcareous, perforate, surface smooth; chambers nearly flat, subrounded on the spiral side, more inflated, subtriangular on the umbilical side, rapidly increasing in size as added, chamber arrangement trochospiral; periphery keeled, sutures arcuate on the spiral side, radial on the umbilical side; aperture interiomarginal, umbilical, low arched, with a plate-like tooth.
Globorotalia menardii (d’Orbigny, 1826)
Figure 14.15-16
1826 Rotalia menardii d’Orbigny, p. 273.
2010 Globorotalia menardii (Parker, Jones and Brady, 1865); Ovechkina, Bylinskaya and Uken, p. 234, fig. 3B, 3E.
Description. Test biconvex, wall calcareous, perforate, surface smooth, no spines; chambers nearly flat, semi-circular on the spiral side, slightly inflated, subtriangular on the umbilical side, rapidly increasing in size as added, chamber arrangement trochospiral; periphery keeled, sutures curved on the spiral side, radial on the umbilical side; aperture low arch, interiomarginal, umbilical, with a plate-like tooth.
Globorotalia neoflexuosa (Srinivasan, Kenette and Bé, 1974)
Figure 15.1
1974 Globorotalia menardii subsp. neoflexuosa Srinivasan, Kenette and Bé, p. 322, pl. 1, figs. 1-2.
Description. Test large, compressed, peripheral view rounded, wall finely perforate, surface smooth; chambers slightly inflated, rapidly increasing in size as added, chamber arrangement low trochospiral, final chamber gently bent towards the umbilicus; periphery with imperforate keel, sutures slightly depressed on the dorsal side, depressed and nearly radial on the ventral side; aperture arch-shaped, bordered by a lip.
Globorotalia tumida (Brady, 1877)
Figure 14.17-18
1877 Pulvinulina menardii d’Orbigny var. tumida Brady, p. 535.
2010 Globorotalia tumida (Brady); Ovechkina, Bylinskaya and Uken, p. 236, fig. 3G, 3H.
Description. Test large, biconvex, wall thick, calcareous, perforate; chambers slightly inflated, reniform on the spiral side, subtriangular and more inflated on the umbilical side, final chamber slightly compressed, chamber arrangement trochospiral; periphery with thick keel, sutures thick, curved on the spiral side, radial on the umbilical side; aperture extraumbilical, interiomarginal with a plate-like tooth.
Globorotalia ungulata Bermúdez, 1961
Figure 14.19-20
1961 Globorotalia ungulata Bermúdez, p. 1304, pl. 15, fig. 6a-b.
2010 Globorotalia ungulata Bermúdez; Ovechkina, Bylinskaya and Uken, p. 236, figs. 3C, 3F, 3I.
Description. Test small, biconvex, wall calcareous, perforate, surface smooth; chambers flat on the spiral side, inflated and subtriangular on the ventral side, chamber arrangement trochospiral, final chamber with a large umbilical flap; periphery with thin keel, sutures curved on the spiral side, radial on the ventral side; aperture interiomarginal, umbilical, low, slit-like opening at the basal end of the large flap with a plate-like lip.
Genus NEOGLOBOQUADRINA Bandy, Frerichs and Vincent, 1967
Neogloboquadrina dutertrei (d’Orbigny, 1839a)
Figure 15.2-3
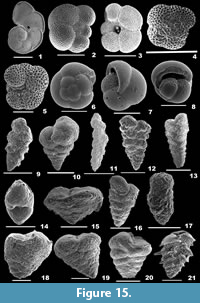 1839a Globigerina dutertrei, d’Orbigny, p. 84, pl. 4, figs. 19-21.
1839a Globigerina dutertrei, d’Orbigny, p. 84, pl. 4, figs. 19-21.
1983 Neogloboquadrina dutertrei (d’Orbigny); Kennett and Srinivasan, p. 198, pl. 48, figs. 7-9.
2010 Neogloboquadrina dutertrei (d’Orbigny); Ovechkina, Bylinskaya and Uken, p. 238, fig. 4E, 4F, 4H, 4I.
Description. Test robust, slightly compressed, wall calcareous, coarsely perforate, surface not ornamented with spines; chambers subglobular, gradually increasing in size as added, chamber arrangement trochospiral; periphery rounded, sutures distinct, depressed; aperture circular opening, umbilical.
Neogloboquadrina incompta (Cifelli, 1961)
Figure 15.4-5
1961 Globigerina incompta Cifelli, p. 83, pl. 4, figs. 1-7.
2010 Neogloboquadrina incompta (Cifelli); Ovechkina, Bylinskaya and Uken, p. 238, fig. 4D, 4G.
Description. Test medium, less compact, wall strongly perforate; chambers globular, chamber arrangement trochospiral; periphery rounded, sutures visible; aperture interiomarginal, umbilical, extending to the periphery, with a slightly extended lip.
Family PULLENIATINIDAE Cushman, 1927
Genus PULLENIATINA Cushman, 1927
Pulleniatina obliquiloculata (Parker and Jones in Carpenter, Parker and Jones, 1862)
Figure 15.6-8
1865 Pullenia sphaeroides d’Orbigny var. obliquiloculata Parker and Jones, pp. 365, 368, pl. 19, fig. 4a-b.
1983 Pulleniatina obliquiloculata (Parker and Jones); Kennet and Srinivasan, p. 202, pl. 49, fig. 2; pl. 50, figs. 6-9.
2010 Pulleniatina obliquiloculata (Parker and Jones); Ovechkina, Bylinskaya and Uken, p. 238, figs. 5A, 5D.
Description. Test rounded, wall calcareous, finely perforate, surface smooth, without spines; chambers spherical, chamber arrangement trochospiral initially, later streptospiral; periphery rounded, sutures disitinct, slightly depressed; aperture low, interiomarginal arch with a thick rim.
Order TEXTULARIIDA Delage and Hérouard, 1896
Suborder TEXTULARIINA Delage and Hérouard, 1896
Superfamily TEXTULARIOIDEA Ehrenberg, 1838
Family TEXTULARIIDAE Ehrenberg, 1838
Subfamily TEXTULARIINAE Ehrenberg, 1838
Genus TEXTULARIA Defrance, 1824
Textularia agglutinans d’Orbigny, 1839b
Figure 15.9
1839b Textularia agglutinans d’Orbigny; p. 144, pl. 1, figs. 17-18, 32-34.
2003 Textularia agglutinans d’Orbigny; Javaux and Scott, p. 22, fig. 5.8-9.
2007 Textularia agglutinans d’Orbigny; Talib and Farroqui, p. 18, pl. 1, fig. 1.
2012 Textularia agglutinans d’Orbigny; Debenay, p. 95.
2012 T extularia agglutinans d’Orbigny; Milker and Schmiedl, p. 38, fig. 10.15-16.
2014 Textularia agglutinans d’Orbigny; Panchang and Nigam, pl. 3, fig. 14a-b.
2015 Textularia agglutinans d’Orbigny; Hanagata and Nobuhara, p. 18, fig. 7.3-4.
Description. Test elongate, slightly compressed, tapering, lateral view narrowly triangular, apertural view subrounded, wall coarsely agglutinated, surface smooth; final chambers slightly inflated, chambers biserially arranged, increasing in size gradually; sutures fairly distinct and slightly depressed, aperture terminal, a low arch or long slit-like depression.
Textularia candeiana d’Orbigny, 1839b
Figure 15.10
1839b Textularia candeiana d’Orbigny, p. 143, pl. 1, figs. 25-27.
2012 Textularia candeiana d’Orbigny; Debenay, p. 96.
2015 Textularia candeiana d’Orbigny; Hanagata and Nobuhara, p. 18, fig. 7.5.
Description. Test narrow, V-shaped, tapering rapidly to the apex, wall agglutinated, surface smooth; chambers many, much compressed in the early stage, rapidly enlarging later, final chambers much inflated; sutures fairly distinct, and depressed; apertural face broadly rounded, aperture terminal, a low arched opening at the base of the final chamber, bordered by a thin lip.
Textularia earlandi Parker, 1952
Figure 15.11
1952 Textularia earlandi Parker, p. 458.
2008 Textularia earlandi Parker; Riveiros and Patterson, p. 14, fig. 5.2a-b.
2014 Textularia earlandi Parker; Panchang and Nigam, pl. 3, figs. 18-19.
Description. Test elongate, much longer than broad, aboral end pointing, apertural end broad, wall agglutinated, surface rough; chambers many, biserially arranged; sutures distinct and depressed; aperture terminal, a small, slit-like depression.
Textularia fistula Cushman, 1911
Figure 15.12
1911 Textularia agglutinans d’Orbigny var. fistula Cushman, p. 10, text-fig. 11.
2012 T extularia fistula Cushman; Debenay, p. 96.
2014 Textularia fistula Cushman; Panchang and Nigam, pl. 3, fig. 20.
Description. Test elongate, triangular, much compressed laterally, wall coarsely agglutinated, surface irregular, rough; chambers many, increasing in size as added, biserially arranged; sutures indistinct initially, horizontal and depressed in later chambers; aperture slit-like depression on the inner margin of the final chamber.
Textularia granulata Costa, 1855
Figure 15.13-14
1855 Textularia sagittula Defrance var. granulata Costa, pl. 23, fig. 11.
2014 T extularia granulata Costa; Panchang and Nigam, pl. 3, fig. 24a-b.
Description. Test elongate, subtriangular, apertural end broadly rounded, tapering towards the aboral end, wall agglutinated, surface smooth; chambers many, gradually increasing in size as added, biserial chamber arrangement; periphery subacute, sutures slightly depressed; aperture terminal, a slit-like small opening at the centre, bordered by a lip.
Textularia pala (Cžjžek, 1848)
Figure 15.15
1848 Textularia pala Cžjžek, p. 148, pl. 13, figs. 25-27.
2006 Textularia pala Cžjžek; Oflaz, p. 135, pl. 1, fig. 5.
2012 Textularia pala Cžjžek; Milker and Schmiedl, p. 40, fig. 10.21-22.
Description. Test triangular, broad than long, compressed laterally, apertural end broad, aboral end tapering, wall finely agglutinated, surface smooth; chambers many, compressed, biserially arranged, rapidly increasing in size as added; periphery slightly undulate, sutures distinct, curved and slightly depressed; aperture interiomarginal, terminal, a low arched opening, bordered by a lip.
Textularia pseudogramen Chapman and Parr, 1937
Figure 15.16
1937 Textularia pseudogramen Chapman and Parr, p. 153.
2012 Textularia pseudogramen Chapman and Parr; Debenay, p. 98.
2014 Textularia pseudogramen Chapman and Parr; Panchang and Nigam, pl. 4, fig. 15a-b.
Description. Test large, elongate, subtriangular, wall coarsely agglutinated, surface rough except apertural face; chambers many, biserially arranged, initially increasing in size as added, final chambers of almost constant width; periphery subacute earlier, thickened later, sutures distinct, depressed; aperture terminal, a low arch opening on the apertural side.
Genus BIGENERINA d’Orbigny, 1826
Bigenerina nodosaria d’Orbigny, 1826
Figure 15.17
1826 Bigenerina nodosaria d’Orbigny, p. 261, pl. 11, figs. 9-12.
2001 Bigenerina nodosaria d’Orbigny; Szarek, p. 94, pl. 8, figs. 12-14.
2002 Bigenerina nodosaria d’Orbigny; Kaminski, Aksu, Box, Hiscott, Filipescu and Al-Salameen, p.170, pl. 1, fig. 9.
2003 Bigenerina nodosaria d’Orbigny; Murray, p. 11, fig. 2.4.
2006 Bigenerina nodosaria d’Orbigny; Oflaz, p. 131, pl. 1, fig. 4.
2010 Bigenerina nodosaria d’Orbigny; Margreth, p. 99, pl. 5, fig. 5a-c.
2012 Bigenerina nodosaria d’Orbigny; Debenay, p. 77.
2012 Bigenerina nodosaria d’Orbigny; Milker and Schmiedl, p. 36, fig. 10.10-12.
2014 Bigenerina nodosaria d’Orbigny; Panchang and Nigam, pl. 3, figs. 4-9.
2015 Bigenerina nodosaria d’Orbigny; Hanagata and Nobuhara, p. 18, fig. 7.1-2.
Description. Test elongate, wall coarsely agglutinated, surface rough, cross section circular; chambers rounded to ovate, or flattened, initial chambers arranged biserially, later uniserially arranged; sutures distinct and slightly depressed; aperture terminal, small and rounded on the apertural face.
Genus SAHULIA Loeblich and Tappan, 1985
Sahulia conica (d’Orbigny, 1839b)
Figure 15.18
1839b Textularia conica d’Orbigny, p. 143, pl. 1, figs. 19-20.
2001 Sahulia conica (d’Orbigny); Szarek, p. 94, pl. 8, figs. 19-21.
2003 Textularia conica (d’Orbigny); Javaux and Scott, p. 21, fig. 5.10-11.
2012 Textularia conica (d’Orbigny); Debenay, p. 96.
2012 Textularia conica (d’Orbigny); Milker and Schmiedl, p. 36, fig. 10.18.
2014 Sahulia conica (d’Orbigny); Panchang and Nigam, pl. 3, figs. 10a-c, 11a-c, 12a-c.
Description. Test broad than long, conical, slightly compressed, outline subtriangular, wall agglutinated, surface smooth or slightly rough; chambers low and broad, biserially arranged, rapidly increasing in size, no visible chamberlets; peripheral margin subacute, sutures nearly horizontal, slightly curved, and depressed; aperture terminal, slit-like, bordered by a lip.
Sahulia kerimbaensis (Said, 1949)
Figure 15.19
1949 Textularia kerimbaensis Said, p. 6, pl. 1, fig. 8.
2007 Textularia aff T. kerimbaensis (Said); Talib and Farroqui, p. 18, pl. 1, fig. 3.
2012 Textularia kerimbaensis (Said); Debenay, p. 97.
2012 Sahulia kerimbaensis (Said); Hemleben, Spindler and Anderson, p. 21, pl. 3, figs. 8-12; pl. 4, figs. 1-8.
Description. Test triangular, outline V-shaped, broad, wall composed of round arenaceous grains, surface smooth; initial chambers triserially arranged, later biserially arranged, rapidly increasing in size as added; periphery subacute, sutures horizontal, curved and depressed; aperture terminal, slit-like in the apertural face, bordered by a distinct lip.
Subfamily SIPHOTEXTULARIINAE Loeblich and Tappan, 1985
Genus KARREROTEXTULARIA Le Calvez, de Klasz, and Brun, 1974
Karrerotextularia flintii (Cushman, 1911)
Figure 15.20
1911 Textularia flintii Cushman, p. 21, text-fig. 36a-b.
2003 Siphotextularia flintii (Cushman); Murray, p. 15, fig. 3.11.
2012 Siphotextularia flintii (Cushman); Debenay, p. 93.
2012 Siphotextularia flintii (Cushman); Milker and Schmiedl, p. 40, fig. 11.1-3.
Description. Test subtriangular, compressed laterally, wall finely agglutinated, surface smooth; chambers inflated, low and broad, increasing in size gradually, arranged biserially; sutures distinct, horizontal and deep, aperture slit-like, at the basal end of the final chamber, bordered by a slightly raised lip.
Family KAMINSKIIDAE Neagu, 1999
Genus SPIRORUTILUS Hottinger, Halicz and Reiss, 1990
Spirorutilus carinatus (d’Orbigny, 1846)
Figure 15.21
1846 Textularia carinata d’Orbigny, p. 247, pl. 14, figs. 32-34.
2001 Spiroplectinella pseudocarinata (Cushman); Szarek, p. 88, pl. 6, figs. 3-6.
2014 Textularia pseudocarinata (Cushman); Panchang and Nigam, pl. 4, fig. 14a-b.
Description. Test triangular, flattened laterally, flanges extending at the sides, wall agglutinated, surface rough; chambers biserially arranged, rapidly increasing in size as added; periphery carinate, sutures distinct, raised ridges; aperture terminal, large, low arched opening, bordered by a rim.
Superfamily EGGERELLOIDEA Cushman, 1937
Family VALVULINIDAE Berthelin, 1880
Subfamily VALVULININAE Berthelin, 1880
Genus CLAVULINA d’Orbigny, 1826
Clavulina angularis d’Orbigny, 1826
Figure 16.1
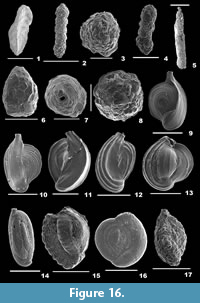 1826 Clavulina angularis d’Orbigny, p. 268, pl. 12, fig. 7.
1826 Clavulina angularis d’Orbigny, p. 268, pl. 12, fig. 7.
1969 Clavulina angularis d’Orbigny; Keller, p. E9, pl. 1, fig. 2; pl. 11, fig. 4.
Description. Test elongate, slender, sharply pointed, tricarinate, parallel sides tapering at the ends, cross section triangular, wall agglutinated, surface smoothly finished; initial chambers distinctly uniserial, final triserial chambers indistinct; periphery acute, sutures curved backwards and depressed; aperture terminal, round in the middle portion of the apertural face.
Family EGGERELLIDAE Cushman, 1937
Subfamily EGGERELLINAE Cushman, 1937
Genus MARTINOTTIELLA Cushman, 1933
Martinottiella cylindrica (d’Orbigny, 1852)
Figure 16.2-3
1852 Clavulina cylindrica d’Orbigny, p. 194.
2012 Clavulina cylindrica (Cushman); Milker and Schmiedl, p. 42, fig. 11.7.
Description. Test elongate, side view cylindrical, basal view subtriangular, wall coarsely agglutinated, surface rough; chambers triserially arranged initially, later subglobular, uniserially arranged, cylindrical in the final stage; sutures distinct, horizontal, and depressed; aperture terminal, a circular opening at the end of a short neck.
