 A new multi-faceted framework for deciphering diplodocid ontogeny
A new multi-faceted framework for deciphering diplodocid ontogeny
Article number: 20.3.43A
https://doi.org/10.26879/674
Copyright Palaeontological Association, September 2017
Author biographies
Plain-language and multi-lingual abstracts
PDF version
Submission: 20 April 2016. Acceptance: 19 June 2017
{flike id=1918}
ABSTRACT
Determining maturity in sauropod dinosaurs histologically is problematic as rapid growth leads to remodeling of Lines of Arrested Growth (LAGs). Although a complimentary system has been devised utilizing several factors including relative amounts of remodeling (Histologic Ontogenetic Stage [HOS]), most assessments of sauropod maturity are based on morphologic indicators. To better assess skeletal maturity and morphologic change through ontogeny, we examined cranial and postcranial material from over 20 diplodocid individuals (Apatosaurus and Diplodocus) from the Upper Jurassic Morrison Formation. Here we describe consistent combinations of morphologic and histologic features that can be used to ascertain maturity. Small diplodocids (femoral lengths ≤120 cm) display non- to weakly bifurcated cervical and dorsal neural spines, acamerate to camerate centra, two to six preserved LAGs in dorsal ribs, and a maximum femoral designation of HOS 7. Larger individuals (femoral length ~125 cm) have more developed internal pneumatic structures, greater neural spine bifurcation, preserve up to eight LAGs, and a femoral designation of HOS 9. In contrast, skeletally mature sauropods (femoral lengths >150 cm) have complex pneumatic structures, extended neural spine bifurcation (also within anterior caudals), and a femoral HOS between 11-13. Further, all of the preserved small diplodocid skulls exhibit a postparietal foramen (previously suggested to be an apomorphy of Dicraeosauridae), which is absent in large skulls (where preserved), suggesting that it is an ontogenetic character. These findings support the hypothesis of significant ontogenetic morphological change in diplodocid sauropods and suggest caution when describing new taxa on the basis of small-bodied holotypes.
D. Cary Woodruff. Royal Ontario Museum; University of Toronto, Toronto, ON, Canada; and Great Plains Dinosaur Museum and Field Station, Malta, MT, United States of America. sauropod4@gmail.com
Denver W. Fowler. Dickinson Museum Center, Dickinson, ND, United States of America. df9465@hotmail.com
John R. Horner. Burke Museum of Natural History and Culture, University of Washington, Seattle, WA, United States of America. johnrhorner@mac.com
Keywords: diplodocid; ontogeny; morphology; histology
Final citation: Woodruff, D. Cary, Fowler, Denver W., and Horner, John R. 2017. A new multi-faceted framework for deciphering diplodocid ontogeny. Palaeontologia Electronica 20.3.43A: 1-53. https://doi.org/10.26879/674
palaeo-electronica.org/content/2017/1918-deciphering-diplodocid-growth
Copyright: August 2017 Palaeontology Association. This is an open access article distributed under the terms of Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0), which permits users to copy and redistribute the material in any medium or format, provided it is not used for commercial purposes and the original author and source are credited, with indications if any changes are made.
creativecommons.org/licenses/by-nc-sa/4.0/
INTRODUCTION
Studies of ontogenetic morphological change in dinosaurs largely rely on comparisons of cranial features (i.e., horns, frills, and crests; Dodson, 1975; Hopson, 1975; Sampson et al., 1997; Carr, 1999; Evans et al., 2005; Horner and Goodwin, 2006; 2009; Scannella and Horner, 2010; Campione and Evans, 2011). As cranial material of sauropods is relatively rare and does not exhibit pronounced display structures (which are often beneficial in recognizing ontogenetic trajectories [e.g., Dodson, 1975]) the work done on them has to depend largely on histology. These histologic studies examine the microstructure of the bone and the degrees of bone remodeling to determine relative maturity of a range of individuals, such as the Age Class assignments of Curry (1999) and the Histologic Ontogenetic Stage (HOS) of Klein and Sander (2008). Yet while the particulars and demarcations of these maturational assessments may vary, all acknowledge that histologically, skeletal maturity occurs at the onset of an external fundamental system (EFS; slowly deposited parallel-fibered or lamellar tissue along the outermost cortex - closely spaced outermost series of LAGs [Huttenlocker et al., 2013; Appendix 1]), which is the histologic indicator of osteogenic cessation (see Padian and Lamm [2013] for a greater discussion of bone tissues throughout maturity).
Recently several sauropod studies have reported morphologic attributes that can be used to denote maturity. Whitlock et al. (2010) documented cranial changes between immature and mature specimens of Diplodocus (or Diplodocinae indeterminate [Tschopp et al., 2015]), which include the development of proportionally larger orbits, rounded and narrower premaxilla, and a more anteriorly situated tooth row. Wedel (2003, 2005, 2009), Schwarz et al. (2007a), and Carballido and Sander (2014) have demonstrated that the complexity of pneumatic architecture has increased through sauropodomorph phylogeny and likewise increased through ontogeny. Salgado (1999) suggested a link between the degree of neural spine bifurcation and ontogeny in diplodocids. Woodruff and Fowler (2012) showed that small (presumably immature) diplodocids exhibited rounded to weakly bifurcated neural spines, whereas neural spines were fully bifurcated in larger (and presumably more mature) individuals. Based on this observation, it was suggested that the degree of bifurcation could be used to infer maturity in diplodocids (and other methodologies have used morphology to predict maturity; such as the Morphologic Ontogenetic Stage [MOS] of Carballido and Sander, 2014; or the ontogenetic trajectory stages of Ikejiri et al., 2005).
While some of these morphologic features have been incorporated into our understanding of sauropod ontogeny, the conclusions of Woodruff and Fowler (2012) have been the subject of ongoing discussion (Wedel and Taylor, 2013; Carballido and Sander, 2014; Hedrick et al., 2014; Woodruff and Foster, 2014; Tschopp et al., 2015). Aspects of this study that are considered contentious include: 1) the unknown precise serial placement for isolated vertebrae, and 2) the proper identification of maturational states to corroborate said morphological interpretations (Wedel and Taylor, 2013; Hedrick et al., 2014). While these are issues that need to be further addressed through continued morphological studies of sauropod growth, it has been suggested that histologic analysis may not be necessary due to specific features attributable to maturity (such as vertebral arch fusion; Wedel and Taylor, 2013). We would argue that histologic analysis is the only repeatedly confirmed methodology to determine a specimen’s maturational state. The current analysis tests the inferred maturational stages of Woodruff and Fowler (2012) using histologic examination. Additional features that may indicate relative maturity are also assessed. A multi-faceted approach, incorporating examination of overall vertebral morphology (cervical, dorsal, and anterior caudal vertebrae), vertebral pneumatic morphology, histologic ontogenetic stage (HOS; Klein and Sander, 2008), histologic and morphologic examination of femora, age determining histology of dorsal ribs, and neural spine histology (of cervical and dorsal vertebrae) was conducted on various members of Diplodocoidea, with primary emphasis on Apatosaurus and Diplodocus (Appendix 2).
MATERIALS AND METHODS
Institutional Abbreviations
All of the specimens examined in this analysis are housed in recognized repositories and are freely available for study by qualified researchers. Repositories in this analysis are as follows: Academy of Natural Sciences of Drexel University, ANS; American Museum of Natural History, AMNH; Carnegie Museum of Natural History, CM; Brigham Young University, BYU; Cincinnati Museum Center, CMC; Dinosaur National Monument, DNM; Houston Museum of Natural Science, HMNS; Great Plains Dinosaur Museum, GPDM; Museum of the Rockies, MOR; Museum of Western Colorado, MWC; National Museum of Nature and Science, NSMT; Sam Nobel Oklahoma Museum of Natural History, OMNH; Sauriermuseum Aathal, SMA; Science Museum of Minnesota, SMM; Texas Memorial Museum, TMM; United States National Museum, USNM; University of Kansas Natural History Museum, KUVP; Yale Peabody Museum, YPM.
Taxonomy and Terminology
The sauropods examined in this analysis all come from the Upper Jurassic Morrison Formation of North America. While numerous genera of Diplodocoidea and even Macronaria were examined, the presented analysis will attempt to restrict a majority of the results and discussion to the Diplodocidea Apatosaurus and Diplodocus. While many maturational hierarchies for sauropods (e.g., Curry, 1999; Ikejiri et al., 2005; Klein and Sander, 2008; Carbadillo and Sander, 2014) and other dinosaurs (Hone et al., 2016) exist, we refrained from initially using such divisions to avoid introducing preconceived ontogenetic hypotheses (i.e., not assigning maturity of a specimen before the analysis). While elemental and body size generally correlates with age - and we were tempted to colloquially refer to small individuals as “younger”, and vice versa - a whole suite of variables (including intraspecific variation) could be concealed by such basic assumptions. Throughout this analysis, for each feature examined the smaller (and presumably more immature) specimens are described first, progressing to larger (presumably more mature).
Histological descriptions follow those established by Francillon‐Vieillot et al. (1990), de Ricqlès et al. (1991), Castanet et al. (1992), and Huttenlocker et al. (2013), while long bone descriptions follow the HOS of Klein and Sander (2008; histological terminology can be found in Appendix 1). Throughout this study the terms “immature” and “mature” are used to refer specifically to skeletal maturity (mature = the presence of an external fundamental system [EFS], immature = lacks an EFS). Of interest, it is important to note the extensive colloquial and defined usages of “immature” and “mature”. Although such informal maturational phrasing can be convenient, with the pervasive usage and varying connotations applied to such terms, definition methodology must be strictly followed (for a good explanation of this situation, see Hone et al. [2016]).
One vertebral feature examined in this analysis is the bifurcated neural spine. Consisting of a paired neural spine (which can be expressed within the cervical, dorsal, and caudal series), bifurcated neural spines are found in several sauropodomorph clades and families, including: Vulcanodontidae, Mamenchisauridae, Diplodocoidea, Camarasauridae, Brachiosauridae, Turiasauria, and Titanosauria (Woodruff, 2016). Throughout this analysis, the term bifurcation shall initially follow that of Janensch’s (1929) definition to indicate a paired neural spine. While the ontogenetic development of this feature is still under study (see Woodruff and Fowler, 2012 and Wedel and Taylor, 2013), Wedel and Taylor (2013) categorized spine apex states and the degrees of bifurcation - 1) unsplit, 2) notched, 3) shallowly bifid, 4) deeply bifid. If bifurcation does develop ontogenetically, then the spine apex would progress from unsplit to bifid. In this analysis we shall follow that any unsplit or rounded spine apex is indeed not bifurcated, while any spine apex that ranges from notched to any degree of bifid is categorized herein as bifurcated.
A recent analysis of Diplodocidea (Tschopp et al., 2015) suggests that this group may be more speciose than previously thought. We will follow the taxonomy of this study and use the multi-faceted approach to determining sauropod ontogeny to examine growth and variation in this group in order to review the proposed distinction of these taxa.
Histologic Methodology
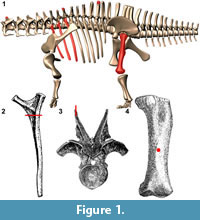 Sauropod elements were photographed from multiple angles (if possible), and all measurements were taken with digital calipers and cloth or retracting measuring tapes. For histological sampling, sites were chosen as follows (Figure 1): for dorsal ribs, sections were removed just distal to the capitulum and tuberculum (following the methods of Waskow and Sander, 2014). If a specimen was small enough, entire transverse sections of the dorsal ribs were taken, while from larger specimens a medial section was removed distal to the capitulum and tuberculum. For limb elements (femora), core sections were removed from the anterior face of the middle of the diaphysis. A longitudinal orientation line was drawn on the periosteal surface, and thin sections were taken perpendicular to this line (Stein and Sander, 2009). If a core broke during drilling, the fragments were pieced back together to the extent that was possible in order to examine microstructure.
Sauropod elements were photographed from multiple angles (if possible), and all measurements were taken with digital calipers and cloth or retracting measuring tapes. For histological sampling, sites were chosen as follows (Figure 1): for dorsal ribs, sections were removed just distal to the capitulum and tuberculum (following the methods of Waskow and Sander, 2014). If a specimen was small enough, entire transverse sections of the dorsal ribs were taken, while from larger specimens a medial section was removed distal to the capitulum and tuberculum. For limb elements (femora), core sections were removed from the anterior face of the middle of the diaphysis. A longitudinal orientation line was drawn on the periosteal surface, and thin sections were taken perpendicular to this line (Stein and Sander, 2009). If a core broke during drilling, the fragments were pieced back together to the extent that was possible in order to examine microstructure.
For samples in this analysis, previously occurring cracks and fractures were exploited if possible. A section of bone containing the desired sample location that had been glued together could be removed (by application of heat or a solvent), from which the section could be molded and cast. If previously occurring fractures could not be exploited, either a rotary tool (Dremel™) with a 3.8 cm diamond cutting wheel or a 10.8 cm diamond bladed tile saw (WorkForce™) were used to make two parallel incisions. However, no bone was cut in its entirety to remove sample material. The bone that is lost due to the cutting blade (called kerf loss) results in adjoining flush edges. During restoration work, these flush edges can result in the element having a different measurement (usually shorter, and depending on the blade width this could be the greater part of a cm discrepancy). When cutting an element, the incisions were made only partially through the bone. From here the desired sample section could be broken out, which produces a jointed edge, allowing for a precise connection for the casted section.
The coring bits pioneered by Sander (1999; 2000) and Stein and Sander (2009) allow for removal of a cylindrical sample of bone, and this can be performed in a variety of locations (such as a collection room, a lab, and even on a mounted specimen). These bits are cylindrical tools consisting of a flat end with sintered or galvanized diamond grit. In our sampling we observed that these flat end bits created a significant degree of friction and often caused flaking or chipping of the periosteal surface. Instead, damage to the periosteal surface was negligible with a serrated core bit. In like manner when sawing across wood grain, sheet woods, or delicate materials such as plexiglass, the presence of smaller and finer blade teeth in tandem produce a smoother cut. In addition to the serrated tip, we found a bit that separated into two sections (known as a two-part bit) to be invaluable. With a traditional bit, the core can become lodged and extremely difficult to remove, and attempted removal can damage or destroy the bone core. However, using a 35 mm two-part bit (Bosch™) allows for a coin (under 35 mm in diameter) to be placed on the bone surface without interfering with the cutting surface. If the core becomes lodged in the bit, the bit can simply be separated, and a larger diameter bolt (such as a carriage bolt) can be inserted into the distal half and used to indirectly push on the bone core via the coin (opposed to direct pressure on the bone surface, which could cause damage; Figure 2).
 In addition to the information pertaining to the core bit itself, we found other useful techniques to aid in the coring process. In the case of a delicate periosteal surface, a thick layer of fossil preservative (such as Vinac/McGean B-15) can be applied to the selected surface. Once dry, a layer of five-minute epoxy resin can be placed on top of the thick preservative layer, which prevents external flaking, and subsequently cored through. Afterwards acetone can be used to clean the area surrounding the bore hole, and the surface of the core if desired. Regarding use of the lubricant, Stein and Sander (2009) suggest making a clay reservoir around the core to contain the lubricant (in most cases water). This keeps the mess to a minimum, but using a squirt bottle to apply a soap and water solution to the bit during coring provides the same effect. The water cools the bit, while the soap provides extra lubrication. It is best to apply frequent lubrication, and always avoid dry bone dust for health and safety reasons, especially with potentially radioactive fossils.
In addition to the information pertaining to the core bit itself, we found other useful techniques to aid in the coring process. In the case of a delicate periosteal surface, a thick layer of fossil preservative (such as Vinac/McGean B-15) can be applied to the selected surface. Once dry, a layer of five-minute epoxy resin can be placed on top of the thick preservative layer, which prevents external flaking, and subsequently cored through. Afterwards acetone can be used to clean the area surrounding the bore hole, and the surface of the core if desired. Regarding use of the lubricant, Stein and Sander (2009) suggest making a clay reservoir around the core to contain the lubricant (in most cases water). This keeps the mess to a minimum, but using a squirt bottle to apply a soap and water solution to the bit during coring provides the same effect. The water cools the bit, while the soap provides extra lubrication. It is best to apply frequent lubrication, and always avoid dry bone dust for health and safety reasons, especially with potentially radioactive fossils.
In the case of a delicate cortex, a stepwise progression is best: core a short distance into the bone (a few mm), extract the bit, and inject some form of penetrating glue into the scored area. Heating the bone with a blow dryer (standard handheld hair dryer) prior to the introduction of glue will aid the bone in absorbing the glue. Allowing for ample drying time (several hours) is imperative; otherwise the bit can become gummed up and will be more susceptible to catching. If the bit and core do become gummed up or glued together, soaking the bit in a cup filled with an adhesive remover (such as CYANO -OFF!™) overnight will dissolve the glue. Doing this stepwise progression will stabilize both the core and surrounding area.