Family PSEUDOGAUDRYINIDAE Loeblich and Tappan, 1985
Subfamily PSEUDOGAUDRYININAE Loeblich and Tappan, 1985
Genus PSEUDOCLAVULINA Cushman, 1936
Pseudoclavulina serventyi (Chapman and Parr, 1935)
Figure 16.4
1935 Clavulina serventyi Chapman and Parr, p. 5, pl. 1, fig. 7.
2001 Pseudoclavulina serventyi (Chapman and Parr); Szarek, p. 97, pl. 10, figs. 16-17.
2012 Pseudoclavulina serventyi (Chapman and Parr); Debenay, p. 88.
2015 Pseudoclavulina serventyi (Chapman and Parr); Hanagata and Nobuhara, p. 20, figs. 7.14-15.
Description. Test elongate, side view subcylindrical, basal view triangular, wall composed of coarse agglutinated material, surface rough; chambers initially small, triserially arranged, later long, subglobular, uniserially arranged; sutures somewhat distinct, horizontal, and depressed; aperture terminal, a circular opening on a round apertural side.
Class MONOTHALAMEA Pawlowski, Holzmann, Berney, Fahrni, Gooday, Cedhagen, Habura, and Bowser, 2003
Order ASTRORHIZIDA Lankester, 1885
Suborder HIPPOCREPININA Saidova, 1981
Superfamily HIPPOCREPINOIDEA Rhumbler, 1895
Family HYPERAMMINIDAE Eimer and Fickert, 1899
Subfamily HYPERAMMININAE Eimer and Fickert, 1899
Genus HYPERAMMINA Brady, 1878
Hyperammina friabilis Brady, 1884
Figure 16.5
1884 Hyperammina friabilis Brady, p. 258, pl. 23, figs. 1-3, 5, 6.
2012 Hyperammina friabilis Brady; Debenay, p. 83.
Description. Test linear, elongate, subcylindrical, tapering towards the apertural side, wall thick, agglutinated but loosely cemented, surface rough; large globular proloculus followed by a long, subcylindrical, second chamber lesser in width; aperture rounded, terminal.
Suborder SACCAMMININA Lankester, 1885
Superfamily SACCAMMINOIDEA Brady, 1884
Family SACCAMMINIDAE Brady, 1884
Subfamily SACCAMMININAE Brady, 1884
Genus LAGENAMMINA Rhumbler, 1911
Lagenammina difflugiformis (Brady, 1879a)
Figure 16.6-7
1879a Reophax difflugiformis Brady, p. 51, pl. 4, fig. 3a-b.
2001 Lagenammina difflugiformis (Brady); Szarek, p. 175, pl. 1, figs. 9-10.
2005 Lagenammina difflugiformis (Brady); Licari and Mackensen, p. 207, pl. 1, fig. 4.
Description. Test pyriform, wall coarsely agglutinated, surface smooth; single chambered, aperture single, subrounded, terminal at the end of a short neck.
Superfamily PSAMMOSPHAEROIDEA Haeckel, 1894
Family PSAMMOSPHAERIDAE Haeckel, 1894
Subfamily PSAMMOSPHAERINAE Haeckel, 1894
Genus PSAMMOSPHAERA Schulze, 1875
Psammosphaera fusca Schulze, 1875
Figure 16.8
1875 Psammosphaera fusca Schulze, p. 113, pl. 2, fig. 8a-f.
2010 Psammosphaera fusca Schulze; Margreth, p. 129, pl. 1, fig. 5a-b (not pl. 5).
2012 Psammosphaera fusca Schulze; Milker and Schmiedl, p. 31, fig. 9.2-3.
Description. Test free or attached, globular, wall coarsely agglutinated, surface roughly finished; chamber single, aperture terminal.
Class TUBOTHALAMEA Pawlowski, Holzman and Tyszka, 2013
Order MILIOLIDA Delage and Hérouard, 1896
Suborder MILIOLINA Delage and Hérouard, 1896
Superfamily MILIOLOIDEA Ehrenberg, 1839
Family CRIBROLINOIDIDAE Haynes, 1981
Genus ADELOSINA d’Orbigny, 1826
Adelosina longirostra (d’Orbigny, 1826)
Figure 16.9-10
1826 Quinqueloculina laevigata d’Orbigny, p. 301.
2014 Adelosina laevigata (d’Orbigny); Panchang and Nigam, pl.5, fig. 19a-c.
Description. Test slightly elongate, central portion convex on the spiral side, wall calcareous, imperforate, surface with many visible striations; periphery carinate; aperture terminal, at the end of a somewhat longer neck, with a small bifid tooth.
Adelosina pulchella (d’Orbigny, 1826)
Figure 16.11-12
1826 Quinqueloculina pulchella d’Orbigny, p. 303.
2006 Adelosina pulchella (d’ Orbigny); Oflaz, p. 149, pl. 2, figs. 8-10.
Description. Test large, side view fusiform, wall calcareous, imperforate, surface ornamented with parallel costae; chambers visible; periphery carinate, sutures distinct, slightly depressed; aperture terminal, at the end of a short neck, weakly bordered by a lip, with a small tooth slightly raised above the aperture.
Genus CRIBROLINOIDES Cushman and LeRoy, 1939
Cribrolinoides curta (Cushman, 1917)
Figure 16.13
1917 Quinqueloculina disparilis d’Orbigny var. curta Cushman, p. 49, pl. 14, fig. 2, text-fig. 30.
2012 Cribrolinoides curtus (Cushman); Debenay, p. 106.
2014 Cribrolinoides curtus (Cushman); Panchang and Nigam, pl. 4, fig. 6a-c.
Description. Test short, robust, side view subcircular, wall calcareous, imperforate, surface ornamented with longitudinal ridges; chambers visible, chamber arrangement quinqueloculine; periphery highly carinate, broadly rounded, sutures distinct; aperture subcircular, with a prominent, projected bifid tooth.
Family MILIAMMINIDAE Saidova, 1981
Genus MILIAMMINA Heron-Allen and Earland, 1930
Miliammina fusca (Brady, 1870)
Figure 16.14
1870 Quinqueloculina fusca Brady, p. 286, pl. 11, figs. 2-3.
2003 Miliammina fusca (Brady); Javaux and Scott, p. 16, fig. 6.7.
2012 Miliammina fusca (Brady); Debenay, p. 86.
2014 Miliammina fusca (Brady); Panchang and Nigam, pl. 39, fig. 10a-b.
Description. Test agglutinated, elongate, cross section ovate, wall composed of well sorted detrital grains in an organic cement, surface rough; chamber arrangement quinqueloculine; aperture terminal, ovate with a simple tooth.
Family RIVEROINIDAE Saidova, 1981
Genus PSEUDOHAUERINELLA McCulloch, 1981
Pseudohauerinella orientalis (Cushman, 1946)
Figure 16.15
1946 Hauerina orientalis (Cushman); Cushman, p. 12, pl. 2, figs. 22-24.
2012 Pseudohauerina orientalis (Cushman); Debenay, p. 114.
Description. Test oval, wall calcareous, imperforate, surface reticulate, ornamented with transverse ribs; chambers inflated, chamber arrangement quinqueloculine, coiling partially evolute; aperture U-shaped in younger forms, cribrate, sieve-like in adults produced by the coalescence of teeth.
Family HAUERINIDAE Schwager, 1876
Subfamily SIGMOILINITINAE Łuczkowska, 1974
Genus SIGMOIHAUERINA Zheng, 1979
Sigmoihauerina bradyi (Cushman, 1917)
Figure 16.16
1917 Hauerina bradyi Cushman; p. 62, pl. 23, fig. 2.
2006 Sigmoihauerina bradyi (Cushman); Oflaz, p. 170, pl. 3, fig. 9.
Description. Test very much flattened, broadly ovate, wall calcareous, imperforate, surface strongly pitted; chambers compressed, final chambers slightly inflated, overlapping the earlier chambers, quinqueloculine chamber arrangement initially, later planispirally arranged; periphery rounded, sutures distinct, slightly depressed; aperture terminal, elliptical with a trematophore.
Subfamily SIPHONAPERTINAE Saidova, 1975
Genus AGGLUTINELLA El-Nakhai, 1983
Agglutinella arenata (Said, 1949)
Figure 16.17
1949 Quinqueloculina anguina Terquem var. arenata Said, p. 9, pl. 1, fig. 25.
1994. Agglutinata arenata (Said); Loeblich and Tappan, p. 306-307, pl. 69, figs. 9-11; pl. 70, figs. 10-15; pl. 74, figs. 10-13.
2002 Agglutinata arenata (Said); Kaminski, Aksu, Box, Hiscott, Filipescu, and Al-Salameen, p. 186.
2012 Quinqueloculina arenata (Said); Debenay, p. 119.
2014 Quinqueloculina arenata (Said); Panchang and Nigam, pl. 7, fig. 20a-b.
Description. Test elongate, wall coarsely agglutinated, surface rough; chambers broadly rounded, tapering towards the ends, chamber arrangement quinqueloculine; periphery rounded, sutures obscured by the arenaceous material; aperture terminal, round at the end of a prominent neck, with a thick, protruding tooth.
Subfamily HAUERININAE Schwager, 1876
Genus CYCLOFORINA Łuczkowska, 1972
Cycloforina semiplicata (McCulloch, 1977)
Figure 17.1-2
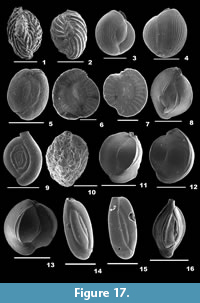 1977 Quinqueloculina semiplicata McCulloch, p. 507, pl. 217, figs. 7, 11, 13, and 17.
1977 Quinqueloculina semiplicata McCulloch, p. 507, pl. 217, figs. 7, 11, 13, and 17.
2014 Cycloforina semiplicata (McCulloch); Panchang and Nigam, pl. 7, fig. 12a-c.
Description. Test elongate, wall calcareous, imperforate, surface ornamented with many transverse, curved ribs; chambers visible, inflated, chamber arrangement quinqueloculine; sutures less distinct, depressed; aperture terminal, round at the end of a neck, bordered by a thin lip, with a small bifid tooth.
Genus FLINTINA Cushman, 1921
Flintina bradyana Cushman, 1921
Fig 17.3-4
1921 Flintina bradyana Cushman, p. 467, pl. 94, fig. 2, text-figs. 38-44.
2012 Flintina bradyana Cushman; Debenay, p. 108.
2014 Flintina bradyana Cushman; Panchang and Nigam, pl. 11, fig. 8a-c.
Description. Test robust, subspherical, wall porcelaneous, imperforate, surface ornamented with long striae; chambers inflated, rapidly increasing in size as added, chamber arrangement triloculine; periphery broadly rounded and highly carinate, sutures distinct, slightly depressed; aperture terminal, ovate, bordered by a thick rim, with a prominent flap-like tooth, well raised above the periphery.
Genus HAUERINA d’Orbigny, 1839
Hauerina earlandi Rasheed, 1971
Figure 17.5
1971 Hauerina earlandi Rasheed, p. 54, pl. 16, fig. 7.
2012 Hauerina earlandi Rasheed; Debenay, p. 108.
Description. Test slightly compressed or globular to subglobular, wall calcareous, imperforate, surface smooth; chamber arrangement triloculine to quinqueloculine initially, planispiral later; periphery rounded, sutures slightly depressed; aperture terminal, a complex trematophore at the end of the final chamber.
Hauerina ornatissima (Karrer, 1868)
Figure 17.6-7
1868 Quinqueloculina ornatissima Karrer, p. 151, pl. 3, fig. 2.
1946 Hauerina ornatissima (Karrer); Cushman, p. 5, pl. 1, fig. 16.
2014 Hauerina ornatissima (Karrer); Panchang and Nigam, pl. 14, fig. 18a-b.
Description. Test oval to circular, compressed, wall calcareous, imperforate, surface of the later chambers ornamented with transverse ribs; chambers quinqueloculine initially, later planispirally arranged; peripheral margin subacute, aperture series of circular openings at the end of the final chamber, bordered by a thick rim.
Genus LACHLANELLA Vella, 1957
Lachlanella barnardi (Rasheed, 1971)
Figure 17.8
1971 Quinqueloculina barnardi Rasheed, pp. 26, 27, pl. 2, fig. 1.
2012 Quinqueloculina barnardi (Rasheed); Debenay, p. 119.
Description. Test robust, cross section triangular, side view oval, wall calcareous, imperforate, surface smooth, very finely striated; chambers visible, chamber arrangement quinqueloculine; peripheral margin acutely rounded to truncated, sutures distinct; aperture elongate, compressed, bordered by a lip, with a long simple tooth slightly raised above the aperture.
Genus MASSILINA Schlumberger, 1893
Massilina laevigata (Cushman and Todd, 1944)
Figure 17.9
1944 Spiroloculina laevigata Cushman and Todd, p. 67, pl. 9, figs. 26-29.
2014 Spiroloculina laevigata (Cushman and Todd); Panchang and Nigam, pl. 6, fig. 19a-b.
Description. Test large, robust, lateral view fusiform, wall calcareous, imperforate, surface smooth; chambers initially compressed, final chambers inflated, chamber arrangement spiroloculine, gradually increasing in size as added; periphery broadly rounded, sutures distinct and very depressed; aperture terminal, round at the end of a short neck, weakly rimmed, with a simple bifid tooth.
Genus QUINQUELOCULINA d’Orbigny, 1826
Quinqueloculina agglutinans d’Orbigny, 1839b
Figure 17.10
1839b Quinqueloculina agglutinans d’Orbigny, p. 195, pl. 12, figs. 11-13.
2001 Agglutinella agglutinans d’Orbigny; Szarek, p. 102, pl. 11, fig. 18.
2003 Quinqueloculina agglutinans d’Orbigny; Javaux and Scott, p. 18, fig. 4.3-4.
2006 Siphonaperta agglutinans d’Orbigny; Oflaz, p. 155, pl. 2, fig. 12.
2012 Quinqueloculina agglutinans d’Orbigny; Debenay, p. 119.
2012 Siphonaperta agglutinans d’Orbigny; Milker and Schmiedl, p. 51, fig. 13.18-19.
2014 Spiroloculina laevigata (Cushman and Todd); Panchang and Nigam, pl. 7, fig. 17a-c.
Description. Test slightly elongate, outline ovate, side view subelliptical, outer wall agglutinated, surface rough; chambers slightly inflated, chamber arrangement quinqueloculine; periphery broadly rounded, sutures visible but mostly hidden by the agglutinated material; aperture terminal, ovate, bordered by a distinct non-agglutinated lip, with a long bifid tooth.
Quinqueloculina auberiana d’Orbigny, 1839b
Figure 17.11-12
1839b Quinqueloculina auberiana d’Orbigny, p. 193, pl. 12, figs. 1-3.
2012 Quinqueloculina auberiana d’Orbigny; Debenay, p. 119.
2012 Quinqueloculina auberiana d’Orbigny; Milker and Schmiedl, p. 56, fig. 15.1-2.
Description. Test broad, oval-shaped, wall calcareous, imperforate, surface smooth; chambers mostly sharply angled or slightly subrounded, faintly carinate; peripheral margin carinate, sutures distinct; aperture terminal, low arch, bordered by a thick rim, with a simple tooth.
Quinqueloculina bicarinata d’Orbigny, 1826
Figure 17.13
1826 Quinqueloculina bicarinata d’Orbigny, p. 302, no. 35.
2001 Quinqueloculina bicarinata d’Orbigny; Szarek, p. 104, pl. 12, fig. 14.
2012 Quinqueloculina bicarinata d’Orbigny; Debenay, p. 120.
2014 Quinqueloculina bicarinata d’Orbigny; Panchang and Nigam, pl. 8, fig. 2a-c.
Description. Test robust, cross section triangular, side view subcircular; chambers carinate at the outer borders, chamber arrangement quinqueloculine; periphery subacute, sutures distinct; aperture terminal, ovate, slightly protruding, bordered by a weak, reversed rim, with a T-shaped tooth.
Quinqueloculina bosciana d’Orbigny, 1839b
Figure 17.14-15
1839b Quinqueloculina bosciana d’Orbigny, p. 191, pl. 11, figs. 22-24.
2006 Quinqueloculina bosciana d’Orbigny; Oflaz, pp. 160-161, pl. 11, fig. 6.
2012 Quinqueloculina bosciana d’Orbigny; Debenay, p. 120.
Description. Test elongate, apertural view suboval, apertural end truncate, aboral end round, wall calcareous, surface smooth; chambers distinct, slightly oblique, chamber arrangement quinqueloculine; periphery rounded, sutures distinct; aperture terminal, circular, on a short, broad neck, with a small bifid tooth.
Quinqueloculina bubnanensis McCulloch, 1977
Figure 18.1-2
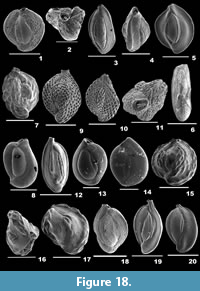 1977 Quinqueloculina bubnanensis McCulloch, pl. 213, fig. 16.
1977 Quinqueloculina bubnanensis McCulloch, pl. 213, fig. 16.
2014 Quinqueloculina bubnanensis McCulloch; Panchang and Nigam, pl. 8, fig. 6a-c.
Description. Test broadly ovate, wall calcareous, imperforate; chambers rapidly increasing in size as added, chamber arrangement quinqueloculine; sutures distinct, depressed; aperture terminal, circular on a short neck, bordered by a distinct lip, with a short, simple tooth.
Quinqueloculina delicatula Vella, 1957
Figure 18.3-4
1957 Quinqueloculina delicatula Vella, p. 27, pl. 4, figs. 77-79.
2012 Quinqueloculina delicatula Vella; Debenay, p. 122.
Description. Test elongate, apertural end extended, truncated by a short neck, aboral end broadly rounded, surface rough, with small pits; chamber arrangement quinqueloculine; peripheral margin convex, sutures distinct, slightly depressed; aperture rounded, bordered by a thick rim and a small bifid tooth.
Quinqueloculina exmouthensis Parker, 2009
Figure 17.16
2009 Quinqueloculina exmouthensis Parker, p. 207, figs. 146a-h, 147a-i, 148a-i.
2012 Quinqueloculina exmouthensis Parker; Debenay, p. 122.
Description. Test elongate, robust, twice longer than wide, lateral view broadly oval, apertural end extended, aboral end rounded, cross section triangular, wall calcareous, surface smooth, with fine striations; chambers visible, margins slightly convex, chamber arrangement quinqueloculine; peripheral margins truncated, sutures distinct, engraved; aperture terminal, round on a long neck, with a small bifid tooth.