All samples used for histological analysis were prepared following the methods and techniques demonstrated in Padian and Lamm (2013). Samples were first embedded in a Silmar two-part epoxy resin (SIL95BA-41), and once cured, two transverse slices (~2-4 mm thick) were cut from the embedded block using a Felker 41-AR tile saw. Slices were then pre-mount ground on the surface to be glued to the glass slide using 320 grit and then 600 grit silicon carbide paper. After this initial grinding, the sample is mounted to frosted glass slides using a Devcon two-part epoxy glue. Then using a Buehler Ecomet 4 Variable Speed Grinder-Polisher, the thin sections were polished using Buehler silicon carbide paper sequentially from a 60 grit to an 800 grit finish. Final slide thickness was approximately ~150 μm, but varied between samples to achieve similar optical clarity. Finished slides were photographed using a Nikon Optiphot-Pol polarizing microscope equipped with a Nikon DS-Fi1 digital camera and compiled with NIS-Elements BR 3.0 software.
In addition to the histologic analyses, Computed Tomography scans (CT scan) of cervical and dorsal vertebrae were performed to examine pneumatic architecture. CT scanning was conducted by Advanced Medical Imaging at Bozeman Deaconess Hospital in Bozeman, MT, U.S.A. using a Toshiba Aquilion 64 CT Scanner. The peak kilovoltage (pKv) for each scan was approximately 135 pKv, and scan resolution (varied due to the dimensions of each vertebra) ranged from every 2 mm up to 5 mm. Scan data were uploaded into the DICOM Viewer OsiriX for multi-plane study, with individual planes being analyzed using the image processing program ImageJ (Abramoff et al., 2004).
Examination Methodology
In exceptional ontogenetic studies, large sample sizes including representatives of multiple growth stages allow for more comprehensive histologic and morphological comparisons (e.g., Dodson, 1975; Carr, 1999; Scannella and Horner, 2010; Horner et al., 2011; Frederickson and Tumarkin-Deratzian, 2014; Woodward et al., 2015). While Morrison diplodocids are by far one of the most common of all North American dinosaur groups, taphonomic biases, stratigraphic resolution, and taxonomic uncertainties generally result in largely incomplete and less well understood individuals.
Several studies have previously documented possible ontogenetic changes in sauropods (including pneumatic architecture, Wedel [2003]; HOS, Klein and Sander [2008]; cranial changes Whitlock et al. [2010]; neural spine bifurcation, Woodruff and Fowler [2012]; MOS, Carballido and Sander [2014]; LAG record, Waskow and Sander [2014]); yet the majority of these analyses examine a single feature (e.g., spine bifurcation [Woodruff and Fowler, 2012]) or a single methodology (i.e., morphology or histology; HOS [Klein and Sander, 2008]). While all of these previous works are invaluable contributions to our understanding of sauropodomorph paleobiology, they represent individual puzzle pieces. Although beneficial, such analyses have little capacity to reflect on the individual sauropod as a whole. Since there are differences in histologic, morphologic, or cladistic approaches, different emphases may lead to conflicting taxonomic interpretations (as in the case of Suuwassea or SMA 0009; Harris [2006a]; Schwarz et al. [2007b]; Whitlock and Harris [2010], Carballido et al. [2012]; Woodruff and Fowler [2012]; Tschopp et al. [2015]).
However, by analyzing a range of skeletal features, and including varied histologic and morphologic examinations, such an encompassing methodology can allow for better resolution of the entire animal. In lieu of a much larger sample of complete diplodocid specimens forming an ontogenetic series, the available material has been used to build an encompassing set of histologic and morphologic features creating a system of supportive characters that we term the Histo-Morph Ontogeny Scale (H-MOS; Appendix 3).
Methodology of Examined Skeletal Features
CT scans of pneumatic architecture. The hypothesis that vertebral pneumatic architecture develops ontogenetically is strongly supported (Wedel, 2003), but has yet to be documented both externally and internally within an ontogenetic series (note Carballido and Sander [2014] do demonstrate external pneumatic architecture among ontogenetic stages of Europasaurus). In order to document this pattern within an ontogenetic series, we examined the internal morphology of cervical and dorsal pneumatic structures from our ontogenetic series using Computed Tomography (CT scan). Only cervical and dorsal vertebrae from known or confidently estimated positions were scanned. The scanned elements consist of two anterior to middle cervical vertebrae from two individuals (MOR 714 [diplodocid indeterminate] and MOR 790 [Diplodocus sp.]), two anterior dorsal vertebrae from two individuals (MOR 790 and MOR 592; both Diplodocus sp.), and three posterior dorsal vertebrae from three individuals (MOR 790, MOR 592 [both Diplodocus sp.], and MOR 957 [Apatosaurus sp.]). Each specimen was examined via frontal and transverse sections. Definitions and usage of pneumatic architecture follow those established by Wedel et al. (2000; Appendix 1).
Vertebral neural spines. To determine if morphological changes in neural spines correspond with changes in microstructure, four neural spine apices (two posterior cervical vertebrae [MOR-790 un-numbered and MOR 592 8-24-90-91; both Diplodocus sp.] and two anterior dorsal vertebrae [MOR 790 8-21-95-238 and MOR 592 8-22-90-15; both Diplodocus sp.]) were coronally sampled to examine histology across the neural spine, and between varying body sizes.
Dorsal ribs. Because sauropods grew so rapidly and their limb bones were generally highly remodeled, lines of arrested growth (LAGs) in limbs are not well preserved, making age determination difficult. Due to the challenges involved with sauropod limb histology, Waskow and Sander (2014) and Waskow and Mateus (2017) recently demonstrated that dorsal ribs record cyclical growth marks. The identification and recognition of recordable growth histories in sauropods finally allows for histological age determination (opposed to maturational inference [HOS] and tabulated longevity calculations). Waskow and Sander (2014) found that sampling dorsal ribs 1-3 within the proximal third of the rib, but distal to the capitulum and tuberculum, recorded the most intact and complete history of growth marks (LAGs).
In this analysis, we sampled the dorsal ribs of four diplodocids (MOR 790 7-24-96-95, MOR 790 7-27-8-96, MOR 592 [all Diplodocus sp.], CM 94 [D. carnegii]). Unfortunately no articulated Apatosaurus or Diplodocus dorsal rib cages were available for this analysis; therefore specimens could only be selected by size and thus hypothesized maturity (smaller - presumably less mature, larger - presumably more mature). For these isolated ribs, individual morphology was heavily scrutinized in order to make sure serial positions were consistent. Further, following Waskow and Sander (2014), we examined the histology of femora associated with the sampled dorsal ribs (note that MOR 592 [Diplodocus sp.] is the only specimen that is represented by a single individual; MOR 790 [Diplodocus sp.] comes from a bone bed which consists of an MNI of 15 [Storrs et al., 2012], and CM 94 [D. carnegii] of an MNI of 2 [Hatcher, 1901]). See sections discussing MOR 790 for how we selected elements/individuals). For isolated dorsal ribs, comparisons to well-documented rib series were used to approximate serial position (Dicraeosaurus - Janensch, 1929; Apatosaurus - Gilmore, 1936).
For Apatosaurus and Diplodocus in this dataset, estimated individual age was based on preserved LAG counts. From the preserved LAG record, Griebeler et al. (2013) and Waskow and Sander (2014) use a retro-calculation method to formulate age estimates. For the retro-calculation, the minimum and maximum distance between LAGs must be measured. Lines are then drawn along the short and long axis, with the origin representing the hypothetical original center of the rib. From there the previously measured distances are marked until reaching the origin and thus attaining a hypothetical maximum age value. While this methodology is convenient, straightforward, and simplistic, this retro-calculation method implies uniform growth rates throughout ontogeny. Whereas some studies suggest that sauropods had a slower initial growth rate (e.g., Erickson et al., 2001; Rogers and Erickson, 2005; Griebeler et al., 2013), others indicate that immature dinosaurs exhibited faster growth early in ontogeny (Curry, 1999; Horner et al., 1999, 2000; Sander, 2000; Erickson et al., 2001; Padian et al., 2001; Horner and Padian, 2004; Sander et al., 2004; Erickson, 2005; Lee and Werning, 2008; Lehman and Woodward, 2008; Woodward and Lehman, 2009; Sander and Tückmantel, 2003; Fowler et al., 2011; Tsuihiji et al., 2011; Campione et al., 2013). If so, the distances between LAGs in immature diplodocids should be greater during early development. Thus the retro-calculation methods likely represent maximum age estimates. However, this retro-calculation method will be beneficial in the analysis of specimens with significantly more remodeling in which LAG counts are obscured and thus maximum age can be estimated.
Femora. To analyze relative maturities using long-bone histology, this analysis relied heavily on the sample set and HOS of Klein and Sander (2008). Two femora from this study’s dataset were sectioned (MOR 790 and MOR 592 [both Diplodocus sp.]) and compared to the Klein and Sander (2008) samples to support previously inferred and hypothesized maturational stages. Histological designations for femora in this analysis follow the numerical and alphabetical designations of Klein and Sander (2008). The histological ontogenetic stage (HOS) of Klein and Sander (2008) follows a 13-part maturational system - HOS 1 being the most immature bone tissue types, HOS 13 the oldest - which consists of a 7-part alphabetical hierarchy denoting bone microstructure - bone Type A having no primary osteons, bone Type G consisting entirely of multi-generational secondary osteons (Klein and Sander, 2008). However, in any analysis using the HOS, one must be aware that HOS values do not correspond to age of the individual; e.g., an HOS of 4 does not correspond to a four year old animal - it represents a demarcated designation in bone tissue morphology.
Body mass estimates were calculated for fifty-four diplodocid femora (Appendix 4) following the allometry-based body mass formula of Mazzetta et al. (2004). Additional mass estimation methods that incorporate multiple elements (such as humeral and femoral circumference; i.e., Campione and Evans, 2012) and histologic data (Sander and Tückmantel, 2003; Lehman and Woodward, 2008; Griebeler et al., 2013) should produce more accurate estimates because these methods incorporate more variables regarding the individual and its life history. However, the diplodocid dataset in this analysis lacks the required inclusive life history variables (i.e., corresponding femur and humerus [Campione and Evans, 2012], or histologic markers - LAGs or polish lines [Sander and Tückmantel, 2003; Griebeler et al., 2013]). As such the most agreeable method that can be incorporated with this dataset is that of Mazzetta et al. (2004). Once mass values were calculated, 10% was deducted to account for pneumaticity (following Wedel, 2003). Since the degree of pneumatization at different body sizes is not known, we decided to deduct a standard value of pneumatization. However, it is important to note that such standardization would imply isometry, which this analysis clearly advocates against; therefore 10% reduction could represent the maximum for immature and the minimum for mature animals.
RESULTS
Morphologic Data
Neural spine bifurcation. As stated previously, one of the two contentious points to the Woodruff and Fowler (2012) findings is whether neural spine bifurcation follows an ontogenetic trajectory (Wedel and Taylor, 2013). Much of this disagreement arises from the use of isolated elements where precise serial placement is at best an approximation (such as the material from the Mother’s Day Quarry); and we agree that serial position is important in regards to spine bifurcation.
 It is unequivocal that the degrees and morphology expressed in neural spine bifurcation vary amongst all of sauropods with this vertebral modification (sensu Woodruff, 2016). Likewise the complex relationship between vertebral column length, column mass, column mobility, and size of the individual animal are all factors that affect and influence bifurcation, even within the same taxon (Woodruff, 2016; Woodruff and Foster, 2017). Therefore we should not think of bifurcation as a static developmental process, and we now know this "defined" pathway as originally proposed by Woodruff and Fowler (2012) is not an entirely accurate reflection. Amongst certain taxa spine bifurcation may progress from rounded to fully bifid in one, notched to shallowly bifid in another, or shallowly to fully bifid in a third. While the initial and final spine condition may vary across taxa - and certainly the mechanics of the cervical columns do vary across taxa - spine morphologies do vary in some capacity from immature to mature individuals. While some of these developmental trajectories may be more dramatic than others (Diplodocus [Woodruff and Fowler, 2012] compared to Barosaurus [Melstrom et al., 2016]), any morphologic change through growth is ontogenetic development in any definition or capacity. Since Woodruff and Fowler's (2012) initial report, the authors have examined material representing a significant portion of known Morrison diplodocids collected to date (and ongoing work by DCW). In addition to the numerous isolated elements, enough associated, formerly articulated, and articulated specimens demonstrate that while there is a great degree in variation in every respect, neural spine bifurcation does indeed develop ontogenetically (Figure 3).
It is unequivocal that the degrees and morphology expressed in neural spine bifurcation vary amongst all of sauropods with this vertebral modification (sensu Woodruff, 2016). Likewise the complex relationship between vertebral column length, column mass, column mobility, and size of the individual animal are all factors that affect and influence bifurcation, even within the same taxon (Woodruff, 2016; Woodruff and Foster, 2017). Therefore we should not think of bifurcation as a static developmental process, and we now know this "defined" pathway as originally proposed by Woodruff and Fowler (2012) is not an entirely accurate reflection. Amongst certain taxa spine bifurcation may progress from rounded to fully bifid in one, notched to shallowly bifid in another, or shallowly to fully bifid in a third. While the initial and final spine condition may vary across taxa - and certainly the mechanics of the cervical columns do vary across taxa - spine morphologies do vary in some capacity from immature to mature individuals. While some of these developmental trajectories may be more dramatic than others (Diplodocus [Woodruff and Fowler, 2012] compared to Barosaurus [Melstrom et al., 2016]), any morphologic change through growth is ontogenetic development in any definition or capacity. Since Woodruff and Fowler's (2012) initial report, the authors have examined material representing a significant portion of known Morrison diplodocids collected to date (and ongoing work by DCW). In addition to the numerous isolated elements, enough associated, formerly articulated, and articulated specimens demonstrate that while there is a great degree in variation in every respect, neural spine bifurcation does indeed develop ontogenetically (Figure 3).
Postparietal foramen. In some sauropods, there is a cranial foramen situated between the posterior portion of the parietals and the anterior portion of the supraoccipital. Known as the postparietal foramen, this feature has been inferred as a synapomorphy of Dicraeosauridae (Salgado and Bonaparte, 1991; Harris, 2006a; Remes, 2009; Whitlock, 2011a), an autapomorphy for the plateosaurian sauropodomorphs Massospondylus and Plateosaurus (Knoll et al., 2012), the basal sauropod Spinophorosaurus (Knoll et al., 2012), and the diplodocids Tornieria (Upchurch et al., 2004a; Remes, 2009), Kaatedocus (Tschopp and Mateus, 2013), and Galeamopus (Tschopp et al., 2015). Yet outside of Kaatedocus, Galeamopus, Tornieria, and Suuwassea (Lovelace et al., 2007; Whitlock and Harris, 2010; Whitlock, 2011a; Woodruff and Fowler, 2012), no members of Diplodocidae or other Late Jurassic sauropods are known to possess this cranial feature; however, given the scarcity of sauropod cranial material, it is possible that this feature may be more widespread in Sauropoda (Harris, 2006a; Whitlock and Harris, 2010; Hedrick et al., 2014).
 Balanoff et al. (2010) reported an Apatosaurus sp. braincase (BYU 17096) with a postparietal foramen, making it the first definitive member of Apatosaurinae to possess this feature. Subsequent examinations of Apatosaurus sp. (MOR 700), and Diplodocus sp. (CMC VP14128, MOR 592, and MOR 7029) reveal that all of these specimens possess a postparietal foramen (Figure 4). In some specimens, such as in Apatosaurus sp. (BYU 17096 and MOR 700) and Galeamopus (SMA 0011), the postparietal foramen appears to be immediately posterior to, or posteriorly connected to the frontoparietal fenestra, while in others the fenestra and foramen are spaced apart. The exact morphology of the foramen can be difficult to discern due to damaged margins, but in general it is ovoid in outline. In some specimens the foramen’s greatest axis is transverse to the long axis of the skull (MOR 700), while in others it appears to be parallel to the long axis of the skull (SMA 0011; note this skull is damaged, so this morphology may be taphonomically altered).
Balanoff et al. (2010) reported an Apatosaurus sp. braincase (BYU 17096) with a postparietal foramen, making it the first definitive member of Apatosaurinae to possess this feature. Subsequent examinations of Apatosaurus sp. (MOR 700), and Diplodocus sp. (CMC VP14128, MOR 592, and MOR 7029) reveal that all of these specimens possess a postparietal foramen (Figure 4). In some specimens, such as in Apatosaurus sp. (BYU 17096 and MOR 700) and Galeamopus (SMA 0011), the postparietal foramen appears to be immediately posterior to, or posteriorly connected to the frontoparietal fenestra, while in others the fenestra and foramen are spaced apart. The exact morphology of the foramen can be difficult to discern due to damaged margins, but in general it is ovoid in outline. In some specimens the foramen’s greatest axis is transverse to the long axis of the skull (MOR 700), while in others it appears to be parallel to the long axis of the skull (SMA 0011; note this skull is damaged, so this morphology may be taphonomically altered).
Macroscopic examination of pneumatic structures in a proposed ontogenetic series. The smallest specimen examined in this study is SMA 0009, a specimen that was initially referred to Diplodocidae (Schwarz et al., 2007b) but that was later recovered as an immature brachiosaurid in phylogenetic analyses (Carballido et al., 2012; Tschopp et al., 2015). We follow Schwarz et al. (2007b) in considering this specimen a diplodocid and note that the forked [a.k.a. “sled”-shaped] chevrons of SMA 0009 (which while coded by Tschopp et al. [2015], such morphology is not observed in Camarasaurus or Brachiosaurus) are further indicators of diplodocid affinity (although it is possible that chevron morphology changes ontogenetically [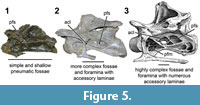 Otero et al., 2012], and “sled”-shaped chevrons are documented in basal sauropodomorphs). SMA 0009 represents an animal with an approximately 50 cm long cervical series. Our description of SMA 0009 follows that of Schwarz et al. (2007b). Macroscopically the cervical vertebrae appear acamerate (fossae do not invade centrum). The pneumatic fossae (not leading to internal chambers) of the cervical series are structurally simple, consisting of a shallow oval that extend for much of the length of each centrum. On the mid-cervical vertebrae the pneumatic fossae have an average depth of 4 mm, while in the posterior cervical the depth increases up to 8 mm. On the anterior and posterior cervical vertebrae, an accessory lamina divides the pneumatic fossae. On the anteriormost cervical vertebrae, this accessory lamina separates an anterior and posterior fossa; on the posteriormost cervical vertebrae these laminae are weakly expressed (Figure 5).
Otero et al., 2012], and “sled”-shaped chevrons are documented in basal sauropodomorphs). SMA 0009 represents an animal with an approximately 50 cm long cervical series. Our description of SMA 0009 follows that of Schwarz et al. (2007b). Macroscopically the cervical vertebrae appear acamerate (fossae do not invade centrum). The pneumatic fossae (not leading to internal chambers) of the cervical series are structurally simple, consisting of a shallow oval that extend for much of the length of each centrum. On the mid-cervical vertebrae the pneumatic fossae have an average depth of 4 mm, while in the posterior cervical the depth increases up to 8 mm. On the anterior and posterior cervical vertebrae, an accessory lamina divides the pneumatic fossae. On the anteriormost cervical vertebrae, this accessory lamina separates an anterior and posterior fossa; on the posteriormost cervical vertebrae these laminae are weakly expressed (Figure 5).
The next size range consists of individuals with a cervical series up to approximately 3 m in length. Individuals representing this size range, such as Kaatedocus SMA 0004, the Apatosaurus CM 3390, all exhibit procamerate to camerate (various degrees of pervading complexity into the centrum) cervical vertebrae. In this size class all of the pneumatic fossae are deepened and well defined. As in the previous size range, the fossae and foramina of the anterior cervicals are shallower than those in the posterior cervical vertebrae. The anterior fossae/foramina range from a depth of ~7 mm to 13 mm, while the posterior ones range from ~ 15 mm to as deep as 24 mm in SMA 0004. Additionally, the primary accessory laminae along with both the posterior centrodiapophyseal lamina and the postzygodiapophyseal laminae are more pronounced (Figure 5).
The next size range collectively represents the largest (and presumably sexually mature) specimens in the dataset. However, within this large size range, there are two discernible groups. The first group represents specimens with cervical series 4-6 m in length, such as the Apatosaurus excelsus (or Brontosaurus excelsus) CM 555 and the Diplodocus sp. specimen MOR 592. As in the smaller specimens, the fossae and foramina continue to increase in size and depth. Likewise, all of the associated laminae continue to grow. The final size range represents the mature condition. The primary difference observed within this collective range is the degree and abundance of fossae and foramina. In examining the Diplodocus carnegii CM 84, the degree and number of foramina and laminae is dramatically increased (Figure 5). The greatest concentrations of these laminae are still in the mid- and posterior cervical, but in this final condition, these features even proceed into the anterior portion of the cervical series. In some of these foramina-rich centra, such as C-11 of CM 84, the degree of laminae gives the appearance of a network of honeycomb.
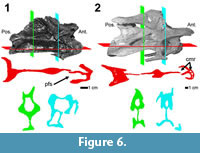 Computed tomography. In the smallest anterior to middle cervical vertebra (diplodocid indeterminate MOR 714 7-22-3-53), the centrum is acamerate and appears to lack any secondary pneumatic structures. In both frontal and transverse views, the portion of the median septum bounded by the fossae has a fairly uniform thickness. Also in frontal view the lateral margins of the condyle are rather bulbous at their extremities. In the anterior to middle cervical vertebra of Diplodocus sp. MOR 790 8-10-96-204, we see in comparison to diplodocid indeterminate MOR 714 7-22-3-53 that the median septum has continued thinning. The most dramatic change is the pneumatic structures of the condyle; in frontal view the condyle has deep excavations, as in diplodocid indeterminate MOR 714 7-22-3-53, but in Diplodocus sp. MOR 790 8-10-96-204, the lateral margins of the condyle have continued to thin and taper. The bulk of the condyle is composed of large camerae (larger rounded cavities with a regular pattern) as observed in other sauropod condyles (Wedel, 2003; Schwarz and Fritsch, 2006; Schwarz et al., 2007a). The posterior portion of the cotyle also seems to possess camerae (Figure 6).
Computed tomography. In the smallest anterior to middle cervical vertebra (diplodocid indeterminate MOR 714 7-22-3-53), the centrum is acamerate and appears to lack any secondary pneumatic structures. In both frontal and transverse views, the portion of the median septum bounded by the fossae has a fairly uniform thickness. Also in frontal view the lateral margins of the condyle are rather bulbous at their extremities. In the anterior to middle cervical vertebra of Diplodocus sp. MOR 790 8-10-96-204, we see in comparison to diplodocid indeterminate MOR 714 7-22-3-53 that the median septum has continued thinning. The most dramatic change is the pneumatic structures of the condyle; in frontal view the condyle has deep excavations, as in diplodocid indeterminate MOR 714 7-22-3-53, but in Diplodocus sp. MOR 790 8-10-96-204, the lateral margins of the condyle have continued to thin and taper. The bulk of the condyle is composed of large camerae (larger rounded cavities with a regular pattern) as observed in other sauropod condyles (Wedel, 2003; Schwarz and Fritsch, 2006; Schwarz et al., 2007a). The posterior portion of the cotyle also seems to possess camerae (Figure 6).
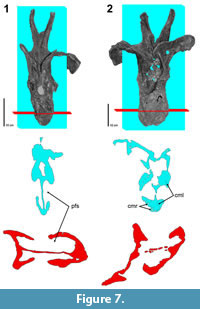 Collectively, the diplodocid dorsal vertebral series represent a more complete trajectory (in total consisting of five scanned vertebrae). In the anterior dorsal, Diplodocus sp. MOR 790 8-21-95-238, the median septum is uniformly thin and the pneumatic fossae extend deeply into the condyle. Both the condyle and cotyle of Diplodocus sp. MOR 790 8-21-95-238 are deeply penetrated by pneumatic fossae. It would also appear that internal pneumatic structures are nearly absent. In frontal view only one possible small camera is observed in the condyle. In the Diplodocus sp. MOR 592 8-22-90-75, there is continuing invasion of the pneumatic fossae into the condyle. Due to lateral shearing, the median septum is highly fragmented. In frontal view it would appear that Diplodocus sp. MOR 592 8-22-90-75 lacks any internal pneumatic structures. However, in transverse view there are several camerae and camellae (smaller angular cavities with an irregular pattern), primarily along the peripheral margins of the centrum. Diplodocus sp. MOR 592 8-22-90-75 also marks the first appearance of pneumatic structures within the neural arch. Those in the arch would likewise appear to consist of camerae and camellae (Figure 7).
Collectively, the diplodocid dorsal vertebral series represent a more complete trajectory (in total consisting of five scanned vertebrae). In the anterior dorsal, Diplodocus sp. MOR 790 8-21-95-238, the median septum is uniformly thin and the pneumatic fossae extend deeply into the condyle. Both the condyle and cotyle of Diplodocus sp. MOR 790 8-21-95-238 are deeply penetrated by pneumatic fossae. It would also appear that internal pneumatic structures are nearly absent. In frontal view only one possible small camera is observed in the condyle. In the Diplodocus sp. MOR 592 8-22-90-75, there is continuing invasion of the pneumatic fossae into the condyle. Due to lateral shearing, the median septum is highly fragmented. In frontal view it would appear that Diplodocus sp. MOR 592 8-22-90-75 lacks any internal pneumatic structures. However, in transverse view there are several camerae and camellae (smaller angular cavities with an irregular pattern), primarily along the peripheral margins of the centrum. Diplodocus sp. MOR 592 8-22-90-75 also marks the first appearance of pneumatic structures within the neural arch. Those in the arch would likewise appear to consist of camerae and camellae (Figure 7).
The posterior dorsal series exhibits perhaps the best ontogenetic development of the pneumatic structures. In examining the series developmentally, the Diplodocus sp. MOR 790 7-8-95-17 is structurally simple. The centrum is acamerate and lacks any sort of internal pneumatic structures (as hypothesized by Wedel, 2003). In addition, the medially shallow lateral fossae are separated by a very thick median septum. In the Diplodocus sp. MOR 592 8-22-90-77, the lateral pneumatic fossae extend medially into the centrum, producing a relatively thinner median septum. In addition to the thinning of the median septum, numerous internal pneumatic structures are present. The cotyle appears to consist of a complex of fairly interconnected camerae with some less numerous camellae, whereas the condyle appears to contain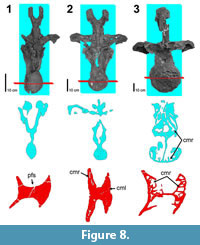 primarily interspersed camellae. There also appear to be some small camerae and camellae in the neural arch. Progressing to the largest posterior dorsal vertebra in this series, Apatosaurus sp. MOR 957 6-29-92 29#1 is significantly larger than Diplodocus sp. MOR 592 8-22-90-77; unfortunately a suitable intermediate specimen was not available for study. The most notable feature of Apatosaurus sp. MOR 957 6-29-92 29#1 is the elaborate and extensive pneumatization. The lateral pneumatic fossae have extended deep into the centrum to produce a median septum that in certain locations is under 2 cm in thickness. In frontal view both the condyle and cotyle are completely composed of large (several cm in greatest length) extensive camerae. In transverse view there is even more apparent pneumatization. The thin median septum and large camerae of the centrum are evident, and the neural arch hosts a series of elongate camerae. These neural arch camerae gently arc medially towards the neural canal, and their overall size decreases dorsally while the dividing septa increase in thickness dorsally (Figure 8).
primarily interspersed camellae. There also appear to be some small camerae and camellae in the neural arch. Progressing to the largest posterior dorsal vertebra in this series, Apatosaurus sp. MOR 957 6-29-92 29#1 is significantly larger than Diplodocus sp. MOR 592 8-22-90-77; unfortunately a suitable intermediate specimen was not available for study. The most notable feature of Apatosaurus sp. MOR 957 6-29-92 29#1 is the elaborate and extensive pneumatization. The lateral pneumatic fossae have extended deep into the centrum to produce a median septum that in certain locations is under 2 cm in thickness. In frontal view both the condyle and cotyle are completely composed of large (several cm in greatest length) extensive camerae. In transverse view there is even more apparent pneumatization. The thin median septum and large camerae of the centrum are evident, and the neural arch hosts a series of elongate camerae. These neural arch camerae gently arc medially towards the neural canal, and their overall size decreases dorsally while the dividing septa increase in thickness dorsally (Figure 8).
Ontogenetic Data
As histology is the only conclusive way to determine bone growth, multiple skeletal elements were sampled histologically.
Neural spine histology. In the posterior cervical of Diplodocus sp. MOR-790 (an un-numbered vertebra here designated un-numbered 1; and anterior dorsal of Diplodocus sp. MOR 790 8-21-95-238) the inter-trabecular spaces are small and generally divided into two size ranges: those well under 1 mm and those equal to or larger than 1 mm (however, in Diplodocus sp. MOR-790 un-numbered; here designated un-numbered 2; a few of the inter-trabecular spaces are up to 3.5 mm in greatest length; Figure 9). In the non-bifurcated Diplodocus sp. MOR-790 un-numbered 2, the apex of the undivided neural spine is more compact, with the bulk of the more cancellous bone loosely oriented in a convex arc across the width of the neural spine.
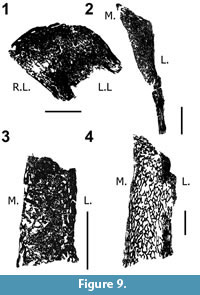 In the Diplodocus sp. MOR 790 8-21-95-238, the lateral margin is primarily compact bone with the more cancellous bone restricted to the medial margin of the split spine. This feature is likewise observed in the posterior cervical (8-24-90-91) and anterior dorsal (8-22-90-15) of Diplodocus sp. MOR 592. In the bifurcated Diplodocus MOR 592 8-24-90-91 there are two ranges of inter-trabecular sizes (far more of the larger spaces), and the spaces are distributed throughout the spine’s apex (Figure 9). In addition, the lateral margin of Diplodocus sp. MOR 592 8-24-90-91 is more compact with the largest inter-trabecular spaces being restricted along the medial border. In Diplodocus sp. MOR 592 8-22-90-15 the inter-trabecular spaces are elongate and generally much larger than those from the previously discussed specimens (approximately 3 mm or larger). The lateral margin of the spine’s apex is primarily compact with a more cancellous medial periphery. A much more complete ontogenetic series is needed to substantiate and correlate these results, but potentially these specimens suggest that throughout ontogeny, inter-trabecular spaces increase in size and orientation, and as bifurcation of the spine develops, so changes the degree and location of these bone types within the spine’s apex.
In the Diplodocus sp. MOR 790 8-21-95-238, the lateral margin is primarily compact bone with the more cancellous bone restricted to the medial margin of the split spine. This feature is likewise observed in the posterior cervical (8-24-90-91) and anterior dorsal (8-22-90-15) of Diplodocus sp. MOR 592. In the bifurcated Diplodocus MOR 592 8-24-90-91 there are two ranges of inter-trabecular sizes (far more of the larger spaces), and the spaces are distributed throughout the spine’s apex (Figure 9). In addition, the lateral margin of Diplodocus sp. MOR 592 8-24-90-91 is more compact with the largest inter-trabecular spaces being restricted along the medial border. In Diplodocus sp. MOR 592 8-22-90-15 the inter-trabecular spaces are elongate and generally much larger than those from the previously discussed specimens (approximately 3 mm or larger). The lateral margin of the spine’s apex is primarily compact with a more cancellous medial periphery. A much more complete ontogenetic series is needed to substantiate and correlate these results, but potentially these specimens suggest that throughout ontogeny, inter-trabecular spaces increase in size and orientation, and as bifurcation of the spine develops, so changes the degree and location of these bone types within the spine’s apex.
Dorsal rib histology. The smallest (and presumably most immature) specimens sampled represent the size extremes recorded from the Mother’s Day Quarry. The Mother’s Day Quarry represents a bone bed of at least 15 immature diplodocids with femoral lengths between 102 and 120 cm. Unfortunately, the Mother’s Day material is largely disarticulated and disassociated, so serial position or association is an approximation. If the femora extremes represent the minimum and maximum size range, we hypothesize that the smallest and largest anteriormost dorsal ribs likewise reflect these size extremes.
Within the smallest Mother’s Day dorsal rib (Diplodocus sp. MOR 790 7-24-96-95; Figure 10), the bone microstructure is predominantly woven (highly disorganized, indicating fast growth). The bone is highly vascularized, and throughout there are numerous resorption cavities (~1 mm in greatest diameter). The vascular canal orientation throughout is principally longitudinal and reticular. In the vicinity of the anterior intercostal ridge, the periosteal cortex vascular canal orientation is uniformly longitudinal, while the medialmost portion transitions from longitudinal to reticular. Secondary osteons are principally located adjacent to the deep cortex trabecular bone and within the anterior intercostal ridge. This varying microstructural anatomy within a single section is why Waskow and Sander (2014) emphasize that dorsal ribs should not be cored; sampling in three different locations could yield three entirely different life history interpretations. While there are no continuous LAGs or indications of growth cessation, there are numerous smaller, semi-opaque, and non-continuous line segments. These segments are primarily concentrated into two distinct regions, and these regions collectively are much thicker than a LAG. Between these segments are smaller regions of slower, but continuous growth; as such we identify these regions as annuli (sensu Francillon‐Vieillot et al., 1990). It must be explicitly stated that an annuli represent a slowing of osteogenesis, not an annual cessation (as in the case of LAGs; Castanet et al., 1992). Therefore, we infer that this individual records a minimum of two annual growth markers.