Quinqueloculina lamarckiana d’Orbigny, 1839b
Figure 18.5
1839b Quinqueloculina lamarckiana d’Orbigny, p. 189, pl. 11, figs. 14-15.
1999 Quinqueloculina lamarckiana d’Orbigny; Nigam and Khare, p. 290, pl. 2, fig. 4.
2003 Quinqueloculina lamarckiana d’Orbigny; Javaux and Scott, p. 20, fig. 4.12-13.
2014 Quinqueloculina lamarckiana d’Orbigny; Panchang and Nigam, pl. 9, fig. 8a-c.
Description. Test broad, flattened at the sides, apertural view triangular, wall calcareous, imperforate, surface smooth; chambers visible, chamber arrangement quinqueloculine; periphery acutely angled, sutures distinct, slightly depressed; aperture terminal, subcircular, at the end of a slightly produced neck, bordered by a rim, with a large, simple tooth.
Quinqueloculina lizardi Baccaert, 1987
Figure 18.6
1987 Quinqueloculina oblonga subsp. lizardi Baccaert, p. 100, pl. 46, fig. 6a-b; pl. 47, fig. 1.
2012 Quinqueloculina lizardi Baccaert; Debenay, p. 123.
Description. Test elongate, compressed laterally, side view elliptical, aboral end rounded, apertural end truncate, wall calcareous, entire surface ornamented with slit-like depressions; chambers rapidly increasing in size as added, chamber arrangement quinqueloculine; periphery broadly oblong, sutures distinct, slightly depressed; aperture terminal, subcircular, bordered by a thick rim, with a small tooth.
Quinqueloculina parkeri (Brady, 1881)
Figure 18.7
1881 Miliolina parkeri Brady, p. 46.
2012 Quinqueloculina parkeri (Brady); Debenay, p. 12.
2014 Quinqueloc ulina parkeri (Brady); Panchang and Nigam, pl. 9, fig. 13a-c.
Description. Test broadly ovate, robust, wall calcareous, imperforate, surface ornamented with prominent transverse ribs; chambers inflated, chamber arrangement quinqueloculine; peripheral margin subacute, sutures depressed; aperture terminal, circular, at the end of a short neck bordered by an everted lip, and in few Lachlanella-type, with a large or thickened single tooth.
Quinqueloculina parvula Schlumberger, 1894
Figure 18.8
1894 Quinqueloculina parvula Schlumberger, p. 255, text-fig. 1, pl. 3, figs. 8-9.
2012 Quinqueloculina parvula Schlumberger; Milker and Schmiedl, p. 59, fig. 15.25-27.
Description. Test oblong, side view ovate, wall calcareous, imperforate, surface ornamented with anastomosing striae; chambers inflated, rapidly increasing in size as added, chamber margins rounded; periphery carinate, sutures distinct and depressed; aperture terminal, large, U-shaped, bordered by a thick rim, with a flap-like, bifid tooth, slightly raised above the periphery.
Quinqueloculina philippinensis Cushman, 1921
Figure 18.9-11
1921 Quinqueloculina kerimbatica (Heron-Allen and Earland) var. philippinensis Cushman, p. 438, pl. 89, figs. 2-3; text-fig. 34.
2001 Quinqueloculina ex gr. philippinensis Cushman; Szarek, p. 104, pl. 12, figs. 17-18.
2007 Quinqueloculina pseudoreticulata Parr; Talib and Farroqui, p. 18, pl. 1, fig. 8a-b.
2012 Quinqueloculina pseudoreticulata Parr; Debenay, p. 125.
2014 Quinqueloculina philippinensis Cushman; Panchang and Nigam, pl. 9, fig. 15a-c.
Description. Test broadly ovate, surface ornamented with regular reticulations, apertural end slightly produced, neck not reticulate; chambers inflated, chamber arrangement quinqueloculine; periphery rounded, sutures distinct; aperture terminal, oval-shaped, bordered by a phialine lip, with a long, bifid tooth.
Quinqueloculina schlumbergeri (Wiesner, 1923)
Figure 18.12
1923 Miliolina schlumbergeri Wiesner, p. 49, pl. 6, fig. 73.
2012 Quinqueloculina schlumbergeri (Wiesner); Debenay, p. 126.
2014 Quinqueloculina schlumbergeri (Wiesner); Panchang and Nigam, pl. 10, fig. 4a-c.
Description. Test elongate-ovate, wall porcelaneous, imperforate, surface smooth, with fine, long striae; chambers visible, chamber arrangement quinqueloculine; peripheral margins thick, acutely angled, sutures prominent; aperture terminal, rounded, bordered by a rim, with a short, bifid tooth.
Quinqueloculina seminula (Linnaeus, 1758)
Figure 18.13-14
1758 Serpula seminulum Linnaeus, p. 786, pl. 2, fig. 1a-c.
2003 Quinqueloculina seminulum (Linnaeus); Murray, p. 17, pl. 4, figs. 11-12.
2007 Quinqueloculina seminulum (Linnaeus); Talib and Farroqui, p. 18, pl.1, fig. 9 a-b.
2010 Quinqueloculina seminula (Linnaeus); Margreth, p. 101, pl. 7, fig. 8a-c.
2012 Quinqueloculina seminula (Linnaeus); Milker and Schmiedl, p. 57, fig. 15.30-31.
2012 Quinqueloculina seminula (Linnaeus); Debenay, p. 126.
Description. Test subrounded, wall calcareous, surface smooth, cross section and side view ovate, apertural end truncated, basal end rounded; chambers visible, chamber arrangement quinqueloculine; periphery rounded, sutures distinct, slightly depressed; aperture terminal, subrounded, bordered by a thick rim, with a simple tooth.
Quinqueloculina subparkeri McCulloch, 1977
Figure 18.15-17
1977 Quinqueloculina subparkeri McCulloch, p. 511, pl. 217, figs. 15-16; pl. 218, figs. 2-6, 12.
2012 Quinqueloculina subparkeri McCulloch; Debenay, p. 127.
Description. Test subcircular, compressed, apertural end truncated, aboral end rounded, wall calcareous, surface ornamented with transverse ribs; chambers visible, chamber arrangement quinqueloculine; peripheral margin undulating, periphery reticulate, sutures depressed; aperture U-shaped, bordered by a slightly everted lip, with a simple, raised tooth.
Quinqueloculina subpolygona Parr, 1945
Figure 18.18
1945 Quinqueloculina subpolygona Parr, p. 196, pl. 12, fig. 2a-c.
2012 Quinqueloculina subpolygona Parr; Debenay, p. 127.
Description. Test elongate, slightly compressed, wall calcareous, surface ornamented with strong costae; chambers distinct, chamber arrangement quinqueloculine; periphery quadrangular, sutures distinct and depressed; aperture terminal, large, bordered by a weakly everted lip, with a bifid tooth.
Quinqueloculina sulcata Fornasini, 1900
Figure 18.19-20
1900 Quinqueloculina sulcata Fornasini, p. 364.
2007 Quinqueloculina sulcata d’Orbigny; Talib and Farroqui, p. 18, pl.1, fig. 10a-b.
2012 Quinqueloculina cf. Q. sulcata d’Orbigny; Debenay, p. 127.
2013 Quinqueloculina sulcata d’Orbigny; Elakkiya and Manivannan, p. 885, fig. 7.11.
Description. Test elongate-oblong, wall calcareous, imperforate, surface ornamented with prominent costae; chamber arrangement quinqueloculine, margins truncate; peripheral margin subacute, periphery carinate, sutures distinct, slightly depressed; aperture terminal, circular at the end of a cylindrical neck, with a bifid tooth, bordered by a distinct rim.
Quinqueloculina undulose costata Terquem 1882
Figure 19.1-2
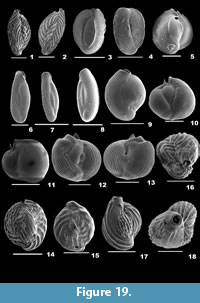 1882 Quinqueloculina undulose costata Terquem, p.185, pl. 20, figs. 18-19.
1882 Quinqueloculina undulose costata Terquem, p.185, pl. 20, figs. 18-19.
2007 Quinqueloculina undulose costata Terquem; Talib and Farroqui, p. 18, pl. 1, fig. 11a-b.
2012 Quinqueloculina undulose costata Terquem; Nisha and Singh, p. 787, pl. 1, fig. 6.
Description. Test broadly ovate, wall calcareous, imperforate, entire surface ornamented with transverse ribs; chambers inflated, chamber arrangement quinqueloculine; peripheral margin carinate, periphery subrounded, sutures indistinct; aperture terminal, circular, at the end of neck, bordered by a weakly everted rim, with a short, raised bifid tooth.
Quinqueloculina venusta Karrer, 1868
Figure 19.3-4
1868 Quinqueloculina venusta Karrer, p. 147, pl. 2, fig. 16.
2012 Quinqueloculina venusta Karrer; Debenay, p. 128.
2014 Quinqueloculina venusta Karrer; Panchang and Nigam, pl. 10, figs. 16a-c, 17a-c.
Description. Test subovate, aboral end rounded, apertural end truncate, wall calcareous, imperforate, surface smooth; chambers flattened, chamber arrangement quinqueloculine; periphery sharply angular, sutures distinct, depressed; aperture terminal, subelliptical, bordered by a thin rim, with a simple tooth.
Quinqueloculina vulgaris d’Orbigny, 1826
Figure 19.5
1826 Quinqueloculina vulgaris d’Orbigny, p. 302, no. 33.
2003 Quinqueloculina vulgaris d’Orbigny; Javaux and Scott, p. 20, fig. 4.20-21.
2007 Quinqueloculina vulgaris d’Orbigny; Talib and Farroqui, p. 18, pl. 1, fig. 12a-b.
Description. Test subcircular, stout, spiral view orbicular, apertural end slightly produced, wall calcareous, imperforate, surface smooth with fine striations; chambers inflated, chamber arrangement quinqueloculine; periphery acutely angled, sutures distinct; aperture elongate, slightly compressed, bordered by a distinctly everted lip with a bifid tooth raised slightly above the periphery of the aperture.
Subfamily MILIOLINELLINAE Vella, 1957
Genus CRIBROMILIOLINELLA Saidova, 1981
Cribromiliolinella milletti (Cushman, 1954)
Figure 19.6-8
1954 Hauerina milletti Cushman in Cushman, Todd and Post, p. 337, pl. 84, fig. 23.
1992 Cribromiliolinella milletti (Cushman); Hatta and Ujiie, p. 69, pl. 9, figs. 4-5.
2012 Edentostomina milletti (Cushman); Debenay, p. 106.
2014 Edentostomina milletti (Cushman); Panchang and Nigam, pl. 5, fig. 13a-b.
Description. Test compressed, longer than width, outline ovate; chambers slightly inflated, planispirally arranged, rapidly increasing in size, one-half coil in length, periphery acute or subrounded, sutures slightly sunken; aperture simple, terminal, elongate or subelliptical at the end of a neck, bordered by a thick lip, with no tooth.
Genus MILIOLINELLA Wiesner, 1931
Miliolinella circularis (Bornemann, 1855)
Figure 19.9
1855 Triloculina circularis Bornemann, p. 349, pl. 19, fig. 4.
2012 Miliolinella circularis (Bornemann); Debenay, p. 109.
2014 Miliolinella circularis (Bornemann); Panchang and Nigam, pl. 11, fig. 10a-b.
Description. Test subovate to round, side view circular, wall calcareous, surface smooth; chambers inflated, three chambers visible, milioline chamber arrangement; periphery rounded, sutures distinct; aperture terminal, a large low arch opening bordered by a well-developed lip with a simple tooth.
Miliolinella subrotunda (Montagu, 1803)
Figure 19.10-11
1803 Vermiculum subrotundum de Montagu, p. 521, pl. 1, fig. 4.
2003 Miliolinella subrotunda (Montagu); Murray, p. 15, pl. 4, fig. 6.
2006 Miliolinella subrotunda (Montagu); Oflaz, p. 168, pl. 11, fig. 7.
2012 Miliolinella subrotunda (Montagu); Debenay, p. 110.
2012 Miliolinella subrotunda (Montagu); Milker and Schmiedl, p. 63, fig. 16.31-32.
Description. Test subcircular, wall porcelaneous, translucent sometimes, surface smooth; chambers inflated, visible, strongly overlapping previous chambers, milioline chamber arrangement, nearly planispiral arrangement in adults; periphery rounded, sutures distinct and depressed; aperture terminal, subrounded, bordered by a distinct lip, with a low arched, broad flap-like tooth.
Miliolinella webbiana (d’Orbigny, 1839b)
Figure 19.12-13
1839b Triloculina webbiana d’Orbigny, p. 140, pl. 3, figs. 13-15.
2012 Miliolinella webbiana (d’Orbigny); Debenay, p. 110.
2012 Miliolinella webbiana (d’Orbigny); Milker and Schmiedl, p. 65, fig. 17.1-2.
Description. Test subcircular, wall porcelaneous, imperforate, surface fully ornamented with long costae; chambers inflated, arranged in quinqueloculine plan initially, later planispirally arranged; periphery rounded, carinate, sutures distinct, depressed; aperture terminal, large, bordered by a lip, with a big, slightly arched, flap-like tooth, slightly raised above the periphery of the aperture.
Genus PARAHAUERINOIDES McCulloch, 1977
Parahauerinoides fragilissima (Brady, 1884)
Figure 20.1
1884 Spiroloculina fragilissima Brady, p. 149, pl. 9, figs. 12-14.
1946 Hauerina fragilissima (Brady); Cushman, p. 9, pl. 2, figs. 1-6, 8.
2001 Hauerina fragilissima (Brady); Szarek, p. 103, pl. 12, fig. 6.
2012 Hauerina fragilissima (Brady); Debenay, p. 108.
Description. Test highly compressed, discoid, outline oval to rounded, wall thin, transparent, surface unevenly pitted; chambers partially involute, rapidly increasing in size as added, chamber arrangement initially quinqueloculine, later planispirally arranged; periphery rounded, sutures distinct, depressed; aperture terminal, cribrate.
Genus PSEUDOTRILOCULINA Cherif, 1970
Pseudotriloculina kerimbatica (Heron-Allen and Earland, 1939)
Figure 19.14-16
1915 Miliolina kerimbatica Heron-Allen and Earland, p. 574, pl. 43, figs. 13-23.
1999 Quinqueloculina kerimbatica (Heron-Allen and Earland); Nigam and Khare, p. 290, pl. 2, fig. 3.
2014 Quinqueloculina kerimbatica (Heron-Allen and Earland); Panchang and Nigam, pl. 9, figs. 4a-b, 5-6.
Description. Test subovate, broad than long, wall calcareous, imperforate, surface ornamented with transverse ribs; chambers inflated, quinqueloculine chamber arrangement; periphery highly carinate, sutures depressed; aperture terminal, circular on a short neck, bordered by a distinct rim, with a short bifid tooth.
Pseudotriloculina linneiana (d’Orbigny, 1839b)
Figure 19.17-18
1839b Triloculina linneiana d’Orbigny, p. 172, pl. 9, figs. 11-13.
2003 Triloculina linneiana (d’Orbigny); Javaux and Scott, p. 23, figs. 5.16-17.
2012 Triloculina linneiana (d’Orbigny); Debenay, p. 116.
Description. Test slightly elongate, tapering at both the ends, wall calcareous, imperforate, surface ornamented by long costae on the final chambers, prominent, raised ridges on the earlier chamber; chambers slightly inflated, arranged in a cryptoquinqueloculine plan; periphery slightly ovate, sutures depressed; aperture terminal, circular on a slightly produced neck, bordered by a thick rim, with a large, bifid tooth.
Pseudotriloculina sp.
Figure 20.2
 Description. Test ovate, slightly compressed, wall calcareous, surface smooth and polished; chambers visible, final two nearly making up the entire test surface, cryptoquinqueloculine chamber arrangement; periphery broadly rounded, sutures distinct, slightly depressed; aperture terminal, high arch opening, bordered by a rim, with a broad flap- like tooth.
Description. Test ovate, slightly compressed, wall calcareous, surface smooth and polished; chambers visible, final two nearly making up the entire test surface, cryptoquinqueloculine chamber arrangement; periphery broadly rounded, sutures distinct, slightly depressed; aperture terminal, high arch opening, bordered by a rim, with a broad flap- like tooth.
Genus PYRGO Defrance, 1824
Pyrgo denticulata (Brady, 1884)
Figure 20.3
1884 Biloculina ringens (Lamarck) var. denticulata Brady, p. 143, pl. 3, figs. 4-5.
2012 Pyrgo denticulata (Brady); Debenay, p. 117.
Description. Test subcircular, wall porcelaneous, surface smooth; chambers biconvex, biloculine chamber arrangement, coiling involute, peripheral margin broadly rounded, aboral margin serrated; aperture terminal, ovate, large opening, bordered by a rim, with a prominently broad T- shaped tooth.
Pyrgo inornata (d’Orbigny, 1846)
Figure 20.4
1846 Biloculina inornata d’Orbigny, p. 266, pl. 16, figs. 7-9.
2010 Pyrgo inornata (d’Orbigny); Margreth, p. 103, pl. 9, fig. 2a-c.
2012 Pyrgo inornata (d’Orbigny); Debenay, p. 117.
Description. Test nearly spherical, outline round, wall porcelaneous, surface smooth; chambers inflated, biconvex, coiling involute, biloculine chamber arrangement; periphery rounded; aperture terminal, oval, bordered by a thick lip, with a broad, flap-like tooth.
Pyrgo oblonga (d’Orbigny, 1839b)
Figure 20.5-6
1839b Biloculina oblonga d’Orbigny, p. 163, pl. 8, figs. 21-23.
2012 Pyrgo oblonga (d’Orbigny); Debenay, p. 117.
2012 Pyrgo oblonga (d’Orbigny); Milker and Schmiedl, p. 66, fig. 17.13.
Description. Test elongate, outline pyriform, more tapering at the apertural end, wall porcelaneous, imperforate, surface smooth; chambers inflated, biloculine chamber arrangement, coiling involute, peripheral margin subacutely rounded; aperture terminal, ovate to subelliptical, bordered by a rim, with a broad, bifid tooth.