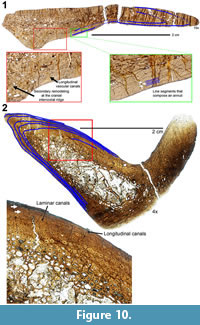 Since no annuli are observed further within the cortex, we would suggest that all - if not the majority - of the growth record is present. Alternatively it could be suggested that portions of the growth record may not be present (due to many possibilities ranging from remodeling to cortical drift). The retrocalculation method of Waskow and Sander (2014) may be applicable in determining maximum age estimates. Waskow and Sander’s (2014) retrocalculation methodology requires measuring the smallest and largest LAGs intervals and then marking off the distance until reaching the rib origin. While this methodology will prove incredibly useful in age estimation, such a methodology implies uniform growth (and the dinosaurian record explicitly argues against such). Using a modified version of Waskow and Sander’s (2014) retrocalculation method, we used the greatest LAG spacing to calculate a maximum age estimate (this technique has been used for other sauropods [Woodruff and Foster, 2017]). Using this methodology we calculate an absolute maximum age of death of 7 years for MOR 790 7-24-96-95 (but we hypothesize this animal’s actual age to be much closer to the histologic value).
Since no annuli are observed further within the cortex, we would suggest that all - if not the majority - of the growth record is present. Alternatively it could be suggested that portions of the growth record may not be present (due to many possibilities ranging from remodeling to cortical drift). The retrocalculation method of Waskow and Sander (2014) may be applicable in determining maximum age estimates. Waskow and Sander’s (2014) retrocalculation methodology requires measuring the smallest and largest LAGs intervals and then marking off the distance until reaching the rib origin. While this methodology will prove incredibly useful in age estimation, such a methodology implies uniform growth (and the dinosaurian record explicitly argues against such). Using a modified version of Waskow and Sander’s (2014) retrocalculation method, we used the greatest LAG spacing to calculate a maximum age estimate (this technique has been used for other sauropods [Woodruff and Foster, 2017]). Using this methodology we calculate an absolute maximum age of death of 7 years for MOR 790 7-24-96-95 (but we hypothesize this animal’s actual age to be much closer to the histologic value).
The largest represented dorsal rib from the Mother’s Day Quarry is Diplodocus sp. MOR 790 7-27-8-96 (Figure 10). Like the smallest Mother’s Day specimen, Diplodocus sp. MOR 790 7-27-8-96 is highly vascularized with numerous resorption cavities (up to ~2 mm in greatest diameter). Adjacent to the deep-cortex trabecular bone is a relatively large (up to several mm in thickness) region of secondary remodeling. Within this lateral remodeled region, up to two generations of secondary osteons are observed. The remainder of the medial cortex is comprised of woven bone with a few secondary osteons infrequently dispersed. Endosteally the vascular canal orientation exhibits longitudinal and laminar regions, while the outermost cortex transitions from laminar to longitudinal canals. Like the smaller MDQ dorsal rib, the anterior intercostal ridge is highly remodeled and consists of Haversian bone comprising up to four generations of secondary osteons. Diplodocus sp. MOR 790 7-27-8-96 does preserve distinct and discernable LAGs. These preserved LAGs are largely restricted to the outer portion of the cortex (note not outermost, and therefore not an EFS), and there are a recorded minimum of six preserved LAGs (there may be up to three additional LAGs in the innermost cortex, but this region is highly remodeled, and these line sections are not continuous nor seen elsewhere, therefore we tentatively do not identify them as such). Using the age estimation methodology outlined above, we estimate a maximum age of death of 10 years for MOR 790 7-27-8-96.
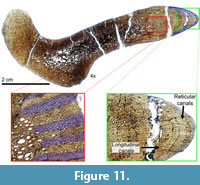 The next size range is represented by the Diplodocus sp. MOR 592 (Figure 11). As in the MDQ dorsal ribs, the dorsal rib of the Diplodocus MOR 592 is highly vascularized with numerous resorption cavities. The anterior intercostal ridge is composed entirely of dense Haversian bone, however, the medial portion of the rib records pertinent life history information. Adjacent to the deep cortex trabecular bone, there is a large (approximately 2 cm) region of highly vascular, longitudinal, woven bone. This region is composed of longitudinal vascular canals arranged in radial rows (sensu Francillon‐Vieillot et al., 1990). Immediately adjacent to this highly vascularized longitudinal region for the remainder of the cortex is an area of less vascularized primary bone. In this outermost region the vascular canal orientation changes from longitudinal to reticular. The dorsal rib of Diplodocus sp. MOR 592 records a minimum of eight observable LAGS; however, no LAGs are visible within or prior to the highly vascularized longitudinal area. In using the aforementioned age estimation technique, we estimate a maximum age of death of 12 years for MOR 592.
The next size range is represented by the Diplodocus sp. MOR 592 (Figure 11). As in the MDQ dorsal ribs, the dorsal rib of the Diplodocus MOR 592 is highly vascularized with numerous resorption cavities. The anterior intercostal ridge is composed entirely of dense Haversian bone, however, the medial portion of the rib records pertinent life history information. Adjacent to the deep cortex trabecular bone, there is a large (approximately 2 cm) region of highly vascular, longitudinal, woven bone. This region is composed of longitudinal vascular canals arranged in radial rows (sensu Francillon‐Vieillot et al., 1990). Immediately adjacent to this highly vascularized longitudinal region for the remainder of the cortex is an area of less vascularized primary bone. In this outermost region the vascular canal orientation changes from longitudinal to reticular. The dorsal rib of Diplodocus sp. MOR 592 records a minimum of eight observable LAGS; however, no LAGs are visible within or prior to the highly vascularized longitudinal area. In using the aforementioned age estimation technique, we estimate a maximum age of death of 12 years for MOR 592.
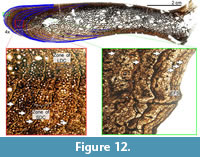 The final and largest specimen is represented by the paratype of Diplodocus carnegii (CM 94; Figure 12). As in all of the aforementioned dorsal ribs, that of CM 94 is highly vascularized and possesses a dense network of deep-cortex trabecular bone. However, unlike the previous dorsal ribs, that of CM 94 has proportionally a much larger and more extensive area of secondary remodeling (laterally up to three generations of secondary osteons) adjacent to the deep cortex trabecular bone. The periosteal portion of the cortex is comprised of fibrolamellar bone. The vascular canal orientation of the entire section is longitudinal. No radial regions are observed in CM 94. Regions of longitudinal vascular canals are bracketed by LAGs; these canals are found in episodic groupings of either predominantly larger (mean canal diameter 86.50 μm) or smaller (mean canal diameter 47.30 μm) vascular canal diameters. Perhaps these bands are representative of episodic growth. Within the outermost four to five mm of the cortex there is a decrease in vascularity, which is represented by the occurrence and size of vascular canals. The dorsal rib of CM 94 records a minimum of 24 countable LAGs. In addition to the high LAG count, the outermost portion of the cortex records 3-4 closely spaced LAGs in low vascularized tissue. We believe that this region is an external fundamental system (EFS; the histologic indicator of growth cessation). The presence of an EFS in the dorsal ribs indicates that CM 94 was skeletally mature (and potentially the same may hold true for the slightly larger holotype CM 84), and therefore these size ranges do indeed represent the previously assumed skeletally mature sizes of D. carnegii. And in calculating the maximum age estimate, we estimate a maximum age of death of 34 years for CM 94.
The final and largest specimen is represented by the paratype of Diplodocus carnegii (CM 94; Figure 12). As in all of the aforementioned dorsal ribs, that of CM 94 is highly vascularized and possesses a dense network of deep-cortex trabecular bone. However, unlike the previous dorsal ribs, that of CM 94 has proportionally a much larger and more extensive area of secondary remodeling (laterally up to three generations of secondary osteons) adjacent to the deep cortex trabecular bone. The periosteal portion of the cortex is comprised of fibrolamellar bone. The vascular canal orientation of the entire section is longitudinal. No radial regions are observed in CM 94. Regions of longitudinal vascular canals are bracketed by LAGs; these canals are found in episodic groupings of either predominantly larger (mean canal diameter 86.50 μm) or smaller (mean canal diameter 47.30 μm) vascular canal diameters. Perhaps these bands are representative of episodic growth. Within the outermost four to five mm of the cortex there is a decrease in vascularity, which is represented by the occurrence and size of vascular canals. The dorsal rib of CM 94 records a minimum of 24 countable LAGs. In addition to the high LAG count, the outermost portion of the cortex records 3-4 closely spaced LAGs in low vascularized tissue. We believe that this region is an external fundamental system (EFS; the histologic indicator of growth cessation). The presence of an EFS in the dorsal ribs indicates that CM 94 was skeletally mature (and potentially the same may hold true for the slightly larger holotype CM 84), and therefore these size ranges do indeed represent the previously assumed skeletally mature sizes of D. carnegii. And in calculating the maximum age estimate, we estimate a maximum age of death of 34 years for CM 94.
Femoral Histology
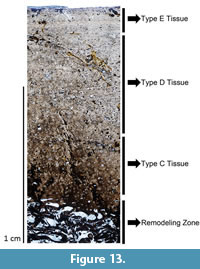 Mother’s Day Quarry femora. The work of Klein and Sander (2008) suggests a linear relationship between femoral length and HOS in sauropods. Thus, this trend indicates that the largest femur from the monospecific Mother’s Day Quarry (Diplodocus sp. MOR 790 7-23-95-122, 120 cm long; Figure 13) represents the oldest animal from the quarry. With a complete cortex 11.29 mm thick, endosteally the medullary region contains large resorption cavities, while periosteally the mineralized tissue grades from Haversian bone into a scattered region of remodeling.
Mother’s Day Quarry femora. The work of Klein and Sander (2008) suggests a linear relationship between femoral length and HOS in sauropods. Thus, this trend indicates that the largest femur from the monospecific Mother’s Day Quarry (Diplodocus sp. MOR 790 7-23-95-122, 120 cm long; Figure 13) represents the oldest animal from the quarry. With a complete cortex 11.29 mm thick, endosteally the medullary region contains large resorption cavities, while periosteally the mineralized tissue grades from Haversian bone into a scattered region of remodeling.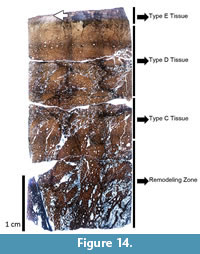 Further periosteally, the remainder of the cortex comprises a very thin unit of Types D and E bone (sensu Klein and Sander 2008). Klein and Sander (2008) report that growth marks may appear in Type E bone (although extremely rare); however, no growth marks are visible in MOR 790 7-23-95-122. The histology indicates that MOR 790 7-23-95-122 presumably the most skeletally mature individual from this locality represents HOS 7 out of 13.
Further periosteally, the remainder of the cortex comprises a very thin unit of Types D and E bone (sensu Klein and Sander 2008). Klein and Sander (2008) report that growth marks may appear in Type E bone (although extremely rare); however, no growth marks are visible in MOR 790 7-23-95-122. The histology indicates that MOR 790 7-23-95-122 presumably the most skeletally mature individual from this locality represents HOS 7 out of 13.
MOR 592 femur. The medullary cavity of the Diplodocus sp. MOR 592 (femur length 124.5 cm) is large with the complete cortex 29.51 mm thick (Figure 14). From the medullary cavity there is a large zone of Haversian bone, while periosteally, remodeling becomes less frequent within the primary tissue. Type E bone predominates for the remainder of the cortex. In the case of the MOR 592 femur, at least one LAG is present along the periosteal margin. MOR 592 representing HOS 9 out of 13 is consistent with femoral length versus HOS correlations found by Klein and Sander (2008). The lack of a cortex consisting entirely of Haversian bone and the lack of an EFS indicates that skeletally, MOR 592 is immature (Figure 14). Klein and Sander (2008) and Gallina (2011, 2012) correlate HOS 9 with sexual maturity in sauropods. While vertebrate reproductive biology favors the onset of sexual maturity prior to skeletal maturity (van Tienhoven, 1983), no histologic indicators of sexual viability was observed (e.g., medullary tissue), therefore, we will not address the reproductive nature of MOR 592. Thus osteohistology supports the conclusions of Woodruff and Fowler (2012) that MOR 592 is skeletally immature.
DISCUSSION
Morphologic Evidence
Pneumatic architecture. Wedel (2005) demonstrated the importance and implications of pneumaticity in sauropod vertebrae (particularly in regards to mass estimations). In regards to quantifying the pneumatic potential, Wedel (2005) proposed the calculation of the air space proportion (ASP) - the volume of air versus the volume of bone. The ASP methodology is simplistic and relies on a two-color image. One color delineates the volume of the bone in cross-section, and the other color represents the area filled by pneumatic structures (see Wedel [2005] for details). A simple calculation comparing the pixel count of bone to air will result in an ASP value. Out of the spectrum of sauropods examined, Wedel (2005) and Wedel and Taylor (2013) had a degree of variability, but noted that most sauropod vertebrae contained approximately 60% empty space by volume, comparable to avian ASP values. Additionally Wedel (2005) noted that the execution of ASP was still in its infancy; the location of the section in a given vertebra and the serial position could greatly affect the calculated ASP. However, Wedel (2003, 2005, 2009) demonstrated that vertebral pneumatic complexity has increased through sauropodomorph phylogeny. Thus ASP increases in tandem with pneumatic complexity.
ASP is an important character, but, in this analysis, we felt cautious about the quantification of ASP. For the CT-scanned vertebrae, we attempted to view transverse slices near the mid-point of the pneumatic foramen and sagittally through the length of the centrum. In viewing the scan data, the anterior and posterior pneumatic foramen slices vary, so in calculating ASP, perhaps an average from multiple fixed locations would give an overall vertebral ASP. Regardless of the quantified ASP, in examining the scan data, one can clearly see (particularly within the dorsal vertebrae) that the pneumatic architecture increases in complexity throughout ontogeny (Wedel, 2003; Schwarz et al., 2007a; Carballido and Sander, 2014). An additional ASP consideration is that patterns in dorsal pneumaticity appear to be associated with avian-style respiration (Wedel, 2003; Schwarz et al., 2007a; Wedel, 2009; Melstrom et al., 2016). Thus differing dorsal ASP between two taxa could be related to variation in the degree of respiratory tissue, rather than to ontogeny or phylogeny. Quantifying the ASP will be important in regards to comparing taxa and calculating evolutionary trends; based on the preliminary data, we see that the complexity follows a developmental trajectory.
Interestingly, while the scans reveal general pneumatic architecture, we would stress they are not a true 1:1 reflection of the actual pneumatic morphology. When the Diplodocus sp. MOR 592 8-22-90-75 was scanned, the results showed that the neural spine had a dense outer cortex with a less dense interior. Within the neural spine there were only a few laterally restricted, small inter-trabecular spaces. Yet thin-sectioning revealed a completely different outcome. The dense periostealmost margin was verified, but the entirety of the internal aspect was instead occupied by cancellous bone with large inter-trabecular spaces. This discrepancy is best explained as an issue with scan resolution (the resolution in this study ranged from 2 mm to 5 mm) ̶ smaller features could become less distinguishable with lower resolution, and density of infilled matrix could likewise cause visual obstruction. Rescanning at finer resolution, intensified contrast, differing can algorithms, or different scan filters could each clarify some of these issues. In addition to these possible adjustments, a comparative study examining the results between scan images and complete histologic sections of each vertebra would be worthwhile.
Cranial foramen. Regarding the significance of the postparietal foramen, Harris (2006a) has suggested that this foramen was used for photoreception (i.e., a pineal eye). While relatively complete diplodocid skulls are rarely preserved, available specimens under ~40 cm in length all possess a postparietal foramen. Larger and presumably more mature diplodocid skulls (such as CM 11161 [Diplodocus longus or diplodocid indeterminate], USNM 2672 [Diplodocus sp. or diplodocid indeterminate], USNM 2673 [Diplodocus sp. or Galeamopus], and CM 11162 [Apatosaurus louisae]; however note that some of these skull are taphonomically altered) all lack this foramen. The presence/absence of this foramen between less and more mature individuals would suggest that, among Diplodocidae, the postparietal foramen is an ontogenetically variable cranial feature (Woodruff and Fowler, 2012; Woodruff et al., 2013; Woodruff and Fowler, 2014; Figure 4).
As the developmental pathway of the postparietal foramen has yet to be examined, it raises the question as to the proper terminology to be used in addressing this feature. Among the immature diplodocid skulls with this feature, it would appear that it initially represents a posterior opening between the sutural margins of the parietals (~1cm), and that through ontogeny this gap is reduced by the fusing parietals. By definition a foramen is a static opening (Grey, 1858), so with regard to the diplodocid condition, foramen is not an appropriate term. Hopson (1979) concluded that this cranial “foramen” was actually a fontanelle. A fontanelle is defined as the temporary membranous gap between developing cranial bones (Grey, 1858). If this feature is indeed ontogenetically variable among diplodocids, then fontanelle would be the more appropriate term. However, the cranial ontogeny of dicraeosaurids is currently unknown. It is possible that this group retains this cranial opening throughout ontogeny (paedomorphic) - thus it is a foramen, while in diplodocids this trajectory could be a peramorphic fontanelle, therefore neither term is suitable for referring to all sauropods. In lieu of a histological examination which could elucidate the proper terminology, we suggest using Balanoff et al.’s (2010) more neutral phrasing of “postparietal aperture”.
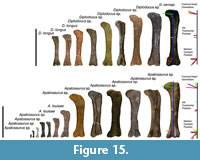 Femoral proportions. The large dataset for femora acquired for diplodocid body-mass estimations has likewise allowed for the examination of femoral ontogeny (Figure 15). Some studies suggest that sauropod limbs may have ontogenetically developed in a more isometric manner (Wilhite and Curtice, 1998; Wilhite, 1999, 2003; Bonnan, 2004; Rogers et al., 2016). Yet examining the dataset, allometric trends are observed. These allometric trends appear to apply to both Diplodocus and Apatosaurus. In many immature individuals the femoral head appears inclined at a greater angle to the long axis of the diaphysis (more pronounced in Diplodocus), and in the larger and more mature specimens the femoral head angle becomes more perpendicularly oriented. Similarly the medial condyle becomes much more pronounced (especially in Apatosaurus) in the larger and more mature individuals. Finally, with regard to femoral proportions, in the immature diplodocids the fourth trochanter is situated proportionally more proximally, while in more mature individuals, this trochanter is generally situated at approximately the mid-point of the diaphysis (Figure 15). A thin-plate spline analysis (such as those used in Wilhite and Curtice, 1998; Wilhite, 1999, 2003; Bonnan, 2004), or a geometric morphometric analysis via a principle component analysis (PCA), would be required to accurately quantify these changes and to possibly detect other proportional changes. Furthermore, since the fourth trochanter is an important attachment site for locomotory muscles (e.g., the m. caudofemoralis longus), the positional change of this anchoring point may affect the moment arm of such muscles, and subsequently, such changes may have gait or other biomechanical implications. While we await such analyses, we hypothesize that these differing femoral changes are indicative of allometric development.
Femoral proportions. The large dataset for femora acquired for diplodocid body-mass estimations has likewise allowed for the examination of femoral ontogeny (Figure 15). Some studies suggest that sauropod limbs may have ontogenetically developed in a more isometric manner (Wilhite and Curtice, 1998; Wilhite, 1999, 2003; Bonnan, 2004; Rogers et al., 2016). Yet examining the dataset, allometric trends are observed. These allometric trends appear to apply to both Diplodocus and Apatosaurus. In many immature individuals the femoral head appears inclined at a greater angle to the long axis of the diaphysis (more pronounced in Diplodocus), and in the larger and more mature specimens the femoral head angle becomes more perpendicularly oriented. Similarly the medial condyle becomes much more pronounced (especially in Apatosaurus) in the larger and more mature individuals. Finally, with regard to femoral proportions, in the immature diplodocids the fourth trochanter is situated proportionally more proximally, while in more mature individuals, this trochanter is generally situated at approximately the mid-point of the diaphysis (Figure 15). A thin-plate spline analysis (such as those used in Wilhite and Curtice, 1998; Wilhite, 1999, 2003; Bonnan, 2004), or a geometric morphometric analysis via a principle component analysis (PCA), would be required to accurately quantify these changes and to possibly detect other proportional changes. Furthermore, since the fourth trochanter is an important attachment site for locomotory muscles (e.g., the m. caudofemoralis longus), the positional change of this anchoring point may affect the moment arm of such muscles, and subsequently, such changes may have gait or other biomechanical implications. While we await such analyses, we hypothesize that these differing femoral changes are indicative of allometric development.
Neural spine bifurcation. As most recently documented by Woodruff (2016) and echoed and noted by others, neural spine bifurcation is a complex vertebral adaptation (Thompson, 1942; Borsuk-Bialynicka, 1977; Bakker, 1986; Salgado and Bonaparte, 1991; Stevens and Parrish, 1999, 2005a; Dodson and Harris, 2001; Christian, 2002; Tsuihiji, 2004; Schwarz et al., 2007a; Senter, 2007; Schwarz-Wings and Frey, 2008; Seymour, 2009; Schwarz-Wings, 2009; Taylor et al., 2009; Wedel and Taylor, 2013). While there is a general consensus for a biomechanical origin, how (and if) it develops is a more contentious point. Amongst the diplodocids of the Morrison Formation that possess bifurcated spines (those represented by several specimens, thus excluding Kaatedocus and Galeamopus), the variabilities and degrees of bifurcation vary considerably - extremely long cervical series with a small degree of bifurcation (Barosaurus) to shorter series with extreme bifurcation (Apatosaurus). The varying degrees of neural spine bifurcation dictate differing vertebral mechanics, and thus the development of this feature should likewise vary. If spine bifurcation is a product of vertebral biomechanics (Woodruff, 2014), then as the vertebral series increases in size (length and mass), then the mechanical stresses enacted upon the series adjust as well. Thus neural spine bifurcation is not considered ontogenetic in our most typical sense - it is not a feature that develops in tandem or coincides with pivotal life history events (i.e., sexual maturity). Instead, neural spine bifurcation can develop as the vertebral series increases in size (i.e., can change as the animal increases in size during growth). While the degrees and specifics pertaining to neural spine development appear to vary amongst these Morrison taxa, the agreeable theme is that the morphology of the bifurcation varies developmentally across ontogeny (i.e., can be absent to weakly expressed in smaller immature animals, while present or enhanced development in larger mature individuals, Figure 3).
Implications Regarding Sauropod Growth Rates
Results of the histologic examination of dorsal ribs indicate that these individuals grew rapidly. An immature diplodocid that is approximately 6 m long is at least six years old (such as Diplodocus sp. MOR 790 7-23-95-122), while a 27 m individual is at least 24 years old (D. carnegii CM 94). These growth rates are not nearly as accelerated as those recorded in some other dinosaurs - such as a 3 m body lengths within the first year in Maiasaura (Woodward et al., 2015) - but they are still consistent with rapid growth rates. In terms of historical perceptions of longevity, sauropods have run the estimation gamut - attaining maximum body size within a decade (Curry, 1999) or taking up to several centuries (and finally sexually mature at 72 years of age; Case, 1978). This variability in longevity estimates has allowed for practically every possible life history strategy to be proposed, and there is little consensus (at least modern studies agree the century estimates to be erroneous; Curry, 1999; Sander, 2000; Erickson et al., 2001; Sander and Tückmantel, 2003; Sander et al., 2004; Rogers and Erickson, 2005; Lehman and Woodward, 2008; Woodward and Lehman, 2009; Griebeler et al., 2013; Waskow and Sander, 2014). Studies that examine multiple aspects of ontogenetic data (e.g., Woodward and Lehman, 2009; Myhrvold, 2013; Hone et al., 2016) represent more comprehensive assessments of growth. For instance, by estimating both limb length and body mass for each LAG interval and applying this data into the von Bertalanffy growth equation, the life history of an individual Apatosaurus could be extrapolated (Woodward and Lehman, 2009).
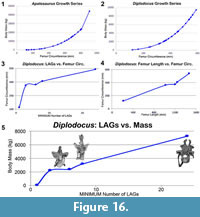 Dorsal rib and femoral data allow for the formulation of hypotheses regarding mass-based ontogenetic trends in diplodocids. The femoral data applied to the Mazzetta et al. (2004) limb-bone allometry-based formula can calculate projected body masses. Morphologic features can be applied to the graph to determine the mass- and age-based timing of ontogenetic events (Figure 16). If the age-determining histology from the Mother’s Day Quarry (Diplodocus sp. MOR 790) specimens is correct, then these animals are collectively under approximately 2,400 kg and around six years of age. The next range approximately represents the change to a taller and incipiently bifurcating spine apex, and this would seem to correlate with animals equal to or greater than 3,500 kg and a minimum of eight years of age. Finally, the mature stage comprises animals with fully bifurcated cervical spine apices and with weights of approximately 7,000 kg and at least 20 years of age. If these categorizations are correct, it would appear that the biomechanical stresses enacted upon the vertebral column change through ontogeny within Diplodocus, thus the degree of neural spine bifurcation was directly correlated with mass.
Dorsal rib and femoral data allow for the formulation of hypotheses regarding mass-based ontogenetic trends in diplodocids. The femoral data applied to the Mazzetta et al. (2004) limb-bone allometry-based formula can calculate projected body masses. Morphologic features can be applied to the graph to determine the mass- and age-based timing of ontogenetic events (Figure 16). If the age-determining histology from the Mother’s Day Quarry (Diplodocus sp. MOR 790) specimens is correct, then these animals are collectively under approximately 2,400 kg and around six years of age. The next range approximately represents the change to a taller and incipiently bifurcating spine apex, and this would seem to correlate with animals equal to or greater than 3,500 kg and a minimum of eight years of age. Finally, the mature stage comprises animals with fully bifurcated cervical spine apices and with weights of approximately 7,000 kg and at least 20 years of age. If these categorizations are correct, it would appear that the biomechanical stresses enacted upon the vertebral column change through ontogeny within Diplodocus, thus the degree of neural spine bifurcation was directly correlated with mass.
The recognition of such has extreme implications towards reconstructing the paleobiology of Diplodocus (and potentially other diplodocids). Immature Diplodocus (collectively under 2,400 kg and six years of age) had short, un-bifurcated neural spines, and quite differing cranial morphologies (such as a rounded “snout” and an elongated tooth row; Whitlock et al., 2010). These cranial changes have been suggested to indicate ontogenetic dietary partitioning (Whitlock et al., 2010), thus the evidence suggests that immature Diplodocus were at least feeding on different vegetation types. The combination of cranial and vertebral changes could be used to suggest that not only were immature Diplodocus feeding on differing plants types, but they might also have employed a differing feeding style (i.e., not lateral sweep feeding). As Woodruff (2016) demonstrated via anatomical comparisons to extant quadrupedal, terrestrial herbivores with bifurcated neural spines, the bifid spines serve as the attachment sites for a split nuchal ligament, with the interspinal ligament occupying the trough of bifurcation. The split nuchal ligament and other soft tissues associated with bifurcated spines, not only provide support against vertebral sagging and torsion, but provide elastic rebound (Woodruff, 2016). The split nuchal ligament provides lateral elastic rebound in the cervical series, which supports the lateral sweep feeding hypothesis of Stevens and Parrish (1999). However, a bifurcating spine affects far more than just the nuchal and interspinal ligament. For instance, increasing the spine height alters the size, area, and attachment of muscles which in turn alter the mass and lever arm of the vertebral series. Therefore all of the anatomical variables of the vertebral column have to change throughout ontogeny. The lack of bifurcated spines in very immature Diplodocus could support the theory that such animals were not laterally sweep feeding, although a mechanical analysis such as performed by Stevens and Parrish (1999) must be conducted to substantiate such.
Regarding growth rate, the morphologic data of this analysis suggests that Apatosaurus achieved a significantly greater body mass throughout ontogeny compared to Diplodocus (Figure 16). An age determining examination of Apatosaurus has yet to be conducted, but we hypothesize that Apatosaurus had a faster growth rate than Diplodocus. If one were to only examine Apatosaurus and Diplodocus, the degree of spine bifurcation would appear to be solely linked to body (or cervical) mass. While based on the relation between spine morphology and body mass, perhaps an immature Apatosaurus possessed greater spine bifurcation earlier in ontogeny than a similarly aged Diplodocus. We hypothesize that body mass is not the only factor influencing spine bifurcation (mobility of the cervical column being perhaps the most significant variable; Stevens and Parrish [1999, 2005a, 2005b]).
Questionable Small-Statured Diplodocid Taxa
Previous studies on diplodocid ontogeny have examined the validity of small-statured taxa such as Suuwassea (ANS 21122; Whitlock and Harris, 2010; Woodruff and Fowler, 2012; Wedel and Taylor, 2013; Hedrick et al., 2014; Tschopp et al., 2015). In the case of Suuwassea, the combination of basal and derived traits has explanatorily ranged from environmental adaptations (Harris, 2006b) to indications of immaturity (Woodruff and Fowler, 2012). Taxonomic placement of Suuwassea has ranged from a basal apatosaurine (Harris and Dodson, 2004), a Laurasian dicraeosaurid (Whitlock and Harris, 2010; Wedel and Taylor, 2013; Tschopp et al., 2015), to an immature diplodocid (Woodruff and Fowler, 2012). Some studies recognize Suuwassea as a valid genus and interpret the holotype as non-juvenile (Wedel and Taylor, 2013; Hedrick et al., 2014).
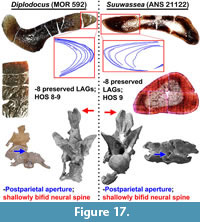 Woodruff and Fowler (2012) interpreted the following morphologic traits as being ontogenetically influenced: narrow spine bifurcation, anterior prominence at the dentary symphysis (a ‘‘chin’’), curvature of the tooth row, decreased vertebral pneumaticity, postparietal “foramen”, unfused scapulocoracoid (yet note the issues regarding fusion), and elongate pedal phalanges. Woodruff and Fowler (2012) stated that until a histologic analysis is performed, the ontogenetic status Suuwassea would not be definitive. Hedrick et al. (2014) examined the histology of Suuwassea, and from analysis of the holotype tibia, they conclude that Suuwassea is HOS 8-9 out of 13 (however it is important to note that the HOS was constructed primarily using femora, thus tibiae may not correspond 1:1). However from this HOS designation, Hedrick et al. (2014) inferred that Suuwassea (ANS 21122) was nearly skeletally mature, had reached sexual maturity, was a valid genus, and that the plesiomorphic characters were not related to immaturity. While we applaud Hedrick et al. (2014) in conducting a histologic analysis to examine life history information, comparison with a specimen exhibiting similar morphologies (MOR 592) suggests an alternative taxonomic interpretation (Figure 17).
Woodruff and Fowler (2012) interpreted the following morphologic traits as being ontogenetically influenced: narrow spine bifurcation, anterior prominence at the dentary symphysis (a ‘‘chin’’), curvature of the tooth row, decreased vertebral pneumaticity, postparietal “foramen”, unfused scapulocoracoid (yet note the issues regarding fusion), and elongate pedal phalanges. Woodruff and Fowler (2012) stated that until a histologic analysis is performed, the ontogenetic status Suuwassea would not be definitive. Hedrick et al. (2014) examined the histology of Suuwassea, and from analysis of the holotype tibia, they conclude that Suuwassea is HOS 8-9 out of 13 (however it is important to note that the HOS was constructed primarily using femora, thus tibiae may not correspond 1:1). However from this HOS designation, Hedrick et al. (2014) inferred that Suuwassea (ANS 21122) was nearly skeletally mature, had reached sexual maturity, was a valid genus, and that the plesiomorphic characters were not related to immaturity. While we applaud Hedrick et al. (2014) in conducting a histologic analysis to examine life history information, comparison with a specimen exhibiting similar morphologies (MOR 592) suggests an alternative taxonomic interpretation (Figure 17).
The Diplodocus sp. MOR 592 displays the same plesiomorphic characters observed in Suuwassea (see above). Originally referred to as Amphicoelias on the basis of a “stovepipe” femur (Wilson and Smith, 1996), and subsequently considered a possible new genus, this specimen has since been alternatively recognized as an immature Diplodocus sp. (Woodruff and Fowler, 2012; Woodruff and Foster, 2014; however note that Whitlock and Harris, 2010 consider it a dicraeosaurid). Aside from the plesiomorphic characters, MOR 592 also has a similar femur length (124.5 cm versus the calculated 135 cm of Suuwassea [Hedrick et al., 2014]). Likewise thin-sectioning of the MOR 592 femur indicates that it is HOS 9 out of 13. Furthermore, thin-sectioning of a dorsal rib of MOR 592 and ANS 21122 records a minimum of eight preserved LAGs in both specimens; and both lack an EFS in any elements histologically examined (Figure 17). Both specimens exhibit similar morphology, and both exhibit similar histology, supporting the hypothesis that they represent similar ontogenetic stages.