Pyrgo williamsoni (Silvestri, 1923)
Figure 20.8
1923 Biloculina williamsoni Silvestri, p. 73.
2003 Pyrgo williamsoni (Silvestri); Murray, p. 17, pl. 4, figs. 7-8.
2010 Pyrgo williamsoni (Silvestri); Margreth, p. 103, pl. 9, fig. 6a-c.
Description. Test ovate, elongate, tapering at the apertural end, aboral end broad, wall porcelaneous, imperforate, surface smooth; chambers added obliquely, biloculine chamber arrangement, coiling involute, periphery subacutely rounded, sutures distinct, slightly depressed; aperture elliptical, with a simple bifid tooth.
Pyrgo sp.
Figure 20.7
Description. Test subspherical, aboral end rounded, apertural end broadly truncate, wall porcelaneous, imperforate, surface smooth; chambers biconvex, biloculine chamber arrangement; periphery subrounded; aperture elongate, curved, with a large flat tooth, bordered by a thick lip.
Genus TRILOCULINA d’Orbigny, 1826
Triloculina echinata d’Orbigny, 1826
Figure 20.9-10
1826 Triloculina echinata d’Orbigny, p. 300, no. 14.
1999 Triloculina echinata d’Orbigny; Nigam and Khare, p. 292, pl. 3, fig. 5.
Description. Test subovate, somewhat broad than long, wall calcareous, imperforate, surface ornamented with regular rows of ridges; chambers slightly inflated, arranged in a triloculine plan; peripheral margin highly carinate, sutures slightly depressed; aperture terminal, round on a slightly produced neck, bordered by a distinct lip, with a short bifid tooth.
Triloculina insignis (Brady, 1881)
Figure 20.11-12
1881 Miliolina insignis Brady, p. 45.
1999 Triloculina insignis (Brady); Nigam and Khare, p. 292, pl. 3, fig. 4.
2014 Triloculina insignis (Brady); Panchang and Nigam, pl. 13, fig. 3a-c.
Description. Test subcircular, wall calcareous, imperforate, surface ornamented with many longitudinal costae; chambers inflated, arranged in a triloculine plan; peripheral margin carinate, periphery subrounded, sutures depressed; aperture terminal, circular opening on a short neck, bordered by a thin, slightly everted rim, with a prominent bifid tooth raised to be seen above the periphery of the aperture.
Triloculina oblonga (Montagu, 1803)
Figure 20.13-14
1803 Vermiculum oblongum de Montagu, p. 522, pl. 14, fig. 9.
2003 Triloculina oblonga (Montagu); Javaux and Scott, p. 23, fig. 5.16-17.
2006 Triloculina oblonga (Montagu); Oflaz, p. 176, pl. 4, fig. 5.
2012 Triloculina oblonga (Montagu); Milker and Schmiedl, p. 67, fig. 17.19-20.
Description. Test oblong, basal end broad, apertural end narrow, lateral view ovate, wall calcareous, imperforate, surface smooth; chambers inflated, arranged in a triloculine plan; periphery rounded, sutures distinct, depressed; aperture terminal, subrounded, on a slightly produced neck, bordered by a thick rim, with a simple, bifid tooth.
Triloculina schreiberiana d’Orbigny, 1839b
Figure 20.15
1839b Triloculina schreiberiana d’Orbigny, p. 174, pl. 9, figs. 20-22.
1997 Triloculina schreiberiana d’Orbigny; Cherif et al., p. 268, pl. 5, figs. 13-14.
Description. Test small, side view subovate, wall calcareous, imperforate, surface smooth; chambers distinct and broad, final chambers slightly inflated, arranged in triloculine plan; periphery broadly rounded; aperture terminal, large, subrounded opening, bordered by a slightly everted lip, with a slightly raised, short, distinct bifid tooth.
Triloculina terquemiana (Brady, 1884)
Figure 20.16-17
1884 Miliolina terquemiana Brady, p. 166, pl. 114, fig. 1a-b.
1999 Triloculina terquemiana (Brady); Nigam and Khare, p. 292, pl. 3, fig. 7.
2007 Triloculina terquemiana (Brady); Talib and Farroqui, p. 18, pl. 1, fig. 15a-b.
2012 Triloculina terquemiana (Brady); Debenay, p. 138.
Description. Test elongate, side view ovate, cross section subtriangular with smooth angles, wall calcareous, imperforate, surface ornamented with long costae; chambers distinct, arranged in triloculine form; peripheral margins somewhat convex; aperture slightly produced, narrow, elongate opening, bordered by a lip, with a long tooth bifid at the tip.
Triloculina tricarinata d’Orbigny, 1826
Figure 20.18
1826 Triloculina tricarinata d’Orbigny, p. 299, pl. 1, fig. 8.
2010 Triloculina trigonula (Lamarck); Margreth, p. 103, pl. 11, fig. 2a-c.
2012 Triloculina tricarinata d’Orbigny; Debenay, p. 138.
2012 Triloculina tricarinata d’Orbigny; Milker and Schmiedl, p. 68, fig. 17.23-24.
Description. Test triangular, slightly longer than wide, wall porcelaneous, imperforate, surface smooth; chambers visible, arranged in triloculine form, peripheral margins carinate, sutures distinct; aperture subrounded, arch-shaped opening bordered by a thick rim, with a short tooth fixed at the base.
Triloculina trigonula (Lamarck, 1804)
Figure 21.1
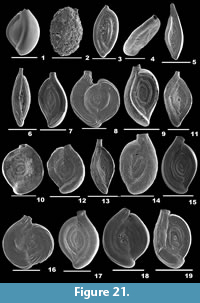 1804 Miliolites trigonula Lamarck, p. 351, pl. 17, fig. 4a-c.
1804 Miliolites trigonula Lamarck, p. 351, pl. 17, fig. 4a-c.
2006 Triloculina trigonula (Lamarck); Oflaz, p. 177, pl. 4, fig. 4.
2007 Triloculina trigonula (Lamarck); Talib and Farroqui, p. 19, pl. 1, fig. 16a-b.
2010 Triloculina trigonula (Lamarck); Margreth, p. 103, pl. 11, fig. 2a-c.
2012 Triloculina trigonula (Lamarck); Debenay, p. 138.
2014 Triloculina trigonula (Lamarck); Panchang and Nigam, pl. 13, fig. 7a-b.
Description. Test subtriangular, slightly longer than broad, wall calcareous, imperforate, surface smooth; chambers inflated, chamber arrangement triloculine; periphery broadly convex with smooth angles, sutures distinct; aperture terminal, narrow, elongate opening, bordered by a thick rim, with a broad, bifid tooth.
Subfamily SIGMOILOPSINAE Vella, 1957
Genus SIGMOILOPSIS Finlay, 1947
Sigmoilopsis schlumbergeri (Silvestri, 1904)
Figure 21.2
1904 Sigmoilina schlumbergeri Silvestri, p. 267, pl. 7, figs. 12-14; p. 481, text-fig. 6; p. 482, text-fig. 7.
2010 Sigmoilopsis schlumbergeri (Silvestri); Margreth, p. 104, pl. 11, fig. 5a-c.
2012 Sigmoilopsis schlumbergeri (Silvestri); Milker and Schmiedl, p. 69, fig. 18.7-8.
2014 Sigmoilopsis schlumbergeri (Silvestri); Panchang and Nigam, pl. 14, figs. 12a-c, 13a-c.
Description. Test oval, wall porcelaneous, outer layer composed of agglutinated material, surface rough; chambers obscured, chamber arrangement sigmoidal initially, later planispirally arranged; periphery broadly rounded, sutures indistinct; aperture terminal, circular on a non-agglutinated, short neck, bordered by a weak rim, with a short tooth.
Genus SPIROSIGMOILINA Parr, 1942
Spirosigmoilina tenuis (Cžjžek, 1848)
Figure 21.3-4
1848 Quinqueloculina tenuis Cžjžek, p. 149, pl. 13, figs. 31-34.
2014 Sigmoilinita tenuis (Cžjžek); Panchang and Nigam, pl. 14, fig. 3a-c.
Description. Test elongate, compressed, wall calcareous, translucent, surface smooth; chambers sigmoidal initially, later arranged in quinqueloculine plan, periphery broadly rounded, sutures very distinct and depressed; aperture terminal, small, circular at the end of a short neck.
Family SPIROLOCULINIDAE Wiesner, 1920
Genus INAEQUALINA Łuczkowska, 1971
Inaequalina affixa (Terquem, 1878)
Figure 21.5
1878 Spiroloculina affixa Terquem, p. 55, pl. 5, fig. 13a-c.
2012 Inaequalina affixa (Terquem); Debenay, p. 108.
2014 Inaequalina affixa (Terquem); Panchang and Nigam, pl. 6, fig. 7a-c.
Description. Test elongate, flat on one side, concave on the other, with tapering ends, surface smooth and glossy; chambers planispirally arranged, rapidly increasing in size as added; periphery carinate; aperture rectangular at the end of a flat and compressed extension of the final chamber without tooth.
Genus SPIROLOCULINA d’Orbigny, 1826
Spiroloculina antillarum d’Orbigny, 1839b
Figure 21.6
1839b Spiroloculina antillarum d’Orbigny, p. 166, pl. 9, figs. 3-4.
2012 Spiroloculina antillarum d’Orbigny; Debenay, p. 132.
Description. Test elongate, twice as long as broad, wall calcareous, imperforate, surface adorned with continuous, long costae throughout; chambers slightly biconcave, ovate in cross section; periphery carinate, sutures distinct, well depressed; aperture terminal, round on a long neck, bordered by a lip, with a small tooth.
Spiroloculina communis Cushman and Todd, 1944
Figure 21.7
1944 Spiroloculina communis Cushman and Todd, p. 63, pl. 9, figs. 4-5, 7-8.
1999 Spiroloculina communis Cushman and Todd; Nigam and Khare, p. 288, pl. 1, fig. 20.
2001 Spiroloculina communis Cushman and Todd; Szarek, p. 101, pl. 11, figs. 6-7.
2012 Spiroloculina communis Cushman and Todd; Debenay, p. 133.
2013 Spiroloculina communis Cushman and Todd; Elakkiya and Manivannan, p. 885, fig. 7.2-3.
2014 Spiroloculina communis Cushman and Todd; Gandhi and Nathan, p. 924, pl. 1, fig. 2a.
Description. Test robust, outline oval, wall porcelaneous, imperforate, surface smooth; chambers equally wide, slightly produced at both the ends; periphery convex and truncated, sutures thin, distinct and slightly depressed; aperture terminal, rounded on a small cylindrical neck, bordered by a lip, with two teeth placed opposite to each other.
Spiroloculina costifera Cushman, 1917
Figure 21.8
1917 Spiroloculina costifera Cushman, p. 34, pl. 6, figs. 1-3.
2014 Spiroloculina costifera Cushman; Babu et al., p. 79, pl. 1, fig. 6.
2014 Spiroloculina costifera Cushman; Gandhi and Nathan, p. 924, pl. 1, fig. 3a.
Description. Test large, subcircular, compressed laterally, wall porcelaneous, imperforate, surface smooth, final chambers ornamented with long costae; chambers arcuate, final chamber slightly detached from the earlier chambers; peripheral margin highly carinate, sutures distinct, slightly depressed; aperture terminal, rounded at the end of a carinate neck, bordered by a rim with a bifid tooth.
Spiroloculina depressa d’Orbigny, 1826
Figure 21.9
1826 Spiroloculina depressa d’Orbigny; p. 298, no.1.
2001 Spiroloculina depressa d’Orbigny; Szarek, p. 101, pl. 11, fig. 17.
2006 Spiroloculina depressa d’Orbigny; Oflaz, pp. 151-152, pl. 11, fig. 6.
Description. Test robust, wall porcelaneous, imperforate, surface smooth; chambers arcuate, gradually increasing in size as added, opposite sides of the chambers very concave; peripheral margin convex, sutures distinct and very much depressed; aperture terminal, circular, at the end of a thick neck, with a simple tooth, bordered by a lip.
Spiroloculina excavata d’Orbigny, 1846
Figure 21.10
1846 Spiroloculina excavata d’Orbigny, p. 271, pl. 16, figs. 19-21.
2002 Spiroloculina excavata d’Orbigny; Kaminski, Aksu, Box, Hiscott, Filipescu and Al-Salameen, p. 170, pl. 1, fig. 11.
2003 Spiroloculina excavata d’Orbigny; Murray, p. 17, fig. 4.13-14.
2005 Spiroloculina excavata d’Orbigny; Debenay, Millet and Angelidis, p. 334, pl. 2, fig. 14.
2012 Spiroloculina excavata d’Orbigny; Milker and Schmiedl, p. 50, fig. 13.3-4.
Description. Test subrounded, wall porcelaneous, imperforate, surface smooth; chambers strongly concave, spiroloculine chamber arrangement; peripheral margin truncate; aperture terminal, elliptical, bordered by a rim, with a short bifid tooth.
Spiroloculina eximia Cushman, 1922a
Figure 21.11
1922a Spiroloculina eximia Cushman, p. 61, pl. 11, fig. 2.
1999 Spiroloculina indica Cushman and Todd; Nigam and Khare, p. 288, pl. 1, fig. 23.
2012 Spiroloculina eximia Cushman; Debenay, p. 133.
Description. Test elongate, outline ellipsoidal, wall porcelaneous, surface smooth; opposite side of the chambers concave, extending towards both the ends; periphery convex, sutures distinct and depressed; aperture terminal, at the end of a cylindrical neck with a small bifid tooth.
Spiroloculina henbesti Petri, 1955
Figure 21.12
1955 Spiroloculina henbesti Petri, p. 82, figs. 4-6.
2002 Spiroloculina henbesti Petri; Gandhi, Rajamanickam and Nigam, p. 52, pl. 1, fig. 3.
Description. Test broad, subcircular, slightly compressed laterally, wall porcelaneous, imperforate, surface smooth; chambers wide, gradually increasing in size as added, final chambers inflated, coiling evolute; periphery rounded, sutures very distinct and depressed; aperture terminal, subrounded, at the end of a very short neck, bordered by a rim, with a simple, bifid tooth.
Spiroloculina indica Cushman and Todd, 1944
Figure 21.13
1944 Spiroloculina indica Cushman and Todd, p. 71, pl. 9, fig. 32a-b.
1999 Spiroloculina indica Cushman and Todd; Nigam and Khare, p. 288, pl. 1, fig. 24.
2007 Spiroloculina indica Cushman and Todd; Talib and Farroqui, p. 18, pl. 1, fig. 6a-b.
Description. Test elongate, asymmetrical, slightly concavo-convex, surface ornamented by fine, longitudinal striations; chambers visible bilaterally, coiling evolute; sutures depressed; aperture round on a long neck, bordered by a rim, with a short bifid tooth.
Spiroloculina inflata Terquem, 1882
Figure 21.14
1882 Spiroloculina inflata Terquem, p. 193.
1987 Spiroloculina cf. S. inflata Terquem; Bhalla and Gaur, p. 123, pl. 1, fig. 5.
Description. Test robust, outline broadly oval, wall porcelaneous, imperforate, surface rough due to minute depressions; chambers inflated, final chambers broader, rapidly increasing in size as added; periphery broadly rounded, sutures distinct, depressed; aperture circular, at the end of a stout neck, bordered by a distinct rim, with a simple, bifid tooth.
Spiroloculina lucida Cushman and Todd, 1944
Figure 21.15
1944 Spiroloculina lucida Cushman and Todd, p. 70, pl. 9, figs. 30-31.
1961 Spiroloculina lucida Cushman and Todd; Huang, p. 85, pl. 2, fig. 31.
Description. Test broad, ovate, side view fusiform, wall porcelaneous, imperforate, surface smooth; chambers many, margins slightly convex, periphery broadly rounded, sutures distinct and depressed; aperture terminal, subcircular on a neck, bordered by a thick rim, with a simple bifid tooth.
Spiroloculina neocircularis McCulloch, 1977
Figure 21.16
1977 Spiroloculina neocircularis McCulloch, p. 544, pl. 228, fig. 14.
2012 Spiroloculina cf. S. neocircularis McCulloch; Debenay, p. 134.
Description. Test nearly circular, broader than long, wall calcareous, imperforate, surface ornamented with oblique ribs, more prominent on the final chambers and the neck; final chambers slightly inflated; periphery broadly rounded and carinate, sutures distinct, slightly depressed; aperture terminal, subrounded, with a simple tooth, bordered by a thin lip.
Spiroloculina nitida d’Orbigny, 1826
Figure 21.17
1826 Spiroloculina nitida d’Orbigny, p. 298.
2002 Spiroloculina nitida d’Orbigny; Gandhi, Rajamanickam and Nigam, p. 52, pl. 1, fig. 5.
2014 Spiroloculina nitida d’Orbigny; Gandhi and Nathan, p. 925, pl. 2, fig. 1a.
Description. Test ovate, side view fusiform, wall porcelaneous, imperforate, surface smooth; chambers gently concave on the inner side, periphery broadly rounded, sutures distinct and depressed; aperture terminal, circular on a neck, bordered by a thick rim, with a simple bifid tooth.
Spiroloculina orbis Cushman, 1921
Figure 21.18
1921 Spiroloculina orbis Cushman, p. 403, pl. 83, fig. 3.
2009 Spiroloculina orbis Cushman; Devi and Rajashekhar, p. 24, fig. 2j.
2013 Spiroloculina orbis Cushman; Elakkiya and Manivannan, p. 885, fig. 7.4-5.
2014 Spiroloculina orbis Cushman; Gandhi and Nathan, p. 924, pl. 1, fig. 4a.
Description. Test broadly ovate, planispiral, wall porcelaneous, imperforate, surface smooth; final chambers of uniform size overlapping the initial ones; periphery broadly rounded, peripheral margin carinate, sutures distinct, somewhat depressed, slightly arcuate; aperture terminal, round at the end of a carinate neck, bordered by a rim, with a small bifid tooth.
Spiroloculina parvula Chapman, 1902
Figure 22.1-2
1902 Spiroloculina parvula Chapman, pl. 36, fig. 5.
2014 Spiroloculina parvula Chapman; Panchang and Nigam, pl. 6, fig. 20a-b.
Description. Test large, outline ovate, wall porcelaneous, surface ornamented with prominent, long ribs; chambers slightly inflated; periphery highly carinate, sutures distinct; aperture terminal, round at the end of the neck, bordered weakly by a thin rim, with a small bifid tooth.
Spiroloculina sp.