We would suggest that an alternative explanation for the plesiomorphic characters of MOR 592 is that it is an immature Diplodocus sp. (in agreement with Woodruff and Fowler, 2012) rather than a Laurasian dicraeosaurid (Whitlock and Harris, 2010). Likewise the abundance of shared features between MOR 592 and ANS 21122 support Suuwassea is an immature individual (Figure 17). While the similarities between MOR 592 and ANS 21122 may be attributable to their ontogenetic stage, the specific vertebral morphologies of Suuwassea resemble those of other immature apatosaurine individuals (such as CM 555 [A. excelsus or Brontosaurus excelsus] or CM 3390 [Apatosaurus sp.]), thus these apparent distinctions can be conversely interpreted as the variation observed in an immature animal of a known taxon. In consideration of the fact that morphological and histologic analyses by separate investigating parties (Woodruff and Fowler, 2012; Hedrick et al., 2014; this current analysis) have reached comparable ontogenetic results, we advocate that the taxonomic status of Suuwassea remains unresolved. We consider the morphology and histology to indicate that, rather than representing a distinct taxon, Suuwassea emilieae (ANS 21122) is more likely an immature Apatosaurus.
The issues discussed above regarding taxonomy should not be trivialized. While MOR 592 could certainly be an immature Diplodocus sp., Suuwassea at this time could equally represent an immature Apatosaurus sp., a distinct taxon, a more or less mature individual or taxon with pedomorphic attributes, and possibly a combination of these conditions. Currently the holotype material does not unanimously support one distinct interpretation. The greater importance in this ongoing discussion is that a specimen exists where alternative taxonomy is derived from differing morphologic interpretations. While the characters used in a phylogenetic analysis are derived from morphologic attributes, this analysis would suggest that many of the characters are ontogenetically dependent. As demonstrated with tyrannosaurs (Carr and Williamson, 2004; Fowler et al., 2011), ceratopsians (Scannella and Horner, 2010; Scannella et al., 2014; Frederickson and Tumarkin-Deratzian, 2014), and hadrosaurs (Campione et al., 2013; Fowler and Horner, 2015), ontogeny (and stratigraphy) does affect taxonomy (likewise echoed in Hone et al., 2016). In animals that undergo an order of magnitude in size change, one could predict that many phylogenetic characters are simultaneously size determinant (ontogenetic in the sense they change as an animal gets larger with age) characters. Such has previously been demonstrated where immature individuals of a derived taxon appear to occupy a more basal position (Campione et al., 2013; Carballido and Sander, 2014). While phylogeny will not be made or broken by a single character, some characters (such neural spine bifurcation or the postparietal aperture) have historically been important for taxonomic distinction (laudably Tschopp et al., 2015 noted the significance of such). The ontogenetic development of the postparietal aperture may appear minor, but the ontogenetic development of another cranial opening was one feature used to re-examine derived Triceratopsini taxonomy (Scannella and Horner, 2010; which is, however, still under debate).
As this analysis calls for the combined efforts of morphologic and histologic observations to better understand ontogeny, perhaps future studies should look to stratigraphy, morphology, and histology as supportive information to enhance phylogenetic studies (such is already being done and is referred to as an ontogram; Frederickson and Tumarkin-Deratzian, 2014). Perhaps the true phylogeny of such conflicting specimens will be resolved from an ontogram-based inclusive approach.
Ontogenetic Compilation
In a like manner to the Nash Equilibrium Theory (Nash, 1950), the individual characters used in this H-MOS may seem insignificant or minor; but, by not forcefully competing (i.e., histologic characters trump morphological characters, or vice versa), each is an important contribution by itself while simultaneously supporting other characters. The design and simplicity of the H-MOS system is that it is not restricted to sauropods; as presented herein, this system has been tailored to a diplodocid series. But the importance and implication of histology and morphology are not restricted to sauropodomorphs. Therefore an H-MOS style system should be adopted and incorporated into all dinosaurian ontogeny studies. In this analysis, the outcome of such a methodology is greater ontogenetic resolution and the predictive capabilities to infer diplodocid maturational stages and conditions.
As an example, let us examine the immature Diplodocus sp. MOR 592. A histologic section of the 124.5 cm long femur identifies it as HOS 9. Morphologic examination shows that the posterior cervical vertebrae are at the initial stage of spine bifurcation, while the dorsal vertebrae exhibit spine bifurcation that is more similar to the adult morphology. CT scans of the vertebrae illustrate a thinning of the median septum and interspersed camellae and camerae within the condyle and cotyle. The calculated body mass is 3,205 kg. Histology of the dorsal ribs record a minimum of eight preserved LAGs with no evidence of an EFS. The combination of these characters helps to define our diplodocid H-MOS Stage 3. Instead of solely using HOS or degree of spine bifurcation to determine a relative maturational state, or dorsal rib data to determine a minimum age, the H-MOS method creates a larger depiction of the histological and morphological attributes of each size range, creating a better representation of the animal as a whole.
Potentially our outlined H-MOS method even has the capacity to be applied to fragmentary and isolated diplodocid elements. While the predictive capabilities based on a single element are less absolute than multi-element analysis, it nonetheless reinforces the notion that each piece of data is significant (i.e., even isolated elements are vital data points). Every specimen helps create a more accurate index. An isolated femur that can be sectioned will reveal HOS. HOS coupled with femur dimensions could contribute to the range (such as HOS range and calculated body mass) of the particular growth stage. Similarly, on an isolated cervical vertebra, the degree of spine bifurcation and pneumaticity can be noted, and the neural spine could be sampled, while the entire vertebra could be CT scanned. While more complete specimens contribute far more data, the H-MOS methodology illustrates that isolated elements should not be overlooked because these seemingly “unimportant” specimens still represent useful data.
As presented, the H-MOS method is in its infancy, and undoubtedly more specimens, characters, ontogenetic divisions, and finer stage resolution will be added to increase its accuracy. Most recently the Remodeling Stages of Mitchell et al. (2017) represent a new histologic consideration, and for certain this new histologic character will be incorporated into future H-MOS works. While this H-MOS seeks to illuminate the life histories of sauropods, some variables such as sexual maturity, ASP, and degrees of intraspecific variation as yet remain unresolved. Documentation in vertebrates shows that sexual maturity typically precedes skeletally maturity (van Tienhoven, 1983). If this holds true for the examined sauropods, perhaps some of the H-MOS Stage 3 specimens were already sexually mature (Griebeler et al., 2013; Waskow and Sander, 2014). Therefore determination of sexual maturity would be a valuable future H-MOS character.
A further factor to consider in any H-MOS style analysis - even the one described herein - are the effects of intraspecific and interspecific variation. Intraspecific variation exists in all species, and only large sample sets (like those demonstrated for Triceratops [100+; Scannella et al., 2014] and Maiasaura [50; Woodward et al., 2015]) will allow for interspecific variation to be recognized and accounted for. Even within the relatively small sample size of this analysis, the degree of intraspecific variation has likely gone unchecked, and could even be affecting our demarcations and maturational inferences. Furthermore, in this analysis we make a collective ontogenetic trajectory for Diplodocus, yet our dataset is comprised of known and unknown species. In all likelihood, interspecific variation did exist amongst species of the same genus, and as addressed before, not accounting for these factors could weaken said system. The reason for such inclusion is twofold: 1) we unfortunately do not have enough specimens of precisely known taxonomy at this time to make a Diplodocus species-level H-MOS, and 2) the goal of initial, preliminary analyses such as this are to first recognize patterns. Initial ontogenetic studies of Triceratops cranial ornamentation noted general patterns such as orbital horn curvature and morphology of epoccipitals (Horner and Goodwin, 2006). And building upon this, subsequent analyses noted that some specimens or features that did not universally conform could be explained as ontogimorphs via anagenesis from T. horridus to T. prorsus (Scannella et al., 2014). In like manner, aspects of the presented H-MOS could later be determined to be affected by variation, or they could represent signals worthy of future exanimation. Undoubtedly future analyses will find flaws in this initial system, but the initial patterns we present serve as the platform for subsequent works.
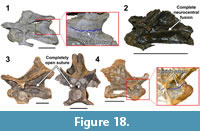 Additionally, further examinations may reveal the validity/invalidity of previously used maturational features. Throughout this current analysis, one may have noted the absence of two popular sauropod maturational characters: neurocentral fusion and Elongation Index. Brochu (1996, 1999), Irmis (2007) and Ikejiri (2012) have demonstrated that crocodilians can have drastically delayed or even a complete lack of vertebral fusion within presacral vertebrae, while Bailleul et al. (2016) demonstrated similar findings in cranial sutures. In all the diplodocids we examined, there was no consistency or pattern to vertebral fusion (Figure 18). In a cervical approximately 10 cm long there can be complete neurocentral fusion, whereas in larger cervicals (~ 25 to 30 cm) fusion can range from completely open to fusing only in the anterior portion or only in the posterior region of the neural arch (Figure 18). As Brochu (1996, 1999), Irmis (2007), Ikejiri (2012), and Bailleul et al. (2016) demonstrated, while sutural patterns may be evident in some crocodilians, (neurocentral) fusion in dinosaurs appears to be sporadic and should only be used with caution when inferring maturity. Conversely, Melstrom et al. (2016) note a pattern in sacral fusion and open dorsal sutures in an immature Barosaurus, and therefore conclude that neurocentral fusion could be a reliable indicator of maturity. Such complete vertebral series will be the way to document such, and perhaps a general fusion pattern exists, but that deviation from said pattern has high plasticity (this could explain the degrees of fusion observed in this study).
Additionally, further examinations may reveal the validity/invalidity of previously used maturational features. Throughout this current analysis, one may have noted the absence of two popular sauropod maturational characters: neurocentral fusion and Elongation Index. Brochu (1996, 1999), Irmis (2007) and Ikejiri (2012) have demonstrated that crocodilians can have drastically delayed or even a complete lack of vertebral fusion within presacral vertebrae, while Bailleul et al. (2016) demonstrated similar findings in cranial sutures. In all the diplodocids we examined, there was no consistency or pattern to vertebral fusion (Figure 18). In a cervical approximately 10 cm long there can be complete neurocentral fusion, whereas in larger cervicals (~ 25 to 30 cm) fusion can range from completely open to fusing only in the anterior portion or only in the posterior region of the neural arch (Figure 18). As Brochu (1996, 1999), Irmis (2007), Ikejiri (2012), and Bailleul et al. (2016) demonstrated, while sutural patterns may be evident in some crocodilians, (neurocentral) fusion in dinosaurs appears to be sporadic and should only be used with caution when inferring maturity. Conversely, Melstrom et al. (2016) note a pattern in sacral fusion and open dorsal sutures in an immature Barosaurus, and therefore conclude that neurocentral fusion could be a reliable indicator of maturity. Such complete vertebral series will be the way to document such, and perhaps a general fusion pattern exists, but that deviation from said pattern has high plasticity (this could explain the degrees of fusion observed in this study).
 The other excluded assessment is that of Elongation Index (EI). Defined as the ratio between the length of the centrum (condyle to cotyle length) divided by the cotyle diameter (cotyle width; Wedel et al., 2000), it has been inferred that EI values might be taxonomically or maturationally specific (Wedel and Taylor, 2013). A simple calculation of EI for 12 diplodocid cervical series indicates that taxonomically, serially, ontogenetically, and individually, EI is extremely variable (see Appendix 5, Appendix 6, Appendix 7, Appendix 8). However, if the EI trends among these diplodocids are legitimate, this suggests that higher EI values collectively could be more indicative of an immature condition (note that this would support the findings of Woodruff and Fowler [2012] that juvenile diplodocid vertebrae primarily increase vertebral length earlier in ontogeny, and later growth is principally directed at width ‒ resulting in a high EI during early ontogeny, and a lower EI in later life). Yet since the nature of EI is so variable, and our understanding of the ontogenetic role of EI is still in its infancy, we would temporarily avoid individual vertebral EI as a means to infer maturity. While variation in neurocentral fusion and EI could easily be explainable and intraspecific variation, and such patterns or trends may be legitimate, at this time we do not possess the supportive data to suggest that these are reliable maturational indicators across ontogeny. It is important to note that Melstrom et al. (2016) note the distribution of EI within the partial Barosaurus vertebral series, and their findings support that it should be a character examined in future analyses.
The other excluded assessment is that of Elongation Index (EI). Defined as the ratio between the length of the centrum (condyle to cotyle length) divided by the cotyle diameter (cotyle width; Wedel et al., 2000), it has been inferred that EI values might be taxonomically or maturationally specific (Wedel and Taylor, 2013). A simple calculation of EI for 12 diplodocid cervical series indicates that taxonomically, serially, ontogenetically, and individually, EI is extremely variable (see Appendix 5, Appendix 6, Appendix 7, Appendix 8). However, if the EI trends among these diplodocids are legitimate, this suggests that higher EI values collectively could be more indicative of an immature condition (note that this would support the findings of Woodruff and Fowler [2012] that juvenile diplodocid vertebrae primarily increase vertebral length earlier in ontogeny, and later growth is principally directed at width ‒ resulting in a high EI during early ontogeny, and a lower EI in later life). Yet since the nature of EI is so variable, and our understanding of the ontogenetic role of EI is still in its infancy, we would temporarily avoid individual vertebral EI as a means to infer maturity. While variation in neurocentral fusion and EI could easily be explainable and intraspecific variation, and such patterns or trends may be legitimate, at this time we do not possess the supportive data to suggest that these are reliable maturational indicators across ontogeny. It is important to note that Melstrom et al. (2016) note the distribution of EI within the partial Barosaurus vertebral series, and their findings support that it should be a character examined in future analyses.
 Likewise, further analyses may indicate that some characters are more maturationally delineated than others (such as sexually maturity versus mass). As Hone et al. (2016) demonstrated, varied life history information only enhances ontogenetic resolution and maturational distinctions. Yet we must remember, that as stated most recently by Goodwin and Evans (2016), ontogeny is developmental progression, and not strictly developmental demarcations; and even though this analysis attempts to categorize sauropod ontogeny, in reality we should expect many of these developmental lines to be gradational.
Likewise, further analyses may indicate that some characters are more maturationally delineated than others (such as sexually maturity versus mass). As Hone et al. (2016) demonstrated, varied life history information only enhances ontogenetic resolution and maturational distinctions. Yet we must remember, that as stated most recently by Goodwin and Evans (2016), ontogeny is developmental progression, and not strictly developmental demarcations; and even though this analysis attempts to categorize sauropod ontogeny, in reality we should expect many of these developmental lines to be gradational.
An additional caution to the H-MOS system would be the recognition of skeletally mature, “small” animals. While it is outside of this study’s scope, the Camarasaurus SMA 0002 provides such a cautionary example. For an “average” body size, SMA 0002 is rather small (calculated mass of 10,634 kg), yet histologic examination by Waskow and Sander (2014) revealed that this specimen was approximately 40 years old at the time of death making it the oldest (and it is also the stratigraphically lowest) Camarasaurus sampled to date (SMA 0002 has an EFS, so theoretically H-MOS 4). Therefore, this specimen (or similar small-statured morphs) could give conflicting morphologic and histologic data. Many of the H-MOS characters appear to be correlated with some aspect of body size, so a smaller, mature animal could have some morphologies lesser expressed (and vice versa). While this current analysis has not identified such a specimen, we propose in the case where a specimen expressed such conflicting morphologies, this would serve as an “alert” to indicate something special about the animal’s life history. The degrees of intraspecific variation will likely affect interpretations but the recognition of variability will only strengthen our understanding of development.
Likewise sample size must be recognized as a factor towards predictive capabilities. The sample size of the Horner et al. (2000) analysis of Maiasaura growth dynamics was not statistically significant (n=six), yet the observed trends and hypotheses of that study served as a starting point and a “stepping stone” for the follow-up analysis of Woodward et al. (2015), which represents the largest single population dinosaur growth study (n=50); and this study confirmed the preliminary data of Horner et al. (2000).
Finally, it may certainly become necessary to have family, genera, or even species specific H-MOS tables (e.g., the body mass estimations of Apatosaurus do not ontogenetically correspond with Diplodocus, so an Apatosaurus mass range will need to be calculated). The argument could be presented that such a system (even this initial H-MOS) or further refined systems are not warranted because such do not uniformly apply, or would inherently be so complex. Single aspects of life history can be simply explained (e.g., HOS or MOS), yet the ultimate goal of the H-MOS is an attempt to better understand the life history of the dinosaur in question (as opposed to only diplodocids; i.e., “DOS”; for a good review of this topic, see Hone et al. [2016]). While the presented H-MOS demarcations do not apply ubiquitously to other sauropod clades (i.e., Macronaria), the methodology of this system can be incorporated throughout all of Sauropodomorpha, and in fact all of Dinosauria. By continually supplementing, adapting, modifying, and applying an H-MOS style system‒and any future subsidiary systems‒our resolution of life history only intensifies.
CONCLUSION
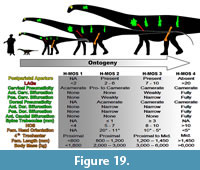 Whereas the recognition of a single feature’s developmental trajectory is important, this analysis indicates that groups of changes are observed during sauropod ontogeny - i.e., individual features appear to substantiate one another in terms of inferred maturational states. The analysis of both morphologic and histologic attributes of multiple cranial and postcranial elements supports a correlation of maturational states through ontogeny. While the understanding of a single character’s development is important, the recognition of a suite of defining histologic and morphologic characters allows for a better recognition of each maturational range. The proposed Histo-Morph Ontogeny Scale incorporates multiple variables and allows for a more complete picture of growth changes in these animals (Figure 19).
Whereas the recognition of a single feature’s developmental trajectory is important, this analysis indicates that groups of changes are observed during sauropod ontogeny - i.e., individual features appear to substantiate one another in terms of inferred maturational states. The analysis of both morphologic and histologic attributes of multiple cranial and postcranial elements supports a correlation of maturational states through ontogeny. While the understanding of a single character’s development is important, the recognition of a suite of defining histologic and morphologic characters allows for a better recognition of each maturational range. The proposed Histo-Morph Ontogeny Scale incorporates multiple variables and allows for a more complete picture of growth changes in these animals (Figure 19).
ACKNOWLEDGEMENTS
We would like to thank G. Ohrstrom and E. and J. Sands for their gracious and continuing support to the MOR Paleontology Department. Without the time and consideration of all the museum staff and collection managers, none of this work would have been possible. We humbly thank K. Siber and the entire staff and family of the Sauriermuseum Aathal; O. Wings and H. Mallison at the Museum für Naturkunde - Leibniz Research Institute for Evolution and Biodiversity; C. Mehling and M. Norell at the American Museum of Natural History; A. Henrici, M. Lamanna, C. Beard, D. Berman, and D. Pickering of the Carnegie Museum of Natural History; T. Daeschler and the Academy of Natural Sciences at Drexel University; S. Lucas and the New Mexico Museum of Natural History and Science; and M. Sander, K. Waskow, N. Klein, and DFG Research Unit 533 from the Steinmann Institut für Geologie, Paläontologie und Mineralogie. K. Ugrin and the staff of Advanced Medical Imaging at Bozeman Deaconess Hospital generously donated their time and use of their CT scanner. H. Woodward and D. Varricchio provided invaluable insight and assistance through manuscript revision and preparation. Further we would like to thank H.D. Sues, E. Tschopp, K. Waskow, R. Wilhite, J. Foster, M. Wedel, M. Taylor, B. Hedrick, J. Harris, E. Prondvai, K. Stein, K. Stevens, J. McIntosh, G. Storrs, J. Scannella, E. Freedman-Fowler, J. Wilson, K. Nordén, B. Baziak, S. Hartman, P. Raia, and two anonymous reviewers for expert advice and assistance, review, and continuing support throughout this project.
REFERENCES
Abramoff, M.D., Magalhaes, P.J., and Ram, S.J. 2004. Image Processing with ImageJ. Biophotonics International, 11(7):36-42.
Bailleul, A.M., Scannella, J.B., Horner, J.R., and Evans, D.C. 2016. Fusion patterns in the skulls of modern archosaurs reveal that sutures are ambiguous maturity indicators for the Dinosauria. PLoS ONE, 11(2):e0147687. doi:10.1371/journal.pone.0147687
Bakker, R.T. 1986. The Dinosaur Heresies. William Morrow and Company, New York.
Balanoff, A.M., Bever, G.S., and Ikejiri, T. 2010. The braincase of Apatosaurus (Dinosauria: Sauropoda) based on computed tomography of a new specimen with comments on variation and evolution in sauropod neuroanatomy. American Museum Novitates, 3677:1-32.
Berman, D.S. and McIntosh, J.S. 1978. Skull and relationships of the Upper Jurassic sauropod Apatosaurus (Reptilia, Saurischia). Bulletin of the Carnegie Museum of Natural History, 8:1-35.
Bonnan, M.F. 2004. Morphometric analysis of humerus and femur shape in Morrison sauropods: implications for functional morphology and paleobiology. Paleobiology, 30:444-470.
Borsuk-Bialynicka, M. 1977. A new camarasaurid sauropod Opisthocoelicaudia skarzynskii, gen. n., sp. n. from the Upper Cretaceous of Mongolia. Acta Palaeontologia Polonica, 37:1-64.
Brochu, C.A. 1996. Closure of neurocentral sutures during crocodilian ontogeny: implications for maturity assessment in fossil archosaurs. Journal of Vertebrate Paleontology, 16:49-62.
Brochu, C.A. 1999. Phylogeny, systematics, and historical biogeography of Alligatoroidea. Society of Vertebrate Paleontology Memoir, 6:9-100.
Campione, N.E., Brink, K.S., Freedman, E.A., McGarrity, C.T., and Evans, D.C. 2013. ‘ Glishadesericksoni ’, an indeterminate juvenile hadrosaurid from the Two Medicine Formation of Montana: implications for hadrosauroid diversity in the latest Cretaceous (Campanian-Maastrichtian) of western North America. Palaeobiodiversity and Palaeoenvironments, 93:65-75.
Campione, N.E. and Evans, D.C. 2011. Cranial growth and variation in edmontosaurs (Dinosauria: Hadrosauridae): implications for latest Cretaceous megaherbivore diversity in North America. PLoS One, 6(9):e25186.
Campione, N.E. and Evans, D.C. 2012. A universal scaling relationship between body mass and proximal limb bone dimensions in quadrupedal terrestrial tetrapods. BMC Biology, 10:60.
Carballido, J.L., Marpmann, J.S., Schwarz‐Wings, D., and Pabst, B. 2012. New information on a juvenile sauropod specimen from the Morrison Formation and the reassessment of its systematic position. Palaeontology, 55:567-582.
Carballido, J.L. and Sander, P.M. 2014. Postcranial axial skeleton of Europasaurusholgeri (Dinosauria, Sauropoda) from the Upper Jurassic of Germany: implications for sauropod ontogeny and phylogenetic relationships of basal Macronaria. Journal of Systematic Palaeontology, 12:335-387.
Carpenter, K. 2013. History, sedimentology, and taphonomy of the Carnegie Quarry, Dinosaur National Monument, Utah. Annals of Carnegie Museum, 81(3):153-232.
Carr, T.D. 1999. Craniofacial ontogeny in tyrannosauridae (Dinosauria, Coelurosauria). Journal of Vertebrate Paleontology, 19:497-520.
Carr, T.D. and Williamson, T.E. 2004. Diversity of late Maastrichtian Tyrannosauridae (Dinosauria: Theropoda) from western North America. Zoological Journal of the Linnean Society, 142:479-523.
Case, T.J. 1978. Speculations on the growth rate and reproduction of some dinosaurs. Paleobiology, 4:320-328.
Castanet, J., Francillon-Vieillot, H., Meunier, F.J., and de Ricqlès, A. 1992. Bone and individual aging, p. 245-283. In Hall, B.K. (ed.), Bone: A Treatise Volume. CRC Press, London.
Christian, A. 2002. Neck posture and overall body design in sauropods. Mitt Museum Naturkunde Berlin Geowissenschaftl Reihe, 5:271-281.
Curry, K.A. 1999. Ontogenetic histology of Apatosaurus (Dinosauria: Sauropoda): new insights on growth rates and longevity. Journal of Vertebrate Paleontology, 19:654-665.
de Ricqlès, A., Meunier, F.J., Castanet, J., and Francillon-Vieillot, H. 1991. Comparative microstructure of bone. Bone, 3:1-78.
Dodson, P. 1975. Taxonomic implications of relative growth in lambeosaurine hadrosaurs. Systematic Biology, 24:37-54.
Dodson, P. and Harris, J.D. 2001. Necks of sauropod dinosaurs: support of a nuchal ligament? Journal of Morphology, 248(3):224.
Erickson, G.M. 2005. Assessing dinosaur growth patterns: a microscopic revolution. Trends in Ecology and Evolution, 20: 677-684.
Erickson, G.M., Rogers, K.C., and Yerby, S.A. 2001. Dinosaurian growth patterns and rapid avian growth rates. Nature, 412:429-433.
Evans, D.C., Forster, C.A., and Reisz, R.R. 2005. The type specimen of Tetragonosaurus erectofrons (Ornithischia: Hadrosauridae) and the identification of juvenile Lambeosaurines, p. 349-366. In Currie, P.J. and Koppelhaus, E.B. (eds.), Dinosaur Provincial Park. Indiana University Press, Bloomington.
Fowler, E.A.F. and Horner, J.R. 2015. A new brachylophosaurin hadrosaur (Dinosauria: Ornithischia) with an intermediate nasal crest from the Campanian Judith River Formation of northcentral Montana. PloS ONE, 10(11):e0141304.
Fowler, D.W., Woodward, H.N., Freedman, E.A., Larson, P.L., and Horner, J.R. 2011. Reanalysis of “Raptorexkriegsteini ”: a juvenile tyrannosaurid dinosaur from Mongolia. PLoS One, 6(6):e21376.
Francillon‐Vieillot, H., de Buffrénil, V., Castanet, J., Géraudie, J., Meunier, F.J., Sire, J.Y., Zylberberg, L., and de Ricqlès, A. 1990. Microstructure and mineralization of vertebrate skeletal tissues, p. 175-234. In Carter, J.G. (ed.), Skeletal Biomineralization: Patterns, Processes and Evolutionary Trends. Van Nostrand Reinhold, New York.
Frederickson, J.A. and Tumarkin-Deratzian, A.R. 2014. Craniofacial ontogeny in Centrosaurusapertus. PeerJ, 2:e252.
Gallina, P.A. 2011. Notes on the axial skeleton of the titanosaur Bonitasaurasalgadoi (Dinosauria-Sauropoda). Anais Da Academia Brasileira de Ciências, 83:235-246.
Gallina, P.A. 2012. Histología ósea del titanosaurio Bonitasaurasalgadoi (Dinosauria: Sauropoda) del Cretácico Superior de Patagonia. Ameghiniana, 49:289302.
Gilmore, C.W. 1925. A nearly complete articulated skeleton of Camarasaurus, a saurischian dinosaur from the Dinosaur National Monument, Utah. Memoirs of the Carnegie Museum, 10:347-384.
Gilmore, C.W. 1936. Osteology of Apatosaurus with special reference to specimens in the Carnegie Museum. Memoirs of the Carnegie Museum, 11:175-300.
Goodwin, M.B. and Evans, D.C. 2016. The early expression of squamosal horns and parietal ornamentation confirmed by new end-stage juvenile Pachycephalosaurus fossils from the Upper Cretaceous Hell Creek Formation, Montana. Journal of Vertebrate Paleontology, e1078343.
Grey, H. 1858. Anatomy: Descriptive and Surgical. Parker and Son, London.
Griebeler, E.M., Klein, N., and Sander, P.M. 2013. Aging, maturation and growth of sauropodomorph dinosaurs as deduced from growth curves using long bone histological data: An assessment of methodological constraints and solutions. PLoS ONE, 8(6):e67012.
Harris, J.D. 2006a. Cranial osteology of Suuwasseaemilieae (Sauropoda: Diplodocoidea: Flagellicaudata) from the Upper Jurassic Morrison Formation of Montana, USA. Journal of Vertebrate Paleontology, 26:88-102.
Harris, J.D. 2006b. The significance of Suuwasseaemilieae (Dinosauria: Sauropoda) for flagellicaudatan intrarelationships and evolution. Journal of Systematic Palaeontology, 4:185-198.
Harris, J.D. and Dodson, P. 2004. A new diplodocoid sauropod dinosaur from the Upper Jurassic Morrison Formation of Montana, USA. Acta Palaeontologica Polonica, 49:197-210.
Hatcher, J.B. 1901. Diplodocus Marsh: its osteology, taxonomy, and probable habits, with a restoration of the skeleton. Memoirs of the Carnegie Museum, 1:1-63.
Hatcher, J.B. 1903. Osteology of Haplocanthosaurus with additional remarks on Diplodocus. Memoirs of the Carnegie Museum, 1:1-75.
Hedrick, B.P., Tumarkin-Deratzian, A.R., and Dodson, P. 2014. Bone microstructure and relative age of the holotype specimen of the diplodocoid sauropod dinosaur Suuwassea emilieae. Acta Palaeontologica Polonica, 59:295-304.
Holland, W.J. 1924. The skull of Diplodocus. Memoirs of the Carnegie Museum, 9:379-403.
Hone, D.W.E., Farke, A.A., and Wedel, M.J. 2016. Ontogeny and the fossil record: What, if anything, is an adult dinosaur? Biology Letters, 12:20150947.
Hopson, J.A. 1975. The evolution of cranial display structures in hadrosaurian dinosaurs. Paleobiology, 1:21-43.
Hopson, J.A. 1979. Paleoneurology, p. 39-146. In Gans, C., Northcutt, R.C., and Ulinski, P. (eds.), Biology of the Reptilia Volume 9 Neurology A. Academic Press, London.
Horner, J.R., de Ricqlès, A., and Padian, K. 1999. Variation in dinosaur skeletochronology indicators: implications for age assessment and physiology. Paleobiology, 25(3):295-304.
Horner, J.R., de Ricqlès, A., and Padian, K. 2000. Long bone histology of the hadrosaurid dinosaur Maiasaurapeeblesorum: growth dynamics and physiology based on an ontogenetic series of skeletal elements. Journal of Vertebrate Paleontology, 20:115-129.
Horner, J.R. and Goodwin, M.B. 2006. Major cranial changes during Triceratops ontogeny. Proceedings of the Royal Society B: Biological Sciences, 273:2757-2761.
Horner, J.R. and Goodwin, M.B. 2009. Extreme cranial ontogeny in the Upper Cretaceous dinosaur Pachycephalosaurus. PLoS One, 4:e7626.
Horner, J.R., Goodwin, M.B., and Myhrvold, N. 2011. Dinosaur census reveals abundant Tyrannosaurus and rare ontogenetic stages in the Upper Cretaceous Hell Creek Formation (Maastrichtian), Montana, USA. PloS One, 6(2):e16574.
Horner, J.R. and Padian, K. 2004. Age and growth dynamics of Tyrannosaurus rex. Proceedings of the Royal Society of London. Series B: Biological Sciences, 271:1875-1880.
Huttenlocker, A.K., Woodward, H.N., and Hall, B.K. 2013. The biology of bone, p. 13-34. In Padian, K. and Lamm, E.T. (eds.), Bone Histology of Fossil Tetrapods: Advancing Methods, Analysis, and Interpretation. University of California Press, Berkeley.
Ikejiri, T. 2012. Histology‐based morphology of the neurocentral synchondrosis in Alligator mississippiensis (Archosauria, Crocodylia). The Anatomical Record, 295:18-31.
Ikejiri, T., Tidwell, V., and Trexler, D.L. 2005. New adult specimens of Camarasaurus lentus highlight ontogenetic variation within the species, p. 154-179. In Tidwell, V. and Carpenter, K. (eds.), Thunder-lizards: The Sauropodomorph Dinosaurs. Indiana University Press, Bloomington.
Irmis, R.B. 2007. Axial skeleton ontogeny in the Parasuchia (Archosauria: Pseudosuchia) and its implications for ontogenetic determination in archosaurs. Journal of Vertebrate Paleontology, 27:350-361.
Janensch, W. 1929. Die Wirbelsaule der Gattung Dicraeosaurus. Palaeontographica, 2:1-34.
Klein, N. and Sander, M. 2008. Ontogenetic stages in the long bone histology of sauropod dinosaurs. Paleobiology, 34:247-263.
Knoll, F., Witmer, L.M., Ortega, F., Ridgely, R.C., and Schwarz-Wings, D. 2012. The braincase of the basal sauropod dinosaur Spinophorosaurus and 3D reconstructions of the cranial endocast and inner ear. PloS One, 7(1):e30060.
Lee, A.H. and Werning, S. 2008. Sexual maturity in growing dinosaurs does not fit reptilian growth models. Proceedings of the National Academy of Sciences, 105(2):582-587.
Lehman, T.M. and Woodward, H.N. 2008. Modelling growth rates for sauropod dinosaurs. Paleobiology, 34:264-281.
Lovelace, D.M., Hartman, S.A., and Wahl, W.R. 2007. Morphology of a specimen of Supersaurus (Dinosauria, Sauropoda) from the Morrison Formation of Wyoming, and a re-evaluation of diplodocid phylogeny. Arquivos do Museu Nacional, Rio de Janeiro, 65:527-544.
Lull, R.S. 1919. The sauropod dinosaur Barosaurus Marsh: redescription of the type specimens in the Peabody Museum, Yale University. Connecticut Academy of Arts and Sciences, 6:1-42.