Figure 21.19
Description. Test ovate, lateral view fusiform, wall porcelaneous, imperforate, surface smooth; chambers angular in cross section, slightly convex; peripheral margin truncate, sutures distinct and depressed; aperture terminal, round on a slightly produced neck, bordered by a distinct rim, with a bifid tooth.
Spiroloculina subimpressa Parr, 1950
Figure 22.3-4
 1950 Spiroloculina subimpressa Parr, p. 291, pl. 6, figs. 12-13.
1950 Spiroloculina subimpressa Parr, p. 291, pl. 6, figs. 12-13.
2012 Spiroloculina subimpressa Parr; Debenay, p. 135.
2013 Spiroloculina subimpressa Parr; Jones, p. 35, pl. 1, fig. 14.
2014 Spiroloculina subimpressa Parr; Panchang and Nigam, pl. 7, fig. 2a-b.
Description. Test broad, subrounded, wall porcelaneous, imperforate, surface smooth; chambers biloculine, coiling evolute; peripheral margin angular and truncated, sutures distinct; aperture terminal, round at the end of short neck, bordered by a lip, with a small bifid tooth.
Family MILIOLIDAE Ehrenberg, 1839
Miliolid sp.
Figure 22.5
2012 Miliolid sp., Milker and Schmiedl, p. 51, fig. 13.14-15.
Description. Test circular, lateral view rounded, strongly biconvex in the central portion, wall calcareous, imperforate, surface smooth with many fine striations; peripheral margin carinate, periphery rounded; aperture terminal, oval at the end of a neck, bordered by a rim, with a short, simple tooth.
Superfamily SORITOIDEA Ehrenberg, 1839
Family PENEROPLIDAE Schultze, 1854
Genus PENEROPLIS de Montfort, 1808
Peneroplis pertusus (Forskål, 1775)
Figure 22.6
1775 Nautilus pertusus Forskål and Niebuhr, p. 125, no. 65.
2006 Peneroplis pertusus (Forskål); Oflaz, p. 185, pl. 4, fig. 10; pl. 5, figs. 1-3; pl. 11, fig. 8.
2008 Peneroplis pertusus (Forskål); Yalçin, Meriç, Avşar, Tetiker, Barut, Yilmaz and Dinçer, p. 370, pl. 3, figs. 6-10.
2012 Peneroplis pertusus (Forskål); Debenay, p. 113.
2012 Peneroplis pertusus (Forskål); Milker and Schmiedl, p. 71, fig. 18.11-12.
2014 Peneroplis pertusus (Forskål); Panchang and Nigam, pl. 15, fig. 3a-b.
Description. Test flattened, umbilical area compressed, wall porcelaneous, surface ornamented with parallel costae perpendicular to the sutures; chambers many, initially planispirally arranged, later uniserially arranged, gradually increasing in size; periphery subrounded, sutures slightly depressed; aperture multiple, terminal, irregular slits in juveniles, a row of rectangular openings in adults.
Peneroplis planatus (Fichtel and Moll, 1798)
Figure 22.7
1798 Nautilus planatus Fichtel and Moll, p. 91, pl. 16, figs. a-c, i; p. 93, pl. 16, figs. d-f; p. 94, pl. 16, figs. g, h.
2006 Peneroplis planatus (Fichtel and Moll); Oflaz, p. 186, pl. 5, figs. 4-5, pl. 11, fig. 9.
2008 Peneroplis planatus (Fichtel and Moll); Yalçin, Meriç, Avşar, Tetiker, Barut, Yilmaz, and Dinçer, p. 370, pl. 3, figs. 11-16.
2012 Peneroplis planatus (Fichtel and Moll); Debenay, p. 114.
Description. Test flattened, wall calcareous, surface ornamented with numerous grooves placed at right angles to the sutures, umbilical region depressed, devoid of ornamentation; chambers distinct, coiling involute and planispiral chamber arrangement initially, final whorl spreading out later, rapidly increasing in size as added; sutures somewhat depressed; aperture a row of irregular, circular openings.
Genus MONALYSIDIUM Chapman, 1900
Monalysidium confusa (McCulloch, 1977)
Figure 22.8
1977 Spirolina confusa McCulloch, p. 231, pl. 100, fig. 9.
2012 Monalysidium confusa (McCulloch); Debenay, p. 111.
Description. Test robust, compressed, wall calcareous, surface ornamented with faint ribs, pitted in regular rows; chambers involute and planispirally arranged initially, later uncoiled, final chambers more inflated, periphery rounded, sutures radial, slightly arcuate initially, nearly horizontal later; aperture centred, areal, bordered by a lip, with teeth.
Superfamily NUBECULARIOIDEA T. R. Jones, 1854
Family OPHTHALMIDIIDAE Weisner, 1920
Genus EDENTOSTOMINA Collins, 1958
Edentostomina cultrata (Brady, 1881)
Figure 22.9-10
1881 Miliolina cultrata Brady, p. 45.
2006 Edentostomina cultrata (Brady); Oflaz, p. 144, pl. 2, fig. 3.
2012 Edentostomina cultrata (Brady); Debenay, p. 106.
Description. Test compressed, slender, outline ovate; chambers long and narrow, later chambers inflated, planispirally arranged, rapidly increasing in size, one-half coil in length; periphery highly carinate; aperture simple, terminal, oval at the end of a neck, rimmed by a reversed lip without a tooth.
Edentostomina rupertiana (Brady, 1881)
Figure 22.11-12
1881 Miliolina rupertiana Brady, p. 46.
1994 Edentostomina rupertiana (Brady); Jones, p. 23, pl. 7, figs. 7-12.
1994 Rupertianella rupertiana (Brady); Loeblich and Tappan, p. 60, pl. 106, figs. 1-14.
Description. Test elongate, compressed, wall calcareous, surface ornamented with long costae with rows of small circular openings in between; chambers visible, chamber arrangement triloculine; peripheral margin acute, sutures distinct, depressed; aperture terminal, ovate, bordered weakly by a very thin lip.
Family FISCHERINIDAE Millett, 1898
Subfamily FISCHERININAE Millett, 1898
Genus PLANISPIRINELLA Weisner, 1931
Planispirinella exigua (Brady, 1879b)
Figure 22.13-14
1879b Hauerina exigua Brady, p. 267.
2006 Planispirinella exigua (Brady); Oflaz, p. 141, pl. 1, figs. 10-11; pl. 11, fig. 3.
2012 Planispirinella exigua (Brady); Debenay, p. 114.
Description. Test disc-shaped, flattened, central part slightly convex, outline circular, surface smooth, slightly convex at the growth lines; chambers rapidly increasing in size as added, coiling evolute; periphery rounded, sutures depressed; aperture terminal, at the end of the tubular chamber, long, slit-like, cribate trematophore.
Superfamily ALVEOLINOIDEA Ehrenberg, 1839
Family ALVEOLINIDAE Ehrenberg, 1839
Genus ALVEOLINELLA H. Douvillé, 1907
Alveolinella quoii (d’Orbigny, 1826)
Figure 22.15
1826 Alveolina quoii d’Orbigny, p. 307, pl. 17, figs. 11-13.
2012 Alveolinella quoii (d’Orbigny); Debenay, p. 102.
Description. Test large, elongate, side view fusiform, wall calcareous, imperforate, surface adorned with numerous costae; chambers narrow, many in adults, planispirally arranged with gradual increase in size as added, thickening at both ends, coiling involute; apertural face with a series of openings placed in many rows.
Genus BORELIS de Montfort, 1808
Borelis melo (Fichtel and Moll, 1798)
Figure 22.16
1798 Nautilus melo Fichtel and Moll, p. 118.
2013 Borelis melo (Fichtel and Moll); Jones, p. 35, pl. 1, fig. 16.
Description. Test subrounded, side view spherical, wall calcareous, imperforate, surface ornamented with horizontal ribs, faint pustules in between low undulating ridges placed along the chamberlet sutures; chambers planispirally arranged, many chamberlets in adults, coiling involute; periphery rounded, sutures well depressed; aperture a series of subrounded to irregularly quadrangular openings placed in a row on the apertural face.
ACKNOWLEDGEMENTS
The authors are thankful to the Ministry of Earth Sciences for their financial support during sample collection. We extend our thanks to the staff members of National Institute of Technology, Chennai for their assistance during the cruises. We are also grateful for the Central Instrumentation Facility in Pondicherry University for helping us avail the SEM images of foraminifera. The present study is funded by University Grants Commission, New Delhi through a Ph.D. grant.
REFERENCES
Albani, A.D. 1981. Pleistocene foraminifera from Botany Bay, New South Wales. Alcheringa, 5:147-160. https://doi.org/10.1080/03115518108565428
Araújo, H.A.B. and Machado, A.J. 2008. Benthic foraminifera associated with the South Bahia Coral Reefs, Brazil. Journal of Foraminiferal Research, 38(1):23-38. https://doi.org/10.2113/gsjfr.38.1.23
Asano, K. 1936. Pseudononion, a new genus of foraminifera found in Muracks-mura, Kamakura-gori, Kanagawa Prefecture. Journal of the Geological Society of Japan, 43:347-348.
Asano, K. 1944. Hanzawaia, a new genus of foraminifera, from the Pliocene of Japan (resume). Journal of the Geological Society of Japan, 51(606):97-99.
Asano, K. 1950. Illustrated Catalogue of Japanese Tertiary Smaller Foraminifera, Part III, Textulariidae. Hosokawa Printing Company, Tokyo.
Asano, K. and Uchio, T. 1951. Illustrated Catalogue of Japanese Tertiary Smaller Foraminifera, Part XIV, Rotaliidae, Pliocene. Hosokawa Printing Company, Tokyo.
Babu, K., Prabhakaran, R., Kumar, R.S., Selvaraj, B., and Subramanian, P. 2014. Taxonomy and distribution of recent benthic foraminifera from the inner shelf of Gulf of Mannar, off Tuticorin, south east coast of India. Journal of Environment and Earth Science, 4(22):75-85.
Baccaert, J. 1987. Distribution Patterns and Taxonomy of Benthic Foraminifera in the Lizard Island Reef Complex, Northern Great Barrier Reef, Australia. Unpublished PhD Thesis, Universite de Liege, C.A.P.S. - Laboratoire de Biosédimentologie, Liège, Belgium.
Bailey, J.W. 1851. Microscopical examination of soundings made by the United States Coast Survey, off the Atlantic coast of the United States. Smithonian Contributions to Knowledge, 2(3):1-15.
Bandy, O.L. 1953. Ecology and paleoecology of some California foraminifera: Part 1, The frequency distribution of Recent Foraminifera off California; Part 2, Foraminiferal evidence of subsidence rates in the Ventura Basin. Journal of Paleontology, 27:161-203.
Bandy, O.L., Frerichs, W.E., and Vincent, E. 1967. Origin, development, and geologic significance of Neogloboquadrina Bandy, Frerichs, and Vincent, gen. nov. Contributions from the Cushman Laboratory for Foraminiferal Research, 18(4):152-157.
Banner, F.T. and Blow, W.H. 1960. The taxonomy, morphology and affinities of the genera included in the subfamily Hastigerininae. Micropaleontology, 6:19-31.
Banner, F.T. and Culver, S.J. 1978. Quaternary Haynesina n. gen. and Paleogene Protelphidium Haynes; their morphology, affinities and distribution. Journal of Foraminiferal Research, 8:177-207. https://doi.org/10.2113/gsjfr.8.3.177
Banner, F.T. and Hodgkinson, R.L. 1991. A revision of the foraminferal subfamily Heterostegininae. Revista Espaňola de Micropaleontolgia, 23:101-140.
Batsch, A.I.G.C. 1791. Sechs Kupfertafeln mit Cochylien des Seessandes, gezeichnet und gestochen von A.J.G.K. Batsch, Jena.
Bermúdez, P.J. 1952. Estudio sistematico de los foraminíferos rotaliformes. Boletín de Geologia Venezuela, 2(4):1-230.
Bermúdez, P.J. 1961. Contribución al estudio de las Globigerinidea de la region Caribe-Antillana (Paleoceno Reciente). Boletín de Geologia, Venezuela, Publicación Especial, 3:1119-1393.
Berthelin, G.1880. Memoire sur les foraminifères fossiles de l'etage Albien de Montdey (Doubs). Memoires de la Societé Geologique de France, 1:1-84.
Berthois, L. and Le Calvez, Y. 1959. Deuxième contribution à l'étude de la sédimentation dans le golfe de Gascogne. Revue des Travaux de l'Institut des Pêches Maritimes, 23(3):323-375.
Bhalla, S.N. and Gaur, K.N. 1987. Recent foraminifera from Colva beach sands, Goa. Journal of the Palaeontological Society of India, 32:122-130.
Blow, W.H. 1959. Age, correlation biostratigraphy of the upper Tocuyo (San Lorenzo) and Pozón Formations, Eastern Falcón, Venezuela. Bulletins of American Paleontology, 39(178):59-251.
Bornemann, J.G. 1855. Die mikroskopische Fauna des Septarienthones von Hermsdorf bei Berlin. Zeitschrift der Deutschen Geologischen Gesellschaft, 7:307-371.
Brady, H.B. 1870. Part II. Analysis and description of foraminifera. Annals and Magazine of Natural History, 4:273-309. https://doi.org/10.1080/00222937008696252
Brady, H.B. 1876. On some foraminifera from the Loo Choo Islands. Proceedings of the Royal Irish Academy, 2(2):589.
Brady, H.B. 1877. Supplementary note on the foraminifera of the Chalk(?) of the New Britain group. Geological Magazine, 4:534-536.
Brady, H.B. 1878. On the reticularian and radiolarian Rhizopoda (Foraminifera and Polycystina) of the North Polar Expedition of 1875-76. Annals and Magazine of Natural History, 5(1):425-550. https://doi.org/10.1080/00222937808682361
Brady, H.B. 1879a. Notes on some reticularian Rhizopoda of the Challenger expedition, Part 1. Quaternary Journal of the Microscopical Science, 19:20-63.
Brady, H.B. 1879b. Notes on some reticularian Rhizopoda of the Challenger expedition, Part 2. Quaternary Journal of the Microscopical Science, 19:261-299.
Brady, H.B. 1881. Notes on some reticularian Rhizopoda of the Challenger expedition, Part 3. Quaternary Journal of the Microscopical Science, 21:31-71.
Brady, H.B. 1884. Report on the foraminifera dredged by H.M.S. Challenger during the years 1873-1876. Report on the Scientific Results of the Voyage of H.M.S. Challenger During the Years 1873-1876, Zoology, 9:1-814.
Brönnimann, P. 1951. Globigerinita naparimaensis n. gen., n. sp. from the Miocene of Trinidad, B.W.I. Contributions from the Cushman Laboratory for Foraminiferal Research, 2:16-18.
Brünnich, M.T. 1772. Zoologiae fundamenta praelectionibus academicis. Accomodata grunde I dyeloeren. Transactions of the Linnean Society of London, 7:241.
Carpenter, W.B., Parker, W.K., and Jones, T.R. 1862. Introduction to the Study of Foraminifera, Volume 32. The Ray Society, London.
Carter, H.J. 1880. Report on the specimens dredged from the Gulf of Mannar. Annual Magazine of National History, 5(5):437-457.
Cavalier-Smith, T. 1981. Eukaryote kingdoms: Seven or nine? BioSystems, 14:461-481. https://doi.org/10.1016/0303-2647(81)90050-2
Cavalier-Smith, T. 2002. The phagotrophic origin of eukaryotes and phylogenetic classification of Protozoa. International Journal of Systematic and Evolutionary Microbiology, 52:297-354. https://doi.org/10.1099/00207713-52-2-297
Cavalier-Smith, T. 2010. Kingdoms Protozoa and Chromista and the eozoan root of the eukaryotic tree. Biology Letters, 6:342-345. https://doi.org/10.1098/rsbl.2009.0948
Chapman, F. 1900. On some new and interesting foraminifera from the Funafuti Atoll, Ellice Islands. Journal of the Linnean Society of London, Zoology, 28:1-27. https://doi.org/10.1111/j.1096-3642.1900.tb01742.x
Chapman, F. 1902. On the foraminifera collected round the Funafuti Atoll from shallow and moderately deep water. Journal of Linnean Society of London, Zoology, 28:379-417. https://doi.org/10.1111/j.1096-3642.1902.tb01547.x
Chapman, F. 1907. Recent foraminifera of Victoria: Some littoral gatherings. Journal Quekett Microscopical Club, 2(10):117-146.
Chapman, F. and Parr, W.J. 1935. Foraminifera and Ostracoda from soundings made by the trawler "Bonthorpe" in the Great Australian Bight. Royal Society of Western Australia, Journal, 21:1-6.
Chapman, F. and Parr, W.J. 1937. Foraminifera. Australasian Antarctic Expedition 1911-14 under the Leadership of Sir Douglas Mawson. Scientific Reports, C, Zoology and Botany, 1(2):1-190.
Chapman, F., Parr, W.J., and Collins, A.C. 1934. Tertiary foraminifera of Victoria, Australia-The Balcombian deposits of Port Phillip, Part III. Journal of the Linnaean Society of London, Zoology, 38:553-577. https://doi.org/10.1111/j.1096-3642.1934.tb00996.x
Cherif, O.H. 1970. Die Miliolacea der Westküste von Naxos (Griechenland) und ihre Lebensbereiche. Dissertation Facultät Natur- und Geisteswissenschaften. Technischen Universität Clausthal, Clausthal, Germany.
Cherif, O.H., Al-Ghadban, A., and Al-Rifaiy, I.A. 1997. Distribution of foraminifera in the Arabian Gulf. Micropaleontology, 43(3):253-280. https://doi.org/10.2307/1485827
Cifelli, R. 1961. Globigerina incompta, a new species of pelagic foraminifera from the North Atlantic. Contributions from the Cushman Laboratory for Foraminiferal Research, 12:83-86.
Collins, A.C. 1958. Foraminifera. Great Barrier Reef Expedition 1928-29, Scientific Reports, 6(6):335-437.
Colom, G. 1956. Los foraminíferos del Burdigaliense de Mallorace. Memorias de la Real Academia del Ciencias y Artes de Barcelona, 32:7-140.
Costa, O.G. 1855. Foraminiferi fossili della marna blu del Vaticano. Memorie della R. Accademia delle Scienze Napoli, 2:113-126.
Culver, S.J., Mallinson, D.J., Corbett, D.R., Leorri, E., Rouf, A.A., Shazili, N.A.Md., Yaacob, R., Whittaker, J.E., Buzas, M.A., and Parham, P.R. 2012. Distribution of foraminifera in the Setiu estuary and lagoon, Terengganu, Malaysia. Journal of Foraminiferal Research, 42(2):109-133. https://doi.org/10.2113/gsjfr.42.2.109
Cushman, J.A. 1910. A Monograph of the Foraminifera of the North Pacific Ocean, Part 1: Astrorhizidae and Lituolidae. Bulletin of the United States National Museum, 71(1):1-134.
Cushman, J.A. 1911. A monograph of the foraminifera of the North Pacific Ocean, Part 2: Textularidae. Bulletin of the United States National Museum, 71(2):1-108.
Cushman, J.A. 1913. A monograph of the foraminifera of the North Pacific Ocean, Part 3, Lagenidae. Bulletin of the United States National Museum, 71(3):1-122.
Cushman, J.A. 1915. A monograph of the foraminifera of the North Pacific Ocean, Part 5: Rotaliidae. Bulletin of the United States National Museum, 71(5):1-87.
Cushman, J.A. 1917. A monograph of the foraminifera of the North Pacific Ocean, Part 6: Miliolidae. Bulletin of the United States National Museum, 71(6):1-108.
Cushman, J.A. 1921. Foraminifera from the north coast of Jamaica. Proceedings of the United States Natural History Museum, 59:47-82. https://doi.org/10.5479/si.00963801.59-2360.47
Cushman, J.A. 1922a. The foraminifera of the Atlantic Ocean, Part 3: Textularidae. Bulletin of the United States National Museum, 104(3):1-143.
Cushman, J.A. 1922b. The foraminifera of the Byram Calcareous Marl at Byram, Mississipi, p. 82-281. In White, D. (ed.), Shorter Contributions to General Geology, US Geological Survey Professional Paper 129E. US Geological Survey, Washington.
Cushman, J.A. 1923. The foraminifera of the Atlantic Ocean, Part 4: Lagenidae. Bulletin of the United States National Museum, 104(4):1-129.
Cushman, J.A. 1924. Samoan Foraminifera. Publications of the Carnegie Institution of Washington, Department of Marine Biology Papers, 21(342):1-75.
Cushman, J.A. 1926. Foraminifera of the typical Monterey of California. Contributions from the Cushman Laboratory for Foraminiferal Research, 2:53-69.
Cushman, J.A. 1927. An outline of a reclassification of the foraminifera. Contributions from the Cushman Laboratory for Foraminiferal Research, 3:1-105.
Cushman, J.A. 1928. Foraminifera: Their classification and economic use. Special publications of the Cushman Laboratory for Foraminiferal Research, 1:1-401.
Cushman, J.A. 1931. The foraminifera of the Atlantic Ocean, Part 8: Rotaliidae, Amphisteginidae, Calcarinidae, Cymbalporettidae, Globorotallidae, Anomalinidae, Planorbulinidae, Pupertiidae and Homotremidae. Bulletin of the United States National Museum, 104(8):1-179.
Cushman, J.A. 1933. The foraminifera of the tropical Pacific collections of the "Albatross," 1899-1900, Part 2: Lagenida to Alveolinellidae. Bulletin of the United States National Museum, 161:1-79.
Cushman, J.A. 1936. New genera and species of the families Verneuilinidae and Valvulinidae and of the subfamily Virgulininae. Special Publications of the Cushman Laboratory for Foraminiferal Research, 6:1-71.
Cushman, J.A. 1937. A monograph of the foraminiferal family Valvulinidae. Special Publications of the Cushman Laboratory for Foraminiferal Research, 8:1-210.
Cushman, J.A. 1939a. A monograph of the foraminiferal family Nonionidae. Professional Papers U.S. Geological Survey, 191:1-100.
Cushman, J.A. 1939b. Notes on some Pliocene foraminifera described by Schwager from Car Nicobar. Journal of the Geological Society of Japan, 46:149-154.
Cushman, J.A. 1944. Foraminifera from the shallow water of the New England coast. Special Publications of the Cushman Laboratory for Foraminiferal Research, 12:1-37.
Cushman, J.A. 1946. The genus Hauerina and its species. Contributions from the Cushman Laboratory for Foraminiferal Research, 22:2-15.
Cushman, J.A. and Brönnimann, P. 1948. Additional new species of arenaceous foraminifera from shallow waters of Trinidad. Contributions from the Cushman Laboratory for Foraminiferal Research, 23:60-72.
Cushman, J.A. and Edwards, P.G. 1937. Astrononion a new genus of the foraminifera, and its species. Contributions from the Cushman Laboratory for Foraminiferal Research, 13:29-36.
Cushman, J.A. and Grant, H.S. 1927. Late Tertiary and Quaternary Elphidium of the west coast of North America. San Diego Society of Natural History, Transactions, 5(6):69-82.
Cushman, J.A. and LeRoy, L.W. 1939. Cribrolinoides, a new genus of the foraminifera, its development and relationships. Contributions from the Cushman Laboratory for Foraminiferal Research, 15:15-19.
Cushman, J.A. and Martin, L.T. 1935. A new genus of foraminifera, Discorbinella, from Monterey Bay, California. Contributions from the Cushman Laboratory for Foraminiferal Research, 11:89-90.
Cushman, J.A. and Moyer, D.A. 1930. Some recent foraminifera from off San Pedro, California. Contributions from the Cushman Laboratory for Foraminiferal Research, 6:49-62.
Cushman, J.A. and Ozawa, Y. 1928. An outline of a revision of the Polymorphinidae. Contributions from the Cushman Laboratory for Foraminiferal Research, 4:13-21.
Cushman, J.A. and Todd, R. 1942. The genus Cancris and its species. Contributions from the Cushman Laboratory for Foraminiferal Research, 18:72-94.
Cushman, J.A. and Todd, R. 1944. The genus Spiroloculina and its species. Special Publications of the Cushman Laboratory for Foraminiferal Research, 11:1-82.
Cushman, J.A., Todd, R., and Post, R.J. 1954. Recent foraminifera of the Marshall Islands, Bikini and nearby atolls, Part II, Oceanography (biologic). Professional Papers U.S. Geological Survey, 260-H:319-384.
Cžjžek, J. 1848. Beitrag zur Kenntnis der fossilen Foraminiferen des Wiener Beckens. Naturwissenschaftliche Abhandlungen, Wien, 2(1):137-150.
d’Archiac, E.J.A. and Haime, J. 1853. Description des Animaux Fossiles du Groupe Nummulitique de l’Inde: Précédé d’un Résumé Géologique et d’une Monographie des Nummulites. Gide et J. Baudry, Paris.
Dawson, J.W. 1860. Notice of Tertiary fossils from Labrador, Maine, etc., and remarks on the climate of Canada in the newer Pliocene or Pleistocene period. Canadian Natural Geology, 5:188-200.
Debenay, J.-P. 2009. Foraminifera, p. 255-280. In Amiard, C. and Rainbow, P.S. (eds.), Environmental Assessment of Estuarine Ecosystems: A Case Study. Taylor and Francis, Boca Raton.
Debenay, J.-P. 2012. A Guide to 1,000 Foraminifera from the Southwestern Pacific, New Caledonia. Publications Scientifiques du Muséum, Muséum National d’Histoire Naturelle, Paris.
Debenay, J.-P., Millet, B., and Angelidis, M.O. 2005. Relationships between foraminiferal assemblages and hydrodynamics in the Gulf of Kalloni, Greece. Journal of Foraminiferal Research, 35:327-343. https://doi.org/10.2113/35.4.327
de Blainville, H.M. 1827. Mémoire sur les Bélemnites, Considérés Zoologiquement et Géologiquement, F.G. Levrault, Strasbourg.
Defrance, J.L.M. 1822. Lenticulites, p. 425-453. In Cuvier, M.F. (ed.), Dictionnaire des Sciences Naturelles, 25 (1aa-1eo). Le Normant, Paris.
Defrance, J.L.M. 1824. Dictionnaire des Sciences Naturelles, 32. F.G. Levrault, Strasbourg.
Delage, Y. and Herouard, E. 1896. Traite de Zoologie Concrete. 1, La Cellule et les Protozoaires. Schleicher Fréres, Paris.
de Montfort, D. 1808. Conchyliologie Systematique et Classification Methodique des Coquilles, 1. F. Schoell, Paris.
Devi, S.G. and Rajashekhar, K.P. 2009. Intertidal foraminifera of India coast - scanning electron photomicrograph-illustrated catalogue. Journal of Threatened Taxa, 1(1):17-36. https://doi.org/10.11609/jott.o1977.17-36
d’Orbigny, A. 1826. Tableau methodique de la classe des cephalopodes. Annales des Sciences Naturelles, 7:96-314.
d’Orbigny, A. 1839a. Voyage dans l'Amérique Méridionale; Foraminifères. P. Bertrand, Strasbourg, Paris.
d’Orbigny, A. 1839b. Foraminiféres des Îles Canaries, p.119-146. In Barker-Webb, P. and Berthelot, S. (eds.), Histoire Naturelle des Îles Canaries, Volume 2, Part 2. Béthune, Paris.
d’Orbigny, A. 1839c. Foraminifères. iI de la Sagra R., Histoire Physique, Politique et Naturelle de l'Ile de Cuba. A. Bertrand, Paris.
d’Orbigny, A. 1846. Foraminiferes Fossiles Du Bassin Tertiaire De Vienne (Autriche). Gide et Comp, Libraires- Editeurs, Paris.
d’Orbigny, A. 1852. Prodome de Paléontologie Stratigraphique Universelle des Animaux Mollusques et Rayonnés Paissant Suite au Cours Élémentaire de Paléontologie et de Géologie Stratigraphiques, 3. Victor Masson, Paris.
Douvillé, H. 1907. Les calcairees à fusulines de l'Indo-Chine. Bulletin de la Société Géologique de France, 4(6):576-587.
Egger, J.G. 1893. Foraminiferen aus Meeresgrundproben, gelothet von 1874 bis 1876 von S.M. Sch. Gazelle. Abhandlungen der Bayrischen Akademie der Wissenschaften, Mathematisch-Physikalische Classe, 18:193-458.
Ehrenberg, C.G. 1838. Über dem blossen Auge unsichtbare Kalkthierchen und Kieselthierschen als Hauptbestandtheile der Kreidegebirge. Bericht über die zur Bekanntmachung geeigneten Verhandlungen der Königlichen Preußischen Akademie der Wissenschaften zu Berlin, 1838:192-200.
Ehrenberg, C.G. 1839. Uber die Bildung der Kreidefelsen und des Kreidemergels durch unsichtbare Organismen. Abhandlungen der königlichen Akademie der Wissenschaften zu Berlin, 1838:59-148.
Eimer, G.H.T. and Fickert, C. 1899. Die Artbildung und Verwandtschaft bei den Foraminiferen. Entwurf einer natürlichen Eintheilung derselben. Zeitschrift für Wissenschaftliche Zoologie, 65:599-708.
Elakkiya, P. and Manivannan, V. 2013. Recent benthic foraminifera from off the coast of Arkattuthurai (near Nagapattinam) south east coast of India. Indian Journal of Geo-Marine Sciences, 42(7):877-887.
El-Nakhal, H.A. 1983. Agglutinella, a new miliolid genus (Foraminiferida). Journal of Foraminiferal Research, 13(2):129-133. https://doi.org/10.2113/gsjfr.13.2.129
Fichtel, L.v. and Moll, J.P.C.v. 1798. Testacea Microscopica, Aliaque Minuta ex Generibus Argonauta et Nautilus, ad Naturam Picta et Descripta (Microscopische und andere kleine Schalthiere aus den Geschlechtern Argonaute und Schiffer). Camesina, Wien.
Figueroa, S., Marchant, M., Gigglio, S., and Ramírez, M. 2006. Benthic foraminifera of continental slope between Concepcion to Guaitecas Islands (36º- 44ºS), Chile (Foraminifera: Lagenina, Miliolina y robertinina), Gayana, 70(1):255-279.
Fleming, J. 1828. A History of British Animals Exhibiting the Descriptive Characters and Systematic Arrangement of the Genera and Species of Quadrupeds, Birds, Fishes, Mollusca and Radiata of the United Kingdom. Bell and Bradfute, Edinburgh.
Finlay, H.J. 1947. New Zealand foraminifera: Key species in stratigraphy. New Zealand Journal of Science and Technology, 28(5):259-292.
Fornasini, C. 1894. Quinto contributo alla conoscenza della microfauna Terziaria Italiana. Memorie della R. Accademie della Scienze dell´Instituto di Bologna, Scienze Naturali, 5(4):201-203.
Fornasini, C. 1900. Intorno odalcuni esexplaridi de foraminiferi adriatict. Memorie della R. Accademie della Scienze dell’ Istituto di Bologna, Italia, 5(8):362.
Forskål, P. and Niebuhr, C. 1775. Descriptiones Animalium, Avium, Amphibiorum, Piscium, Insectorum, Vermium [Microform]: Quae in Itinere Orientali /Observavit Petrus Forskål. Post Mortem Auctoris Edidit Carsten Niebuhr; Adjuncta est Materia Medica Kahirina Atque Tabula Maris Rubri Geographia. Mölleri, Aulae Typographi, Copenhagen.
Gadi, S.D. and Rajashekhar, K.P. 2007. Changes in inter-tidal foraminifera following tsunami inundation of Indian coast. Indian Journal of Marine Sciences, 36(1):35-42.
Galloway, J.J. 1933. A Manual of Foraminifera. Principia Press, Bloomington.
Ganapati, P.N. and Satyavati, P. 1958. Report on the foraminifera in bottom sediments in the Bay of Bengal off the East Coast of India. Andhra University Memoirs in Oceanography, 62(2):91-99.
Gandhi, S. and Nathan, S. 2014. Benthic foraminifera and geochemical studies with influence on pollution studies along the coast of Cuddalore, Tamil Nadu-ITS, India. Arab Journal of Geo Sciences, 7:917-925. https://doi.org/10.1007/s12517-012-0775-3
Gandhi, S., Rajamanickam, G.V.M., and Nigam, R. 2002. Taxonomy and distribution of benthic foraminifera from the sediments off Palk Strait, Tamil Nadu, east coast of India. Journal of Palaeontological Society of India, 47:47-64.
Gmelin, J.F. 1791. Systema Naturae Linnaei, Tome 1, Part 6, Editio 13. G.E. Beer, Leipzig.
Gnanamuthu, C.P. 1943. The foraminifera of Krusadi Island (In the Gulf of Mannar). Bulletin of the Madras Government Museum, 1:1-21.
Gronovius, L.T. 1781. Zoophylacii Gronoviani. Theodorus Haak et Soc., Leyden, Netherlands.
Guérin-Méneville, F.E. 1843. Crustacés. In Cuvier, G. (ed.), Iconographie du Règne Animal de G. Cuvier: ou, Représentation d'aprés Nature de l'une des Espèces les Plus Remarquables, et Souvent non Encore Figurées, de Chaque Genre D’amimaux. Avec un Texte Descriptif mis au Courant de la Science. J.B. Baillière, Paris.
Haeckel, E. 1894. Systematische Phylogenie. Entwurf eines Natürlichen Systems der Organismen auf Grund ihrer Stammesgeschichte. Systematische Phylogenie der Protisten und Pflanzen, 1. Georg Reimer, Berlin.
Haman, D. 1984. Saidovina, new name for Loxostomina Saidova, 1975 (non Sellier de Civrieux, 1968) and the status of Loxostomella Saidova, 1975 (Foraminiferida). Proceedings of the Biological Society of Washington, 97:419.
Hanagata, S. and Nobuhara, T. 2015. Illustrated guide to Pliocene foraminifera from Miyakojima, Ryukyu Island Arc, with comments on biostratigraphy. Palaeontologia Electronica, 18.1.3A:1-140. https://doi.org/10.26879/444
palaeo-electronica.org/content/2015/1016-foraminifera-of-miyakojima
Haque, A.F.M.M. 1956. The foraminifera of the Ranikot and the Laki of the Nammal Gorge, Salt Range. Palaeontologia Pakistanika, Geological Survey of Pakistan, 1:1-300.
Hatta, A. and Ujiie, H. 1992. Benthic foraminifera from Coral Seas between Ishigaki and Iriomote Islands, Southern Ryukyu Island Arc, Northwestern Pacific. Bulletin of the College of Science, University of the Ryukyus, 53:49-119.
Haynes, J.R. 1973. Cardigan Bay Recent Foraminifera (Cruises of the RV Antur, 1962-1964). Bulletin of the British Museum (Natural History), Zoology, Supplement, 4:1-245.
Haynes, J.R. 1981. Foraminifera. John Wiley and Sons, New York.
Hayward, B.W., Hollis, C.J., and Grenfell, H.R. 1997. Recent Elphidiidae (Foraminiferida) of the south-west Pacific and fossil Elphidiidae of New Zealand. Institute of Geological and Nuclear Sciences, Monograph, 16:1-166.
Hayward, B.W., Cedhagen, T., Kaminski, M., and Gross, O. 2017. World Modern Foraminifera Database. Available at http://www.marinespecies.org/foraminifera. Accessed on July 2017.
He, Y., Hu, L.Y., and Wang, K.L. 1965. Quaternary foraminifera in East Jiangsu Province. Memoirs of Nanjing Institute of Geology and Palaeontology, Academia Sinica, 4:51-162. (In Chinese)
Hemleben, C., Spindler, M., and Anderson, O.R. 2012. Modern Planktonic Foraminifera. Springer, New York. https://doi.org/10.1007/978-1-4612-3544-6
Heron-Allen, E. and Earland, A. 1915. The foraminifera of the Kerimba Archipelago (Portugese East Africa), Part II. Transactions of Zoological Society of London, 20(17):543-794.
Heron-Allen, E. and Earland, A. 1930. Some new foraminifera from the South Atlantic, Part 3. Journal of Royal Microscopical Society, 50:38-45.
Hofker, J. 1927. The Foraminifera of the Siboga Expedition, Part 1, Tinoporidae, Rotaliidae, Nummulitidae, Amphisteginidae, Siboga Expeditie, Monographie IV. Brill, Leiden.