Mannion, P.D., Upchurch, P., Mateus, O., Barnes, R.N., and Jones, M.E. 2012. New information on the anatomy and systematic position of Dinheirosaurus lourinhanensis (Sauropoda: Diplodocoidea) from the Late Jurassic of Portugal, with a review of European diplodocoids. Journal of Systematic Palaeontology, 10:521-551.
Mazzetta, G.V., Christiansen, P., and Farina, R.A. 2004. Giants and bizarres: body size of some southern South American Cretaceous dinosaurs. Historical Biology, 16(2-4):71-83.
McIntosh, J.S. 1981. Annotated catalogue of the dinosaurs (Reptilia, Archosauria) in the collections of Carnegie Museum of Natural History. Bulletin of the Carnegie Museum of Natural History, 18:1-67.
McIntosh, J.S. 1990. Species determination in sauropod dinosaurs with tentative suggestions for their classification, p. 53-69. Carpenter, K. and Currie, P.J. (eds.), Dinosaur Systematics: Approaches and Perspectives. Cambridge University Press.
McIntosh, J.S. 2005. The genus Barosaurus Marsh (Sauropoda, Diplodocidae), p. 38-77. In Tidwell, V. and Carpenter, K. (eds.), Thunder-Lizards: The Sauropodomorph Dinosaurs: Indiana University Press, Bloomington.
McIntosh, J.S., Miles, C.A., Cloward, K.C., and Parker, J.R. 1996. A new nearly complete skeleton of Camarasaurus. Bulletin of the Gunma Museum of Natural History, 1-87.
McIntosh, J.S. and Williams, M.E. 1988. A new species of sauropod dinosaur, Haplocanthosaurus delfsi sp. nov., from the Upper Jurassic Morrison Fm. of Colorado. Kirtlandia, 43:3-26.
Melstrom, K.M., D'emic, M.D., Chure, D., and Wilson, J.A. 2016. A juvenile sauropod dinosaur from the Late Jurassic of Utah, U.S.A., presents further evidence of an avian style air-sac system. Journal of Vertebrate Paleontology (advance online publication). DOI:10.1080/02724634.2016.1111898
Michelis, I. 2004. Taphonomie des Howe Quarry’s (Morrison-Formation, Oberer Jura), Bighorn County, Wyoming, USA. Unpublished PhD Thesis, Institute of Palaeontology, University of Bonn, Germany.
Mitchell, J., Sander, P.M., and Stein, K. 2017. Can secondary osteons be used as ontogenetic indicators in sauropods? Extending the histological ontogenetic stages into senescence. Paleobiology, 43: 1-22.
Mook, C.C. 1917. The fore and hind limbs of Diplodocus. Bulletin of the American Museum of Natural History, 37:815-819.
Myers, T.S. 2004. Taphonomy of the Mother's Day Quarry: Implications for Gregarious Behavior in Sauropod Dinosaurs. Unpublished PhD dissertation, University of Cincinnati, Cincinnati, Ohio, USA.
Myhrvold, N.P. 2013. Revisiting the estimation of dinosaur growth rates. PLoS One, 8(12):e81917.
Nash, J.F. 1950. Equilibrium points in n-person games. Proceedings of the National Academy of Sciences, 36:48-9.
Ostrom, J.H. and McIntosh, J.S. 1966. Marsh’s Dinosaurs. Yale University Press, New Haven and London.
Otero, A., Gallina, P.A., Canale, J.I., and Haluza, A. 2012. Sauropod haemal arches: morphotypes, new classification and phylogenetic aspects. Historical Biology, 24:243-256.
Padian, K., de Ricqlès, A.J., and Horner, J.R. 2001. Dinosaurian growth rates and bird origins. Nature, 412(6845):405-408.
Padian, K. and Lamm, E.T. (eds.) 2013. Bone Histology of Fossil Tetrapods: Advancing Methods, Analysis, and Interpretation. University of California Press, Berkeley.
Remes, K. 2009. Taxonomy of Late Jurassic diplodocid sauropods from Tendaguru (Tanzania). Fossil Record, 12:23-46.
Rogers, K.C. and Erickson, G.M. 2005. Sauropod histology, p. 303-326. In Curry Rogers, C. and Wilson, J.A. (eds), The Sauropods, Evolution and Paleobiology. University of California Press, Berkeley, California.
Rogers, K.C., Whitney, M., D’Emic, M., and Bagley, B. 2016. Precocity in a tiny titanosaur from the Cretaceous of Madagascar. Science, 352(6284):450-453.
Salgado, L. 1999. The macroevolution of the Diplodocimorpha (Dinosauria; Sauropoda): a developmental model. Ameghiniana, 36:203-216.
Salgado, L. and Bonaparte, J.F. 1991. Un nuevo sauropodo Dicraeosauridae, Amargasauruscazaui gen. et sp. nov., de la Formacion La Amarga, Neocomiano de la provincia del Neuquen, Argentina. Ameghiniana, 28:333-346.
Sampson, S.D., Ryan, M.J., and Tanke, D.H. 1997. Craniofacial ontogeny in centrosaurine dinosaurs (Ornithischia: Ceratopsidae): taxonomic and behavioral implications. Zoological Journal of the Linnean Society, 121:293-337.
Sander, P.M. 1999. Life history of Tendaguru sauropods as inferred from long bone histology. Fossil Record, 2(1):103-112.
Sander, P.M. 2000. Longbone histology of the Tendaguru sauropods: implications for growth and biology. Paleobiology, 26(3):466-488.
Sander, P.M., Klein, N., Buffetaut, E., Cuny, G., Suteethorn, V., and Le Loeuff, J. 2004. Adaptive radiation in sauropod dinosaurs: bone histology indicates rapid evolution of giant body size through acceleration. Organisms Diversity and Evolution, 4:165-173.
Sander, P.M. and Tückmantel, C. 2003. Bone lamina thickness, bone apposition rates, and age estimates in sauropod humeri and femora. Palaontologische Zeitschrift, 77:161-172.
Scannella, J.B., Fowler, D.W., Goodwin, M.B., and Horner, J.R. 2014. Evolutionary trends in Triceratops from the Hell Creek Formation, Montana. Proceedings of the National Academy of Sciences, 111:10245-10250.
Scannella, J.B. and Horner, J.R. 2010. Torosaurus Marsh, 1891, is Triceratops Marsh, 1889 (Ceratopsidae: Chasmosaurinae): synonymy through ontogeny. Journal of Vertebrate Paleontology, 30:1157-1168.
Schwarz, D., Frey, E., and Meyer, C.A. 2007a. Pneumaticity and soft-tissue reconstructions in the neck of diplodocid and dicraeosaurid sauropods. Acta Palaeontologica Polonica, 52(1):167.
Schwarz, D. and Fritsch, G. 2006. Pneumatic structures in the cervical vertebrae of the Late Jurassic Tendaguru sauropods Brachiosaurus brancai and Dicraeosaurus. Eclogae Geologicae Helvetiae, 99:65-78.
Schwarz, D., Ikejiri, T., Breithaupt, B.H., Sander, P.M., and Klein, N. 2007b. A nearly complete skeleton of an early juvenile diplodocid (Dinosauria: Sauropoda) from the lower Morrison formation (Late Jurassic) of North Central Wyoming and its implications for early ontogeny and pneumaticity in sauropods. Historical Biology, 19:225-253.
Schwarz-Wings, D. 2009. Reconstruction of the thoracic epaxial musculature of diplodocid and dicraeosaurid sauropods. Journal of Vertebrate Paleontology, 29:517-534.
Schwarz-Wings, D. and Frey, E. 2008. Is there an option for a pneumatic stabilization of sauropod necks? -- an experimental and anatomical approach. Palaeontologia Electronica, 11:1-26.
Senter, P. 2007. Necks for sex: sexual selection as an explanation for sauropod dinosaur neck elongation. Journal of Zoology, 271:45-53.
Seymour, R.S. 2009. Raising the sauropod neck: it costs more to get less. Biology Letters, 5:317-319.
Stein, K. and Sander, M. 2009. Histological core drilling: a less destructive method for studying bone histology, p. 69-80. In Brown, M.A., Kane, J.F., and Parker, W.G. (eds.), Methods In Fossil Preparation: Proceedings of the First Annual Fossil Preparation and Collections Symposium.
Stevens, K.A. and Parrish, J.M. 1999. Neck posture and feeding habits of two Jurassic sauropod dinosaurs. Science, 284:798-800.
Stevens, K.A. and Parrish, M.J. 2005a. Digital reconstructions of sauropod dinosaurs and implications for feeding, p. 178-200. In Curry Rogers, K. and Wilson, J.A. (eds.), The Sauropods, Evolution and Paleobiology. University of California Press, Berkeley, California.
Stevens, K.A., and Parrish, J.M. 2005b. Neck posture, dentition and feeding strategies in Jurassic sauropod dinosaurs, p. 212-232. In Tidwell, V. and Carpenter, K. (eds.), Thunder-lizards: The Sauropodomorph Dinosaurs. Indiana University Press, Bloomington, Indiana.
Storrs, G.W., Oser, S.E., and Aull, M. 2012. Further analysis of a Late Jurassic dinosaur bone-bed from the Morrison Formation of Montana, USA, with a computed three-dimensional reconstruction. Earth and Environmental Science Transactions of the Royal Society of Edinburgh, 103(3-4):443-458.
Taylor, M.P., Wedel, M.J., and Naish, D. 2009. Head and neck posture in sauropod dinosaurs inferred from extant animals. Acta Palaeontologica Polonica, 54:213-220.
Thompson, D’A.W. 1942. On Growth and Form, 2nd ed. Cambridge University Press, Cambridge, UK.
Tschopp, E. and Mateus, O. 2013. The skull and neck of a new flagellicaudatan sauropod from the Morrison Formation and its implication for the evolution and ontogeny of diplodocid dinosaurs. Journal of Systematic Palaeontology, 11:1-36.
Tschopp, E., Mateus, O.V., and Benson, R.B.J. 2015. A specimen-level phylogenetic analysis and taxonomic revision of Diplodocidae (Dinosauria, Sauropoda). PeerJ, 3:e857.
Tsuihiji, T. 2004. The ligament system in the neck of Rheaamericana and its implication for the bifurcated neural spines of sauropod dinosaurs. Journal of Vertebrate Paleontology, 24:165-172.
Tsuihiji, T., Watabe, M., Tsogtbaatar, K., Tsubamoto, T., Barsbold, R., Suzuki, S., Lee, A.H., Ridgely, R.C., Kawahara, Y., and Witmer, L.M. 2011. Cranial osteology of a juvenile specimen of Tarbosaurus bataar (Theropoda, Tyrannosauridae) from the Nemegt Formation (Upper Cretaceous) of Bugin Tsav, Mongolia. Journal of Vertebrate Paleontology, 31:497-517.
Turner, C.E. and Peterson, F. 1999. Biostratigraphy of dinosaurs in the upper Jurassic Morrison Formation of the western interior, USA, p. 77-114. In Gillette, D.D. (ed.), Vertebrate paleontology in Utah. Utah Geological Survey Miscellaneous Publication, Salt Lake City, Utah.
Upchurch, P., Barrett, P.M., and Dodson, P. 2004a. Sauropoda, p. 259-322. In Weishampel, D.B., Dodson, P., and Osmolska, H. (eds.), The Dinosauria. Second edition. University of California Press, Berkeley, California.
Upchurch, P., Tomida, Y., and Barrett, P.M. 2004b. A new specimen of Apatosaurus ajax (Sauropoda: Diplodocidae) from the Morrison Formation (Upper Jurassic) of Wyoming, USA. National Science Museum Monographs, 26:1-118.
van Tienhoven, A. 1983. Reproductive Physiology of Vertebrates. Cornell University Press, London.
Waskow, K. and Sander, P.M. 2014. Growth record and histological variation in the dorsal ribs of Camarasaurus sp. (Sauropoda). Journal of Vertebrate Paleontology, 34:852-869.
Waskow, K. and Mateus O. 2017. Dorsal rib histology of dinosaurs and a crocodile from western Portugal: Skeletochronological implications on age determination and life history traits. Comptes Rendus Palevol (advance online publication) doi: http://doi.org/10.1016/j.crpv.2017.01.003
Wedel, M.J., Cifelli, R.L., and Sanders, R.K. 2000. Osteology, paleobiology, and relationships of the sauropod dinosaur Sauroposeidon. Acta Palaeontologica Polonica, 45:343-388.
Wedel, M.J. 2003. The evolution of vertebral pneumaticity in sauropod dinosaurs. Journal of Vertebrate Paleontology, 23:344-357.
Wedel, M.J. 2005. Postcranial skeletal pneumaticity in sauropods and its implications for mass estimates, p. 201-228. In Curry Rogers, K. and Wilson, J.A. (eds.), The Sauropods: Evolution and Paleobiology. University of California Press, Berkeley, California.
Wedel, M.J. 2009. Evidence for bird-like air sacs in saurischian dinosaurs. Journal of Experimental Zoology A, 311:1-18.
Wedel, M.J. and Taylor, M.P. 2013. Neural spine bifurcation in sauropod dinosaurs of the Morrison Formation: ontogenetic and phylogenetic implications. Palarch’s Journal of Vertebrate Palaeontology, 10(1):1-34.
Whitlock, J.A. 2011a. A phylogenetic analysis of Diplodocoidea (Saurischia: Sauropoda). Zoological Journal of the Linnean Society, 161:872-915.
Whitlock, J.A. 2011b. Inferences of diplodocoid (Sauropoda: Dinosauria) feeding behavior from snout shape and microwear analyses. PLoS One, 6(4):e18304.
Whitlock, J.A. and Harris, J.D. 2010. The dentary of Suuwassea emilieae (Sauropoda: Diplodocoidea). Journal of Vertebrate Paleontology, 30:1637-1641.
Whitlock, J.A., Wilson, J.A., and Lamanna, M.C. 2010. Description of a nearly complete juvenile skull of Diplodocus (Sauropoda: Diplodocoidea) from the Late Jurassic of North America. Journal of Vertebrate Paleontology, 30:442-457.
Wilson, J.A. and Smith, M. 1996. New remains of Amphicoelias Cope (Dinosauria: Sauropoda) from the Upper Jurassic of Montana and diplodocoid phylogeny. Journal of Vertebrate Paleontology, 16(Suppl. 3):73A.
Wilhite, R. 1999. Ontogenetic variation in the appendicular skeleton of the genus Camarasaurus. Master Thesis, Brigham Young University, Provo, Utah, USA.
Wilhite, R. 2003. Biomechanical reconstruction of the appendicular skeleton in three North American Jurassic sauropods. PhD Thesis, Louisiana State University, Baton Rouge, Louisiana, USA.
Wilhite, R. and Curtice, B. 1998. Ontogenetic variation in sauropod dinosaurs. Journal of Vertebrate Paleontology, 18(Suppl. 3):86A.
Woodruff, D.C. 2014. The anatomy of the bifurcated neural spine and its occurrence within Tetrapoda. Journal of Morphology, 275:1053-1065.
Woodruff, D.C. 2016. Nuchal ligament reconstructions in diplodocid sauropods support horizontal neck feeding postures. Historical Biology (advance online publication). DOI:10.1080/08912963.2016.1158257
Woodruff, D.C. and Foster, J.R. 2014. The fragile legacy of Amphicoelias fragillimus (Dinosauria: Sauropoda; Morrison Formation-latest Jurassic). Volumina Jurassica, 12(2):211-220.
Woodruff, D.C. and Foster, J.R. 2017. The first specimen of Camarasaurus (Dinosauria: Sauropoda) From Montana: The northernmost occurrence of the genus. PLoS ONE, 12(5):e0177423. doi.org/10.1371/journal.pone.0177423
Woodruff, D.C. and Fowler, D.W. 2012. Ontogenetic influence on neural spine bifurcation in Diplodocoidea (Dinosauria: Sauropoda): a critical phylogenetic character. Journal of Morphology, 273:754-764.
Woodruff, D.C. and Fowler, D.W. 2014. The effects Of ontogeny in regards to morrison sauropod diversity. Mid-Mesozoic: The age of dinosaurs in transition. Society of Vertebrate Paleontology 2014 Abstract Volume.
Woodruff, D.C., Fowler, D.W., and Horner, J.R. 2013. Changes in vertebral morphology associated with histologic data support significant change through ontogeny in diplodocid sauropods. Society of Vertebrate Paleontology 2013 Abstract Volume.
Woodward, H.N., Freedman Fowler, E.A., Farlow, J.O., and Horner, J.R. 2015. Maiasaura, a model organism for extinct vertebrate population biology: a statistically robust assessment of growth dynamics and survivorship. Paleobiology, 41:503-527.
Woodward, H.N. and Lehman, T.M. 2009. Bone histology and microanatomy of Alamosaurus sanjuanensis (Sauropoda: Titanosauria) from the Maastrichtian of Big Bend National Park, Texas. Journal of Vertebrate Paleontology, 29:807-821.