Hofker, J. 1932. Notizen über die Foraminiferen des Golfes von Neapel III, Die Foraminiferenfauna des Ammontatura. Pubblicazioni della Stazione Zoologica di Napoli, 12:61-144.
Hofker, J. 1950. Wonderful animals of the sea: Foraminifera. Amsterdam Naturalist, 1(3):60-79.
Hofker, J. 1951. The Foraminifera of the Siboga Expedition, Part 3, Siboga Expeditie, Monographie IVb. Brill, Leiden.
Hofker, J. 1956. Tertiary foraminifera of coastal Ecuador: Part II. Additional notes on the Eocene species. Journal of Paleontology, 30:891-958.
Hofker, J. 1976. Further studies on Caribbean foraminifera. Studies on the Fauna of Curaçao and Other Caribbean Islands, 49(162):1-256.
Höglund, H. 1947. Foraminifera in the Gullmar fjord and the Skagerak. Zoologiska Bidrag Fran Uppsala, 26:3-328.
Hohenegger, J. 2004. Depth coenoclines and environmental consideration of Western Pacific larger foraminifera. Journal of Foraminiferal Research, 34:9-33. https://doi.org/10.2113/0340009
Hohenegger, J. 2011. Large Foraminifera: Greenhouse Constructions and Gardeners in the Oceanic Microcosm. The Kagoshima University Museum, Kagoshima, Japan.
Hohenegger, J. 2014. Species as the basic units in evolution and biodiversity: Recognition of species in the recent and geological past as exemplified by larger foraminifera. Gondwana Research, 25:707-728. https://doi.org/10.1016/j.gr.2013.09.009
Horton, B.P., Culver, S.J., Hardbattle, M.I.J., Larcombe, P., Milne, G.A., Morigi, C., Whittaker, J.E., and Woodroffe, S.A. 2007. Reconstructing Holocene sea-level change for the central Great Barrier Reef (Australia) using subtidal foraminifera. Journal of Foraminiferal Research, 37:327-343. https://doi.org/10.2113/gsjfr.37.4.327
Hottinger, L.Z., Reiss, Z., and Halicz, E. 1990. Comments on Neoeponides (Foraminifera). Revue de Palébiologie, 9:335-340.
Huang, T. 1961. Smaller foraminifera from the beach sands at Tanmenkang, Pachao-Tao, Penghu. Proceedings of the Geological Society of China, 4:83-90.
Jain, S.P. and Bhatia, S.B. 1978. Recent benthonic foraminifera from Mandvi, Kutch, p. 153-174. In Rasheed, D.A (ed.), Proceedings 7th Indian Colloquium on Micropalaeontology and Stratigraphy, Department of Geology, Madras University, Madras.
Javaux, E.J. and Scott, D.B. 2003. Illustration of modern benthic foraminifera from Bermuda and remarks on distribution in other subtropical/tropical areas. Palaeontologia Electronica, 6.1.4A:1-29. https://palaeo-electronica.org/2003_1/benthic/issue1_03.htm
Jones, R.W. 1994. The Challenger Foraminifera, 1st Edition. Oxford University Press, New York.
Jones, R.W. 2013. Supplemental notes on the Challenger foraminifera, p. 31-45. In Bowden, A.J., Gregory, F.J., and Henderson, A.S. (eds.), Landmarks in Foraminiferal Micropalaeontology: History and Development. The Micropalaeontological Society, Special Publication, 6. The Geological Society, London.
Jones, T.R. 1854. Notes on the Entomostraca of the Woolwich and Reading Series. The Quarterly Journal of the Geological Society of London, 10:160-162.
Jones, T.R. 1875. Foraminifera, p. 316-320. In Griffith, J.W. and Henfrey, A. (eds.), The Micrographic Dictionary: A Guide to the Examination and Investigation of the Structure and Nature of Microscopic Objects. Volume 1. Van Voorst, London.
Kaminski, M. 2004. The year 2000 classification of the agglutinated foraminifera, p. 237-255. In Bubík, M. and Kaminski, M.A. (eds.), Proceedings of the Sixth International Workshop on Agglutinated Foraminifera. Grzybowski Foundation Special Publication, Volume 8. Grzybowski Foundation, Prague.
Kaminski, M.A., Aksu, A., Box, M., Hiscott, R.N., Filipescu, S., and Al-Salameen, M. 2002. Late Glacial to Holocene benthic foraminifera in the Marmara Sea: Implications for Black Sea-Mediterranean Sea connections following the last deglaciation. Marine Geology, 190:165-202. https://doi.org/10.1016/s0025-3227(02)00347-x
Kaminski, M.A. and Cetean, C.G. 2011. Lectotypes of type species of agglutinated foraminiferal genera in the collections of the Natural History Museum, London. Part 2. Hippocrepinina and Hormosinina, p. 61-69. In Kaminski, M.A. and Filipescu, S. (eds.), Proceedings of the Eighth International Workshop on Agglutinated Foraminifera. Grzybowski Foundation Special Publication, 16. Grzybowski Foundation, Prague.
Karrer, F. 1865. Die Foraminiferen-Fauna der Tertiären Grünsandsteines der Orakei Bay bei Auckland. Novara Expedition 1857-1859, Geologischer Theil, Wien, 1(2):69-86.
Karrer, F. 1868. Die Miocene Foraminiferenfauna von Kostej im Banat. Sitzungeberichte der Kaiserlichen Akademie der Wissenschaften, Mathematisch Naturwissenschaftliche Classe, 58:121-195.
Kathal, P.K. and Singh, V.K. 2010. First report on some recent benthic foraminifera from the East coast of India. Journal of the Geological Society of India, 76:69-74.
Kennett, J.P. and Srinivasan, S. 1983. Neogene Planktonic Foraminifera, A Phylogenetic Atlas. Hutchinson Ross Publishing Company, Stroudsburg, Pennsylvania.
Kisel'man, E.N. 1972. Verkhnemelovye, Paleotsenovy e Foraminifery Novogo Roda Spiroplectinella. Trudy Sibirskogo Nauchno-Issledovatel’skogo Instituta Geologii Geofiziki i Mineral’nogo Syr’ya (SNIIGGIMS). Ministerstva Geologii i Okhrany Nedr SSSR, Novosibirsk, 146:134-140. (In Russian)
LaFond, E.C. 1966. Bay of Bengal, p. 110-118. In Fairbridge, R.W. (ed.), The Encyclopedia of Oceanography. Van Nostrand Reinhold, New York.
Lamarck, J.B. 1804. Suite des mémoires sur les fossils des environs de Paris. Annales Muséum National d’Histoire Naturelle, 5:179-188.
Lankester, E.R. 1885. Protozoa, p. 831-865. In Baynes, T.S. (ed.), The Encyclopedia Britannica, 9th edition, Volume 19. J.M. Stoddard Co., Ltd., Philadelphia.
Lankford, R.R. and Phleger, F.B. 1973. Foraminifera from the nearshore turbulent zone, western North America. Journal of Foraminiferal Research, 3:101-132. https://doi.org/10.2113/gsjfr.3.3.101
Larsen, A.R. 1976. Studies of recent Amphistegina, taxonomy and some ecological aspects. Israel Journal of Earth-Sciences, 25:1-26.
Le Calvez, Y. 1949. Révision des foraminifères Lutétiens du Bassin de Paris. Rotaliidae et familles affines. Mémoires du Service de la Carte Géologique Détaillée de la France, 2:1-54.
Le Calvez, Y., de Klasz, I., and Brun, L. 1974. Nouvelle contribution a la connaissance des microfaunes du Gabon. Revista Española de Micropaleontología, 6:381-400.
Lehmann, R. 1961. Strukturanalyse einiger Gattungen der Subfamilie Orbitolitinae. Eclogae Geologicae Helvetiae, 54:597-667.
Lei, Y.L., Li, T.G., Bi, H., Cui, W.L., Song, W.P., Li, J.Y., and Li, C.C. 2015. Responses of benthic foraminifera to the 2011 oil spill in the Bohai Sea, PR China. Marine Pollution Bulletin, 96:245-260. https://doi.org/10.1016/j.marpolbul.2015.05.020
Leymerie, A. 1846. Memoire sue de terrain a Nummulites (epicretace') des corbieres et de la montagne Noire. Memoires de la societe Geologique de France, 21:337-373.
Licari, L. and Mackensen, A. 2005. Benthic foraminifera off West Africa (1°N to 32°S): Do live assemblages from the topmost sediment reliably record environmental variability? Marine Micropaleontology, 55:205-233. https://doi.org/10.1016/j.marmicro.2005.03.001
Linnaeus, C. 1758. Systema Naturae per Regna tria Naturae, Secundum Classes, Ordines, Genera, Species, cum Characteribus, Differentiis, Synonymis, Locis. 1, 10th edition. L. Salvii, Holmiae (Stockholm).
Loeblich, A.R. and Tappan, H. 1953. Studies of Arctic foraminifera. Smithsonian Miscellaneous Collections, 121(7):1-150.
Loeblich, A.R., Jr. and Tappan, H. 1961. Suprageneric classification of the Rhizopodea. Journal of Paleontology, 35(2):245-330.
Loeblich, A.R., Jr. and Tappan, H. 1964. Foraminiferal classification and evolution. Journal of the Geological Society of India, 5:5-39.
Loeblich, A.R. and Tappan, H. 1985. Some new and redefined genera and families of agglutinated foraminifera II. Journal of Foraminiferal Research, 15:175-217. https://doi.org/10.2113/gsjfr.15.3.175
Loeblich, A.R, Jr. and Tappan, H. 1987. Foraminiferal Genera and Their Classification. Van Nostrand Reinhold, New York.
Loeblich, A.R., Jr. and Tappan, H. 1994. Foraminifera of the Sahul Shelf and Timor Sea. Special Publications of the Cushman Laboratory for Foraminiferal Research, 31:1-661.
Łuczkowska E. 1971. A new zone with Praeorbulina indigena (Foraminiferida, Globigerinidae) in the Upper Badenian (Tortonian s.s.) of Central Paratethys. Rocznik Polskiego Towarzystwa Geologicznego, 40(3-4):45-448.
Łuczkowska, E. 1972. Miliolidae (Foraminiferida) from Miocene of Poland, Part I. Revision of the classification. Acta Palaeontologica Polonica, 17:341-371.
Łuczkowska, E. 1974. Miliolidae (Foraminiferida) from Miocene of Poland, Part II. Biostratigraphy, palaeoecology and systematics. Acta Palaeontologica Polonica, 19:3-176.
Lutze, G.F. 1974. Benthische Foraminiferen in Oberflächen-Sedimenten des Persischen Golfes. Teil 1: Arten. Meteor Forschungsergebnisse, C17:1-66.
Margreth, S. 2010. Benthic Foraminifera Associated to Cold-Water Coral Ecosystems. Unpublished Ph.D. Thesis, Department of Geosciences, University of Fribourg, Fribourg, Switzerland.
Mathews, R.D. 1945. Rectuvigerina, a new genus of foraminifera from a restudy of Siphogenerina. Journal of Paleontology, 19:588-606.
Maync, W. 1952. Critical taxonomic study and nomenclatural revision of the Lituolidae based upon the prototype of the family, Lituola nautiloidea Lamarck, 1804. Contributions from the Cushman Laboratory for Foraminiferal Research, 3:35-56.
McCulloch, I. 1977. Qualitative Observations on Recent Foraminiferal Tests with Emphasis on the Eastern Pacific, Parts I-III. University of Southern California, Los Angeles.
McCulloch, I. 1981. Qualitative Observations on Recent Foraminiferal Tests. Pt. IV: With Emphasis on the Allan Hancock Collection, North Atlantic. University of Southern California, Los Angeles.
Meriç, E., Avşar, N., and Yokeş, B. 2008. Some alien foraminifers along the Aegean and southwestern coasts of Turkey. Micropaleontology, 54(3-4):307-349.
Meriç, E., Yokes, M.B., Avsar, K.N., Kirki-Elmas, E., Dinçer, F., and Karhan, S.U. 2013. First report of Pararotalia calcariformata from the Hatay coastline (Turkey—north-eastern Mediterranean). Marine Biodiversity Records, 6:e31. https://doi.org/10.1017/s1755267212001108
Mikhalevich, V. I. 1992. Makrosistema Foraminifer (The Macrosystem of the Foraminifera). Unpublished PhD Thesis, Zoological Institute of the Russian Academy of Sciences, St. Petersburg, Russia. (In Russian)
Mikhalevich, V.I. 2004. On the heterogeneity of the former Textulariina. Grzybowski Foundation Special Publication, 8:317-349.
Milker, Y. and Schiemdl, G. 2012. A taxonomic guide to modern benthic shelf foraminifera of the western Mediterranean Sea. Palaeontologia Electronica, 15.2.16A:1-134. https://doi.org/10.26879/271
palaeo-electronica.org/content/2012-issue-2-articles/223-taxonomy-foraminifera
Milker, Y., Schmiedl, G., Betzler, C., Römer, M., Jaramillo-Vogel, D., and Siccha, M. 2009. Distribution of recent benthic foraminifera in neritic carbonate environments of the Western Mediterranean Sea. Marine Micropalaeontology, 73:207-225. https://doi.org/10.1016/j.marmicro.2009.10.003
Millett, F.W. 1898. Report on the recent foraminifera of the Malay Archipelago contained in anchor-mud collected by Mr. A. Durrand, F.R.M.S. Part I. Journal of the Royal Microscopical Society, 1898:258-269.
Millett, F.W. 1900a. Report on the Recent foraminifera of the Malay Archipelago contained in anchor-mud collected by Mr. A. Durrand, F.R.M.S. Part VIII. Journal of the Royal Microscopical Society, 1900:273-281.
Millett, F.W. 1900b. Report on the Recent foraminifera of the Malay Archipelago contained in anchor-mud collected by Mr. A. Durrand, F.R.M.S. Part IX. Journal of the Royal Microscopical Society, 1900:539-549.
Millett, F.W. 1904. Report on the Recent foraminifera of the Malay Archipelago contained in anchor-mud collected by Mr. A. Durrand, F.R.M.S. Part XVI. Journal of the Royal Microscopical Society, 1904:489-506.
Montagu, G. 1803. Testacea Britannica or Natural History of British Shells Marine, Land and Fresh Water, Including the Most Minute. J.S. Hollis, Romsey, England.
Murray, J.W. 1971. An Atlas of British Recent Foraminiferids. Heinemann Educational Books, London.
Murray, J.W. 2003. An illustrated guide to the benthic foraminifera of the Hebridean shelf, west of Scotland, with notes on their mode of life. Palaeontologia Electronica, 5.2.5A:1-31.
https://palaeo-electronica.org/2002_2/guide/issue2_02.htm
Murthy, K.S.R., Subrahmanyam, A.S., Murty, G.P.S., Sarma, K.V.L.N.S., Subrahmanyam, V., Mohana Rao, K., Suneetha Rani, P., Anuradha, A., Adilakshmi, B., and Sri Devi, T. 2006. Factors guiding tsunami surge at the Nagapattinam-Cuddalore shelf, Tamil Nadu, East coast of India. Current Science, 90(11):1535-1538.
Naidu, T.Y., Kamalakaram, S., Bhaskara Rao, V., and Kaladhar, R. 1990. Living foraminiferal diversity in marginal water bodies east coast of India. Journal of the Palaeontological Society of India, 35:33-39.
Nandhakumar, S. and Rao, R. N. 2008. Benthic foraminiferal studies from inner shelf sediments off north Chennai, south-east coast of India. Proceedings of the 5th International Conference on Environmental Micropalaeontology, Microbiology and Meiobenthology, EMMM'2008. University of Madras, Madras.
Natland, M.L. 1938. New species of foraminifera from off the west coast of North America and from the later Tertiary of the Los Angeles Basin. Scripps Institute of Oceanography Bulletin, Technical Series, 4(5):146.
Neagu, T. 1999. Barbuspira complanata n.g., n.sp., a new cornuspirid foraminifera from lower Valanginian, Cernavoda-Pod (Southern Dobrogea)-Romania. Acta Paleontologica Romaniae, 2:305-307.
Nigam, R. and Khare, N. 1999. Spatial and temporal distribution of foraminifera in sediments off the central west coast of India and use of their test morphologies for the reconstruction of paleomonsoonal precipitation. Micropaleontology, 45:285-303. https://doi.org/10.2307/1486138
Nisha, N.R. and Singh, A.D. 2012. Benthic foraminiferal biofacies on the shelf and upper continental slope off North Kerala (Southwest India). Journal of the Geological Society of India, 80:783-801.
Norman, A.M. 1892. Museum Normanianum. Durham, England.
Oflaz, S.A. 2006. Taxonomy and Distribution of the Benthic Foraminifera in the Gulf of Iskenderun, Eastern Mediterranean. Unpublished MSc Thesis, Middle East Technical University, Ankara, Turkey.
Ovechkina, M.N., Bylinskaya, M.E., and Uken, R. 2010. Planktonic foraminiferal assemblage in surface sediments from the Thukela Shelf, South Africa. African Invertebrates, 51(2):231-254. https://doi.org/10.5733/afin.051.0202
Panchang, R. 2008. Ecology and Distribution of Benthic Foraminifera off Myanmar. Unpublished Ph.D. Thesis, National Institute of Oceanography, Goa, India.
Panchang, R. and Nigam, R. 2014. Benthic ecological mapping of the Ayeyarwady Delta Shelf off Myanmar, using foraminiferal assemblages. Journal of the Palaeontological Society of India, 59(2):121-168.
Pannikar, N.K. and Jayaraman, R. 1966. Biological and oceanographic differences between the Arabian Sea and the Bay of Bengal as observed from the Indian region. Proceedings of the Indian Academy of Science, 64B:231-240.
Papp, A. and Schmidt, M.E. 1985. The fossil foraminifera of the Tertiary Basin of Vienna: Revision of the monograph by Alcide d’Orbigny (1846). Abhandlungen der Geologischen Bundesanstalt (Wien), 37:1-311.
Parker, F.L. 1952. Foraminiferal distribution in the Long Island Sound - Buzzards Bay area. Bulletin of the Museum of Comparative Zoology, Harvard College, 106(10):428-473.
Parker, F.L. 1953. Formainifera species off Portsmouth, New Hampshire. Bulletin of the Museum of Comparative Zoology, Harvard College, 106(9):391-423.
Parker, F.L. 1962. Planktonic foraminiferal species in Pacific sediments. Micropalaeontology, 8(2):219-254.
Parker, J.H. 2009. Taxonomy of foraminifera from Ningaloo Reef, Western Australia. Memoirs of the Association of Australasian Palaeontologists, 36:1-810.
Parker, W.K. and Jones, T.R. 1865. On some foraminifera from the North Atlantic and Arctic Oceans, including Davis Straits and Baffin's Bay. Philosophical Transactions of the Royal Society of London, 155:325-441. https://doi.org/10.1098/rstl.1865.0006
Parr, W.J. 1942. New genera of foraminifera from the Tertiary of Victoria. Mining and Geological Journal, 2:361-363.
Parr, W.J. 1945. Recent foraminifera from Barwon Heads, Victoria. Proceedings of the Royal Society of Victoria, 56(2):189-227.
Parr, W.J. 1950. Foraminifera. Reports B.A.N.Z. Antarctic Research Expedition 1929-1931, Series B (Zoology, Botany), 5(6):232-392.
Patterson, R.T. and Richardson, R.H. 1988. Eight new genera of unilocular foraminifera (Lagenidae). Transactions of the American Microscopical Society, 107:240-258. https://doi.org/10.2307/3226501
Pawlowski, J., Holzmann, M., Berney, C., Fahrni, J.F., Gooday, A.J., Cedhagen, T., Habura, A., and Bowser, S.S. 2003. The evolution of early foraminifera. Proceedings of the National Academy of Science of the United States of America, 100:11494-11498. https://doi.org/10.1073/pnas.2035132100
Pawlowski, J., Holzmann, M., and Tyszka, J. 2013. New supraordinal classification of foraminifera: Molecules meet morphology. Marine Micropaleontology, 100:1-10. https://doi.org/10.1016/j.marmicro.2013.04.002
Perelis, L. and Reiss, Z. 1975. Cibicididae in recent sediments from the Gulf of Elat. Israel Journal of Earth Sciences, 24:73-96.
Petri, S.I. 1955. Recent foraminifera from Sao Paulo, Brazil. Contributions from the Cushman Laboratory for Foraminiferal Research, 6 (2):82-86.
Pilarczyk, J.E., Reinhardt, E.G., Boyce, J.I., Schwarcz, H.P., and Donato, S.V. 2011. Assessing surficial foraminiferal distributions as an overwash indicator in Sur Lagoon, Sultanate of Oman. Marine Micropaleontology, 80:62-73. https://doi.org/10.1016/j.marmicro.2011.06.001
Podobina, V.M. 1978. Sistematika i filogeniya Gaplofragmiidey. Tomsk Universitet, Tomsk. (In Russian)
Polski, W. 1959. Foraminiferal biofacies off the north Asiatic Coast. Journal of Paleontology, 33:569-587.
Puri, H.S. 1954. Contribution to the study of the Miocene of the Florida Panhandle Part II-Foraminifera. Florida Geological Survey Geological Bulletin, 36:5-213.
Rajasekhar, N. 1981. Some Recent Foraminifera from Off Tuticorin, Tamil Nadu State. Unpublished Master of Philosophy Thesis, University of Madras, Madras, India.
Ramesh, R. K. 2005. Recent Benthic Foraminifera From the Inner Shelf Sediments off Pondicherry and Cuddalore, Southeast Coast of India. Unpublished PhD Thesis, Madras University, Madras, India.
Rao, M.S. and Vedantam, D. 1968. Distribution of foraminifera in Vishakhapatnam, Bay of Bengal. Bulletin National Institute of Science India, 38:491-501.
Rasheed, D.A. 1971. Some foraminifera belonging to Miliolidae and Opthalmidiidae from the Coral Sea, south of Papua (New Guinea), Part 2. Journal of the Madras University, B, 37-38:19-68.
Reiss, Z. 1960. Structure of so-called Eponides and some other rotaliiform foraminifera. Bulletin Israel Geological Survey, 29:1-28.
Reiss, Z. 1963. Reclassification of perforate foraminifera. Bulletin of the Geological Survey of Israel, 35:1-111.
Renema, W. and Hohenegger, J. 2005. On the identity of Calcarina spengleri (Gmelin 1791). Journal of Foraminiferal Research, 35(1):15-21. https://doi.org/10.2113/35.1.15
Reuss, A.E. 1848. Die fossilen Polyparien des Wiener Tertiärbeckens, p. 1-109. In Haidinger, W. (ed.), Naturwissenschaftliche Abhandlungen. Wilhelm Braumüller, Wien.
Reuss, A.E. 1850. Neue Foraminiferen aus den Schichten des österreichischen Tertiärbeckens. Denkschriften der Mathematisch-Naturwissenschaftlichen Classe der Kaiserlichen Akademie der Wissenschaften, 1849(1):360-395.
Reuss, A.E. 1858. Über die Foraminiferen von Pietzbühl. Zeitschrift der Deutschen Geologischen Gesellschaft, 10:433-438.
Reuss, A.E. 1860. Die Foraminiferen der Westphälischen Kreideformation. Sitzungsberichte der K. Akademie der Wissenschaften in Wien, Mathematisch-Naturwissenschaftliche Classe, 40:147-238.
Reuss, A.E. 1861. Paläontologische Beiträge. Sitzungsberichte der Kaiserlichen Akademie der Wissenschaften in Wien, Mathematisch Naturwissenschaftliche Classe, 44:301-342.
Reuss, A.E. 1862. Entwurf einer systematischen Zusammenstellung der Foraminiferen. Sitzungsberichte der Kaiserliche Akademie der Wissenschaften, Mathematisch-Naturwissenschaftliche Klasse, 44:355-396.
Revets, S.A. 1996. The generic revision of five families of Rotaliine foraminifera. Special Publications of the Cushman Laboratory for Foraminiferal Research, 34:1-108.
Rhumbler, L. 1895. Entwurf eines natürlichen Systems der Thalamophoren. Nachrichten von der Gesellschaft der Wissenschaften zu Göttingen, Mathematisch-Physikalische Klasse, 1:51-98.
Rhumbler, L. 1911. Die Foraminiferen (Thalamophoren) der Plankton-Expedition. Part I - Die allgemeinen Organisationsverhältnisse der Foraminiferen. Ergebnisse der Plankton-Expedition der Humoldt-Stiftung, Kiel und Leipzig, 3:1-331.
Riveiros, N.V. and Patterson, T.R. 2008. An illustrated guide to Fjord foraminifera from the Seymour-belize inlet complex, Northern British Columbia, Canada. Palaeontologia Electronica, 11.1.2A:1-45.
https://palaeo-electronica.org/2008_1/145/index.html
Rzehak, A. 1886. Die Foraminiferenfauna der Neogenformation der Umgebung von Mähr.-Ostrau. Naturforschender Verein Brünn, Verhandlungen, Brünn, 1885(24):77-126.
Said, R. 1949. Foraminifera from the northern Red Sea. Special Publications of the Cushman Laboratory for Foraminiferal Research, 26:1-44.
Saidova, K.M. 1975. Bentosnye Foraminifery Tikhogo Okeana. [Benthonic foraminifera of the Pacific Ocean]. Institut Okeanologii, Akademiya Nauk SSSR, Moscow. (In Russian)
Saidova, K.M. 1981. O Sovremennom Sostoyanii Sistemy Nadvidovykh Taksonov Kaynozoyskikh Bentosnykh Foraminifer. [On up-to-date system of supraspecific taxonomy of Cenozoic benthonic foraminifera]. Institut Okeanologii, Akademiya Nauk SSSR, Moscow. (In Russian)
Saito, T., Thompson, P.R., and Breger, D. 1981. Recent and Pleistocene Planktonic Foraminifera. University of Tokyo Press, Tokyo.
Sarojini, D. 1958. Studies on Littoral Foraminiferal from the Bay of Bengal. Unpublished PhD Thesis, Andhra University, Waltair, India.
Schiebel, R. and Hemleben, C. 2017. Planktic Foraminifers in the Modern Ocean. Springer Verlag, Berlin. https://doi.org/10.1007/978-3-662-50297-6
Schlumberger, C. 1881. Compte rendu sommaire d’une exploration zoologique, faite dans le Méditerranée, à bord du navire de l’etat ‘le Travailleur’. Compte Rendu Hebdomadaire des Séances de l’Académie des Sciences, 93. l’Académie des Sciences, Paris.
Schlumberger, C. 1893. Monographie des Miliolidoe du golfe de Marseille. Mémoires de la Société Zoologique de France, 6:199-228.
Schlumberger, C. 1894. Note sur les foraminifères des mers arctiques russes. Mémoires de la Société Zoologique de France, 7:252-258.
Schultze, M.S. 1854. Über den Organismus der Polythalamien (Foraminiferen), nebst Bermerkungen über die Rhizopoden im Allgemeinen. W. Engelmann, Leipzig.
Schulze, F.E. 1875. Zoologische Ergebnisse der Nordseefahrt vom 21. Juli bis 9. September 1872, I Rhizopoden. II. Jahresberichte der Kommission zur Untersuchung der Deutschen Meere in Kiel, 1874:99-114.
Schwager, C. 1866. Fossile Foraminiferen von Kar Nikobar. Reise der Österreichischen Fregatte Novara um die Erde in den Jahren 1857, 1858, 1859 unter den Befehren des Commodore B. von W ü llerstorf - Urbair, Geologischer Theil, 2:187-268.
Schwager, C. 1876. Saggio di una classificazione dei foraminiferi avuto riguardo alleloro famiglie naturali, Bollettino del Reale Comatato Geologico d'Italia, 7:475-485.
Schwager, C. 1877. Quadro del proposto sistema di classificazione dei foraminiferi con guscio. Bollettino del Reale Comatato Geologico d'Italia, 8:18-27.
Scott, D. B., Takayanagi, Y., Hasegawa, S., and Saito, T. 2000. Illustration and taxonomic reevaluation of Neogene foraminifera described in Japan. Paleontologia Electronica, 3.2.5A:1-41. https://palaeo-electronica.org/2000_2/foram/issue2_00.htm
Seguenza, G. 1862. Dei terreni Terziarii del Distretto di Messina, Parte II, Descrizione dei Foraminiferi Monothalamici Delle Marne Mioceniche del Distretto di Messina. Capra, T., Messina.
Seibold, I. 1972. Preliminary report on the transport of foraminifera into Cochin back water. Journal of the Marine Biological Association of India, 14(2):452-455.
Seibold, I. 1975. Benthonic foraminifera from the coast and lagoon of Cochin (South India). Revista Espaňola de Micropalaeontologia, 7:175-213.
Sellier de Civrieux, J.M. 1969. Cuatro géneros nuevos de foraminíferos del Mar Caribe. Boletín del Instituto Oceanográfico, Universidad de Oriente, Cumana, 7(1):149-193.
Setty, M.G.A.P. 1976a. Foraminiferal assemblages at continental margin off Pondicherry, p. 23-28. In (editor unknown), Proceedings of the Workshop on the Coastal Sedimentation of India, South of Latitude 18 Degrees. (publisher unknown).
Setty, M.G.A.P. 1976b. Recent foraminifera from the shelf off Madras, p. 31-36. In (editor unknown), Proceedings of I Seminar. Indian Geological Congress, New Delhi.
Setty, M.G.A.P. 1978. Shelf edge regime and foraminifera off Pondicherry, Bay of Bengal. Indian Journal of Marine Sciences, 7:302-304.
Setty, M.G.A.P. and Rao, M.S. 1978. Recent foraminifera largely cosmopolitan fauna dominated by miliolids, Ammonia, Cibicides and Elphidium. Recent Research in Geology, 7:53-57.
Sigal, J. 1952. Ordre des Foraminifera, p. 133-301. In Piveteau, J. (ed.), Traité de Paléontologie, Volume1. Masson, Paris.
Silvestri, A. 1903. Dimorfismo e nomenclatura d’una Spiroplecta. Altre notizie sulla struttura della Siphogenerina columellaris. Atti della Pontificia Accademia Romana dei Nuovi Lincei, 56:59-66.
Silvestri, A. 1904. Ricerche strutturali su alcune forme dei Trubi di Bonfornello (Palermo). Memorie della Pontificia Accademia Romana dei Nuovi Lincei, 22:235-276.
Silvestri, A. 1923. Lo stipite della Elissoforme e le sue affinitá. Memorie della Pontificia Accademia della Scienze, Nuovi Lincei, 2(6):231-270.
Sivakumar, K. 2002. Ecology and Distribution of Recent Benthic Foraminifera from the Shelf Sediments of Bay of Bengal, off Chennai, Tamil Nadu. Unpublished PhD Thesis, National College Tiruchirapalli, Tamil Nadu, India.
Srinivasan, M.S., Kennett, J.P., and Bè, A.W.H. 1974. Globorotalia menardii neoflexuosa new sub-species from the northern Indian Ocean. Deep Sea Research, 21:321-324.
Subba Rao, M. 1960. Organic matter in the marine sediments off east coast of India. Bulletin American Association of Petroleum Geologists, 44:1705-1713.
Szarek, 2001. Biodiversity and Biogeography of Recent Benthic Foraminiferal Assemblages in the South-western South China Sea (Sunda Shelf). Dissertation zur Erlangung des Doktorgrades der Mathematisch-Naturwissenschaftlichen Fakultät der Christian-Albrechts-Universität, Kiel, Germany.
Takayanagi, Y. 1955. Recent foraminifera from Matsukawaura and its vicinity. Contributions from the Institute of Geology and Paleontology, Tohoku University, 45:18-52.
Talib, A. and Farroqui, M.Y. 2007. Distribution of recent foraminifera in the littoral sediments of Dwarka, Saurashtra coast, Gujarat. Journal of the Palaeontological Society of India, 52(1):17-25.
Terquem, O. 1875. Essai sur le classement des animaux qui vivent sur la plage et dans les environs de Dunquerque. Memoirs de la Society Dunkerquoise, 19:405-447.
Terquem, O. 1878. Les foraminiféres et les entomostracés ostracodes du Pliocène supérieur de l'ile de Rhodes, premiére section-foraminiféres. Memoires de la Societe Geologique de France, 3(3):1-135.
Terquem, O. 1882. Les foraminiferes de l’Eocene des environs de Paris. Memories de la Societe Geologique de France, 3, 2(3):1-193.
Uchio, T. 1962. Influence of the River Shinano on foraminifera and sediment grain size distributions. Publications of the Seto Marine Biological Laboratory, 10:363-392.
Ujiié, H. and Rifardi, D. 1993. Some benthic foraminifera from the Oura River. Estuary and its Environs, Okinawa. Bulletin of the College of Science, University of the Ryukyus, 5:121-243.
Vella, P. 1957. Studies in New Zealand foraminifera. Paleontological Bulletin, Wellington, 28:1-64.
Vénec-Peyré, M.-T. and Poignant, A. 2010. Status of foraminiferal collections from the Tertiary Basin of Vienna, and revision of Dentalina spinosa d’Orbigny, 1846 and Nonionina boueana d’Orbigny, 1846. Geodiversitas, 32(3):479-491. https://doi.org/10.5252/g2010n3a5
Voloshinova, N.A. 1958. O novoy sistematike Nonionid (A new systematic of Nonionids). Trudy Vsesoyuznogo Neftyanogo. Nauchnoissledovatel’skogo Geologorazvedochnogo Instituta, 115:117-191. (In Russian)
Walker, G. and Boys, W. 1784. Testacea Minuta Rariora, Nuperrime Detecta in Arena Littoris Sandvicensis (A collection of the minute and rare shells lately discovered in the sand of the seashore near Sandwich). G. Walker, London.
Walker, G. and Jacob, E. 1798. An arrangement and description of minute and rare shells, p. 629-645. In Adams, G. and Kanmacher, F. (eds.), Essays on the Microscope. Dillon and Keating, London.
Wedekind, P.R. 1937. Einführung in die Grundlagen der historischen Geologie, Band II. Mikrobiostratigraphie die Korallen- und Foraminiferenzeit. Ferdinand Enke, Stuttgart.
Wiesner, H. 1920. Zur Systematik der Miliolideen. Zoologischer Anzeiger, 51:13-20.
Wiesner, H. 1923. Die Miliolideen der östlichen Adria. Privately printed, Prag-Bubanec.
Wiesner, H. 1931. Die Foraminiferen der deutschen Siidpolar-Expedition 1901-1903. Deutsche Südpolar-Expedition 1901-1903 im Auftrage des Reichsamtes des Innern Herausgegeben von Erich von Drygalski Leiter Expedition, Zoology, 20:53-165.
Williamson, W.C. 1848. On the Recent British species of the genus Lagena. The Annals and Magazine of Natural History, 2, 1:1-20. https://doi.org/10.1080/03745485809494465
Williamson, W.C. 1858. On the Recent Foraminifera of Great Britain. Ray Society, London.
Wiseman, J.D.H. and Bennett, H. 1940. The distribution of organic carbon and nitrogen in the sediments from the Arabian Sea. Scientific Reports of the John Murray Expedition, 3(4):193-221.
Yalçin, H., Meriç, E., Avşar, N., Tetiker, S., Barut, I.F., Yilmaz, S., and Dinçer, F. 2008. Mineralogical and geochemical features of colored benthic foraminifers from Aegean and southwestern coasts of Turkey. Micropaleontology, 54:351-370.
Yordanova, E.K. and Hohenegger, J. 2002. Taphonomy of larger foraminifera: Relationships between living individuals and empty tests on flat reef slopes (Sesoko Island, Japan). Facies, 46:169-204. https://doi.org/10.1007/bf02668080
Yordanova, E.K. and Hohenegger, J. 2007. Studies on settling, traction and entrainment of larger benthic foraminiferal tests: Implications for accumulation in shallow marine sediments. Sedimentology, 54:1273-1306. https://doi.org/10.1111/j.1365-3091.2007.00881.x
Zheng, S.-Y. 1979. The recent foraminifera of the Xisha Islands, Guangdong Province, China, Part II. Studia Marina Sinica, 15:101-232.
Zweig-Strykowski, M. and Reiss, Z. 1976 (“1975”). Bolivinitidae form the Gulf of Elat. Israel Journal of Earth Sciences, 24:97-111.

